Abstract
Producer reports from ranches over 2,438 meters in southwest Colorado suggest that the mortality of preweaned beef calves may be substantially higher than the national average despite the selection of low pulmonary pressure herd sires for over 20 years. Diagnostic investigations of this death loss problem have been limited due to the extensive mountainous terrain over which these calves are grazed with their dams. The objective of the current study was to determine the causes of calf mortality on 5 high-altitude ranches in Colorado that have been selectively breeding sires with low pulmonary pressure (<45 mmHg) for over 20 years. Calves were followed from branding (6 weeks of age) in the spring to weaning in the fall (7 months of age). Clinical signs were recorded, and blood samples were taken from sick calves. Postmortem examinations were performed, and select tissue samples were submitted for aerobic culture and/or histopathology. On the principal study ranch, 9.6% (59/612) of the calves that were branded in the spring either died or were presumed dead by weaning in the fall. In total, 28 necropsies were performed: 14 calves (50%) had lesions consistent with pulmonary hypertension and right-sided heart failure, and 14 calves (50%) died from bronchopneumonia. Remodeling of the pulmonary arterial system, indicative of pulmonary hypertension, was evident in the former and to varying degrees in the latter. There is a need to better characterize the additional risk factors that complicate pulmonary arterial pressure testing of herd sires as a strategy to control pulmonary hypertension.
Pulmonary hypertension is a disease syndrome that is problematic for cattle producers in mountainous regions of the United States. The condition, most commonly known as “brisket disease” or “high-altitude disease,” is thought to be due to an exuberant vasoconstrictive response of the pulmonary arteries to hypoxia that results in pulmonary hypertension culminating in congestive heart failure. 11 A blunted pulmonary pressor response is beneficial for mammals at high altitude. 8 Studies suggest the pulmonary pressor response to be moderately heritable. 25 This means that managers of high-altitude herds that select sires with low pulmonary arterial pressures, typically <45 mmHg, 11 should be able to reduce the incidence of high-altitude disease within their herds. 22 A description of pulmonary artery pressure (PAP) testing can be found elsewhere. 11
Prior to the initiation of sire selection based on PAP measurement, ranchers observed calves with typical signs of brisket disease, which included rapid onset brisket edema, jugular swelling, exophthalmia, and ascites. Affected calves were known colloquially as “belly-draggers.” Personal communication with producers that have initiated and maintained a breeding program based on herd sires with low PAP indicate that they rarely see calves showing these typical signs of high altitude disease.
Producer reports from ranches over 2,438 meters (8,000 ft) in southwest Colorado suggest that the mortality of preweaned beef calves may be substantially higher than the national average (6.4%; U.S. Department of Agriculture, Animal and Plant Health Inspection Service, Veterinary Services, National Animal Health Monitoring System: 1997, Beef ’97, Part 1: reference of 1997 beef cow-calf management practices. Fort Collins, CO. Available at: http://www.aphis.usda.gov/animal_health/nahms/beefcowcalf/downloads/beef97/Beef97_dr_PartI.pdf; USDA, APHIS, VS, NAHMS: 1997, Part II: reference of 1997 beef cow-calf health and management practices. Fort Collins, CO. Available at: http://www.aphis.usda.gov/animal_health/nahms/beefcowcalf/downloads/beef97/Beef97_dr_PartII.pdf) despite the selection of low PAP herd sires for over 20 years. Records from a ranch in southwestern Colorado that has used only bulls with low PAPs as sires for over 25 years indicate that, on average, 11.9% (±0.8% from 2006 to 2010) of calves that are turned out onto summer grazing either died or were presumed dead before weaning in the fall. This suggests that either selective breeding of low PAP scoring sires is an insufficient management tool in reducing the occurrence of pulmonary hypertension and/or calves are dying from other diseases unrelated to PAP testing per se. Producers believe the latter scenario to be the case, as the sick calves that they see over the summer period show signs more consistent with respiratory distress than the “belly-draggers” affected by pulmonary hypertension. Producer descriptions of sick calves include rapid breathing rate, dull expression, fever, roughened hair coats, drooped ears, and a stiff-gaited walk, usually followed by death. Calves summer-pastured with their dams at high altitude graze over a large area of rugged mountainous terrain and are seldom observed by producers or veterinarians between branding (approximately 6 weeks of age) in the spring and weaning in the fall when the calves are approximately 7 months of age. The current study was a diagnostic investigation of preweaned beef calf mortality on 5 high-altitude ranches in Colorado that have been selectively breeding sires with low PAP (<45 mmHg) for over 20 years.
Materials and methods
Study site
Postmortem examinations of calves were performed on 5 ranches in southwestern Colorado that have selected bulls with low PAP for at least 20 years. Calves on the 5 ranches consisted of predominantly British-based breeds (Leachman Stabilizer composite, pure Hereford, and Aberdeen Angus) and were pastured 2,438–3,505 m (8,000–11,500 ft) during the summer months.
Calves on the principal study ranch were extensively monitored to maximize the number of calves observed. Calves were observed from horseback 6 days per week from July 10 to October 15, 2010. Nighttime temperatures began to drop below freezing early in September at 2,730 m (8,958 ft). The maximum daily temperature would usually fall between 24–27°C. During this period, cow–calf pairs were rotated through 32,375 ha (80,000 acres) of mountainous terrain. Once sampled, treated sick calves remained in their respective herds. Calves were tagged at birth (March 20 to April 25, 2010). A clostridial vaccine containing Mannheimia haemolytica bacterin a and a growth hormone implant b were given to all calves at branding (May 15–17). Cows were given a prebreeding modified live respiratory disease vaccine according to the manufacturer’s instructions. c Salt and mineral blocks d were provided free choice year-round. Calves were not given a respiratory disease vaccine until 2 weeks prior to weaning. c Ear notch samples are routinely taken annually from all calves kept as replacement heifers for individual Bovine viral diarrhea virus (BVDV) enzyme-linked immunosorbent assay testing. Cows on the remaining 4 ranches were given a modified live vaccine prebreeding with 1 of 2 products offering protection against Bovine herpesvirus 1 (infectious bovine rhinotracheitis [IBR]), BVDV, Bovine respiratory syncytial virus (BRSV), and Bovine parainfluenza virus 3 (BPIV-3),c,e and calves on these ranches were vaccinated at both branding and weaning with 1 of 2 products.c,e No calves persistently infected with BVDV have been detected in these herds to date.
