Abstract
Brucella abortus RB51 is the vaccine strain currently licensed for immunizing cattle against brucellosis in the United States. Most cattle are vaccinated as heifer calves at 4–12 months of age. Adult cattle may be vaccinated in selected high-risk situations. Two herds of pregnant adult cattle in the brucellosis-endemic area of Wyoming were vaccinated with a standard label dose (1.0–3.4 × 1010 organisms) of RB51. Reproductive losses in the vaccinated herds were 5.3% (herd A) and 0.6% (herd B) and included abortions, stillbirths, premature calves, and unbred cows (presumed early abortion). Brucella abortus was cultured from multiple tissues of aborted and premature calves (7/9), and from placenta. Isolates were identified as B. abortus strain RB51 by standard strain typing procedures and a species-specific polymerase chain reaction. Bronchopneumonia with intralesional bacteria and placentitis were observed microscopically. There was no evidence of involvement of other infectious or toxic causes of abortion. Producers, veterinarians, and laboratory staff should be alert to the risk of abortion when pregnant cattle are vaccinated with RB51, to potential human exposure, and to the importance of distinguishing field from vaccinal strains of B. abortus.
Brucellosis is one of the most important historical causes of bovine abortion in the United States. The Federal-State Cooperative Brucellosis Eradication Program was started in 1934 by the U.S. Department of Agriculture (USDA) and resulted in the virtual eradication of brucellosis in domestic cattle in the United States (USDA, Animal and Plant Health Inspection Service: 2012, National Bovine Brucellosis Surveillance Plan). The disease remains endemic in free-ranging elk (Cervus elaphus nelsoni) and bison (Bison bison) in the Greater Yellowstone Area (GYA) of Idaho, Montana, and Wyoming. 16 This wildlife reservoir is a risk to livestock in and around the GYA. Owners of cattle herds in these at-risk areas may use adult vaccination along with other approved procedures to prevent the introduction of Brucella abortus and to control its spread should brucellosis occur. This requires that a whole herd vaccination plan is developed and approved by the state veterinarian and Animal and Plant Health Inspection Service (APHIS) Area Veterinarian in Charge (USDA, APHIS: 2003, Brucellosis eradication: uniform methods and rules). Brucella abortus strain RB51 was licensed for use in cattle by APHIS in 1996, replacing the previously used strain 19 (S19). Experimental studies indicate that strain RB51 is less abortigenic in cattle than S19, does not produce clinical signs of disease, is cleared from the bloodstream within 3 days of vaccination, and is absent from nasal secretions, saliva, and urine. The vaccine is considered safe in cattle older than 3 months. The preferred vaccination strategy is to vaccinate calves at 4–12 months of age with a label dose of 1.0–3.4 × 1010 organisms (USDA, APHIS: 2003, Brucellosis eradication: uniform methods and rules). Protection from Brucella infection is similar across the dosage range (i.e., larger vaccine doses do not confer greater protection against challenge). 10 Adult vaccination is not routinely used for disease control as there are risks associated with RB51 use in sexually mature animals. Brucella abortus RB51 can infect placenta, mammary gland, and fetal tissues, inducing placentitis and, in some cases, preterm birth. 11 In specific high-risk situations, it may be appropriate to vaccinate cattle older than 12 months (USDA, APHIS: 2003, Brucellosis eradication: uniform methods and rules).
Sublette County in Wyoming is located in the Designated Surveillance Area of the GYA and is considered a high-risk area for brucellosis transmission. Thirteen of Wyoming’s 23 winter elk feedgrounds, which result in high concentrations of elk, are located in the county. The feedgrounds represent a risk for the transmission of brucellosis from elk to cattle. The last major outbreak of bovine brucellosis in Wyoming occurred in Sublette County in late 2003, resulting in 31 reactor cattle. Limited information is available about reproductive losses associated with RB51 used under field conditions.8,15 Observational field studies using large populations represent the most meaningful test of the efficacy of vaccines and adverse side effects, but interpretation can be confounded by many factors. 7 RB51 was associated with an abortion rate of 0.52% when 1,000 herds and approximately 40,000 cattle were vacated in the Extremadura region of western Spain. 15
Personnel at the Wyoming State Veterinary Laboratory (WSVL; Laramie, Wyoming) diagnosed 3 herd episodes of abortion due to RB51 vaccination of pregnant cattle in 2006–2007, 2008–2009, and 2011. The current report describes the first 2 episodes (2007 and 2009).
