Abstract
Serum amyloid A (SAA) is reported not only as a marker for the presence of inflammation but also as a prognostic indicator in human beings. In cats, however, there is no report on the association between SAA concentration and prognosis. The objective of the current study was to evaluate SAA concentration as a prognostic marker in diseased cats. A total of 175 cats with neoplastic diseases, inflammatory diseases, and other diseases were retrospectively recruited, and the medical records of these cats, including follow-up data on mortality, were reviewed. Cats were divided into 2 groups according to SAA concentration, and differences in survival between each group were assessed. Median survival time of cats in the elevated SAA (>0.82 mg/l) group was significantly shorter than that in the nonelevated SAA (≤0.82 mg/l) group (P < 0.001). Furthermore, by multivariate analysis, SAA concentration was shown as a significant and independent prognostic marker in cats with various diseases (P = 0.015). Serum amyloid A concentration in diseased cats is a useful predictive indicator of prognosis regardless of diagnosis.
Serum amyloid A (SAA) is one of the major acute phase proteins in many species including human beings7,10 and cats. 5 SAA concentration may increase up to 1,000-fold in inflammatory status in human beings 7 and cats. 14 Therefore, measurement of SAA concentration is considered useful to detect the presence of inflammation.11,14 In human medicine, SAA is not only a marker of existing inflammation but also a prognostic marker for several diseases including inflammatory diseases, neoplasia, and also noninflammatory diseases such as diabetes.3,4,6 In cats, SAA concentration increased not only in inflammatory diseases but also in neoplastic and noninflammatory diseases, such as diabetes mellitus and hyperthyroidism. 14 The objective of the current study was to evaluate the relationship between SAA concentration and prognosis in cats.
Medical records of cats referred to the Veterinary Medical Center of the University of Tokyo (VMC-UT) from April 2006 through July 2007 were reviewed. Follow-up data on mortality were collected until March 2012 from medical records, referring veterinarians, and telephone interviews with owners. Inclusion criteria for the study were confirmed diagnosis and sufficient clinicopathological data including SAA concentration at the time of arrival at the VMC-UT. Cats were excluded from the study if there was only a 1-day follow-up period because of insufficient information for survival analysis. Cats that had unrelated and symptomatic concurrent diseases were also excluded. Cats with incidentally detected, asymptomatic concurrent diseases such as chronic renal failure and cardiomyopathy were included in the present study.
Information including signalment, diagnosis, white blood cell count (WBC), packed cell volume (PCV), platelet count, and SAA concentration were collected from medical records. The data of the blood assays obtained on the day of first examination at VMC-UT was used for analysis. WBC, PCV, and platelet count were obtained using an automatic analyzer. a WBC was considered abnormal when the value was below 5,500 μl or above 19,500 μl. Anemia was defined as a PCV value below 24%, and platelet count was considered abnormal when the value was below 300,000 μl. As described in a previous study, SAA concentrations were measured in the cats by using a commercially available turbidimetric immunoassay for human SAA b with an automated chemical analyzer.c,14 The reference range of feline SAA was selected as ≤0.82 mg/l on the basis of a previous study. 14
Cats were divided into 2 groups according to SAA concentration. The nonelevated SAA group included the cats whose SAA concentration was within the reference range (≤0.82 mg/l), and the elevated SAA group included the cats whose SAA concentration was above the reference range (>0.82 mg/l). Furthermore, cats were divided into 3 categories according to diagnosis: neoplastic diseases, inflammatory diseases, and other diseases. The inflammatory diseases category included infectious diseases such as feline infectious peritonitis.
Statistical analyses were performed with a statistical software package. d To determine differences between nonelevated and elevated SAA groups, the chi-square test was used for categorical data. The Mann–Whitney U-test was used to analyze continuous data. Differences in survival between each group were assessed using the Kaplan–Meier product limit method and the generalized Wilcoxon test. Correlation between SAA concentration and survival time was assessed using the Spearman rank correlation coefficient. Multivariate analyses were performed using the Cox proportional hazards model for any variables that were significant on the univariate analysis. Variables with P values <0.05 were considered statistically significant.
