Abstract
Plants of the genus Taxus are common ornamental shrubs that contain cardiotoxic alkaloids. Gross lesions consistent with heart failure are frequently reported in fatal cases; however, microscopic lesions in the heart have not been well characterized. The current report describes 2 related outbreaks in which 7 of 30, 250-kg calves died after confirmed exposure to clippings of Japanese yew (Taxus cuspidata). Three calves died 24 hr after initial exposure, with no significant gross or histologic lesions. Leaves of the yew plant were identified within the rumen contents, and Taxus alkaloids were confirmed by gas chromatography–mass spectrometry. Following the initial diagnosis, the yew clippings were burned. Two days later, the remaining calves were reintroduced to the enclosure. Within 24 hr, 3 additional calves began to show clinical signs of depression (3/3) or labored breathing (1/3), and by the fourth day, these 3 calves and an additional calf were found dead. Partially burnt yew leaves were found during close inspection of the enclosure. Two of 3 calves submitted for necropsy were severely autolyzed; the third had pulmonary edema and mild fibrinous pleural effusion. Histologic lesions in the latter included multifocal cardiac myocyte hypereosinophilia, sarcolemma fragmentation, pyknosis, karyolysis, myocyte loss, and a mild interstitial lymphoplasmacytic infiltrate with edema. Moderate fibrinosuppurative interstitial pneumonia was the only other significant finding. Cardiac changes were attributed to damage from the initial exposure to Taxus 6 days prior to death.
Plants of Taxaceae, or yew family, are widely recognized as palatable and highly toxic to livestock. Known since antiquity as the “tree of death,” 3 some species of yew cause death so rapidly in susceptible animals that the offending plant may be found within the oral cavity at necropsy. Most cases of poisoning involve unexpected access to the plant which, given its hardy nature, is often utilized as a low lying ornamental shrub. Toxicoses may be more frequent in winter months when concentrations of the toxic principals are highest and the relative palatability of the evergreen leaves increases. All parts of the plant, save the red, fleshy aril, are toxic; toxicity is not diminished by drying. Taxus species are found worldwide, particularly within the midlatitudes of the northern hemisphere with species indigenous to Europe, Asia, and North America (Farr K: 2008, Genus level approach to Taxus species. In: Proceedings of the Convention on International Trades in Endangered Species of Wild Fauna and Flora, p. 21, Nov. 17–21, Cancun, Mexico). The English yew, Taxus baccata, and the Japanese yew, Taxus cuspidata, are the most commonly cultivated species in North America. 8
The toxicity of yew species is attributed to a group of 10 or more toxic alkaloids, the most toxic of which are taxine A and B. 15 Taxine B is in greater concentrations and is more toxic than taxine A in most species of yew. The primary mechanism of toxicity is antagonism of calcium channels in cardiac myocytes, 16 resulting in dose-dependent depression of atrioventricular conduction, marked slowing of the ventricular contraction rate, arrhythmias, and death due to cardiac arrest in diastole. Taxine is thought to have lesser effects on other organs, particularly involuntary smooth muscle, with relaxation of intestinal tone and decreased intestinal and uterine contraction in experimentally induced disease. 15 In addition, yew species also contain nitriles, ephedrine, and volatile irritant oils, which may be associated with gastrointestinal disease in subacute cases of toxicity, 8 as well as bioflavonoids that experimentally depress the central nervous system. 3
Fatal toxicosis has been reported in a variety of animal species, including human beings; however, the majority of cases occur in domestic livestock, especially cattle and horses. The oral lethal dose of yew varies significantly by species; horses are particularly sensitive, with an estimated minimal lethal dose (LDmin) of 0.2–0.4 g of yew leaves per kg of body weight. Goats and chickens are relatively more resistant (12 g/kg and 16.5 g/kg, respectively). 16 Cattle are of intermediate susceptibility with an estimated LDmin of 2.0 g/kg. Wild ruminants, particularly white-tailed deer (Odocoileus virginianus), are apparently resistant to the toxic effects, possibly due to increased ruminal detoxification of taxines. 15 Although less commonly reported, Taxus species are equally toxic to dogs and cats, and findings are similar to those in farm animals.
