Abstract
In February 2012, 12 farmed mule deer (Odocoileus hemionus) were moved from a facility in southwestern Oklahoma to a facility in southeastern Oklahoma that housed 100 farmed white-tailed deer (Odocoileus virginianus). Between the third and fifth weeks, 9 of the 12 mule deer had died, 4 of which were submitted for necropsy. The deer were heavily infested with Amblyomma americanum (lone star ticks). Hematologic data from 1 deer revealed severe anemia, leukocytosis, and intraerythrocytic hemoparasites consistent with Theileria spp. Microscopically, the liver, lymph nodes, and spleen contained multifocally distributed, enlarged monocytic cells whose cytoplasm was replaced by developing meronts in various stages of merogony. It appears that, upon arrival, the Theileria cervi–naïve mule deer became infested with large numbers of Theileria-infected lone star ticks leading to massive exposure of the mule deer to sporozoites of the protozoan, resulting in an acute hemolytic crisis and fatalities. The merogonic stages of T. cervi are also described. The lack of earlier reports of merogony may be due to the fact that only a single, short-lived, merogonic cycle follows exposure to sporozoites and thus merogonic stages are demonstrable for only a short period. Polymerase chain reaction testing of paraffin-embedded tissue yielded a 507-bp amplicon sequence that was 100% identical with the sequence of T. cervi previously reported from white-tailed deer in Oklahoma and from elk in Wisconsin and Indiana.
Theileria spp. are cosmopolitan apicomplexan hemoparasites of domestic and wild ungulates. Some species, such as T. parva and T. annulata, are highly pathogenic parasites of cattle; infection results in high morbidity and mortality in naïve cattle. 11 Other bovine isolates, such as T. buffeli, T. sergenti, and T. mutans, cause mild to moderate clinical disease. 11 In 1962, a Theileria species was recognized in the blood of white-tailed deer (Odocoileus virginianus) in the United States.8,14 Subsequently, Theileria cervi infections have been reported in white-tailed deer from states including Texas, Oklahoma, Missouri, Arkansas, Alabama, Florida, Georgia, South Carolina, Maryland, and Virginia,4,6 as well as in mule deer (Odocoileus hemionus), elk (or wapiti; Cervus elaphus canadensis), sika deer (Cervus nippon nippon), and axis deer (or chital; Axis axis).2,10,15 Fallow deer (Cervus dama), however, were resistant to experimental infections. 7 Although approximately 50% of the white-tailed deer population in enzootic areas may be infected with T. cervi, reports of clinical disease are rare.1,12 However, it has been observed that cervid theileriosis and mortality can occur when population densities of deer are high, nutrition is poor, and other diseases coincide with the T. cervi infection.1,4,12 Fawn mortality can occur when parasitemia is high.1,16
It is well established that Amblyomma americanum is the definitive host and biologic vector of T. cervi.9,10,13 Furthermore, white-tailed deer are a major host for A. americanum. 5 Both white-tailed deer and A. americanum are populous species in the eastern two thirds of Oklahoma and Texas, but the distribution of both is greatly diminished in the western one third of both states. 3
The current report addresses a high-morbidity, high-mortality outbreak of theileriosis in mule deer shortly after they were translocated from far western to southeastern Oklahoma and describes merogonic development of the parasite within tissues of 4 naturally infected mule deer. Circumstances leading to the disease outbreak and evidence of the mechanism by which the etiologic agent caused disease are highlighted.
In February 2012, 12 young farmed mule deer were moved from a facility in Beckham County, western Oklahoma to a facility in Bryan County, southeastern Oklahoma. The mule deer were placed in a newly established, previously unoccupied, 3 acre, partially forested pen that had been added adjacent to an existing 9-pen facility, sharing a common fence with a pen occupied by approximately 100-resident, tick-infested white-tailed deer. A corn-based product and Bermuda hay were provided as nutritional supplements to available browse and pasturage. The attending veterinarian characterized husbandry as “very good.” However, the nutritional condition of the mule deer on arrival was judged “moderately poor.”
