Abstract
A free-ranging, young adult, female American white pelican (Pelecanus erythrorhynchos), found dead on the grounds of the San Diego Zoo Safari Park in Southern California, had severe multifocal to coalescing necrotizing hepatitis and splenitis on postmortem examination. Histologically, within the large areas of necrosis were myriad pleomorphic, 5–20 µm in diameter, protozoal organisms with 1 to multiple nuclei. Ultrastructurally, the organisms were consistent with a trichomonad flagellate. Polymerase chain reaction and sequencing of the small subunit ribosomal RNA gene identified nucleotide sequences with 99% identity to Tetratrichomonas gallinarum, which is a common inhabitant of the intestinal tract of galliform and anseriform birds that has occasionally been associated with disease, including typhlitis and hepatitis. Damage to the cecal mucosa in the pelican from trematodes and secondary bacterial infection could have allowed invasion and systemic dissemination of the organism. Exposure of the pelican to a variety of native and exotic anseriform and galliform birds at the zoological institution could have led to cross-species infection and severe manifestation of disease in a novel host.
Tetratrichomonas gallinarum is a trichomonad flagellate protozoan that is most commonly isolated from the intestinal tract (cecum) of galliform and anseriform birds, where its role as a pathogen remains unclear. In turkeys and some species of ducks, it has been described as a cause of typhlitis and hepatitis similar to histomoniasis.1,8 However, other experimental infections in turkeys have shown no clinical significance and only mild to absent changes in the cecum.2,6 Disease in chickens infected with T. gallinarum also appears to be rare.2,4,5 Sequence comparison of T. gallinarum isolates from multiple species of galliform and anseriform birds by polymerase chain reaction (PCR) detected differences in nucleotide distance of up to 8.9%, which exceeded differences between some species of trichomonads. 3 This marked heterogeneity among T. gallinarum isolates could account for some of the differences in pathogenicity between studies. The current report describes a systemic infection due to a Tetratrichomonas sp. with closest sequence identity to T. gallinarum resulting in hepatitis and splenitis in a pelican.
A free-ranging, female American white pelican (Pelecanus erythrorhynchos), with adult plumage but small ovary and prominent bursa of Fabricius (young adult), was found dead on the grounds of the San Diego Zoo Safari Park near Escondido, California. Because this was a native bird that was not part of the zoological collection, no history was available. A necropsy performed the same day showed that the liver and spleen were enlarged and dark red with approximately 60–75% of the capsular surface and parenchyma replaced by multifocal to coalescing, 1–3 mm, irregular, slightly bulging, yellow, dry, soft to firm foci (Fig. 1). Myriad endoparasites were present, including lice in the oral pouch (Piagetiella sp.), trematodes in the trachea (probable Clinostomum sp.), and ascarid nematodes in the esophagus and proventriculus (probable Contracaecum sp.). The parasites were identified to probable genus level based on their morphology, the host, and their location therein. The proventriculus and ventriculus contained green granular fluid, and the small intestine and colon contained thick tan to green fluid and dark green clumped fecal material, respectively. Adipose stores were small to absent. Tissue sections collected at necropsy were fixed in 10% neutral buffered formalin, processed routinely, embedded in paraffin, sectioned at 5 µm, and stained with hematoxylin and eosin.