Clinical signs and blood sampling
Signs of disease were recorded on a standardized clinical evaluation form. After clinical signs were recorded calves were roped or, whenever possible, restrained in a nearby chute or corral for blood sampling. Calves showing signs consistent with respiratory disease were treated with tulathromycin f and flunixin meglumine. g Blood samples were taken from sick calves on the principal study ranch for biochemistry, complete blood cell count, and serology. After the calf had been restrained, a blood sample was obtained from the jugular vein using a 6-ml syringe and 3.81-cm (1.5 inches) 16-gauge hypodermic needle. Approximately, 2 ml was placed in an ethylenediamine tetra-acetic acid tube and 4 ml in a plain glass “red top” tube. After allowing sufficient time (30–60 min) for the blood to clot at body temperature, the blood samples were placed in the nearest running stream under shade or inside a plastic resealable zipper bag containing cool stream water, which was replaced every few hours. The clotted blood was spun at 1,100 × g for 10 min in a portable centrifuge machine h no later than 10 hr from the time of collection.
When a chute was available for calf restraint, arterial blood-gas analysis was performed on the principal study ranch using a handheld analyzer. i Calves were given 15 min to recover from the exertion involved in chute restraint. A blood sample was collected from the coccygeal artery using a 20-gauge, 2.54-cm (1 inch) heparinized hypodermic needle attached to a syringe. The coccygeal artery is a suitable site for collecting arterial samples in calves.6,18
Contemporary healthy calves from the same herd served as controls. The calves were sampled for arterial blood-gas (n = 17), biochemistry (n = 20), and serology (n = 8) at 2,730 m (9,000 ft). Calcium-chelated whole blood was obtained from healthy calves (n = 27) when the calves were approximately 6 months old.
Postmortem examination and tissue sample collection
A standardized postmortem evaluation form was used to record gross pathological observations. Postmortem examinations were performed on calves found within approximately 48 hr of death. Calves determined to be moribund and in sufficient distress and/or pain to warrant euthanasia were euthanized with a single shot from a .22 caliber pistol using landmarks on the cranium previously described. 17 Standard tissue sections preserved in 10% buffered neutral formalin included the apex of the right cranioventral lung lobe, the base of the right middle lung lobe, the dorsal aspect of the right diaphragmatic lung lobe, the pulmonary artery and aorta proximal to the closed ductus arteriosus, the free wall of the right and left ventricles and interventricular septum, spleen, liver, and both kidneys. Sections <2 cm thick were placed in a resealable plastic zipper bag containing a sufficient amount of formalin to coat the tissue surfaces (5–10 ml). Samples were later immersed in a 10:1 volume ratio of formalin to tissue no later than the night after the postmortem. After complete fixation, retrimmed tissues were embedded in paraffin, sectioned at 5 μm, and routinely stained with hematoxylin and eosin. Selected sections were stained with von Kossa stain for mineralization and Verhoeff–van Giesen (VVG) stain for elastic fibers. Pulmonary lesions were semiquantitatively evaluated as follows: 0 = no lesion, 1+ = mild, 2+ = moderate, 3+ = severe. The primary objective of the current study was to obtain a diagnosis of the cause of death. Usually a gross diagnosis of bronchopneumonia was sufficient for this purpose. Therefore, only a limited number of pneumonic lung samples were submitted for histopathology evaluation.
Lung lesions consistent with bacterial pneumonia were sampled for aerobic culture. Lung samples, with margins ≥4 cm, were collected at junctions adjacent to normal tissue using a sterile technique. Tissues were stored at 5°C and submitted for culture within 24 hr if possible. After 24 hr, nonsubmitted tissues were discarded.
Statistical analysis
All statistical analyses were performed using commercial software. j Type I error rate of 5% was used. Kruskal–Wallis rank tests were used for group average comparisons, as sample sizes were small. The Bonferroni method was then used to adjust the comparison-wise error rate to match the study-wise error rate (5%).
Results
On the principal study ranch, 612 calves were turned out with their dams onto summer grazing. Of those, 59 (9.6%) either died or were presumed to be dead by the time of weaning: 33 (66%) were found dead or were euthanized and 26 (44%) were not found. The corpses of 11 calves were too deteriorated to perform a postmortem. In total, 28 necropsies were performed: 22 on the principal study ranch and 6 on the other 4 ranches. Of the 28 necropsies, 14 calves (50%) lacked significant bronchopneumonia and had lesions consistent with pulmonary hypertension and right-sided heart failure, and 14 calves (50%) had gross lesions of significant bronchopneumonia.
The main clinical signs and gross pathology of the lungs are indicated in Table 1. Not shown in the table are clinical signs that were shared by calves with either pneumonia or pulmonary hypertension. Such signs included lethargy, tachypnea, drooped ears, and a rough hair coat. Sporadic coughing, ataxia, distension of the jugular vein, and diarrhea were sometimes observed in calves with either pneumonia or pulmonary hypertension. Calves with chronic pneumonia or pulmonary hypertension also showed distension of the jugular vein and diarrhea. Only calves with pulmonary hypertension showed brisket edema, exophthalmia, and ascites. Unlike calves with pulmonary hypertension, calves with acute pneumonia showed open-mouth breathing particularly on exertion. Calves with chronic pneumonia had the lowest body condition scores (<3/9).
Clinical signs and gross postmortem appearance of the lungs.