Adult cattle of 4 beef herds totaling 1,600 pregnant cows at approximately 4 months gestation were vaccinated in November 2006 with a full calfhood dose (1.0–3.4 × 1010 organisms) of B. abortus strain RB51 in Sublette County, Wyoming. The herd owners were advised that vaccinating pregnant cattle could result in reproductive losses. All cows were tested for pregnancy in the fall, and only pregnant animals were retained. No ill effects were recognized until 101 days postvaccination (February 2007) when the owner of herd A (280 cows, 80 heifers) noted a late-term abortion. All heifers had been calfhood vaccinated with RB51 in October 2006. The aborting cow retained the placenta for 1 week. Other cattle in herd A aborted or delivered premature weak or stillborn calves over the following 49 days, with most losses at 126–135 days postvaccination. Total losses were 9 abortions (7 cows, 2 heifers), 5 stillborn or premature weak live born calves (2 stillbirths, 3 premature), and 5 assumed abortions from 4 cows and 1 heifer that were not pregnant at calving (total: 19/360; 5.3%). Historical average losses in this herd due to reproductive wastage between pregnancy check time and birth ranged from 0.2% to 0.5% annually. Samples from 4 calves (animals 1–4 in Table 1) were submitted to the WSVL for necropsy and associated testing. Samples from another 3 fetuses (animals 5–7 in Table 1) were submitted to the National Veterinary Services Laboratories (NVSL; USDA, Ames, Iowa). No abortions or other losses were recognized by the owners of the other 3 herds that were vaccinated at the same time as herd A. These 3 herds were considered to be at a higher risk of brucellosis because they were geographically located near an affected index herd where multiple cattle were identified as B. abortus reactors in 2003. For that reason, and unlike herd A, unbred cattle in these herds received an additional spring vaccination (March–May 2006) of RB51.