A total of 175 cats with various diseases were included. Patient characteristics are summarized in Table 1. There were 110 cats in the nonelevated SAA group and 65 cats in the elevated SAA group. Median SAA concentration in each group was 0.15 mg/l (range: 0–0.8 mg/l) and 18.1 mg/l (range: 0.83–88.3 mg/l), respectively. There were no significant differences between the 2 groups regarding age, sex, and platelet count. However, body weights of the elevated SAA group were significantly lower than those of the nonelevated SAA group (P = 0.047). Furthermore, in the elevated SAA group, 32 cats (49.2%) had abnormal WBC and 19 cats (29.2%) had anemia. These percentages were significantly higher than those in the nonelevated SAA group (P = 0.002 and 0.001, respectively).
Study population and clinical characteristics of the cats included in the current study.*
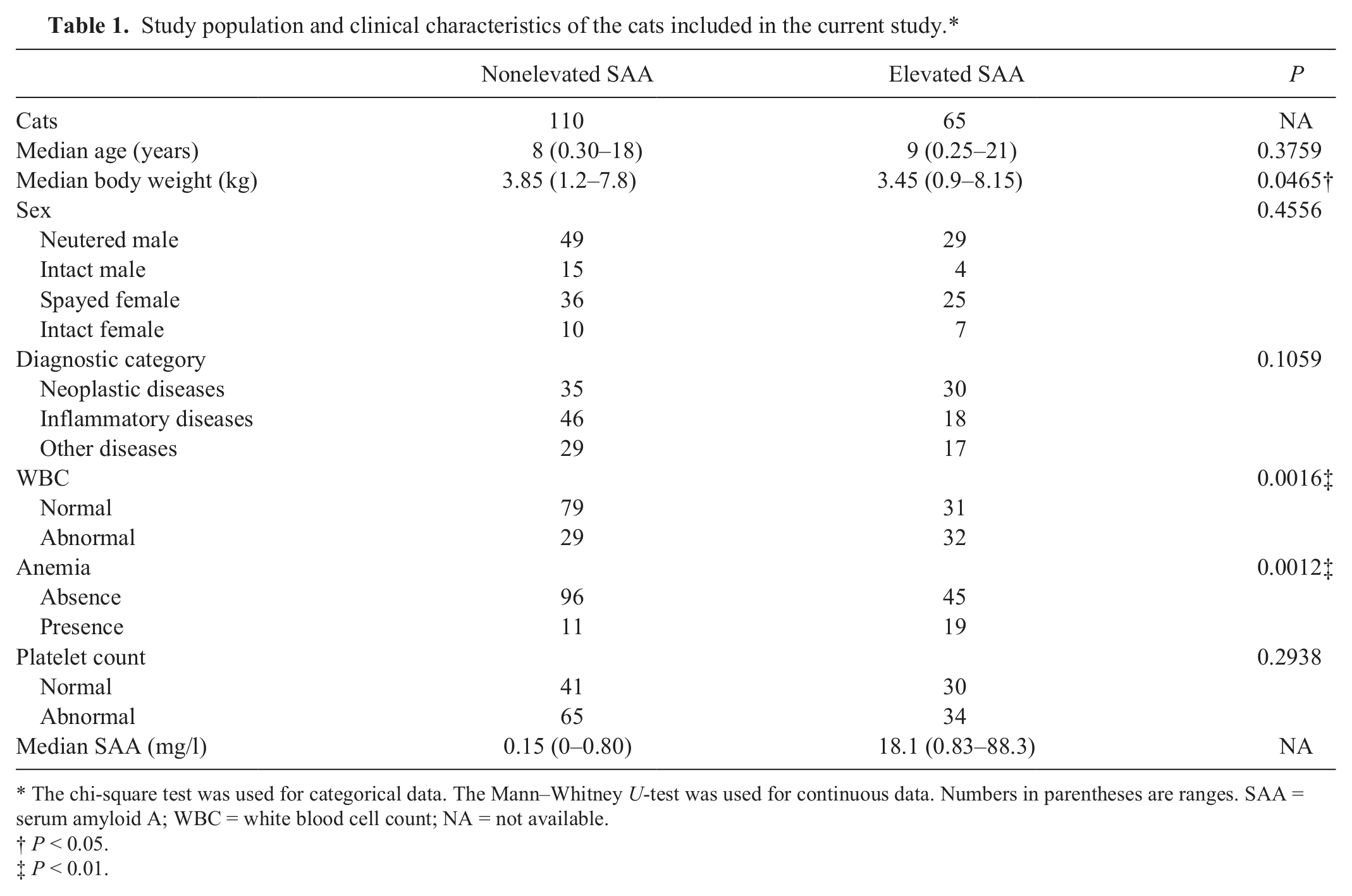
The chi-square test was used for categorical data. The Mann–Whitney U-test was used for continuous data. Numbers in parentheses are ranges. SAA = serum amyloid A; WBC = white blood cell count; NA = not available.