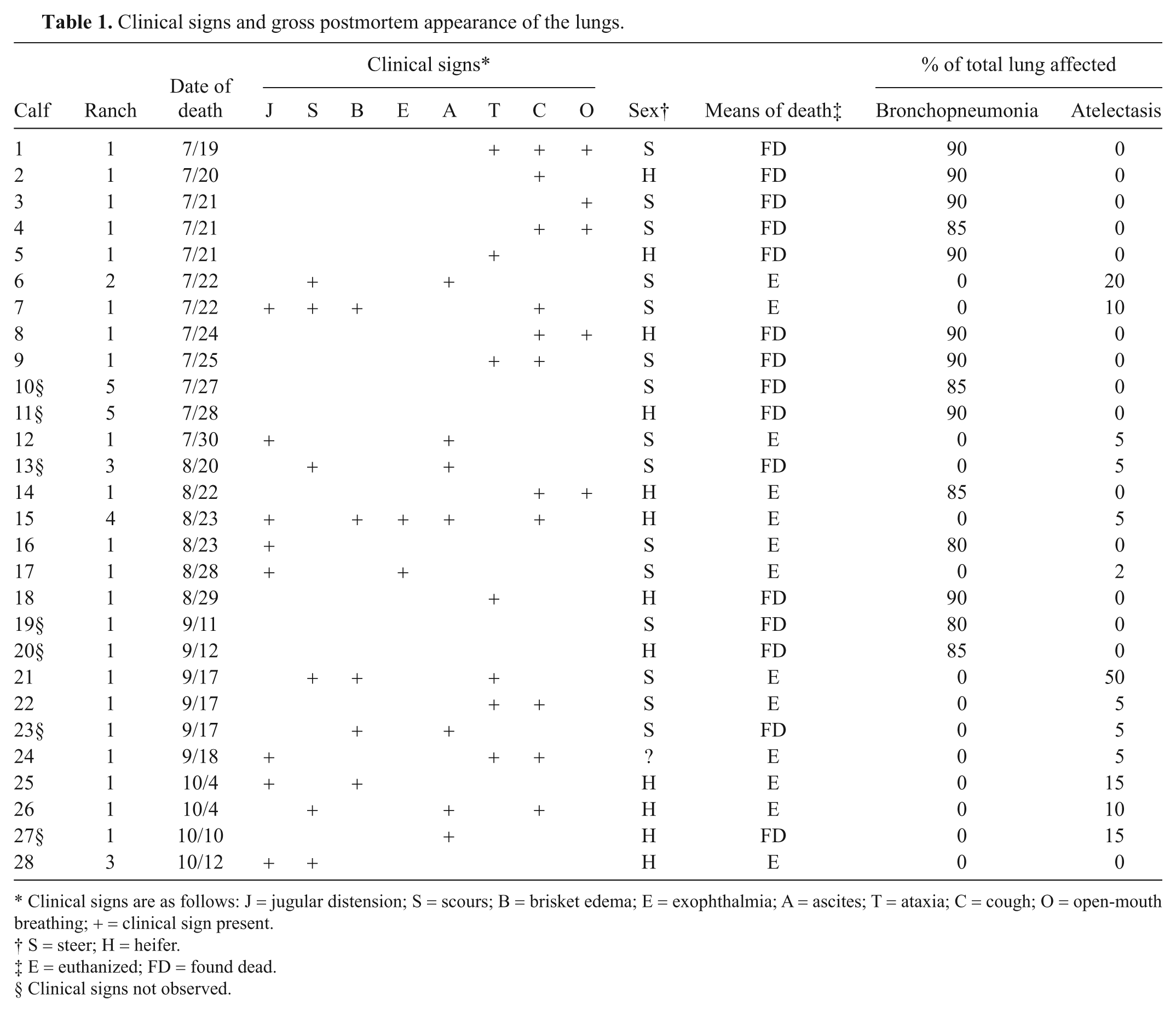
Clinical signs are as follows: J = jugular distension; S = scours; B = brisket edema; E = exophthalmia; A = ascites; T = ataxia; C = cough; O = open-mouth breathing; + = clinical sign present.
S = steer; H = heifer.
E = euthanized; FD = found dead.
Clinical signs not observed.
There were 2 basic patterns of macroscopic changes. One pattern, indicative of pulmonary hypertension, featured combinations of jugular distension; right cardiac ventricular hypertrophy; pericardial, pleural, and/or peritoneal effusion; chronic passive liver congestion; and edema of the mesenteric lymph nodes, colonic mucosa, and subcutis of the brisket area. The lungs of these calves had the normal spongy texture throughout all lobes and were homogeneously pink or mottled red and pink. Regions of atelectasis were common, especially along margins of the middle and cranial lobes (Table 1). Occasional thrombi were present in medium-sized pulmonary arteries.
The other main pattern featured changes indicative of acute or chronic pneumonia. Acute pneumonia was characterized by cranioventral consolidation that usually affected more than 1 lobe and was often associated with fibrinous pleuritis. The affected cut surfaces were diffusely red-black or mottled red and pink. Calves with chronic pneumonia had parenchymal consolidation, pleuritis with fibrous adhesions, and abscessation.
Significant histologic changes were evident in the lungs and are summarized in Table 2. The calves that had gross evidence of heart failure had pulmonary vascular remodeling consistent with pulmonary hypertension. In minimally affected calves, pulmonary arterioles and small muscular arteries had hypertrophy of the tunica media, often accompanied by diminished luminal area (Fig. 1). More severely affected small vessels had adventitial thickening composed of disorderly connective tissue fibers. With VVG staining, it was evident that the adventitia was rich in elastic fibers (Fig. 2). The adventitial proliferation made the small vessels more conspicuous when examined at low magnifications.
Pulmonary lesions in calves dying on summer pasture at high altitude.