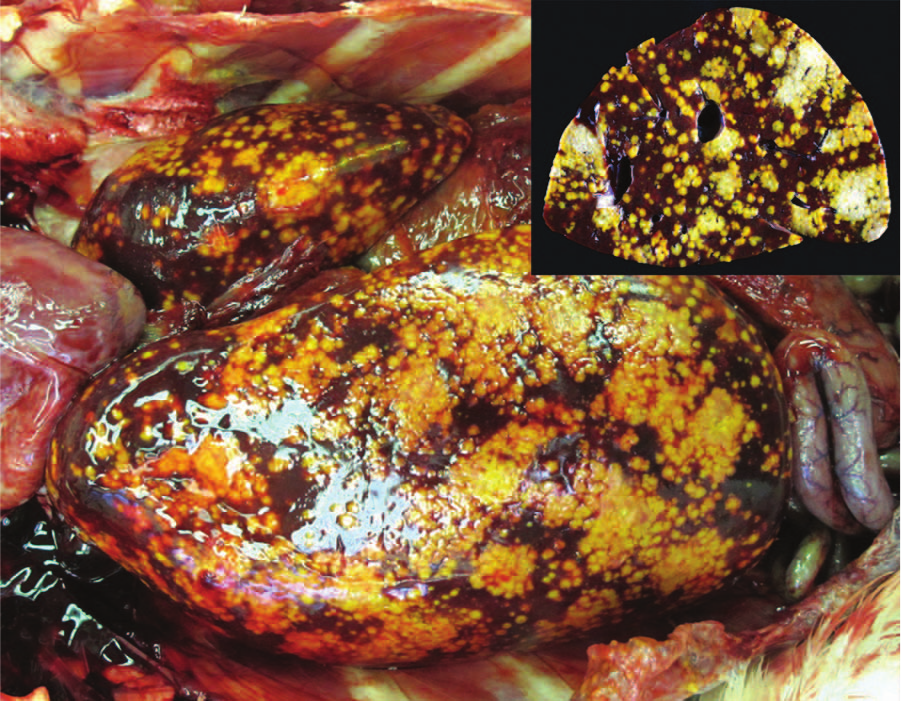
Liver; pelican. The liver is enlarged with multifocal to coalescing, irregular, tan, slightly bulging foci disseminated across the capsular surface and within the parenchyma on section (inset).
Histologic examination of the liver and spleen showed severe disruption of the parenchyma by multifocal to coalescing areas of lytic necrosis (Fig. 2). Mixed with cellular debris and fibrin within the necrotic foci were myriad round to pleomorphic organisms that were amphophilic to basophilic, approximately 5–20 µm wide, and had 1 to multiple round nuclei (Figs. 3, 4). These organisms were also present in rare small foci of necrosis in the lung, heart, pectoral muscle, bone marrow, proventriculus, and small and large intestinal mucosal lamina propria. Segments of small intestinal, cecal, and colonic mucosa were heavily colonized by trematode parasites approximately 300–400 µm long that invaded crypts and lamina propria and disrupted mucosal epithelium. There was associated mild to moderate infiltration of the lamina propria with lymphocytes and histiocytes, villous blunting, and rare crypt abscesses. The cecal mucosa was also multifocally disrupted by erosion and heterophilic inflammation with intralesional bacteria.
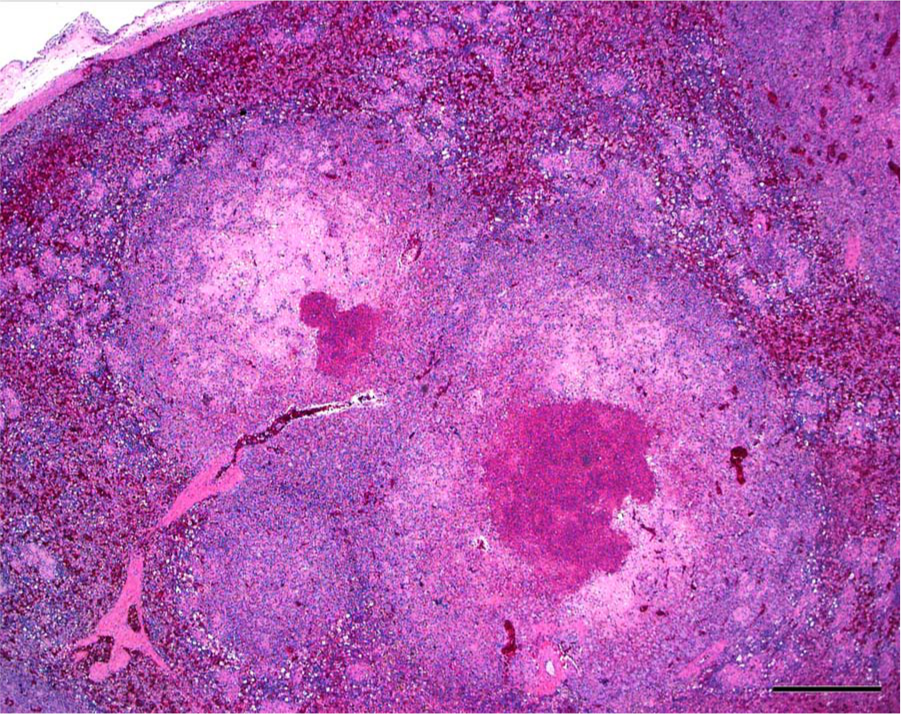
Spleen; pelican. Large foci of lytic necrosis disrupt the parenchyma. Hematoxylin and eosin. Bar = 500 µm.