Within 3 weeks of arrival at the new facility, a poorly defined illness was recognized. By mid-March 2012, 4 young adult mule deer had died and were submitted to the Oklahoma Animal Disease Diagnostic Laboratory (Stillwater, Oklahoma) for necropsy. Within the following 2 weeks, a total of 9 of the original 12 had succumbed to the illness. Throughout this period, neither illness nor death was recognized in the resident white-tailed deer.
Gross necropsy revealed the mule deer to be in moderately poor body condition and heavily infested with ticks confirmed as A. americanum. The first deer examined by necropsy was severely autolyzed, but had a chronic fibrous pleuritis. The second deer submitted had ulcerative abomasitis, splenomegaly, and a mildly congested liver. The third and fourth deer had 1–2 mm, multifocal grayish lesions visible on the capsular and cut surfaces of their livers. The fourth deer also had a prominently enlarged brownish spleen; the serum was yellowish and the viscosity of its blood seemed low. The alimentary tract, heart, adrenal glands, lungs, thyroid glands, trachea, esophagus, skeletal muscle, and brain of each deer were grossly unremarkable.
Specimens of brain, lung, heart, liver, spleen, rumen, abomasum, adrenal glands, thyroid glands, esophagus, trachea, skeletal muscle, small and large intestine, kidney, and lymph node were collected from each, fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned at 5 µm, and routinely stained with hematoxylin and eosin. Selected sections were variously stained with Perls stain for iron and von Kossa stain for calcium. Furthermore, whole blood from an ill deer was submitted for analysis.
Salient histologic findings included randomly distributed, ovoid, intracellular, 275 μm × 325 μm structures (mean size: 125 μm × 150 µm) representing various stages of merogony within the liver, lymph nodes, and spleen. Developing meronts replaced the cytoplasm of greatly hypertrophied monocytic host cells that had single, greatly enlarged, elongate, serpentine nuclei with coarsely stippled chromatin and indistinct nucleoli (Fig. 1A). The cytoplasm of infected monocytes initially appeared to contain