Bacteriologic, virologic, and histologic results from aborted and premature bovine calves from 2 Wyoming cattle herds (A and B).*
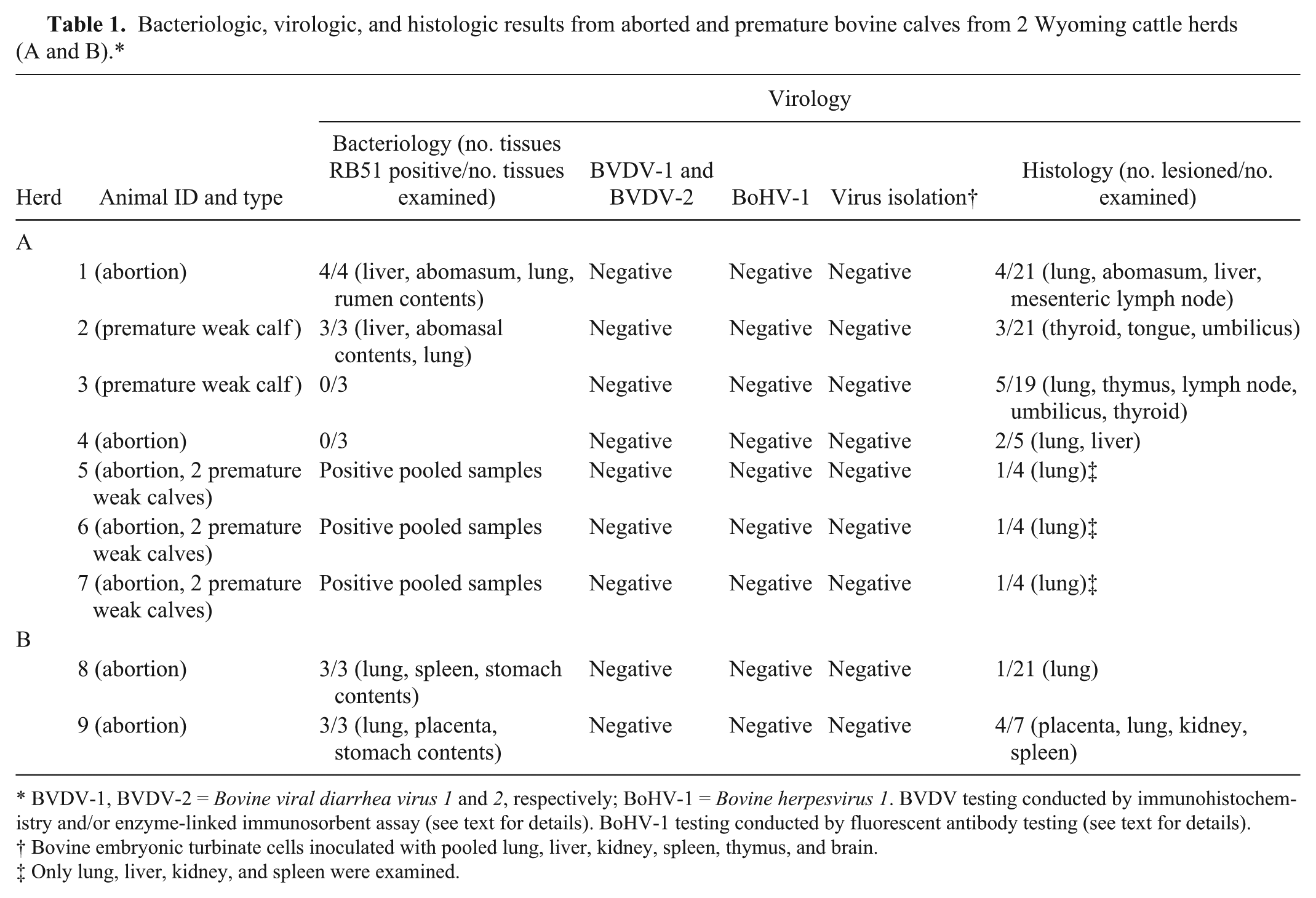
BVDV-1, BVDV-2 = Bovine viral diarrhea virus 1 and 2, respectively; BoHV-1 = Bovine herpesvirus 1. BVDV testing conducted by immunohistochemistry and/or enzyme-linked immunosorbent assay (see text for details). BoHV-1 testing conducted by fluorescent antibody testing (see text for details).
Bovine embryonic turbinate cells inoculated with pooled lung, liver, kidney, spleen, thymus, and brain.
Only lung, liver, kidney, and spleen were examined.
A second abortion episode following RB51 vaccination of pregnant cattle occurred in March 2009 on another Wyoming ranch, identified as herd B. Two abortions and 1 weak calf were observed, all from first-calf, 3-year-old pregnant heifers in herd B, which consisted of 475 cattle, a reproductive loss of 0.6% (3/475). All cattle 2 years or older received a standard label dose of the vaccine at approximately 3–5 months of gestation in November 2008. Herd management plans dictated that all cattle in the herd were vaccinated as heifer calves (<1 year old) in the fall of their birth year, and given an adult booster vaccination in the spring (age of 1+ years). In 2006, the herd plan was changed to biennial fall booster vaccination. As a result, the 3-year-old cows were calfhood vaccinated in spring 2006 and received their first adult booster in November 2008 with no vaccination in the interim. Both aborted calves were submitted to WSVL for necropsy along with a sample of placenta (animals 8 and 9 in Table 1).
Necropsy examinations were performed on aborted fetuses and neonatal calves (animals 1–9 in Table 1). Tissues were collected and fixed for 24–48 hr in 10% neutral buffered formalin for histology. A set of samples (abomasal fluid, lung, liver) was collected fresh for bacterial culture. 17 Sections of fixed tissues were embedded in paraffin, sectioned at 5 µm, and stained with hematoxylin and eosin for examination by light microscopy.