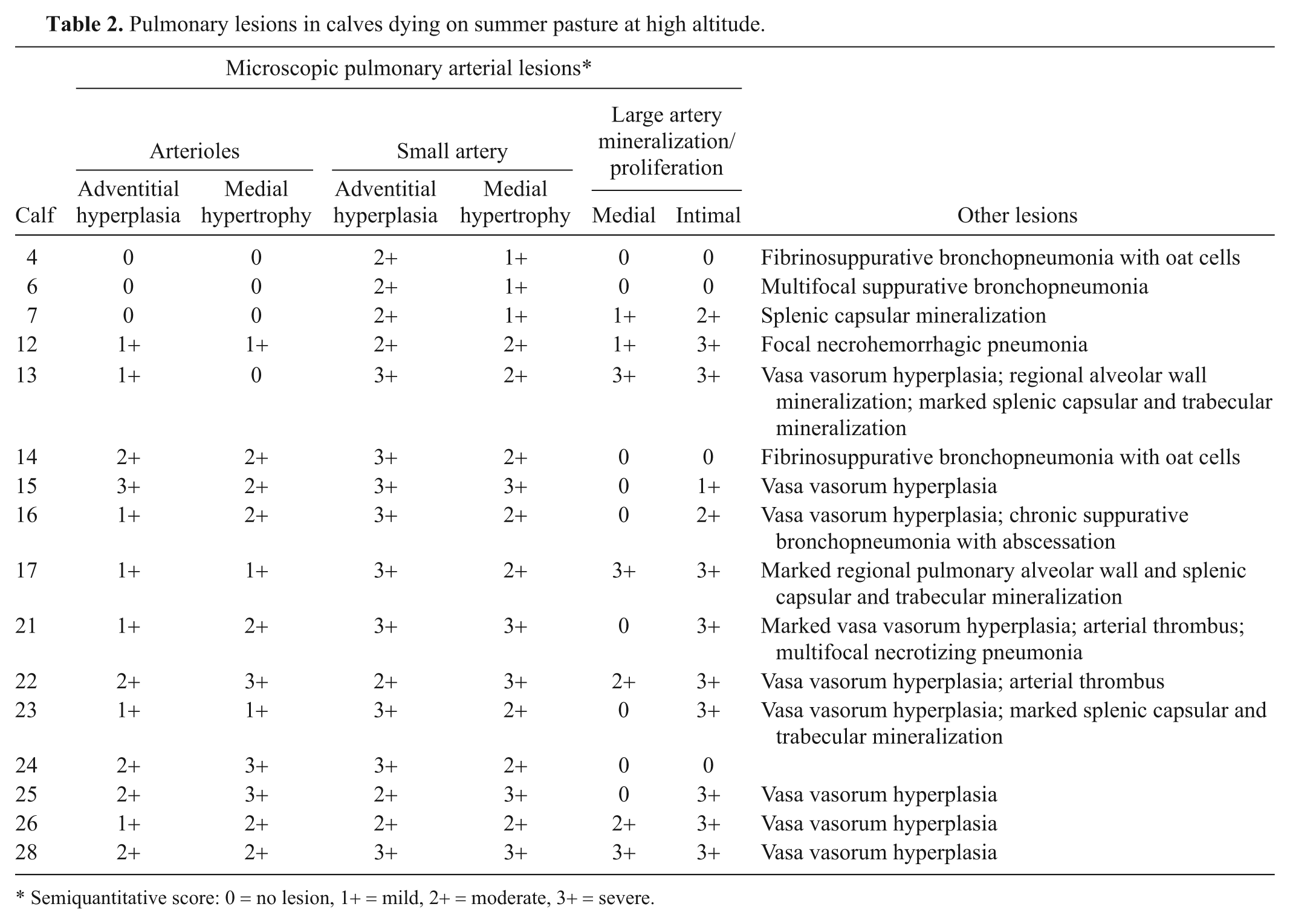
Semiquantitative score: 0 = no lesion, 1+ = mild, 2+ = moderate, 3+ = severe.
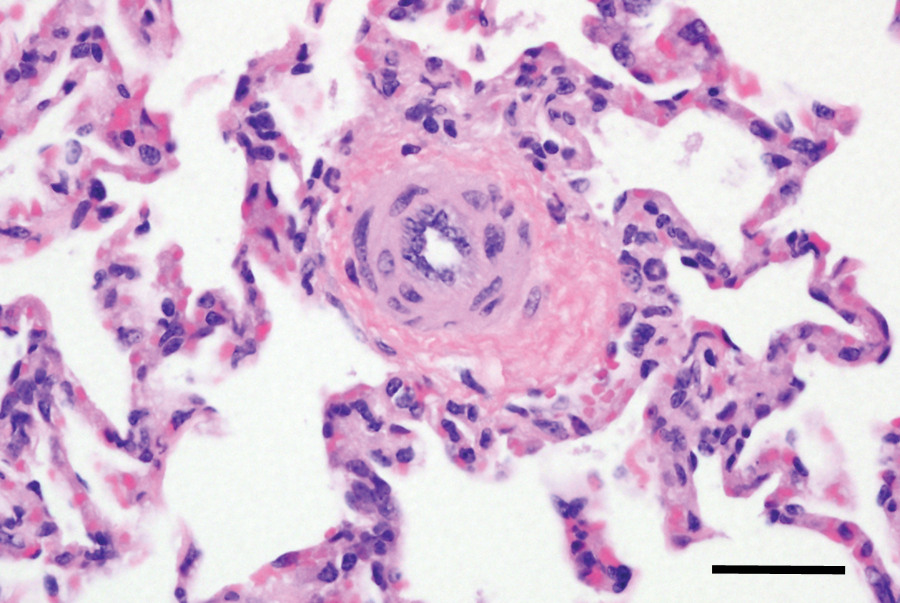
Lung from calf no. 14 with acute pneumonia. Pulmonary arteriole with medial hypertrophy and adventitial proliferation. Hematoxylin and eosin stain. Bar = 40 μm.