finely granular basophilic material that, with further development, became discrete, 1–2 µm, pinpoint, basophilic structures (developing merozoites; Fig. 1B). As maturation of merozoites progressed, basophilic structures aggregated into clusters (cytomeres; Fig. 1C), and degeneration of the host cell led to rupture and release of encysted zoites into the surrounding tissue where they incited a robust pyogranulomatous inflammatory response, a zone of hemorrhage, a narrow zone of necrosis, and eventually fibrosis (Fig. 1D).
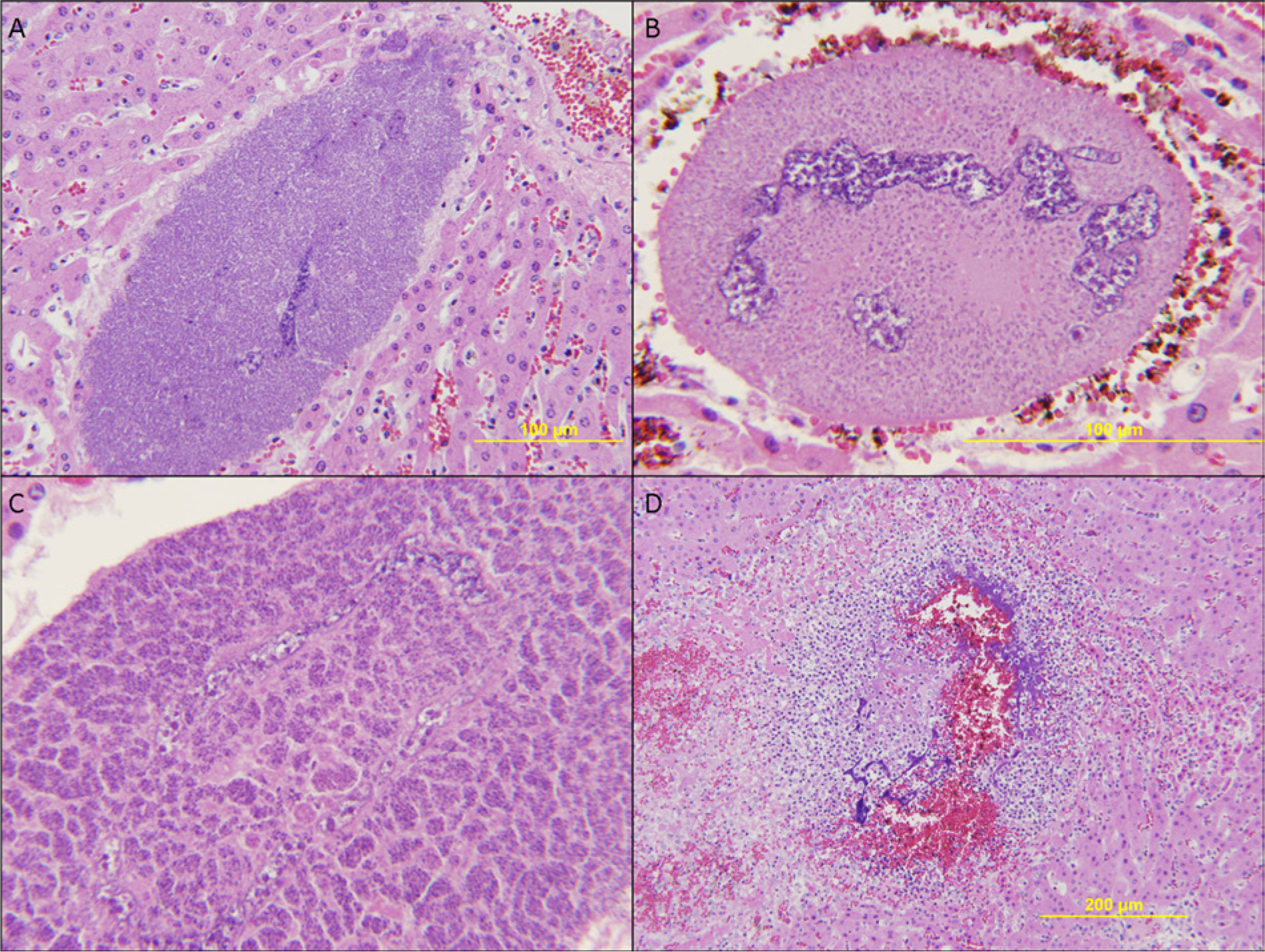
Mule deer (Odocoileus hemionus).
In addition to active parasitic cysts and inflammatory lesions associated with resolution of the cysts, each of the 4 mule deer had prominent splenic hemosiderosis and lesser degrees of hemosiderosis in the liver and lung. Three deer had hemoglobin casts in renal tubular lumens and pinocytized hemoglobin in renal tubular epithelial cells. The second deer had especially severe hemoglobinuric nephrosis and renal failure as evidenced by severe ulcerative and mineralized uremic abomasitis. Histologic examination of multiple specimens of skeletal muscle from each animal did not reveal myodegeneration, further indicating that the iron-containing pigment in the kidney was hemoglobin rather than myoglobin.
Giemsa-stained impression smears from the liver of the first mule deer revealed intraerythrocytic hemoparasites consistent with Theileria species as well as tortuous, elongated monocytic host cell nuclei and associated aggregates of small zoites within the cytoplasm of the monocytic host cells. Analysis of the whole blood submitted from a fifth deer revealed severe anemia, as well as leukocytosis and numerous intraerythrocytic hemoparasites in both ring and paperclip forms that infected approximately 15% of the examined erythrocytes.
At the time of the outbreak in the Oklahoma mule deer, a captive white-tailed deer in Kentucky was submitted to Murray State University (Hopkinsville, Kentucky) for necropsy. Interestingly, collaborative histologic review of stained paraffin-embedded tissue sections revealed identical lesions to those observed in the Oklahoma mule deer.
