Abstract
Bycatch (accidental drowning in fishing nets) is a significant problem for some marine mammal species, but can be difficult to diagnose as there are no pathognomonic gross or histological lesions. In human medicine, biomarkers such as S100B are increasingly being used to investigate hypoxic-ischemic syndromes, but, to the authors’ knowledge, studies using this marker have not been reported for marine mammal species. The aims of the current study were to determine baseline postmortem S100B levels in a pinniped species, and to determine whether S100B levels were stable over a postmortem interval of 48 hr. Aqueous humor, which is simple to collect and avoids many of the problems associated with postmortem collection of blood, was used as a surrogate for serum. S100B was detected in the aqueous humor of acute deaths (<15 min) and was stable for up to 48 hr, with a wider variation in values at the 48-hr time interval.
Death due to drowning can be difficult to definitively diagnose in human beings, as there are no pathognomonic gross or histological lesions. 16 A similar problem exists in marine mammals, where accidental capture in fishing nets (“bycatch”) or entanglement in derelict fishing gear can be an important and contentious cause of death (Kuiken T: 1994, Review of the criteria for the diagnosis of bycatch in cetaceans. European Cetacean Society Newsletter, special issue 26:38–43). 13 The ultimate mechanism of death in bycaught marine mammals is widespread hypoxia–ischemia and, while in human medicine there is increasing emphasis on the use of biomarker proteins to identify hypoxic-ischemic brain damage,3,9 similar studies have not yet been published for marine mammal species.
S100B is a dimeric calcium-binding protein that is part of the S100 family. As a group, these proteins play a role in a wide range of processes including cell growth, cell survival, and regulation of transcription. 12 S100B is found in highest concentrations in the nervous system, predominantly in astrocytes. Lower levels are found in some nonneural sites, including adipose tissue and melanocytes. Hypoxic-ischemic brain injury results in increased levels of S100B in cerebrospinal fluid (CSF) due to both leakage from damaged cells and active secretion. 12 Damage to the blood–brain barrier results in movement of S100B into serum, and levels in serum and CSF are both diagnostic and correlated with prognosis.3,12 In human beings, serum S100B increases within minutes of the onset of hypoxia,1,11 suggesting that serum levels could be of use in the diagnosis of hypoxic-ischemic brain injury in bycaught marine mammals. S100B proteins are highly conserved across vertebrate species, 12 and commercial kits designed for use in human beings have been successfully used in a number of other species, including pigs, 8 rabbits, 17 and rats. 18
In addition to CSF and serum, S100B is found in a range of other biological fluids following brain damage, including urine, amniotic fluid, and saliva. 12 Although aqueous humor levels of S100B have not been studied, this body fluid has been established as a valid surrogate for serum biochemistry in a variety of species, including pinnipeds.5,7,10,15 Aqueous humor can be rapidly and easily collected from marine mammals postmortem and can avoid potential problems associated with hemolysis that commonly occurs in serum samples collected after death. 15
Sample quality is of particular importance in wildlife investigations, where prolonged postmortem intervals can compromise diagnosis. 14 Accordingly, the aims of the current study were first to determine baseline S100B levels in the aqueous humor of northern fur seals (Callorhinus ursinus) using a commercially available kit; and second, to determine whether S100B levels in aqueous humor were stable over a postmortem interval of 48 hr.
Aqueous humor samples were collected from 16 subadult male northern fur seals that were killed as part of the annual Aleut harvest on St Paul Island, Pribilof Islands, Alaska, in July 2010. Samples were collected under authority of United States Marine Mammal Permit no. 782-1708 issued to the National Marine Mammal Laboratory in Seattle, Washington. The fur seals were killed by the traditional method of acute blunt trauma to the head, and all died instantaneously. The head of each animal was removed, and aqueous humor was aspirated from the anterior chamber of the left eye using a sterile 20-gauge needle at <15 min (n = 9) or 48 hr (n = 7) after death. Hemorrhagic samples were discarded, and a replacement sample was collected from the right eye. For the 48-hr group, heads were kept at ambient temperature (4–10°C) until collection. Samples were centrifuged, transferred into cryovials, and held at −80°C until analysis.
Assays were conducted using an anti-bovine S100B enzyme-linked immunosorbent assay (ELISA) kit a designed for use in human beings. Analysis was performed following the manufacturer’s instructions. All samples were evaluated in duplicate, and the mean of the 2 values was determined for each case. Statistical analyses were conducted using commercial software, b with group comparisons made using the Mann–Whitney rank sum test for nonparametric data. Results are shown in Table 1. There was no significant difference in mean S100B concentration between the control group and the 48-hr postmortem interval group (P = 0.112; Fig. 1), suggesting that S100B protein is stable in the aqueous humor for up to 2 days after death.
S100B concentrations in aqueous humor samples collected from northern fur seals (Callorhinus ursinus) at <15 min (controls) and 48 hr after death.