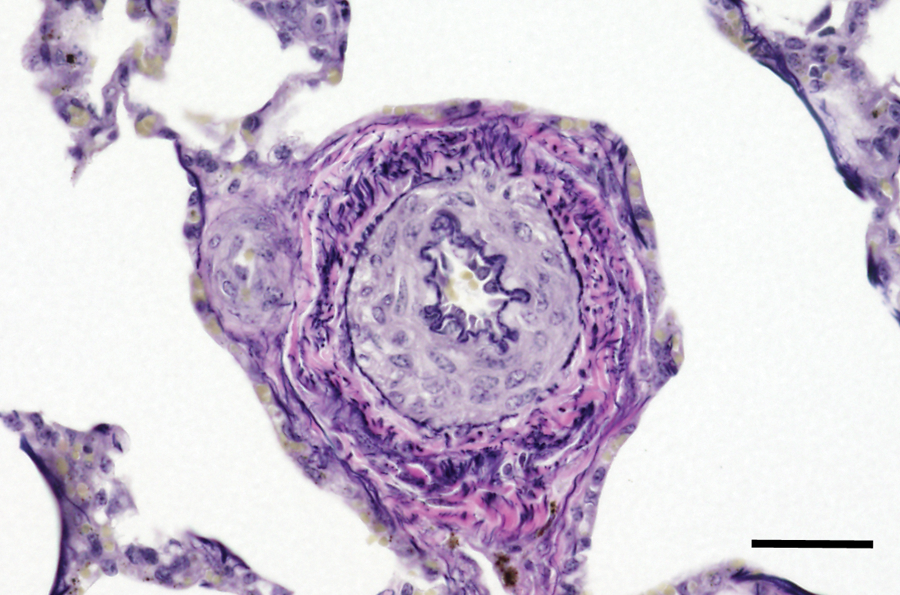
Lung from calf no. 15 without pneumonia but with evidence of right-sided heart failure. Pulmonary arteriole with medial hypertrophy and adventitial proliferation. The thickened adventitia is rich in elastin fibers. Verhoeff–van Giesen stain. Bar = 40 μm.
In calves with more advanced disease, the medium-sized arteries (200–600 μm) had intimal thickening and similar but more severe changes of the tunica media and adventitia (Fig. 3). The adventitia was often thickened up to 60 μm, with increased numbers of elastic fibers. In some of these arteries, the adventitia had prominent small vessels, indicative of vasa vasorum hyperplasia (Fig. 3). More severely affected medium-sized arteries had intimal proliferation characterized by thickening with loose connective tissue containing spindle cells with large, pale, euchromatic nuclei. Occasional mononuclear leukocytes were present. In some cases, the intimal thickening occupied a significant fraction of the luminal area (Fig. 4). Vasa vasorum hyperplasia was sometimes present in the thickened intimal and medial layers.
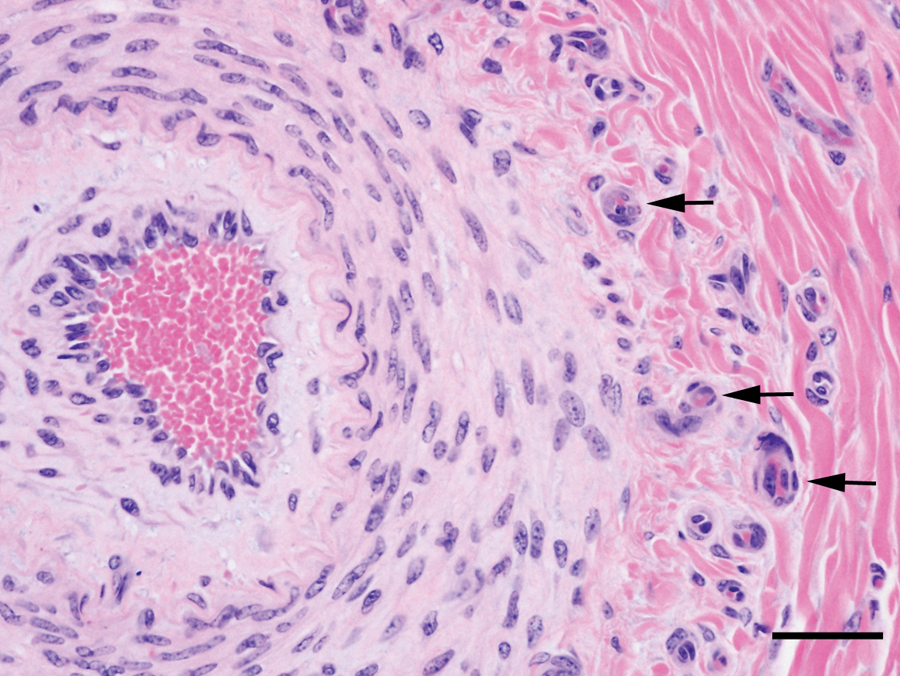
Lung from calf no. 22 without pneumonia but with evidence of right-sided heart failure. Medium-sized pulmonary artery showing intimal and adventitial proliferation and medial hypertrophy. Note prominent adventitial vasa vasorum hyperplasia (arrows). Hematoxylin and eosin stain. Bar = 40 μm.
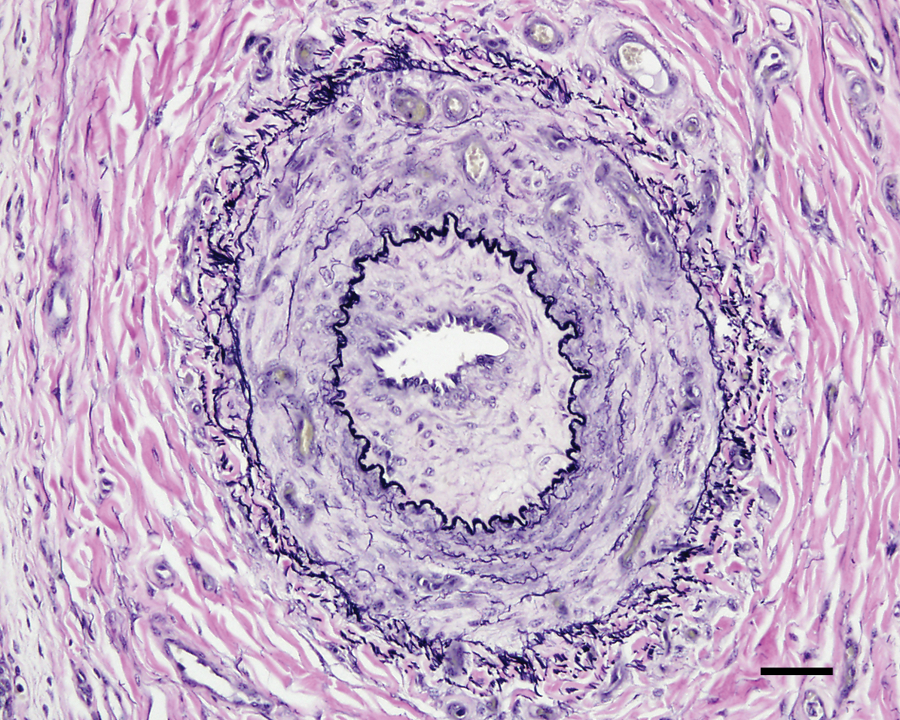
Lung from calf no. 22 without pneumonia but with evidence of right-sided heart failure. Medium-sized artery with intimal and adventitial proliferation, medial hypertrophy, and hyperplasia of the vasa vasorum within the media and adventitia. Verhoeff–van Giesen stain. Bar = 50 μm.