The most common syndrome of Taxus intoxication in farm animals is acute collapse and death, often within minutes.5,6 Less frequently, animals, particularly cattle, may survive a few hours to rarely days, 1 and occasional cases of survival without apparent ill effect are reported. 7 Animals that survive for even a short time may exhibit dyspnea, tremors, and collapse. In cases of subacute toxicosis, gastroenteritis, manifesting as congestion and edema in the stomach and small intestine, may be the primary finding. 6 Signs of depression, ataxia, muscle tremors, and seizures have been reported in the dog, but are typically attributed to circulatory failure. Subacute cases may present with gastroenteritis and diarrhea. 2
Given the rapid progression of disease, definitive clinical, gross, or histologic changes are typically not described. Historically, diagnosis of Taxus toxicosis relies on confirmed exposure, or identification of the plant within the gastrointestinal tract at necropsy. When described, gross lesions of generalized organ congestion and pulmonary edema are attributable to circulatory failure; rarely, nonspecific hemorrhage or evidence of gastroenteritis are identified. 15 The current report describes a myocardial lesion in a young beef calf as a manifestation of subacute Taxus toxicosis.
Three Angus calves (cases 1–3) from a herd of 75 were found dead without premonitory signs. Affected calves weighed 227–272 kg (approximately 8 months of age) and were kept in a fenced enclosure that they had occupied for several months. Several calves in the herd had been diagnosed with coccidiosis 3 days prior, and amprolium a had been added to the drinking water of all calves, per labeled dosing instruction. Calves had previously been vaccinated twice for Clostridium chauvoei, respiratory bacterial agents including Mannheimia haemolytica and Pasteurella multocida, and otherwise had no significant medical history. Lesions identified during necropsy of the calves at the Oklahoma Animal Disease Diagnostic Laboratory (Stillwater, Oklahoma) included pulmonary edema (3/3) and pericardial effusion (1/3); these findings were considered possibly due to acute heart failure. Given the sudden death of multiple animals without premonitory signs, a toxin was suspected. Aqueous humor was negative for nitrites by nitrite test strips, b and a diphenylamine spot test for nitrates was negative. Bovine herpesvirus 1 was not identified on fluorescent antibody testing of the trachea of 2 of the 3 animals. Visual examination of rumen contents revealed multiple leaves and small twigs morphologically consistent with Japanese yew.
Survey of the enclosure identified a large pile of Japanese yew trimmings stacked into a pile of brush and miscellaneous farm debris. Based on necropsy findings, animals were immediately removed from the enclosure, and the yew trimmings were burned. Two days later, the remaining calves were returned to the enclosure. Within 12 hr and progressing over the next 24 hr, 3 calves (cases 4–6) began to show clinical signs of general depression (3/3) and labored breathing (1/3). Suspecting pneumonia, animals were individually treated with oxytetracycline c without improvement. On the morning of the fifth day, 1 calf (case 4) was found dead and another markedly depressed animal (case 7) was euthanized and necropsied on site. The morning of the sixth day, 2 additional calves (cases 5 and 6) were found dead. Calves 4–6 were submitted for necropsy along with portions of fresh lung from calf 7. Survey of the enclosure and burn pile at this time identified numerous, partially incinerated, Japanese yew branches and leaves.
During gross necropsy examination of calves 1–3, marked pulmonary atelectasis (3/3), mild interlobular pulmonary edema and tracheal hemorrhage (1/3), moderate thymic edema (3/3), and moderate pericardial hemorrhagic effusion (1/3) were observed. Within the rumen of 1 calf (case 1) partially masticated Japanese yew leaves were identified within the rumen contents. Seven days after submittal of cases 1–3, postmortem examination was completed on 3 additional animals (cases 4–6). At necropsy, advanced autolysis was present in calves 4 and 5. Gross findings were limited to the abomasal mucosa and included congestion with scattered petechial hemorrhages in calf 5. Gross findings in the third calf (case 6) were limited to pulmonary edema and mild fibrinous pleural effusion. Sections of fresh lung (case 7) were submitted and were grossly unremarkable. Despite careful examination of rumen and abomasal contents of calves 4–6, Taxus leaves were not identified. Gas chromatography–mass spectrometry (GC-MS) was completed, as previously described, 1 at the California Animal Health and Food Safety Laboratory (Davis, California) on ruminal contents from case 1 and abomasal contents from case 6; taxine alkaloids A and B were identified in calf 1, but not in calf 6.