P < 0.05.
P < 0.01.
According to diagnostic category, 65, 64, and 46 cats were categorized in neoplastic diseases, inflammatory diseases, and other diseases, respectively. Diagnosis of each category and the number of cats that had elevated SAA concentration are shown in Table 2. Twenty-two cats had asymptomatic concurrent diseases such as chronic renal failure and cardiomyopathy. These cats were categorized according to the symptomatic diagnosis; the concurrent diseases were not reflected in the data. The distribution of diseases was analyzed based on diagnostic categories, and there was no significant difference between the nonelevated SAA and elevated SAA groups (P = 0.106).
Diagnosis and number of cats in each diagnostic category in the current study.*
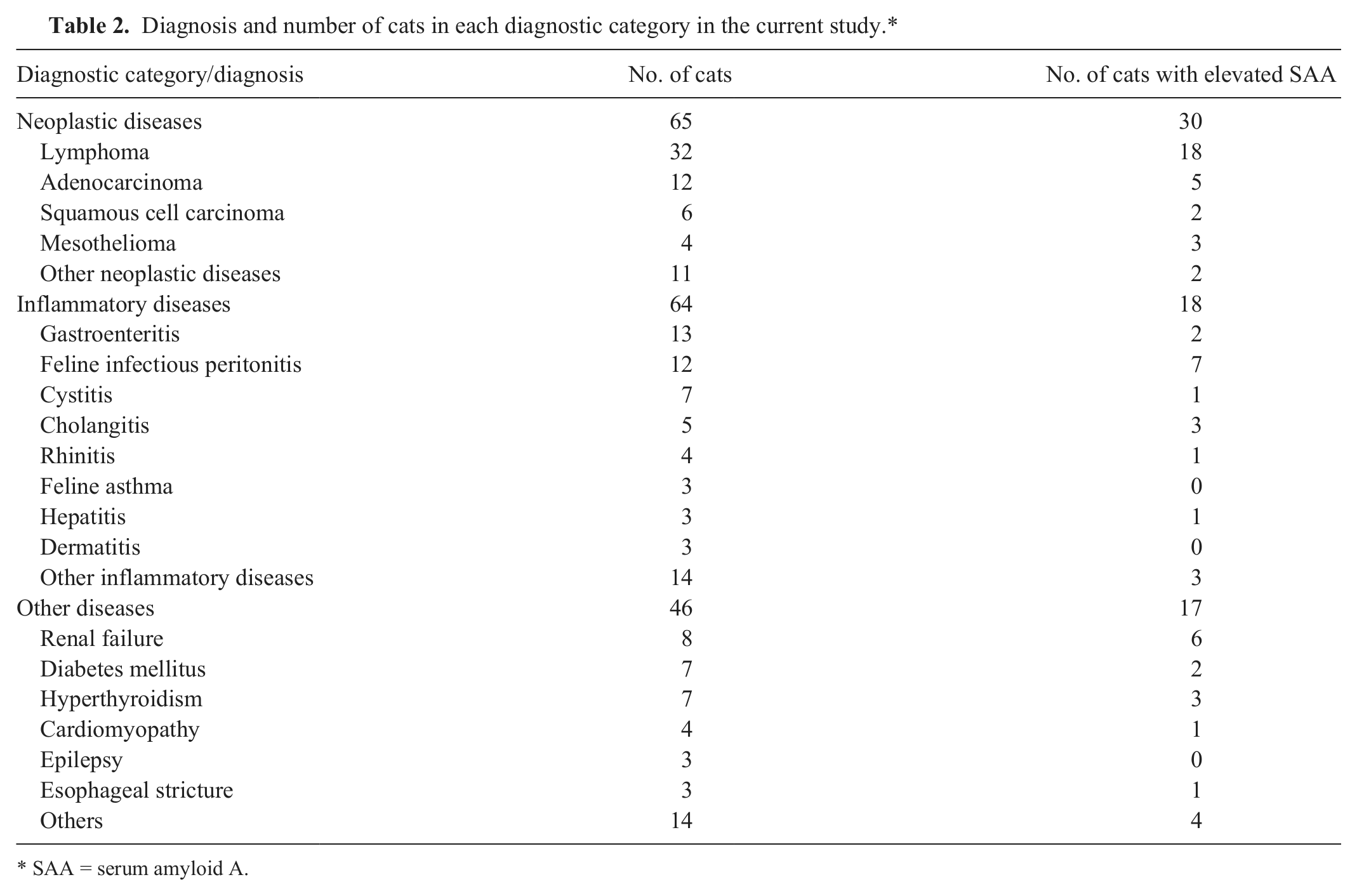
SAA = serum amyloid A.
Median follow-up time for all cats was 42 days (range: 1–2,104 days). Seventy-seven cats (70%) in the nonelevated SAA group and 29 cats (44.6%) in the elevated SAA group were alive at the time of last contact. Reasons for premature cessation of hospital visits were not always available from medical