Whole blood in ethylenediamine tetra-acetic acid from the fourth and fifth Oklahoma mule deer and the Kentucky white-tailed deer was extracted for total DNA; nested polymerase chain reaction (PCR) assay was performed as previously described to amplify a hypervariable region of the 18S ribosomal RNA (rRNA) gene (V4) of Theileria spp. and some other apicomplexa. 16 Owing to difficulties encountered with direct sequencing attempts from whole blood, cloning into plasmid vectors was performed on gel-purified amplicons. A total of 11 approximately 500–base pair (bp) sequences were obtained from the blood of the 3 deer. Sequences were compared with data available in the National Center for Biotechnology Information database (GenBank), and identity ranged from 95.8–100% to T. cervi sequences documented from white-tailed deer in Texas and a Wisconsin elk (AY735135, U97054, U97055, and U97056) and 95.8–99.8% to T. buffeli (AF162432) from a Missouri cow.
While the PCR and subsequent amplicon sequencing of whole blood from the mule and white-tailed deer supported the premise that the deer were likely infected with Theileria sp., the tests did not absolutely establish the identity of merogonic forms in tissue. Consequently, DNA was extracted from five 10-µm sections of paraffin-embedded tissues from the second and fourth mule deer that contained numerous meronts. Again, PCR amplification of the V4 variable region of the 18S rRNA gene was performed, and correctly sized amplicons were obtained from the tissues of both deer. 16 Amplicons (507 bp) from the fourth deer were sequenced from plasmid vectors, and were 100% identical to the sequence of T. cervi previously reported from white-tailed deer in Oklahoma and from elk in Wisconsin and Indiana (GenBank accession nos. AY735122, AY735135, and AY735120). The next closest species match was only 96% homologous to a sequence documented as T. ovis (JQ737135), providing strong support that the merogenic tissue forms were T. cervi. Tests for Bluetongue virus and Epizootic hemorrhagic disease virus by PCR and chronic wasting disease by immunohistochemical staining were uniformly negative.
The high morbidity and mortality in this group of mule deer are unusual manifestations of T. cervi infection. Considering that T. cervi infection and A. americanum population densities are associated, 10 and morbidity and mortality are related to the parasitic infection, 1 a hypothesis for pathogenesis has been proposed. Evidence of hemolysis was evident in each of the necropsied deer. The following conditions were found: splenic hemosiderosis in all deer; hemoglobinuric nephrosis in 3 of the 4 deer; phagocytized iron-containing pigment in cells of the liver and lung of all 4 deer; and piroplasms in 3 deer. Severe regenerative anemia was found in the 1 deer from which hematologic data was available. The data indicates that severe intravascular hemolysis was the demonstrable common pathogenic mechanism for illness in the deer of the outbreak described herein.
Because T. cervi infection is typically subclinical,1,12 the high morbidity and mortality in the outbreak described herein was unusual. It is unlikely that, prior to translocation, these mule deer had been exposed to significant numbers of lone star ticks or T. cervi as they were from a geographic area relatively free of A. americanum. Several factors contributed to the severity of the outbreak. The deer were essentially naïve to T. cervi, and were translocated from a geographic area (western Oklahoma) where the definitive host tick A. americanum is absent or rare, 3 to an established deer farm in southeastern Oklahoma inhabited by white-tailed deer heavily infested with A. americanum and presumably chronically infected with T. cervi. The history suggested that, upon arrival, the mule deer, naïve to T. cervi infection, became immediately infested with large numbers of Theileria-infected lone star ticks resulting in massive exposure of the mule deer to sporozoites of the protozoan. With completion of merogony, merozoites massively infected erythrocytes, precipitating an acute hemolytic process and the fatal disease outbreak. Similar disease was not recognized in the white-tailed deer.
The absence of descriptions of merogonic stages of T. cervi was unexpected as the parasite was first identified in 1962. However, as only a single merogonic cycle follows exposure to sporozoites, merogonic stages are demonstrable only during a short period following exposure. This and the ease of diagnosis by examination of stained blood smears help explain the long interval between original recognition of T. cervi infection and description of the merogonic stages.
Footnotes
Acknowledgements
The authors would like to thank Jeff Gruntmeir for his assistance with the polymerase chain reaction testing of blood samples and Betty Handlin for assistance with photography.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