Histologic findings in cases 1–3 were unremarkable. The presence of small numbers of coccidia within the colonic mucosa supported the clinical diagnosis of coccidiosis. Pulmonary and thymic edema were confirmed, presumably reflecting an acute cardiovascular insult. Myocardial lesions were not present. Due to advanced autolysis, microscopic examination of cases 4 and 5 was limited to the heart, and tissues examined in case 6 included lung, liver, heart, spleen, and kidney. Autolysis precluded histologic interpretation of the heart in cases 4 and 5. Microscopic cardiac changes in case 6 were characterized by loss of approximately 10–20% of myocytes as multiple, punctate clusters of acute to subacute myocardial necrosis characterized by myocardiocyte hypereosinophilia, sarcolemmal fragmentation, pyknosis, karyolysis, and myocyte dropout with minimal fibroblast and sarcolemmal cell hypertrophy (Fig. 1). Regionally, the interstitium was minimally infiltrated by perivascular to diffuse lymphocytes, plasma cells, and expanded by edema (Fig. 2). Staining with reticulin and trichrome stains confirmed retention of reticulin network with a mild decrease in reticulin staining intensity (Fig. 3) and lack of significant fibrosis and collapse (Fig. 4). Regionally, the pulmonary interstitium was moderately expanded by fibrin, edema, and ectatic lymphatics containing intravascular fibrin. Regionally, alveoli were flooded by fibrin and edema, and contained increased numbers of alveolar macrophages. Intrapulmonary perivascular connective tissues were expanded by edema. Significant findings were not observed in the kidney, spleen, or liver of calf 6.
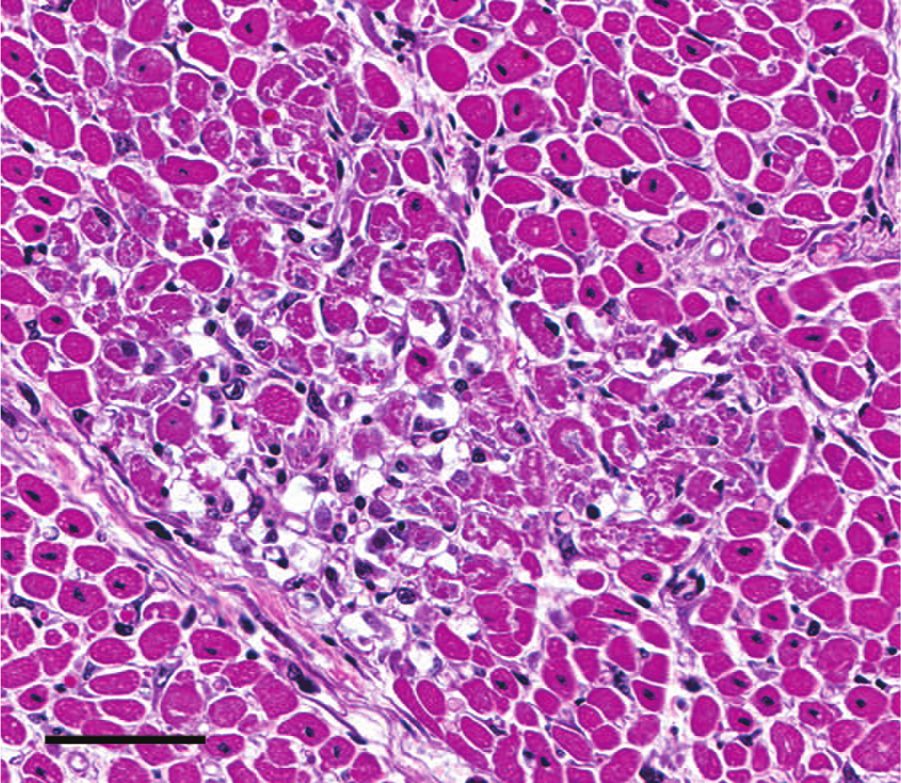
Heart; case 6. Within the affected foci, myocytes are variably hypereosinophilic with loss of striations, fragmentation and vacuolation of the sarcolemma, pyknosis, karyolysis, and regional dropout. Note lack of appreciable inflammatory infiltrate. 40× hematoxylin and eosin. Bar = 70 μm.