Some large elastic arteries (900–2,000 μm) had segmental areas of mineralization predominately in the intima, but a few had mineralization in the tunica media (Fig. 5). Two calves (nos. 21 and 22) showed thrombi associated with intimal proliferation and mineralization. The same 2 calves were the only calves showing macroscopic evidence of pulmonary thrombi. In addition, no. 21 had a 6-cm thrombus occluding the caudal vena cava at the level of the liver. In some calves, there were regions of pulmonary alveolar wall mineralization without an associated inflammatory response (Fig. 5).
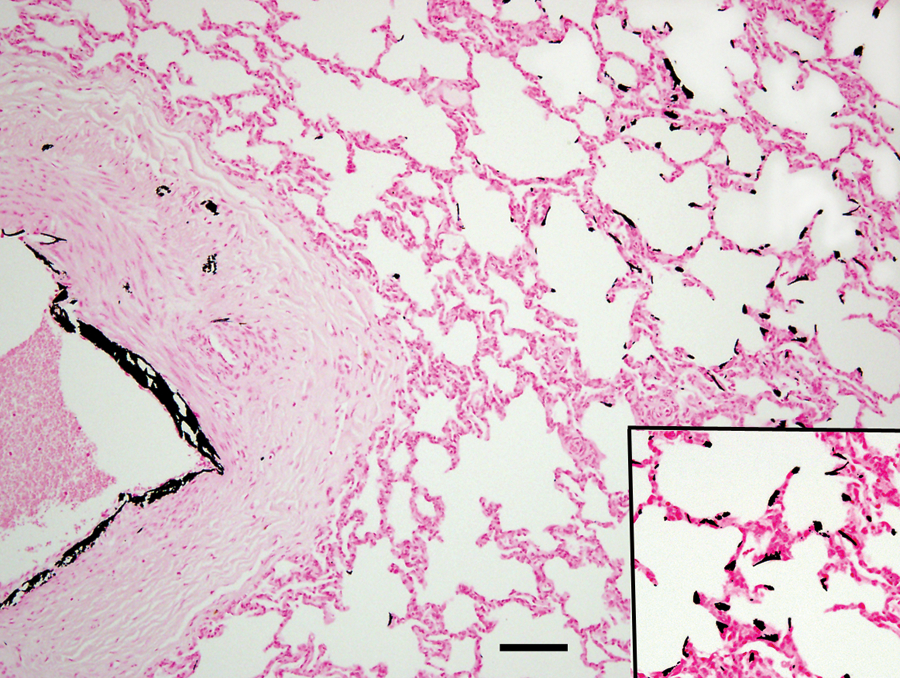
Lung from calf no. 17 without pneumonia but with evidence of right-sided heart failure. Large pulmonary artery with intimal and medial mineralization. Inset: mineralization of alveolar walls. Von Kossa stain. Bar = 100 μm.
Extrapulmonary sites of mineralization were also evident. A few calves had mineralization in the arches of the aorta and pulmonary artery. The mineral was localized to a zone in the outer one-third of the wall. The splenic capsule and trabeculae commonly had multifocal mineralization. A few small foci of mineralization were evident in renal cortical stroma. The livers of calves with evidence of right heart failure had well-developed centrilobular fibrosis indicative of chronic passive congestion.
Tissues from 3 of the 14 calves with macroscopic evidence of pneumonia (Table 1) were evaluated histologically: 2 calves with acute pneumonia and 1 with chronic pneumonia. Lesions consisted of fibrinosuppurative pleuritis, heavy accumulations of polymorphonuclear cells in airways, and varying proportions of polymorphonuclear and mononuclear cells in alveolar spaces. There were some areas of hemorrhage and necrosis. “Oat” cells, indicative of leukotoxin injury of leukocytes, were present in 2 calves. One calf had pulmonary abscesses with heavy fibrous, focally mineralized capsules. All 3 calves showed mild to moderate adventitial proliferation of small to medium-sized pulmonary arteries. The calf with chronic pneumonia and 1 of the 2 calves with acute pneumonia showed medial hypertrophy of the small to medium-sized arteries and arterioles (Table 2, Fig. 1). The calf with chronic pneumonia had intimal mineralization of some of the large pulmonary arteries.
Aerobic culture of lung tissue from 3 calves produced a light to moderate growth of Pasteurella multocida and moderate growth of M. haemolytica. In addition, 1 of the 3 calves showed moderate growth of Arcanobacterium pyogenes. The latter calf had fibrosis and abscessation consistent with chronic pneumonia. Antibody titers to respiratory viruses (BRSV, BVDV, IBR, and BPIV-3) were elevated ≥1:256 in 3 out of 11 calves that died of pneumonia: BRSV (n = 2) and BPIV-3 (n = 1). None of the 8 normal calves had antibody titers to these viruses ≥1:256.
Blood-gas, biochemistry, and complete blood cell count analyses
Blood-gas, biochemistry, and complete blood cell count analyses were performed on the principal study ranch only. Blood-gas analysis was performed on 5 calves with pulmonary hypertension. These calves had significantly lower arterial oxygen pressures than healthy control calves (P = 0.007) and calves with bronchopneumonia (P = 0.003) sampled at a similar elevation (Table 3). Arterial pH was lower for calves with pulmonary hypertension than healthy controls (P = 0.007) and calves with bronchopneumonia (P = 0.009). There were no significant differences in arterial pH or blood-gas tensions between calves with bronchopneumonia and controls (Table 3). Rectal temperature was not significantly different between calves with pulmonary hypertension, bronchopneumonia, and controls. The high blood lactate levels, even in the apparently healthy controls, indicate that a substantial degree of anaerobic respiration is occurring (Table 3).