Gross and microscopic findings for animals whose tissues or carcasses were submitted from both herds are summarized (Table 1). Six of 9 calves had mild to moderate neutrophilic bronchopneumonia with scant intralesional bacteria (animals 1, 3, 4–7, 9; Table 1; Fig. 1). Multifocal necrotizing placentitis with intralesional bacteria and necrosis of villi was present in 1 animal (animal 9 in Table 1; Fig. 2). Less consistent lesions were omphalitis (animals 2 and 3), lymphadenitis (animals 1 and 3), catarrhal abomasitis with intralesional bacteria (animal 1), and pleocellular portal hepatitis (animals 1 and 4). Other changes were mild hyperplastic goiter (animals 2 and 3), lingual edema (animal 2), mild thymic atrophy (animal 3), fetal atelectasis without pneumonia (animal 8), and multifocal lymphocytic interstitial nephritis with mild lymphoid hyperplasia in spleen (animal 9).
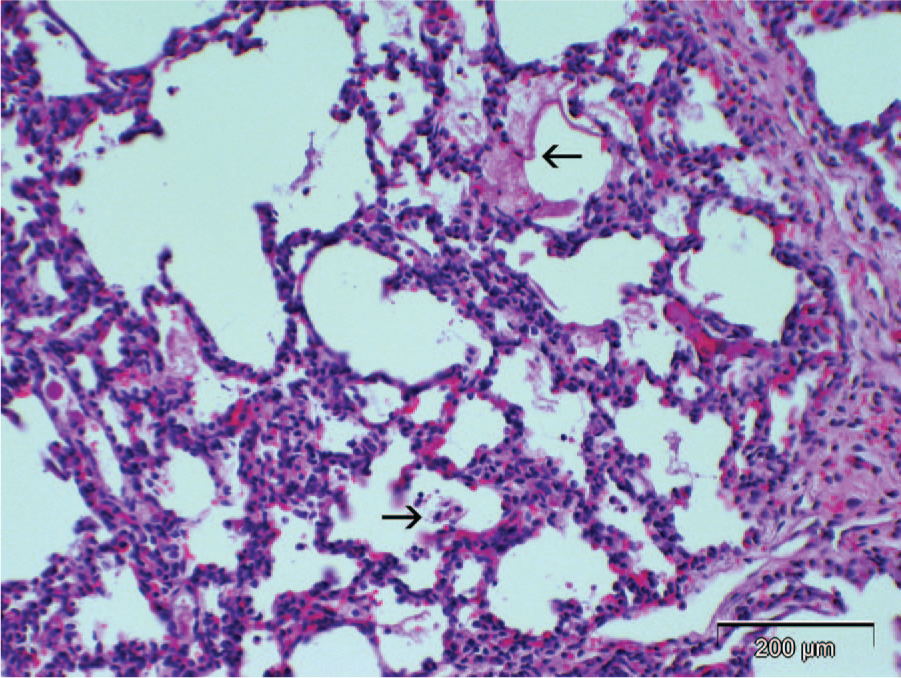
Aborted fetal calf; lung. Mild diffuse interstitial pneumonia with minimal intra-alveolar fibrinous exudate (arrows). Brucella abortus strain RB51 was isolated from lung of calf. Hematoxylin and eosin. Bar = 200 µm.