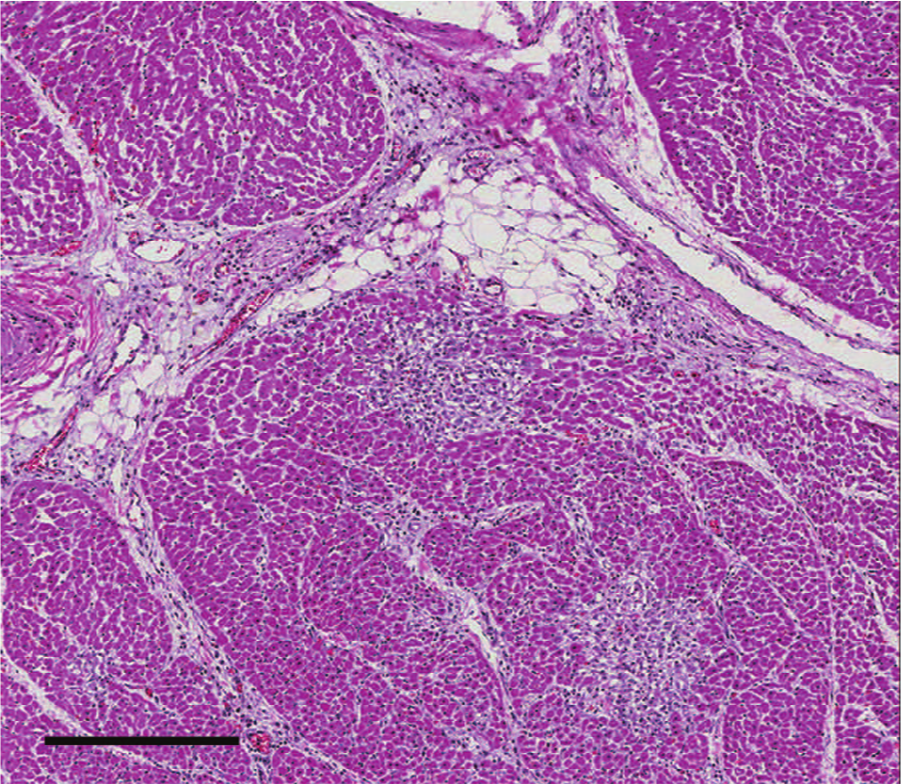
Heart; case 6. Small foci of myocardial necrosis and mild infiltration by predominantly perivascular lymphocytes, plasma cells, and few macrophages. 7.4× hematoxylin and eosin. Bar = 300 μm.