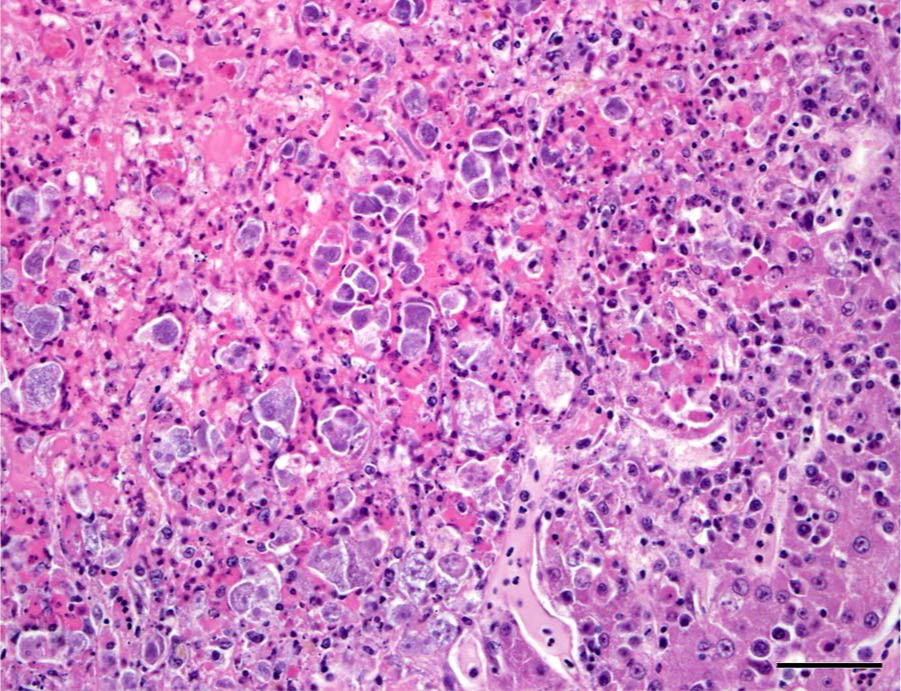
Liver; pelican. Within foci of necrosis are myriad, pleomorphic, basophilic organisms. Hematoxylin and eosin. Bar = 50 µm.
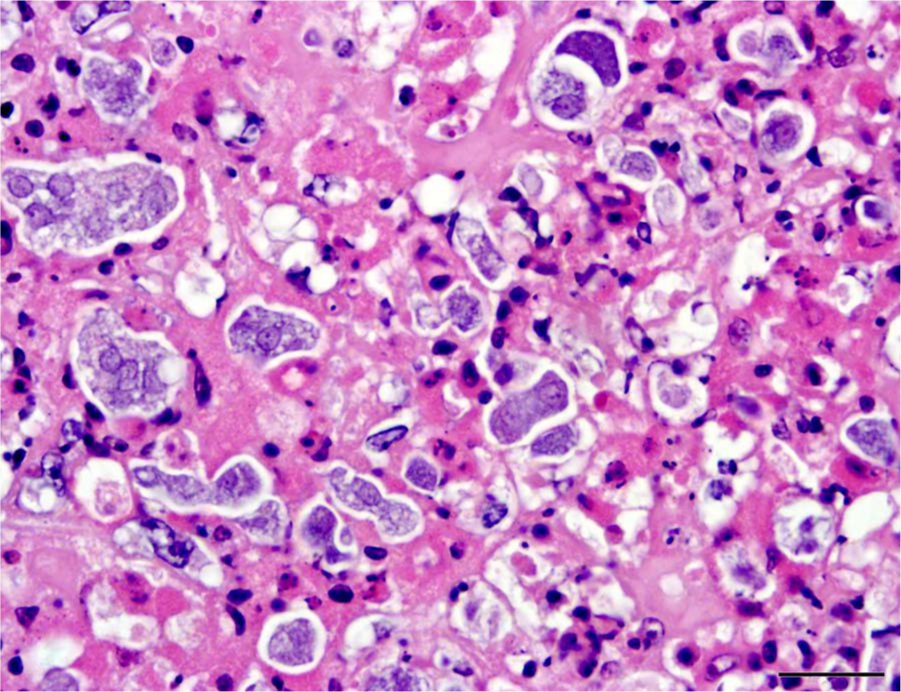
Liver; pelican. The organisms are round to irregular, approximately 5–20 µm in diameter, and have 1 to multiple round nuclei. Hematoxylin and eosin. Bar = 20 µm.