records; those cats were censored from the survival analysis at the time of the last arrival. Median survival time for the nonelevated SAA group was 571 days compared with 72 days for the elevated SAA group (P < 0.001; Fig. 1). No correlation was observed between the magnitude of SAA concentration and survival time (data not shown). With the Cox proportional hazards model, age, diagnostic category, abnormal WBC, presence of anemia, and elevated SAA concentration were shown to be significant prognostic factors by univariate analysis. These 5 parameters were included in the multivariate analysis, and elevated SAA concentration was shown to be a significant and independent prognostic factor (hazard ratio: 1.39, 95% confidence interval: 1.07–1.81, P = 0.015).
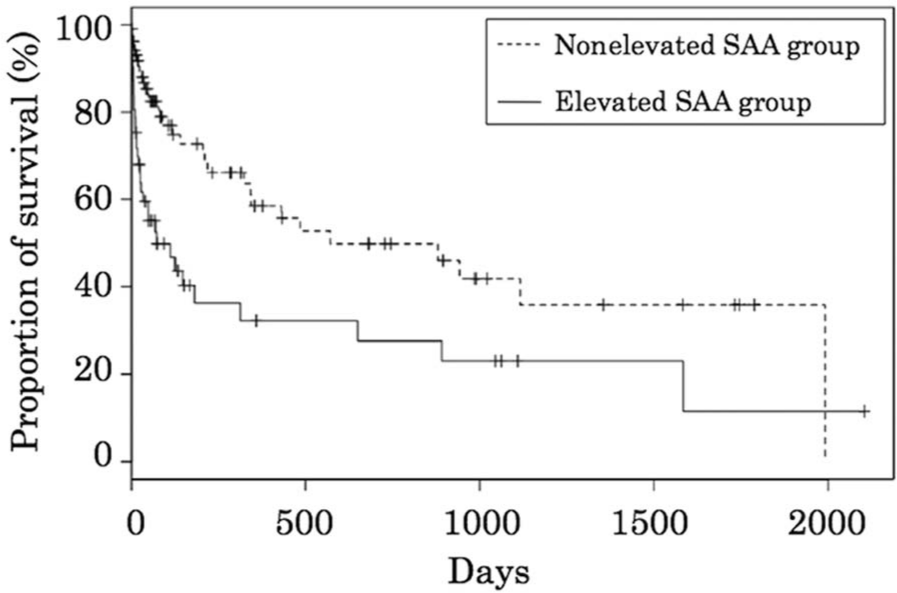
Kaplan–Meier survival curves of cats categorized according to serum amyloid A (SAA) level. The nonelevated SAA group (≤0.82 mg/l, broken line) and elevated SAA group (>0.82 mg/l, solid line) are shown. The significance of the difference between the curves was calculated by the generalized Wilcoxon test (P = 0.0001).