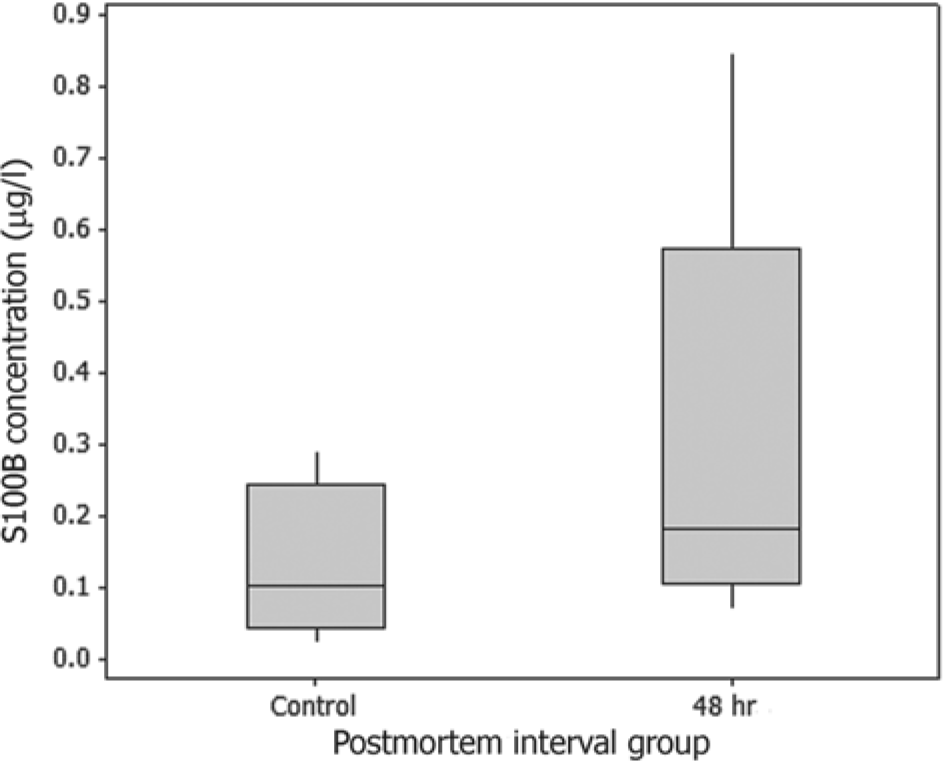
Comparison between aqueous humor S100B concentrations for northern fur seals (Callorhinus ursinus) at <15 min (control) and 48 hr after death.
S100B concentrations for normal human adults vary depending on the specific assay used 4 and on the biological fluid being tested, 12 thus reference ranges are not generally specified, and direct comparisons can rarely be made between studies. More frequently, comparisons are made between mean S100B concentrations of study and control subjects 4 or between baseline levels and clinical states for individual subjects.1,11 Aqueous humor S100B concentrations in drowned pinnipeds are yet to be established, and the biomarker potential of this protein would depend on the magnitude of the increase.
One potential difficulty with interpretation of subsequent studies in pinnipeds arises from the comparatively high variation between individual values in the 48-hr group (range = 0.07–0.86 μg/l; standard error of the mean = 0.11). This variability could compromise detection of subtle increases in S100B when using aqueous humor collected from cases with a prolonged postmortem interval. The source of the variability in the 48-hr group is uncertain, but it is possible that S100B is released from surrounding cells as they autolyze, producing an artifactual increase in concentration within the aqueous humor. S100B is found within normal melanocytes, 2 thus the iris and ciliary body are potential sources for contamination of ocular fluids. Ideally, future studies in pinnipeds should use aqueous humor collected from recently dead cadavers.
While the magnitude and speed of increase in S100B levels are unknown for asphyxiated pinnipeds, a previous study 6 found that severely brain-damaged asphyxiated human neonates had urinary S100B concentrations 30–60 times higher than normal neonates, and a concentration of >1.0 μg/l was 100% specific and sensitive for a fatal outcome. Urine levels exceeded this cutoff at the first postnatal urination, implying a rapid shift of S100B from the serum into the urine. Similarly, a previous study 1 found that serum concentrations increased significantly within 5–10 min of apnea in breath-hold divers. The findings suggest that elevated body fluid levels of S100B are a potentially useful diagnostic marker of drowning in pinnipeds.
There are several limitations to the present study, most of which are a consequence of the limitations imposed by the nature of the source population, that is, fur seals killed as part of a subsistence harvest. First, the sample size was small, and expansion of the study to incorporate other sites and years would enhance the validity of the findings presented herein. Second, the method of euthanasia (blunt trauma to the head) may have resulted in elevations in S100B, despite the lack of any observed period of survival. Future studies could be directed toward the following: establishing the diagnostic utility of S100B in other pinniped body fluids such as urine and serum; the possibility of assessing this marker in blubber, with the potential to assess progression or recovery from hypoxic events in live-sampled individuals; and evaluation of S100B levels in animals that have been euthanized without sustaining blunt trauma. In addition, evaluation of S100B levels in brain tissue of pinnipeds and cetaceans may be of interest, as these diving mammals are repeatedly exposed to hypoxic conditions and may therefore have quantitative differences from terrestrial species.
In summary, a commercial ELISA assay was able to detect an S100B-like protein in the aqueous humor of northern fur seals, at concentrations broadly similar to those published for the body fluids of other species. Samples collected immediately after death were not significantly different from those collected at 48 hr postmortem. Further investigations to evaluate S100B concentrations in the aqueous humor of accidentally drowned pinnipeds are warranted.
Footnotes
Acknowledgements
The authors gratefully acknowledge the assistance of the people of St Paul Island, Pribilof Islands, and Dr. Tom Gelatt, of the National Marine Mammal Laboratory, for assistance with permits and administrative requirements. All work was conducted under United States Marine Mammal Permit no. 782-1708.
a.
BioVendor, Tumova, Brno, Czech Republic.
b.
Minitab Inc., State College, PA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was partly funded by the McGeorge Research Fund, Massey University.