For transmission electron microscopy, formalin-fixed liver was post-fixed in 0.166 M of cacodylate-buffered, 3% glutaraldehyde with 1% tannic acid solution,
a
followed by a second post-fixation treatment in 1% osmium tetroxide.
a
Ultrathin 80-nm sections were stained with uranyl acetate and lead citrate,
a
and examined with a transmission electron microscope.
b
Within areas of necrosis were unicellular microorganisms, which measured approximately 10.3 × 6.3 µm and had 4 anterior flagella, 1 recurrent flagellum, basal bodies, pelta, axostyle, costa, hydrogenosomes, and glycogen granules (Fig. 5). Occasionally, segments of external flagella were detected in the intercellular space. Nuclei and organelles were sometimes seen in close proximity, suggestive of internal replication, but could not be confirmed to be within the same cell due to inadequate sample preservation. For scanning electron microscopy, the liver tissue was dehydrated in a graded ethanol series, critical point dried under CO2, sputter coated with gold, and examined with a
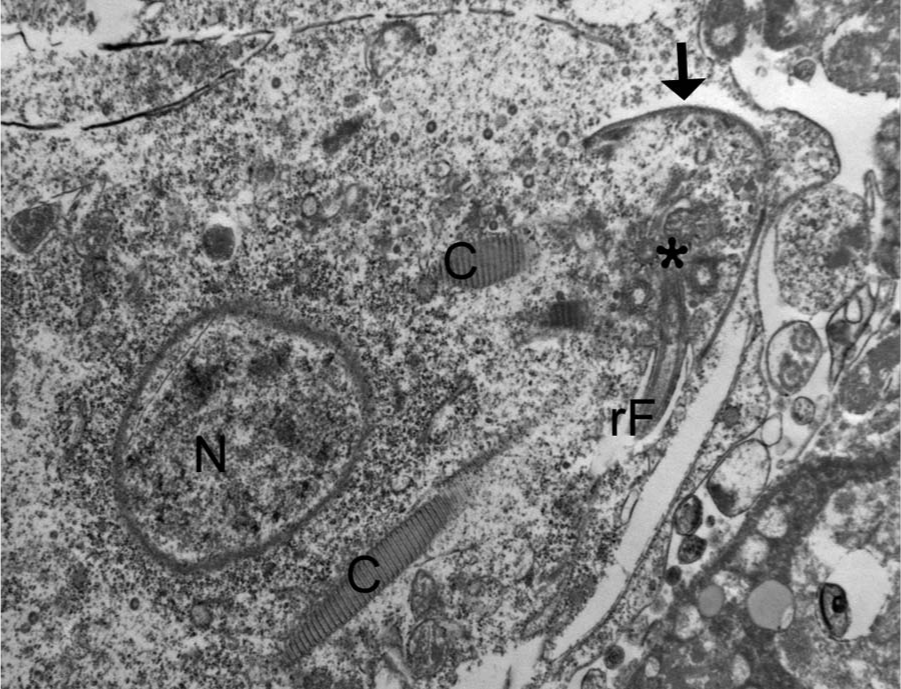
Liver; pelican. The organisms have features consistent with a trichomonad flagellate, including a nucleus (N), basal bodies (asterisk), a recurrent flagellum (rF), costa (C), and pelta/axostyle complex (arrow). Transmission electron micrograph.