Selected clinical values obtained from calves on the principal study ranch.
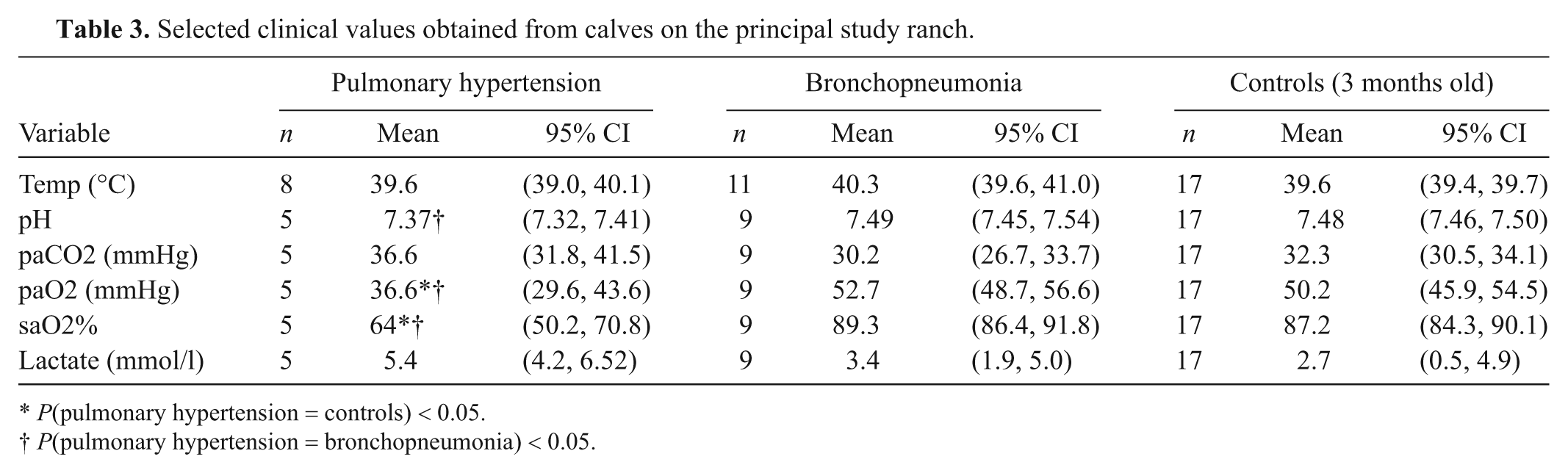
P(pulmonary hypertension = controls) < 0.05.
P(pulmonary hypertension = bronchopneumonia) < 0.05.
Biochemical analysis was performed on 10 calves with pulmonary hypertension and 10 calves with bronchopneumonia. Calves with pulmonary hypertension had significantly lower sodium (130.2 ± 1.4 mmol/l, P < 0.01), chloride (89.0 ± 0.8 mmol/l, P < 0.01), bicarbonate (17.5 ± 1.2 mmol/l), albumin (29.1 ± 1.1 g/l), iron (690 ± 130 μg/l), and total calcium (2.36 ± 0.06 mmol/l) than healthy controls (P < 0.01). Relative to healthy controls, significant elevations were found in total bilirubin (11.3 ± 2.6 mg/l, P < 0.01); the liver enzymes aspartate aminotransferase (AST; 150.0 ± 26.8 IU/l, P < 0.05) and gamma-glutamyl transferase (GGT; 265.2 ± 74.0 IU/l, P < 0.01); creatinine (164.4 ± 17.0 μmol/l, P < 0.01); phosphorus (3.87 ± 0.37 mmol/l, P < 0.05); creatine kinase (1504.6 ± 624.5 IU/l, P < 0.05); and globulin (32.4 ± 2.3 g/l, P < 0.05). Sorbitol dehydrogenase was not significantly increased (46.1 ± 18.7 IU/l) over healthy controls.
Relative to healthy controls, calves with bronchopneumonia, like calves with pulmonary hypertension, showed significantly lower sodium (138.8 ± 1.53 mmol/l, P < 0.01), chloride (97.2 ± 1.58 mmol/l, P < 0.05), bicarbonate (16.5 ± 2.8 mmol/l, P < 0.01), and iron (872 ± 227 μg/l, P < 0.01). Unlike calves with pulmonary hypertension, levels of phosphorus, total calcium, creatinine, albumin, liver enzymes (AST and GGT), total bilirubin, and creatine kinase were not significantly different from controls. Globulin (33.4 ± 1.9 g/l) and glucose (1.8 ± 0.4 g/l) were significantly greater than control values.
Complete blood cell counts were performed on 8 calves with pulmonary hypertension and 10 calves with bronchopneumonia. Mean cell hemoglobin concentration was significantly lower for calves with pulmonary hypertension (369 ± 5.1 g/l) and calves with bronchopneumonia (355 ± 4.8 g/l) than control calves (378 ± 1.5 g/l, P < 0.01). There were no other statistically significant differences in the cellular characteristics of whole blood.
Discussion
Pulmonary hypertension in cattle, known descriptively as “brisket disease,” “high-altitude disease,” “dropsy,” and “fat steer disease,” was first reported to occur 100 years ago at altitudes over 2,134 m (7,000 ft) in Colorado. 10 It was later determined that, in response to chronic alveolar hypoxia associated with high altitude, vasomotor tone initiates and the high ensuing intravascular pressures advances medial hypertrophy. 2 In human beings, pulmonary hypertension syndrome is classified into 5 groups. 23 Alveolar hypoxia–induced pulmonary hypertension is group 3. 23 It is becoming increasingly appreciated that changes in the structure of larger pulmonary arteries induced by chronic hypoxia contribute to right ventricular workload and altered flow dynamics that could contribute to downstream arteriolar remodeling 27 and inflammation. 15 It is evident that although much has been learned regarding bovine pulmonary hypertension, the pathogenesis is far from clear. There may be limitations to PAP testing as a screening tool. Studies using a neonatal bovine model of the human condition are ongoing 28 and demonstrate the true complexity of the syndrome.
