Abstract
A 6-month-old, neutered male, mixed-breed dog was examined for a 2-month persistent fever, nonhealing dermal metacarpal area wound, and leukocytosis (47.0–198.0 × 103/μl). Serum chemistry findings included hypoalbuminemia, hyperglobulinemia, hyperphosphatemia, and hyperphosphatasemia. Complete blood cell count results revealed a moderate microcytic, hypochromic nonregenerative anemia with a profound leukocytosis (198.5 × 103/μl), characterized by neutrophilia with toxicity and hypersegmentation, and significant band cells. Tick-borne disease titers (genera Anaplasma, Ehrlichia, and Borrelia) were negative, as were polymerase chain reaction for other infectious agents (genera Hepatozoon, Mycobacterium, Mycoplasma; and Canine distemper virus). No agents were identified in a deep dermal biopsy (conventional and special histochemical stains) of the chronic draining, metacarpal region lesion. Cytology of the draining tract revealed numerous mixed bacteria and a surprising lack of neutrophils. Chronic occult blood loss with iron deficiency was considered a possible cause of the anemia. Differentials for the leukon were chronic established inflammation (occult infectious agent), chronic neutrophilic leukemia, paraneoplastic leukocytosis (neoplastic source of granulocyte colony-stimulating factor [CSF] or granulocyte-macrophage CSF), and leukocyte adhesion deficiency (LAD). The possibility of a LAD disorder was further investigated because of the noted hypersegmented neutrophils, absence of neutrophils in the cytology sample, the animal’s young age, and persistence of clinical and laboratory signs. Flow cytometry of blood neutrophils showed a 60% reduction in surface expression of the β2-integrin (CD18) subunit, whereas neutrophil function tests (oxidative burst and phagocytosis) were normal. Genetic testing revealed a homozygous missense mutation in the β2-integrin subunit gene, previously recognized only in purebred Irish Setters, leading to a diagnosis of LAD type 1 disorder in this mixed-breed dog.
Keywords
A 6-month-old, 15.9 kg, neutered male, mixed-breed dog was referred to the Virginia-Maryland Regional College of Veterinary Medicine (VMRCVM; Virginia Tech, Blacksburg, VA) for fluctuating fever, delayed wound healing, jaw swelling, and persistent leukocytosis of at least 2-month duration. The dog had been found and taken to a rescue shelter when 6 weeks old, where he presumptively developed kennel cough and a protracted upper respiratory tract infection. At 4 months of age, he was neutered, experienced delayed wound healing, and subsequently developed swelling of his distal front limbs and swelling of the jaw with pain on opening his mouth. His fever fluctuated between 39.4°C to 41.1°C over this 2-month period, and his white blood cell count varied from 47.0 to 198.0 × 103/μl.
On examination at VMRCVM, the dog’s temperature was 39.4°C, pulse 150 bpm, and respiration 20 bpm. Multiple retained deciduous teeth and a fetid mouth odor were noted. The dog was grade IV/IV lame on the left forelimb with open draining wounds over the dorsal aspect of the third and fourth metacarpals, and there was a firm swelling over the caudal aspect of the left mandible.
Cytologic examination of a scraping from the draining metacarpal region wound (Fig. 1) revealed mixed bacteria identified as Escherichia coli and Streptococcus canis on culture. Noteworthy serum chemistry findings a included hypoalbuminemia (2.3 g/dl, reference interval [RI]: 2.8–3.7 g/dl), hyperglobulinemia (4.2 g/dl, RI: 2.1–3.8 g/dl), hyperphosphatemia (5.0 mg/dl, RI: 1.9–4.4 mg/dl), and hyperphosphatasemia (675 U/l, RI: 8–70 U/l). A microscopic image of a blood smear (Fig. 2) made at time of VMRCVM admission and concurrent complete blood cell count (CBC) results, b as well as 30 days prior, are shown in Table 1.

Cytology sample prepared from a scraping of the draining metacarpal region wound showing numerous mixed types of bacteria and a few degenerate cells. Modified Wright stain. Bar = 10 μm.