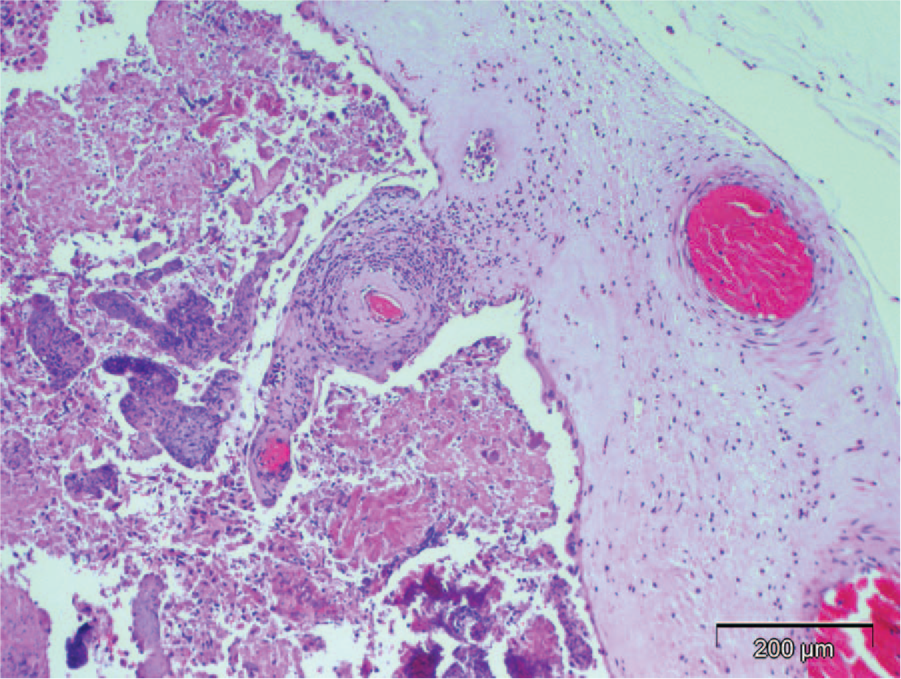
Adult cow; placenta. Necrosuppurative placentitis with intralesional bacteria (Brucella abortus), vasculitis, and mineralized cellular debris. The organism was confirmed as strain RB51 by a B. abortus species–specific polymerase chain reaction. Placental lesions of iatrogenic abortion due to RB51 are indistinguishable from those due to field strains of brucellosis. Hematoxylin and eosin. Bar = 200 µm.
Lung, liver, and abomasal contents were plated on Columbia blood agar a for attempted bacterial isolation; plates were incubated at 37°C in 10% CO2 atmosphere. Brucella isolates were identified using NVSL criteria along with standard and differential Brucella culture techniques. 2 Isolates with the characteristics of B. abortus were submitted to NVSL for confirmation. Brucella abortus was isolated from 1 premature calf, 6 aborted fetuses, and 1 placental sample (7/9 in Table 1; total from both herds).
A cell suspension was made from bacterial cultures in a methanol b –saline solution (3:1) and used as a template for the B. abortus species–specific polymerase chain reaction (BaSS-PCR). 6 The PCR differentiates isolates of B. abortus as field strain, S19, or RB51 on the basis of amplicon size. Bacteria isolated in culture were confirmed by BaSS-PCR as B. abortus strain RB51 (Fig. 3; Table 2). Confirmation of B. abortus RB51 was made at NVSL using similar criteria.
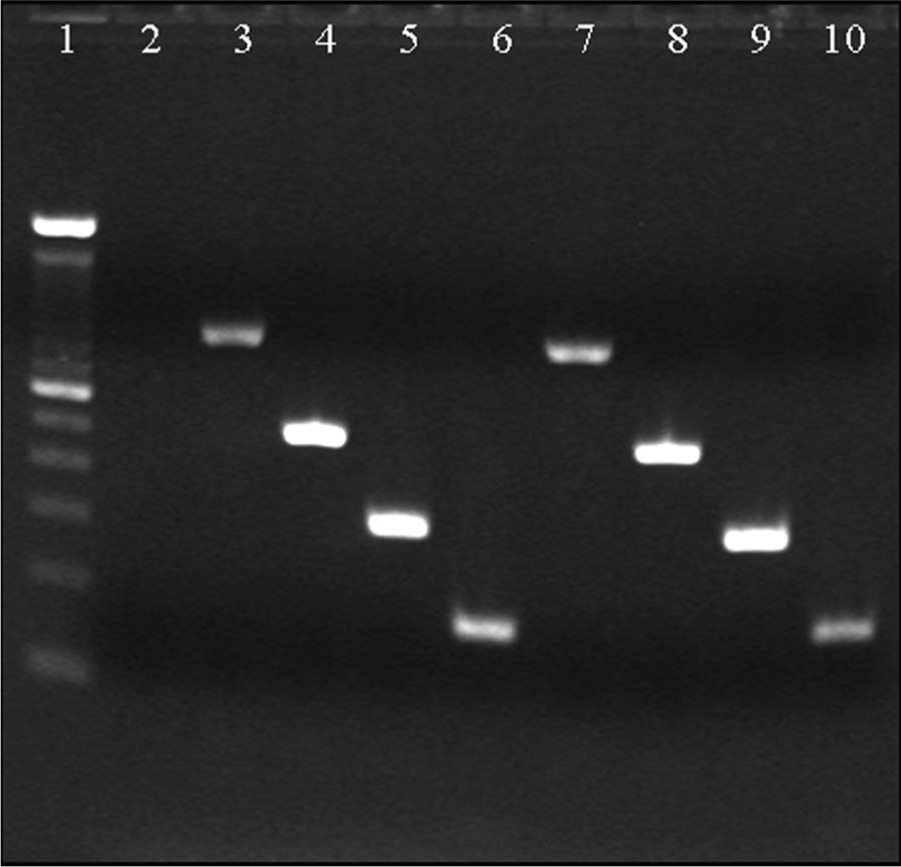
Resultant gel electrophoresis from polymerase chain reaction (PCR) of bacterial cultures to confirm the identity of Brucella abortus strain RB51. Lane 1: 100-bp DNA ladder; lane 2: negative control; lanes 3–6: RB51 positive control; lanes 7–10: DNA extracted from cultured bacteria. Amplification of all 4 products identifies the bacterial DNA as B. abortus RB51 (Table 2). The primer set for each locus (#1–4) was used in separate PCR reactions and visualized in separate lanes of the gel during electrophoresis.
Primer combinations for polymerase chain reaction (PCR) for the detection of Brucella abortus by the B. abortus species–specific PCR (BaSS-PCR) and expected product sizes.*
The BaSS-PCR test identifies B. abortus and distinguishes between biovars 1, 2, and 4, strain 19, and RB51. Identification of B. abortus RB51 is determined by the amplification of all 4 products.