It is speculated that, similar to the syndrome in human beings, multiple, but not mutually exclusive, subcategories of bovine pulmonary hypertension may exist. This may explain the apparent change in the clinical signs associated with pulmonary hypertension over the years from the classic, rapid-onset condition producing “belly-draggers” to a slower more insidious-onset pulmonary hypertension associated with vague clinical signs that share similarities with pneumonia as documented in the current study. Other unknown risk factors likely acting in concert with low alveolar oxygen tension may play a greater role than previously thought. This would explain the persistence of pulmonary hypertension in high-altitude herds that have bred only bulls with low PAPs and its occurrence at lower altitudes and in older animals. 16
Despite selection of bulls with low PAP as herd sires, half of the calves examined postmortem over the summer period had lesions consistent with pulmonary hypertension. There are 2 possible explanations for this. First, screening for PAPs in the maternal line was not performed on these ranches. A dam with a high PAP may predispose her calf to developing pulmonary hypertension. However, analysis of herd records over a 5-year period from the principal study ranch found that the risk of failing to wean a calf did not differ among cows. This suggests that screening of dams for PAP may not reduce calf death loss. Second, PAP measurement and screening cannot, by itself, eliminate the occurrence of pulmonary hypertension if the disease syndrome is not solely attributable to alveolar hypoxia. Other, as yet undetermined, risk factors may be acting in concert with, or independently of, alveolar hypoxia. One key risk factor that has a prominent role in the initiation and/or development of pulmonary hypertension is inflammation. 21 Inflammation can be triggered by chronic alveolar hypoxia, 5 as well as infection, 7 vascular injury due to aberrant blood flow, 15 toxins, 12 and autoimmune conditions. 20 Swainsonine contained in locoweed (genera Astragalus and Oxytropis) is a mediator of pulmonary hypertension in calves 13 but was not observed to be a risk factor during the present study.
Due to genetic selection for various production traits, modern cattle have small lungs relative to their body size and metabolic demands, which may increase their risk of cardiopulmonary disease. The alveolar surface area of the bovine lung expressed as a proportion of body weight or per unit of basal oxygen consumption is less than half of the mammalian average. 29 This means that an unusually high demand is placed on the bovine pulmonary system, thus predisposing cattle to respiratory disease and pulmonary hypertension. 29 It has been previously documented that pulmonary arterial pressure is positively correlated with growth rate 25 and therefore metabolic oxygen demand. Further studies are necessary to determine the relationship among oxygen uptake, metabolic demand, and cardiopulmonary disease.
The presence of pulmonary intimal lesions in bovine pulmonary hypertension is not a new finding, 2 but why they occur is poorly understood. Endothelial injury and increased pulmonary blood flow are both necessary for their development.4,19 In systemic-pulmonary shunt studies of pulmonary hypertension, intimal lesions develop early in the course of disease.9,24 Medial and intimal mineralization of pulmonary arteries may occur due to vessel injury. Mineralization of alveoli, splenic trabeculae, and mediae of the renal arteries has been previously documented, 2 but why this occurs is unclear. Plants capable of causing enzootic calcinosis, such as Cestrum diurnum and Trisetum flavescens, were not found on the premises of the principal study ranch in the present study.
Aspects of the blood biochemical and cellular changes associated with pulmonary hypertension have been previously described.1,3 For an unknown reason, low serum iron levels have been associated with pulmonary hypertension in both calves 3 and human beings. 26 Hematinic fortification in the diet of calves for several years did not reduce the incidence of pulmonary hypertension. 3 Hyperventilation, as indicated by hypocapnia and the associated muscular exertion, may diminish the usefulness of rectal temperature as a means to distinguish between infectious and noninfectious causes of disease.
In summary, there is a need to better characterize the pathogenesis of bovine pulmonary hypertension to improve selection tools for producers. The similarity of clinical signs shown by calves with pneumonia and pulmonary hypertension combined with the inability to distinguish disease categories based on rectal temperature presents a considerable diagnostic challenge to producers and may mean that the true prevalence of pulmonary hypertension is underestimated. Historically, the focus of research has been on events in the precapillary arterioles such as development of medial hypertrophy, thought to occur in response to chronic alveolar hypoxia. However, more proximal remodeling in the larger pulmonary arteries may have a greater role in the pathogenesis of bovine pulmonary hypertension than previously appreciated.14,27 Other, as yet unknown, risk factors may dictate the course of disease pathogenesis.
Footnotes
Acknowledgements
The authors would like to thank Barbara East and Bryan Neves for greatly assisting the collection of field samples; Bill Trampe for use of facilities and access to study calves; Dr Elizabeth Frazer for initiating the study and continued support; and Todd Bass for preparation of histopathology specimens.
a.
One Shot Ultra 8, Pfizer Animal Health, Inc, Madison, NJ.
b.
Synovex C, Pfizer Animal Health, Inc, Madison, NJ.
c.
BoviShield Gold 5: IBR, BVD, BRSV and PI-3, Pfizer Animal Health Inc., Madison, NJ.
d.
Cattle feed 12% block with copper, Colorado Agri-Feed, Colorado Springs, CO.
e.
Pyramid 5: IBR, BVD, BRSV and PI-3, Boehringer Ingelheim Vetmedica Inc., St. Joseph, MO.
f.
Draxxin, Pfizer Animal Health Inc., Madison, NJ.
g.
Banamine, Merck & Co. Inc., Whitehouse Station, NJ.
h.
Mobilespin, Vulcon Technologies Inc., Grandview, MO.
i.
VetScan i-STAT 1, Abaxis North America, Union City, CA.
j.
STATA version 12, StataCorp LP, College Station, TX.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Funding for this research came from CSU College of Veterinary Medicine and Biomedical Sciences College Research Council and Integrated Livestock Management producer donations.