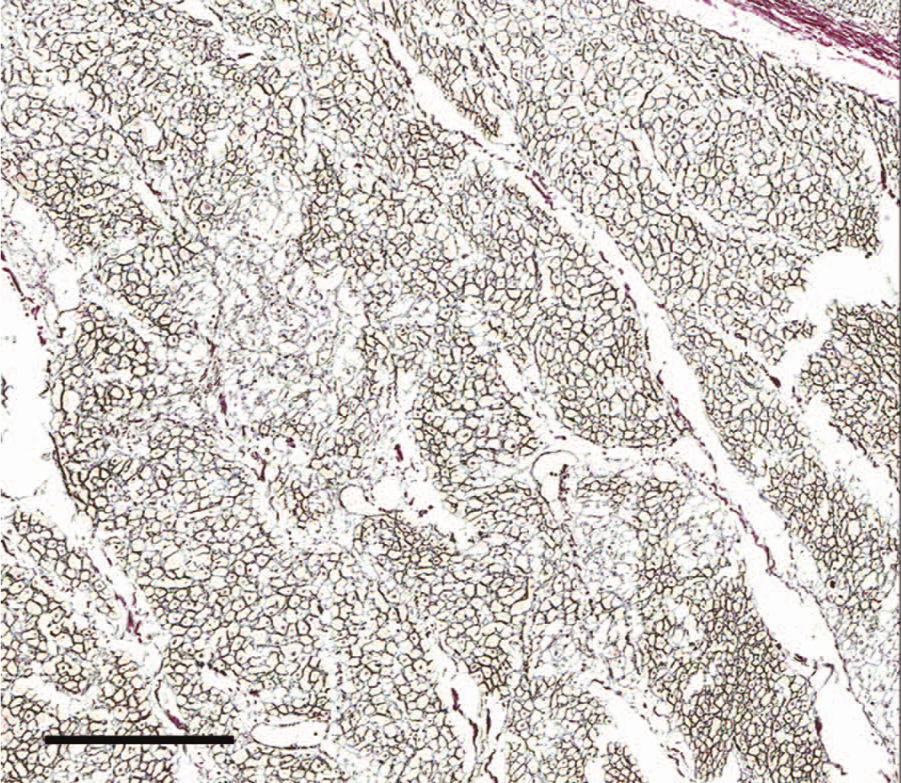
Heart; case 6. Minimal decrease of staining intensity within affected areas reflects retention without significant stromal collapse or loss of reticulin. 10× reticulin. Bar = 300 μm.
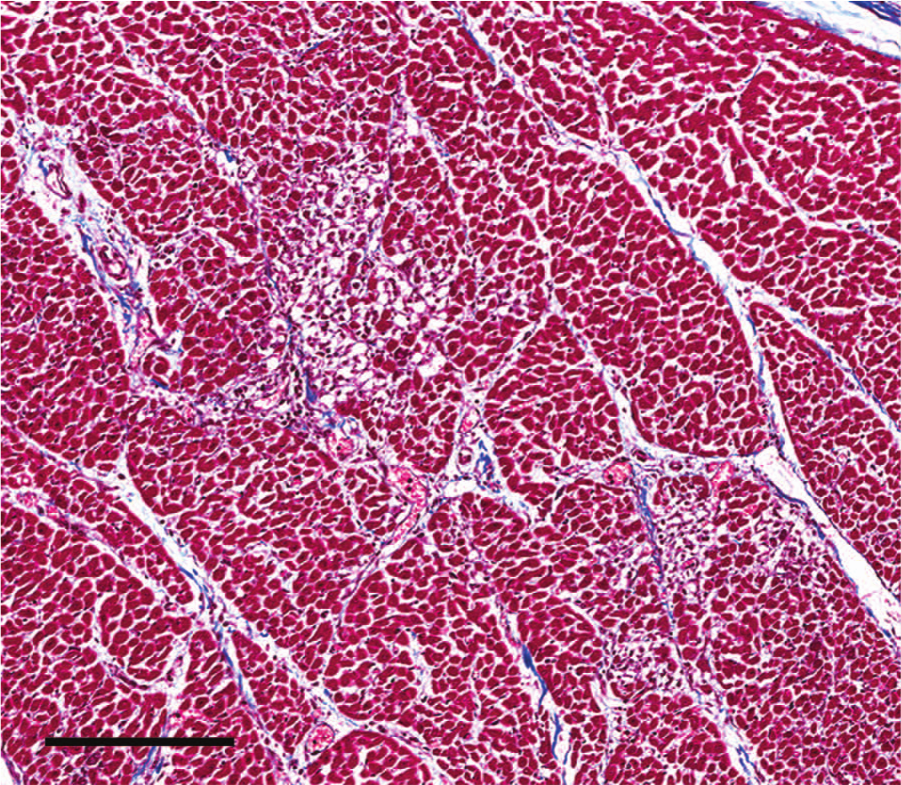
Heart; case 6. No significant increase of mature collagen within the affected regions. 10× trichrome. Bar = 300 μm.