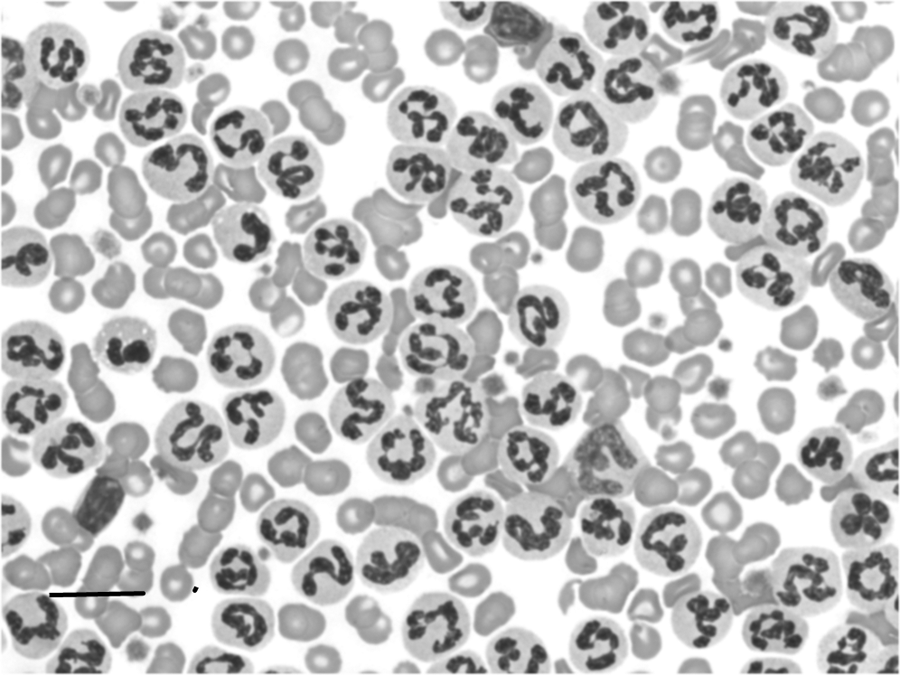
Blood smear from the mixed-breed dog at 6 months of age showing the 198,500 white blood cells/μl with marked neutrophilia and neutrophil hypersegmentation. Modified Wright stain. Bar = 20 μm.
Serial hematology results from mixed-breed dog at 5 and 6 months of age.*
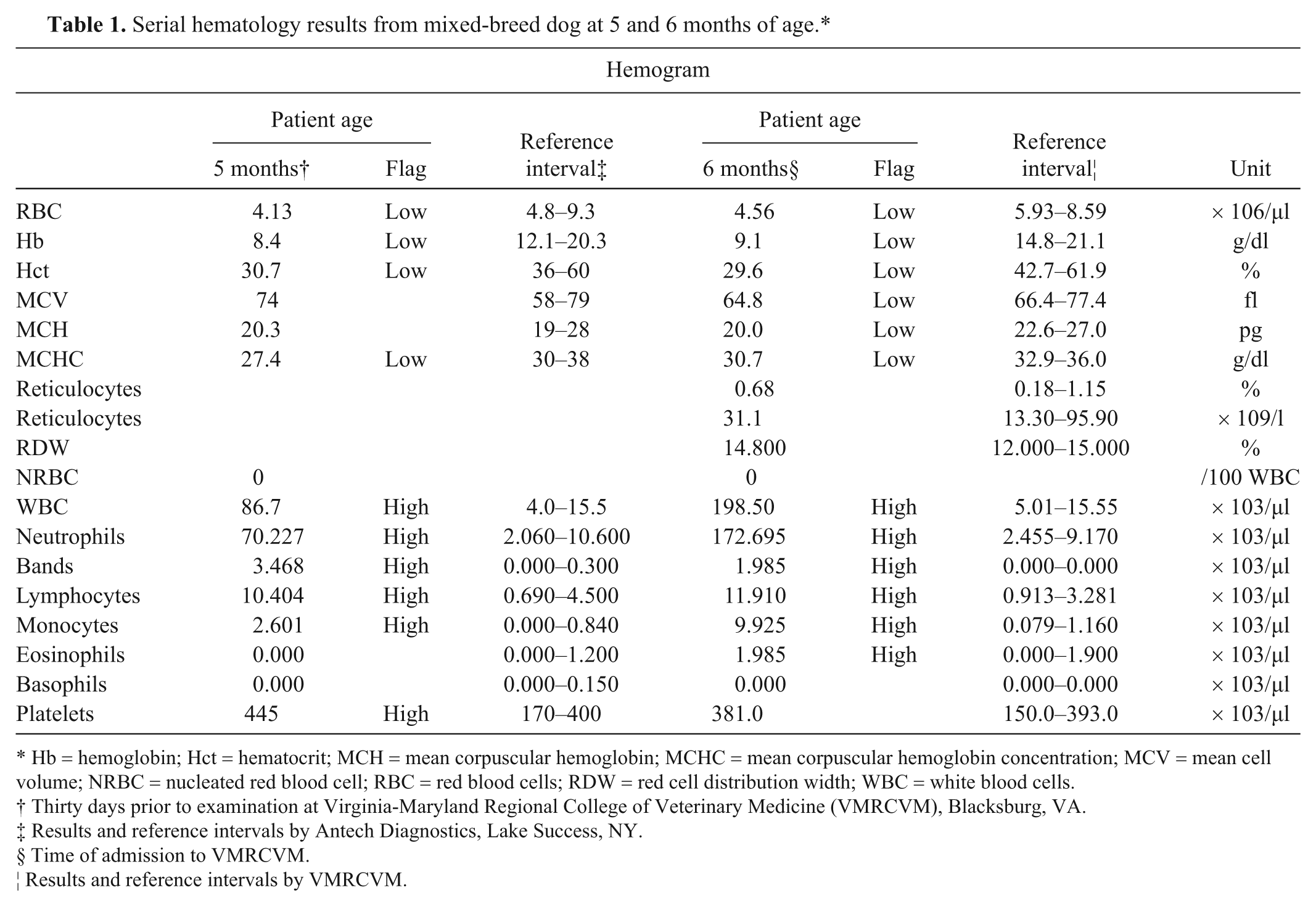
Hb = hemoglobin; Hct = hematocrit; MCH = mean corpuscular hemoglobin; MCHC = mean corpuscular hemoglobin concentration; MCV = mean cell volume; NRBC = nucleated red blood cell; RBC = red blood cells; RDW = red cell distribution width; WBC = white blood cells.
Thirty days prior to examination at Virginia-Maryland Regional College of Veterinary Medicine (VMRCVM), Blacksburg, VA.
Results and reference intervals by Antech Diagnostics, Lake Success, NY.
Time of admission to VMRCVM.
Results and reference intervals by VMRCVM.
Additional diagnostics were performed on the dog. Blood samples were collected for infectious disease testing. Titers or polymerase chain reaction (PCR) c for Anaplasma phagocytophilum, Ehrlichia canis, and Borrelia burgdorferi infection were negative. PCR d for Hepatozoon americanum and H. canis and Canine distemper virus were negative. Histologic examination of a surgical biopsy from the draining wound over the metacarpal region indicated chronic marked multifocal to coalescing histocytic, lymphocytic, neutrophilic dermatitis. Ziehl–Neelsen, periodic acid–Schiff, and Brown–Brenn modified Gram