Virus isolation was attempted from pooled tissues. No viruses were isolated from bovine embryonic turbinate following inoculation with pooled samples of lung, liver, kidney, spleen, thymus, and brain from carcasses submitted to WSVL. Sections of brain, kidney, liver, lung, spleen, and heart from 1 aborted fetus (animal 1) and aural skin from the premature calves (animals 3 and 4) were stained for Bovine viral diarrhea virus 1 and 2 (BVDV-1 and -2) antigen by immunohistochemistry using a monoclonal antibody c as previously described, 4 with negative results. Skin from all calves was tested for BVDV with the ear notch antigen-capture enzyme-linked immunosorbent assay using a commercially available kit d according to the manufacturer’s instructions, again with negative results. A direct fluorescent antibody test for Bovine herpesvirus 1 using specific polyclonal antibody e was negative in sections of liver, lung, and spleen in all animals.
Fresh and fixed tissues from 3 additional aborted fetuses in herd A were submitted to NVSL by the USDA veterinarian for bacteriology, virus isolation, and histology (animals 5–7; Table 1). Microscopic examination of fixed tissues exhibited mild exudative pneumonia consisting of minimal to moderate numbers of neutrophils and scant aggregates of mononuclear inflammatory cells in alveoli. There were no lesions in kidney, spleen, or liver in any of the calves. There was no evidence of viral infection in bovine turbinate cell cultures. Cells were negative for BVDV using an indirect fluorescent antibody assay. Brucella abortus RB51 was isolated by bacterial culture from pooled tissues (lung, liver, kidney, spleen) from each of the 3 fetuses.
There are few reports of RB51-induced abortion in commercial herds.15,18,20 The results of experimental studies indicate that vaccination with RB51 in pregnant cattle is generally safe.5,11,12,14,18 Total reproductive losses in the 2 herds were 5.3% (19/360, herd A) and 0.6% (3/475, herd B). In herd A, losses comprised abortion, stillbirths, or premature weak calves, as well as some open cattle. Open cattle are included in the present analysis because all cows retained in the herd were confirmed pregnant by a veterinarian in the fall, indicating subsequent fetal loss. Typical reproductive loss among retained animals between pregnancy check time and birth was 1–2 of 360 animals (0.2–0.5%), according to the owner. The occurrence of 5 cattle that were pregnant in the fall and open at calving time was atypically high. It is likely that most of the calves examined from herd A died due to infection with B. abortus RB51. Of those 7 animals, RB51 was isolated from 5 (Table 1). No other pathogens were isolated. Of the 2 remaining animals, 1 had bronchopneumonia (animal 3; Table 1), which is a feature consistent with brucellosis. Omphalitis and lymphadenitis in this animal were also consistent with bacterial abortion. No lesions and no bacteria were found in the remaining fetus (animal 4; Table 1). This might represent intercurrent abortion unrelated to RB51, or failure to detect the organism in submitted samples.