In the current case series, calves presented both as acute deaths associated with initial exposure to yew clippings, and a week later, as a second outbreak of mortality. Typical of most cases of Taxus toxicosis, cases 1–3 died acutely without specific gross or histologic lesions. In cases with similar initial presentation and nonspecific signs of heart failure, a variety of toxins known to cause heart failure should be considered. Given the composition of a typical farm burn pile, nitrates, arsenic, and other heavy metals were a primary consideration. Many cases involving heavy metal toxins may have specific gross or histologic lesions that suggest cause of death. In addition, depending on the season and the local flora, enclosures should be evaluated for possible toxic plants; cardiotoxic plants including Nerium spp. (oleanders), Rhododendron spp. (rhododendrons, azaleas), Digitalis spp. (foxglove), Asclepias spp. (milkweeds), Kalmia spp. (laurels), and Taxus spp. (yew), among others, may be of specific consideration. Although lesions of plant toxicities may be less clearly defined than those of heavy metals, the time course tends to be more protracted, and gastrointestinal disease is a common concurrent finding. While gastrointestinal edema, congestion, and even myocardial hemorrhage 12 are reported with yew toxicity, clinical manifestations of diarrhea, vomiting, or abdominal pain are much less common than in those of other cardiotoxic plants. As in the currents case, presence of the offending plant in the gastrointestinal tract supports the diagnosis of plant toxicosis.
In the case of calf 6, gross lesions were consistent with heart failure. The lack of significant inflammation colocalized to the areas of myocyte loss, absence of fibrosis, and retention of the reticulin network in calf 6 suggest apoptosis may have been responsible for myocyte loss, rather than necrosis. Antagonism of calcium channels has been associated both with increased and decreased rates of apoptosis; 9 however, it is unknown if calcium channel perturbation by taxine contributed to apoptosis in the current case. Although it is currently believed that apoptosis is antiphlogistic and that necrosis triggers inflammation and an immune response, this paradigm is shifting. Moreover, some calcium channel blockers have been shown to have some antifibrotic properties, 13 which may have contributed to the lack of a fibroblastic response in this case. Additionally complicating the picture, only 1 of 3 hearts were preserved well enough to interpret; it is unknown the roles that dosage, duration of clinical signs, and time until death may have on development of these cardiac lesions.
In an effort to delineate the potential role of apoptosis in the development of the cardiac lesion, terminal deoxynucleotidyl transferase 2’-deoxyuridine 5’-triphosphate (dUTP) nick end labeling (TUNEL) and caspase-3 immunohistochemical staining were completed, as previously described10,11 by the Michigan State University Diagnostic Center for Population and Animal Health (Lansing, Michigan). While cardiac myocytes within affected regions had strong cytoplasmic and nuclear TUNEL staining, no myocytes stained positively for cleaved caspase-3. Although detection of fragmented DNA strand ends, as achieved by TUNEL staining, has previously been considered a marker of apoptosis, necrotic cells may also generate fragmented DNA, and as such staining with caspase-3 is preferred for its greater specificity. 4 The lack of staining for cleaved caspase-3 in affected myocytes indicates that apoptosis may not have had a primary role in the development of this cardiac lesion; however, a caspase-independent mechanism of apoptotic cell death cannot be entirely ruled out.
The only other significant lesion in calf 6 was moderate, regionally extensive pulmonary edema and fibrinosuppurative interstitial pneumonia characterized by mild expansion of alveolar septa by fibrin, edema, and low numbers of neutrophils. The cause of this pneumonia was not definitively determined. Hemodynamic alterations and vascular leakage secondary to subacute heart failure cannot be excluded. Exposure to an unknown toxin resulting in endothelial damage, or a concurrent systemic septic process, were both initially considered; the absence of microscopic lesions in other tissues decreased the likelihood of the latter. Smoke exposure associated with burning the pile of yew and other potential inhalant toxins was also considered; however, a bronchocentric pulmonary lesion would be anticipated.