In the present study, SAA concentration at the time of first examination at the hospital was demonstrated as a significant prognostic factor in cats with neoplasia, inflammatory diseases, and other diseases. From the survival curve analysis, SAA concentration seems to affect survival, particularly short-term survival. However, the prognosis was affected only by whether SAA concentration was elevated. The magnitude of SAA concentration at the first visit did not correlate with prognosis in the current study. Further investigation is warranted to evaluate the association between serial changes of SAA concentration and prognosis.
Although cats with neoplastic diseases were included in the present study, and neoplastic diseases are generally considered to have poorer prognoses than non-neoplastic diseases, SAA concentration was shown as an independent prognostic marker from diagnostic category by the Cox proportional hazards model analysis. Age, abnormal WBC, and anemia were demonstrated as prognostic marker only by univariate analysis. It was considered that age was related to diagnostic category and that abnormal WBC and anemia were related to SAA concentration, respectively.
There are several reasons why SAA concentration could function as a prognostic marker. First, increased inflammatory markers, such as SAA, suggest that the diseases are poorly controlled, and chronic inflammatory status is considered to affect prognosis directly. The association between SAA concentration and prognosis in human patients with rheumatoid arthritis was reported. 4 Furthermore, C-reactive protein (CRP), as well as SAA, is one of the major acute phase reactants in human beings, and elevated CRP concentration was found to be a prognostic factor in rheumatoid arthritis. 12 Because both SAA and CRP were reported as prognostic markers in patients with rheumatoid arthritis, the theory that these markers reflect the activity of the diseases is plausible. Second, SAA protein is known as a precursor of amyloid A fibrils, and sustained high SAA levels may give rise to secondary amyloidosis, so called reactive amyloid A amyloidosis. 8 The development of amyloid A amyloidosis is considered an important prognostic factor. 5 Third, there are several reports that SAA protein promotes cytokine production 13 and leukocyte migration. 1 These functions of SAA protein might affect the prognoses. However, all of these facts were reported in human medicine, and it remains unclear whether it can be applied to cats.
A previous study demonstrated that cats with metabolic or endocrine diseases often showed high SAA concentration. 14 Also in human patients with diabetes and metabolic syndrome, high SAA concentrations were reported, and SAA concentration was considered to be a risk factor of these diseases.3,9 Serum amyloid A concentration appears to be related to the development of cardiovascular disorders and nephropathy 15 in diabetes, and these concurrent disorders are considered to affect prognosis.
The limitations of the current study are mainly due to the retrospective study design. Cats with various diseases were included in the study, and diagnostic criteria of these diseases were not constant; furthermore, treatment before and after diagnosis differed among cats and might have affected the prognosis. Cats with neoplastic diseases were more likely to return for follow-up visits than cats with other non-neoplastic diseases; therefore, mortality data of the cats with neoplasia were more abundant compared with the cats with other diseases. This might have led to a biased result. In human medicine, it was reported that mean SAA levels during the treatment period were significantly related to the survival time of patients with lung cancer. 2 Although the change in SAA concentration, an increase or decrease, is considered important in prognosis, serial data about each patient could not be collected in the present study.
In conclusion, SAA concentration was demonstrated as a significant and independent prognostic factor in diseased cats by retrospective analysis. The presence of systemic inflammation may have deleterious effects on the prognosis of cats as well as human beings. Future investigation should preferably evaluate the relationship between serial SAA concentrations and prognosis in cats with a single disease in a prospective study.
Footnotes
a.
PocH-100, Sysmex Co., Kobe, Japan.
b.
LZ-SAA, Eiken Chemical Co. Ltd., Tokyo, Japan.
c.
TBA-80FR NEO2, Toshiba Medical Systems, Tokyo, Japan.
d.
JMP version 5.0.1J, SAS Institute, Cary, NC.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a Grant-in-Aid for Scientific Research from the Japan Society for the Promotion of Science and the Ministry of Education, Culture, Sports, Science and Technology of the Japanese Government.