stains of the biopsy sample did not reveal the presence of acid-fast, fungal, or other bacteria in the deep portions of the section; PCRs e for Mycobacterium and Mycoplasma were also negative. Radiographs of the lung indicated a mild interstitial pattern. Mandibular radiographs were interpreted as being compatible with craniomandibular osteopathy, whereas changes noted in the left and right radius and ulna were felt to be compatible with hypertrophic osteodystrophy.
Findings from the CBC at time of admission to VMRCVM included the presence of a mild to moderate microcytic, hypochromic nonregenerative anemia with a profound leukocytosis, neutrophilia, significant band cells, lymphocytosis, monocytosis, and slight eosinophilia. Examination of the blood smear confirmed the lack of significant polychromasia with mild rouleaux (Fig. 2). There was mild poikilocytosis with occasional target cells and rare echinocytes; slide estimate of white blood cell numbers agreed with reported values. There was a notable orderly left shift back to the band cell stage with mild toxicity characterized by the presence of Döhle bodies, slight basophilia, and rare vacuolation of both segmented neutrophils and band cells. Hypersegmentation was a notable feature of the neutrophils along with occasional gigantism. Lymphocytes were occasionally reactive. Platelets appeared adequate to mildly increased with occasional macroplatelets seen and a few clumps noted at the feathered edge.