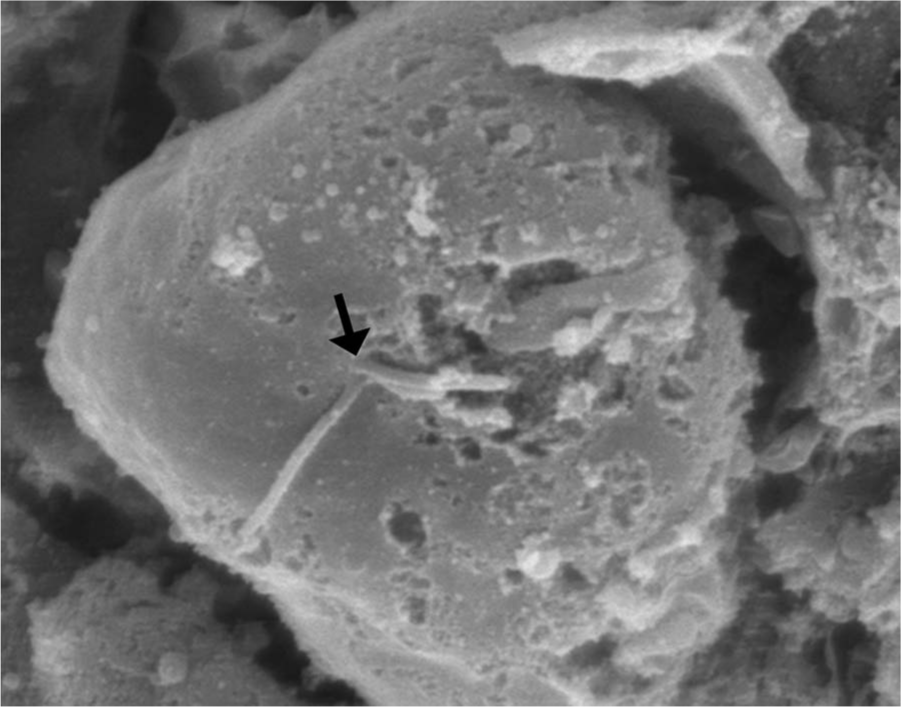
Liver; pelican. Broken anterior flagella (arrow) are rarely identified on organisms. Scanning electron micrograph.
DNA was extracted from frozen liver collected at the time of necropsy using a commercial kit, d with final elution in 100 µl of kit buffer AE. Initially, a trichomonad-specific PCR assay was performed targeting the 16S ribosomal RNA (rRNA) using primers 16Sl and 16Sr. 3 The 20-µl reaction mix was composed of 0.4 µM of primer, 10 µl of DNA polymerase, e 1 µl of extracted DNA, and nuclease-free water to a final volume of 20 µl. The PCR cycling parameters were 95°C for 5 min; followed by 45 cycles of 95°C for 45 sec, 48°C for 45 sec, and 72°C for 1 min 30 sec; with a final extension at 72°C for 10 min. The PCR product was visualized on a 0.8% agarose gel, purified using a DNA gel extraction system, f and directly sequenced g resulting in a 1,512-bp product.
A second PCR assay was performed with primers 16Sl and ITSF. 3 The PCR cycling parameters were 95°C for 5 min; followed by 40 cycles of 95°C for 1 min, 55°C for 45 sec, and 72°C for 1 min; with a final extension at 72°C for 10 min. The PCR products were visualized on a 0.8% agarose gel and purified using a DNA gel extraction system. f The purified PCR product was directly sequenced g and included 2 primer walking steps to extend the sequencing length to a 1,870-bp product. The sequencing products of both PCR assays were compared to the GenBank database using the Basic Local Alignment Tool (BLAST; http://www.ncbi.nlm.nih.gov/blast/Blast.cgi). Both products were 99% identical to the small subunit rRNA gene of several strains of Tetratrichomonas gallinarum. The next closest matches were to Tetratrichomonas sp. (up to 96%) and Trichomonas sp. (up to 94%).