It is noteworthy that of the 4 herds vaccinated with RB51 in 2008, abortions were recognized in only one (herd A). Unlike the other 3 herds, animals in herd A were vaccinated just once (in the fall); cattle in other 3 herds were vaccinated twice (spring and fall).
The only detailed published account of bovine abortion following RB51 vaccination affected a 14.5-month-old heifer that was vaccinated with the calfhood dose of RB51 while pregnant at 8 months of age. 19 A tentative diagnosis of brucellosis was made on the basis of placentitis and fetal pneumonia observed in the heifer and her calf. Brucella abortus bacteria were identified by histology, immunohistochemistry, bacteriologic culture on special media, and PCR, and confirmed as B. abortus strain RB51. The bacterium was isolated from fetal lung and placenta, indicating a B. abortus RB51–induced placentitis and fetal infection in the heifer. A report from Iran describes abortion in dairy cows vaccinated with the reduced dose of 1.0–3.4 × 109 colony-forming units (CFU) of RB51 during pregnancy. 20 Four dairy cows aborted, of which only 2 had been vaccinated. All 4 had purulent uterine discharge and metritis after abortion, and B. abortus RB51 was isolated from 1 aborted fetus via bacteriologic culture and confirmed by PCR. The authors speculated that horizontal transmission occurred between vaccinated cattle and herdmates. 20 In a region of Spain where brucellosis was endemic, strain RB51 was cultured from 78 out of 897 fetuses, placentas, or vaginas of aborting cattle following vaccination of 14,893 pregnant cattle. 15
Several studies have been conducted to determine the safety and efficacy of strain RB51 in different reproductive stages and ages of cattle. Ten heifers were inoculated intravenously with 1 × 1010 CFU RB51 in the sixth month of gestation and were euthanized and examined at postinoculation week 8 or at full term in a previous study to assess the placental tropism and abortigenicity of RB51. 11 One near-term heifer delivered a premature weak calf, and B. abortus RB51 was cultured from liver, lung, spleen, and bronchial lymph nodes of most heifers and their calves. Lesions were consistent with brucellosis, and RB51 was cultured from all tissues with lesions. 11 Overall, the study indicated that RB51 can infect maternal and fetal tissues, and cause placentitis and premature birth. 11 A subsequent vaccination study using RB51 during pregnancy showed that a lower dose of RB51 (109 CFU) did not induce abortion or cause brucellosis. 12 Cattle vaccinated with S19 as calves and revaccinated with RB51 while pregnant did not show clinical signs. 14 No abortions were observed in calves vaccinated with S19 and boosted with 1 × 109 CFU RB51 either 2 or 3 times late in gestation. 18 A dose of 3 × 1010 CFU of RB51 was demonstrated to be safe for pregnant heifers vaccinated in midgestation. 5 Brucella abortus RB51 was not recovered from tissues of heifers or their calves. No abortions occurred among 8 adult cattle vaccinated with 1.6 × 1010 CFU of RB51 at day 60 of gestation and then challenged by conjunctival route with virulent strain 2308 (3 × 108 CFU). 13
The use of the label dosage indicated for calves in adult or pregnant cattle has not been thoroughly studied, particularly in field situations, due to paucity of reports from commercial herds. 10 Reduced dosage regimes have been evaluated in adult cattle that were vaccinated prior to breeding. 9 Consequently, it is likely that the size of the dose and the timing can influence the outcome of vaccination for pregnant cattle.