An interesting aspect of this case series is that cases 4–7 may have survived an initial, sublethal exposure to yew leaves, and the microscopic lesion may have remained unidentified if not for a second exposure to the incompletely burned Taxus leaves. Although the possibility remains that these 4 animals died from cardiac decompensation 6 days after the initial exposure, the rapid, synchronous clinical illness of 4 animals following re-exposure to the partially burnt plant advocates for additional toxicosis on previously compromised myocardium. Although re-exposure could not be confirmed via identification of leaves or burnt leaf fragments in the ruminal contents, or GC-MS identification of toxic alkaloids, it is postulated that minimal taxine would have resulted in death in these already compromised animals. Furthermore, only abomasal contents were available for GC-MS analysis in calf 6, and death may have occurred before measureable levels of taxine were present in the abomasal compartment.
Although the acute death in the first 3 calves of this report is rather typical, the microscopic cardiac lesions in the cases of subacute to chronic Taxus toxicity are noteworthy. The subacute form of Taxus toxicity occurs infrequently, and is encountered most often in cattle as gastroenteritis, attributed primarily to the volatile irritant oils produced by the plant. Reported gross lesions most frequently include pulmonary edema attributed to heart failure and submucosal congestion, and edema of the stomach, small, and/or rarely large intestine typical of nonspecific gastroenteritis. Rarely, epicardial or gastric mucosal hemorrhages are reported. Histologic lesions are typically nonspecific and supportive of congestion and edema in the lungs and gastrointestinal tract with variable pulmonary atelectasis. In 2013, a report of extensive cardiac fibrosis suggestive of previous cardiac necrosis was reported in a Holstein calf with prolonged exposure to yew clippings. 1 Prior to this, in only 1 previous report of Taxus toxicosis is a specific microscopic cardiac lesion described. 14 In that case, a 2-year-old Thoroughbred colt with gross lesion of ecchymotic endocardial hemorrhage and submucosal edema and congestion in the gastrointestinal tract was associated with an acute, multifocal contraction band necrosis and mild, neutrophilic, and lymphocytic interstitial myocarditis. In both reports, Taxus toxicosis was confirmed by presence of plant material and/or presence of taxine alkaloids by GC-MS.
While the duration of clinical disease may vary significantly, acute to peracute death typifies yew toxicosis. Protracted clinical disease may be dependent on the amount of plant eaten and the relative concentration of toxins. Toxin concentrations are known to be higher in winter months, in older plants, and in particular species such as T. baccata and T. cuspidata. 16 Traditionally, diagnosis of Taxus toxicity relies on a supportive clinical history singly or most often in concert with gross evidence of foliage within stomach contents of acutely affected animals. Rarely is there the opportunity to examine the hearts of animals that have apparently survived the initial insult. The incidence of sublethal cardiac lesions in surviving cattle is unknown.
When investigating cases of unexpected death in cattle, perhaps following a course of apparent respiratory distress, the presence of multifocal cardiomyocyte loss should lead to an investigation for a possible cardiotoxic cause. Although the cardiac lesion in the affected calf in the current report is not pathognomonic for yew toxicosis, Taxus should be a differential diagnosis in any animal with similar cardiac lesion. In rare cases, it is possible that this lesion may be the only remaining diagnostic support of Taxus toxicity days after exposure to, and intestinal elimination of, the offending plant.
Footnotes
Acknowledgements
The authors would like to thank Dr. Matti Kuipel for immunohistochemistry support.
a.
Merial Ltd., Duluth, GA.
b.
EM Quant, EMD Chemicals Inc., Gibbstown, NJ.
c.
Norbrook Laboratory International, Newry, Down, Northern Island.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) declared that they received no financial support for their research and/or authorship of this article.