The appearance of the hepatic lesions on gross and histologic examination was similar to those of histomoniasis, caused by the trichomonad Histomonas meleagridis. Macroscopic differentiation of trichomoniasis due to Tetratrichomonas gallinarum (formerly Trichomonas gallinarum) and histomoniasis in turkeys has been described. 1 Liver lesions of pure T. gallinarum infections were granular, pinpoint to approximately 2 cm in diameter, irregularly shaped, and even with, or slightly bulging from, the hepatic surface. This was in contrast to the more consistently larger, circular, slightly depressed lesions of H. meleagridis. 1 While the hepatic lesions in the pelican in the current report were consistent with the description of lesions due to T. gallinarum in turkeys, no known cases of histomoniasis in pelicans were available for comparison. The pleomorphic size and shape of the organisms histologically was comparable to that of H. meleagridis in its amoeboid phase, the form that occurs in tissues. 5 Therefore, a similar change in morphology between organisms in the intestinal lumen or culture and those in tissue might occur with T. gallinarum. The presence of basal bodies and sometimes flagella in the organisms was confirmed with electron microscopy and was consistent with the PCR results. The presence of apparently multinucleated forms on routine histology in the present case was unusual and was not clearly appreciated on electron microscopy, possibly due to necrosis and postmortem changes. However, replication of these organisms by binary fission, preceded by duplication of the nucleus and organelles, and dense crowding of the organisms could account for this appearance.
The liver and spleen were most severely affected with necrotizing lesions effacing large areas of the parenchyma. Smaller foci of necrosis with intralesional organisms were also occasionally seen in the lung, heart, skeletal muscle, bone marrow, proventriculus, and small and large intestine. This appearance was consistent with hematogenous dissemination of the infection. The likely origin of these trichomonads was the lower intestinal tract, although no areas of colonization of the lumen or crypts of any segment of intestine were seen. However, in addition to the variety of endoparasites noted grossly, myriad small trematodes invaded and multifocally disrupted the small intestinal, colonic, and cecal mucosa. Secondary bacterial infections were also present in the ceca, and it is possible that this damage to the intestinal mucosa allowed invasion and dissemination of the flagellates. Poor immune function could also have been a contributing factor, as the pelican described herein was young, heavily parasitized, and in poor nutritional condition. In a previous report of T. gallinarum infection in ducks, all affected ducks were juveniles or subadults. 8
The organism in the pelican most closely matched T. gallinarum, but was not molecularly identical to any previously described strain. As previously noted, isolates of T. gallinarum have a high degree of molecular polymorphism, which could indicate the presence of multiple species or subspecies within this group, many of which could be host-adapted. 3 Infection of a non-galliform or -anseriform bird (such as a pelican), or cross-infections between the orders Galliformes and Anseriformes, might be more likely to result in disease. A previous report of encephalitis due to T. gallinarum occurred in a mockingbird (a passerine bird), and in the previous outbreak in ducks, a turkey was a possible source of the parasite.7,8 The pelican in the current report could have been exposed to a variety of native and exotic anseriform and possibly galliform birds on an open pond, and may have had other exposure opportunities prior to arriving at this zoological institution. A disseminated form of this infection has not previously been documented in any free-ranging or collection birds at this institution. However, the current case underscores the potential risk for Tetratrichomonas sp. infections across a broad avian host range, particularly when native and exotic species are allowed to mix in a zoological setting.
Footnotes
Acknowledgements
The authors would like to thank Megan Varney, Yvonne Cates, Cindy Spiva-Evans, and Jennifer Burchell for necropsy, histology, and laboratory support, and Don Ariyakumar and Dean Muldoon for electron microscopy preparation.
a.
Electron Microscopy Sciences, Hatfield, PA.
b.
JEM 1200 EXII, JEOL Ltd., Tokyo, Japan.
c.
Hitachi S3500N, Hitachi Ltd., Tokyo, Japan.
d.
DNeasy Blood and Tissue Kit, Qiagen Inc., Valencia, CA.
e.
MyTaq HS Red Mix, Bioline USA Inc., Taunton, MA.
f.
Ultra-free DA Centrifugal Unit, Millipore Corp., Billerica, MA.
g.
Eton Biosciences Inc., San Diego, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