The leukon and thrombon findings were interpreted to be most compatible with chronic antigenic stimulation and inflammation, and a reactive thrombocytosis. However, the dysplasia noted in the neutrophils prompted consideration of other conditions, which included chronic neutrophilic leukemia, paraneoplastic leukocytosis, and leukocyte adhesion deficiency (LAD). Other causes of neutrophilia such as steroid-related, physiological response, and estrogen toxicosis were felt unlikely considering the magnitude and duration of the increase. Furthermore, several of the serum chemistry changes were compatible with chronic inflammation with hypoalbuminemia due to albumin being a negative acute phase protein and the concurrent gammopathy, presumably polyclonal, being associated with chronic antigenic stimulation. Changes associated with the erythron were most compatible with interference of hemoglobin synthesis as seen most commonly with absolute iron deficiency anemia; however, the lack of clinical or other laboratory evidence of chronic blood loss argued against this possibility, and hypochromasia was not a notable feature of the blood smear. Other causes of microcytic anemia such as liver disease, breed, age, copper deficiency, and B6 deficiency were deemed unlikely based on signalment and serum chemistry data. Therefore, in the context of the noted leukogram and in spite of the fact that a normocytic normochromic anemia is much more common, it was felt that the best explanation for the microcytosis and hypochromasia was a relative iron deficiency related to anemia of chronic disease, which was further supported by the finding of abundant marrow iron stores as noted below with the marrow findings.
Core bone marrow biopsy indicated a hypercellular marrow with myeloid hyperplasia. Findings from bone marrow cytology included a myeloid-to-erythroid ratio of 7:1 with 5% blast cells overall and abundant iron stores; there was no dysplasia or dyscrasia in the myeloid or erythroid cell lines (Fig. 3). Megakaryocytes were present in increased numbers (4–5/particle), whereas lymphocytes were less than 5% of the nucleated cells, and plasma cells were rare. The lack of dysplasia and dyscrasia in the marrow along with the low percentage of blast cell stages greatly decreased the probability of a myeloid leukemia as an explanation for the persistent neutrophilia.
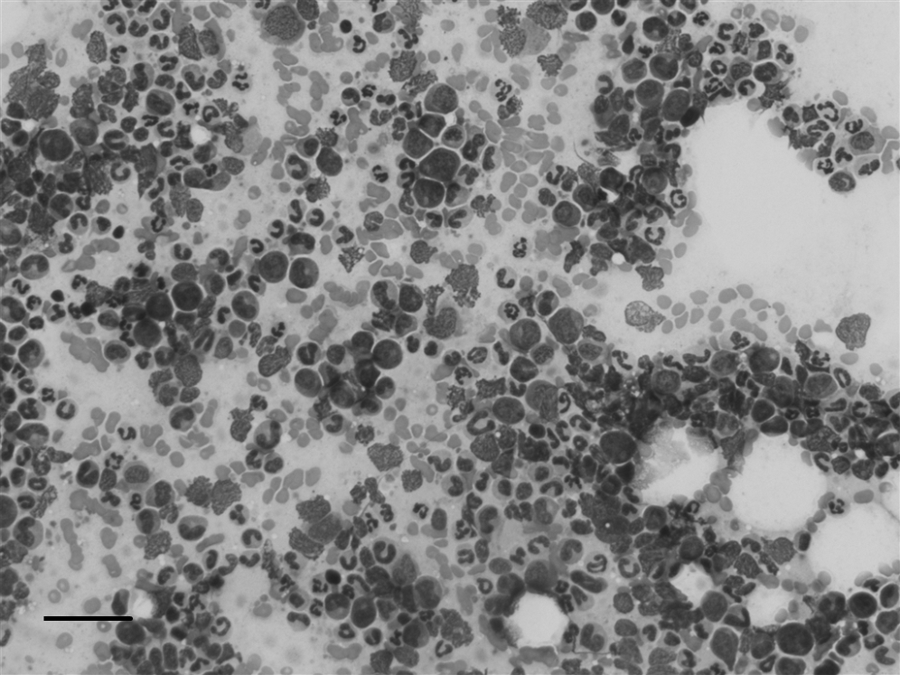
Bone marrow aspirate from the mixed-breed dog at 6 months of age showing the myeloid hyperplasia with orderly progression, large neutrophil storage pool, and a low percentage of blast precursors. Modified Wright stain. Bar = 50 μm.