The success rate for determining the causative agent of bovine abortion, particularly in beef cattle, is relatively low (i.e., 30–40%). 7 In the current study, both BVDV and BoHV-1 were excluded through standard virology diagnostic tests. Histological findings in both episodes of abortion were similar to those observed in experimental infection of cattle with RB51. 11 In some tissues, RB51 was not isolated, and no cause of abortion was established in the submitted samples. Brucella abortus can be difficult to recover in culture, due to the bacterium’s slow growth, requirement for specialized media, and specific incubation conditions.1,3 Condition, time of collection, size, and sterility of samples submitted influence the likelihood of recovering the organism.1,3 One difficulty for determining the precise incidence of abortion and recovering carcasses for diagnostic workup is the location and size of ranches in Wyoming. Typically, pregnant cattle are not observed daily until they are near term. Pastures comprise many hundreds of acres of rough terrain, often with the presence of scavengers and predators.
The conclusion that RB51 vaccine induced abortion and reproductive loss is supported by the presence of fetal pneumonia, omphalitis, placentitis, histological evidence of bacteria in fetal tissues, and isolation of B. abortus strain RB51 from multiple tissues of multiple animals. Lesions due to RB51 in placental and fetal tissues are similar to those due to field strains of B. abortus. 11 Abortion in RB51-vaccinated adult cattle, while rare, illustrates the need for additional evaluation of the dose, efficacy, and risks associated with RB51 vaccination. The current report details different vaccination schedules and herd plans for brucellosis management. The occurrence of abortion and reproductive loss in herds with different vaccination strategies highlights the observation that several factors may play a role in vaccine-induced loss, including immune status, current health status, other diseases and vaccines, and the vaccine strategy with regards to relative risk and geographic proximity to a B. abortus reservoir. Occurrence of vaccine-induced abortions highlights the merit of diagnostic laboratories possessing appropriate diagnostic tests to differentiate vaccine from field strains of B. abortus in areas where strain RB51 is used. RB51 is zoonotic, and poses some risk to owners experiencing RB51-induced abortions in their herds, and to diagnostic personnel processing fetuses and other samples. 2
Although some production losses due to abortion or premature birth are normal, veterinarians and cattle producers should be concerned when losses occur in excess of 3–5% per year. 7 The vaccination of adult pregnant cattle with standard doses of RB51 presents a risk of calf loss due to abortion or premature birth. The simplest way to minimize the risk of abortion due to RB51 vaccine in adult cattle is to vaccinate them before they are rebred. While vaccine-induced reproductive loss is presumably rare, producers in brucellosis endemic areas need to balance the risk of vaccination-induced abortion with the risks of a potential brucellosis outbreak. In high risk locations, such as the GYA, the costs and risk associated with a whole herd brucellosis outbreak may outweigh the potential reproductive losses associated with vaccinating pregnant cattle. A reduced dosage of vaccine (1 × 106–109) has been shown to be safe in healthy cattle,9,12,18 provide protection against infection and abortion, and may be the best approach when adult vaccination is deemed necessary. However, several field reports appear to challenge the results of experimental studies.8,15,20 Changing the time during gestation when the vaccine is administered might reduce vaccine-associated losses, as it appears that abortions are less likely when the vaccine is administered earlier in gestation (i.e., before midgestation). The incidence of RB51-induced abortions and calf losses in Wyoming cattle indicates that vaccination of adult pregnant cattle with RB51 should be approached with caution and may not be appropriate in all situations.
Footnotes
Acknowledgements
The authors thank Paula Jaeger, Mercedes Thelen, Jackie Cavendar, and BreAnna Bonner for their technical assistance, and the cooperating herd owners for background information. Christina Loiacono is thanked for performing the diagnostic histology on the samples submitted to NVSL.
a.
Hardy Diagnostics, Santa Maria, CA.
b.
Sigma-Aldrich, St. Louis, MO.
c.
Monoclonal antibody 15.C.5, Syracuse Bioanalytical Inc., Ithaca, NY.
d.
Bovine Viral Diarrhea Antigen Test Kit, IDEXX Herdchek; IDEXX Laboratories, Westbrook, ME.
e.
VMRD, Pullman, WA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Portions of this work were supported by the USDA Animal Health Program WYO-397-05.