Alternate causes for the myeloid hyperplasia were then considered. Because of the lack of evidence of any intrathoracic or intraabdominal masses in the radiographic images, it was felt that the likelihood of paraneoplastic leukocytosis related to neoplastic production of granulocyte colony-stimulating factor (CSF) or granulocyte-macrophage CSF was low. Although hypertrophic osteodystrophy can be associated with an inflammatory leukogram, the degree of the neutrophilia seemed disproportionate for such a response. Many of the common infectious causes of marked leukocytosis had already been excluded via PCR and serology. These considerations, along with knowledge of the dog’s young age, marked hypersegmentation of neutrophils, and lack of extravascular inflammatory cells in the cytology sample of the metacarpal region wound suggested a LAD-type disorder.
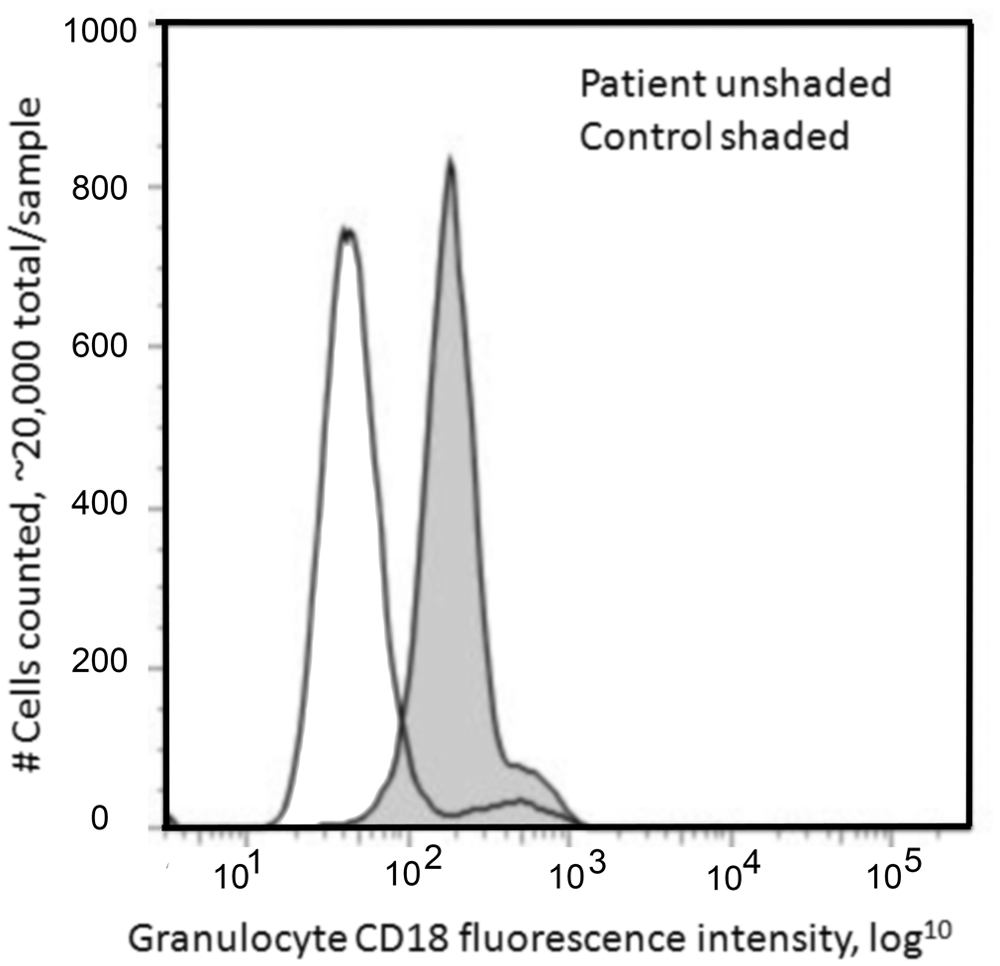
Further diagnostics to investigate the possibility of canine LAD included neutrophil function assays, flow cytometry of neutrophils to assess cluster of differentiation (CD)11/CD18 integrin expression, and genetic testing for presence of a point mutation related to the CD18 domain. A neutrophil functional assay f composed of 2 parts, neutrophil oxidative burst g and phagocytic activity, h was conducted. All of the patient’s neutrophil function test results exceeded those of an adult, clinically healthy, control dog, which had a normal CBC. The oxidative burst was measured in response to stimulus phorbol 12–myristate 13–acetate (PMA) and a particulate stimulant E. coli. The oxidative PMA result was 48% for the patient and 39% for the control dog, with the estimated normal response range being 60% to 93%. The decreased result for both the patient and control dogs was attributed to a processing delay due to shipment of the samples to the testing laboratory. The oxidative E. coli result was 58% for the patient and 54% for the control dog, with the estimated normal range being 30% to 95%. The phagocytosis activity for the patient was 46% and 18% for the control dog, with the estimated normal response being 50% to 99%. For the flow cytometry analysis, i leukocytes were counted and standardized to a concentration of approximately 4 × 106 cells/ml, then stained with fluorescein isothiocyanate–labeled rat, antihuman, CD18 monoclonal antibody. j Flow cytometry results k on approximately 20,000 events per sample showed a significant median fluorescence sample difference l (Mann–Whitney test, P < 0.05, 95% confidence interval median difference: −104.14 to −101.65) for the control (median fluorescence 178.06) and patient (median fluorescence 72.33), representing a 60% median reduction in granulocyte CD18 fluorescence intensity in the case dog compared to the control dog (Fig. 4). Genetic testing m for the Irish Setter mutation of the β2-integrin gene responsible for canine LAD in this breed was homozygous positive. Confirmation of the carrier status requires DNA testing using oligonucleotide ligation assay, pyrosequencing, or DNA pyrosequencing. 6 The method used for identification of the LAD mutation in the current case was a commercialized analysis based on pyrosequencing with PCR amplification using primers directed at the recognized Irish Setter DNA mutation region, restrictase enzyme cleavage, and then gel visualization as described previously. 10 These findings support a diagnosis of canine LAD type 1 disorder in the dog described herein, as seen in Irish Setters. In light of this diagnosis, the cause of discrepancy between the decreased numbers of neutrophils seen in the cytology sample versus the increased number seen in the biopsy sample became clear. The leukemoid response and intravascular restricted neutrophils gave the histologic impression of neutrophilic inflammation at the biopsy site, whereas the cytology sample reflected the ineffective neutrophil diapedesis associated with the adhesion molecule deficiency.
Neutrophils found within the luminal space of blood vessels of dogs are approximately equally distributed between 2 functional pools, a circulating pool and a marginal pool. 7 It is the population within the freely circulating pool that is sampled when a vascular blood sample is drawn and whose number is reported as part of a CBC. The marginal neutrophil pool population is located in close association with the vascular endothelium and remains occult to such sampling. This close, neutrophil, endothelial marginal pool association is mediated through a variety of surface adhesion molecules.
One group of adhesion molecules is referred to as selectins. P- and E-selectins are expressed on endothelium and interact with oligosaccharide ligands (sialyl-Lexis X) found on neutrophils allowing them to “roll” and form weak adhesion bonds with the endothelium.6,7,12 Assisted by this initial tethering of neutrophils through the selectin-ligand interaction, another group of adhesion molecules, referred to as integrins, facilitate neutrophil arrest and adhesion and transmigration in response to a wide variety of chemotactic signals. Although there are a variety of integrin proteins, it is the heterodimeric CD11/CD18, β2-integrin receptor, found on all leukocytes, and its interaction with the immunoglobulin intercellular adhesion molecule 1 (ICAM-1) ligand molecule, found on endothelium, that is most important for neutrophil arrest and migration.6,12
It is important to note that the affinity and expression of these various adhesion molecules (selectins and integrins) as well as their respective ligands increase in response to inflammatory molecular signaling (interleukin 1, tumor necrosis factor, etc.). This up-regulation of affinity and expression is crucial to facilitate recruitment of neutrophils from the vascular circulatory pool into the marginal pool in preparation of vascular transmigration in response to tissue inflammation. 12 Failure in any of these neutrophil rolling, arrest adhesion, or migration steps leads to the presence of an ineffective and often profoundly inflammatory leukogram characterized by a marked leukocytosis, neutrophilia, and left shift, and diminished neutrophil tissue trafficking at sites of inflammation.
LAD disorders have been identified in mice, cattle, dogs, and human beings.6,14 At least 3 variants of the disorder have been identified. Type I is the most common form in which there is a deficiency of the β2-integrin (CD18) subunit.4,6,8 Type II is due to a defect in fucose metabolism leading to a deficiency of the sialyl-Lewis X ligand for selectins on leukocytes, affecting their ability to tether and roll on endothelial cells.6,12 Type III (variant) is due to a defect in kindlin-3 mediated activation of β1-, β2-, and β3-type integrins expressed on leukocytes and platelets.1,6 In 2010, a LAD variant was described in a German Shepherd Dog due to a mutation in the canine kindlin-3 gene. 1 The dog experienced significant bleeding and infections, and had persistently high leukocyte counts.
The cause of LAD in Irish Setters and Irish Red and White Setters in both Europe and the United States has been identified as a type I missense mutation at base pair position 107 in the β2-integrin subunit gene (ITGB2) that results in an amino acid change from cysteine to serine at amino acid 36 (Cys36Ser) in the β2-integrin subunit protein.3-5,8-11 The occurrence of LAD disease in mixed-breed dogs is rare, but a few reports do exist. One such case deals with a purpose-bred, F2 mixed-breed breeding colony established from a pair of heterozygous-affected Irish Setters. 2 Another case involved 2 littermates with decreased expression of CD18(β2-integrin). 11 However, in that report, 11 the affected littermates had reduced neutrophil oxidative burst and phagocytic capacity (in contrast to typical LAD cases) and lacked the specific Irish Setter mutation. Although the pedigree from the dog of the present case is unknown, its external phenotype was of mixed breed with a dolichocephalic head, drop ears, and a medium-length black and brown coat.
Typical clinical signs for dogs affected with LAD disease include development of omphalitis and bacterial infections accompanied by fever occurring soon after birth. Affected dogs typically develop a progressive marked neutrophilic leukocytosis, accompanied by development of severe gingivitis, lymphadenopathy along with poor wound healing, pyrexia, and anorexia. Hypertrophic osteodystrophy has also been described in dogs with LAD disease.2,6,13 Affected dogs usually die secondary to bacterial infections or are euthanized at a young age. The animal described herein was successfully managed by use of oral antibiotic therapy for several months following the LAD diagnosis before its condition declined further and was euthanized at the owner’s request. No necropsy was performed on this patient.
Footnotes
a.
AU480, Beckman Coulter Inc., Brea, CA.
b.
Advia 120, Siemens Healthcare Diagnostics Inc., Deerfield, IL.
c.
IDEXX Laboratories, Westbrook, ME.
d.
Molecular Diagnostics, College of Veterinary Medicine, Auburn, AL.
e.
Washington Animal Disease Diagnostic Laboratory, College of Veterinary Medicine, Pullman, WA.
f.
University of Tennessee, College of Veterinary Medicine, Knoxville, TN.
g.
BurstTest Kit (PhagoBurst; catalog no. 10-02000), Glycotope Biotechnology GmbH, Heidelberg, Germany.
h.
PhagoTest Kit (catalog no. 10-0100), Glycotope Biotechnology GmbH, Heidelberg, Germany.
i.
FACS ARIA cell sorter, BD Biosciences, San Jose, CA.
j.
Catalog no. MCA503PET (batch no. 0708, clone YFC118.3, 1:400 dilution), AbD Serotec, Raleigh, NC.
k.
FlowJo 7 for PC, Tree Star Inc., Ashland, OR.
l.
Minitab 15 for PC, Minitab Inc., State College, PA.
m.
OptiGen LLC, Ithaca, NY.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
