Abstract
Nonlethal sampling is becoming a common method to diagnose fish diseases, especially with the availability of molecular testing. Viral hemorrhagic septicemia virus (VHSV) is a viral pathogen of finfish distributed worldwide. Although VHSV has been known to occur in some parts of the world for decades, a new genotype, IVb, recently emerged in the Laurentian Great Lakes of northeastern North America. Golden shiners (Notemigonus crysoleucas; Mitchill, 1814) and fathead minnows (Pimephales promelas; Rafinesque, 1820) were exposed to VHSV-IVb doses between 102 and 106 plaque forming units per fish by intraperitoneal injection at 10°C. Both species experienced significant mortality after exposure, ranging from 38% to 52% in golden shiners and from 35% to 95% in fathead minnows. In golden shiners, a fin or gill sample was somewhat less sensitive at detecting VHSV-IVb by quantitative reverse transcription polymerase chain reaction (qRT-PCR) than a pooled organ sample (consisting of liver, anterior and posterior kidney, spleen, and heart), however the relative sensitivity increased when a fin and gill sample were tested in parallel. In fathead minnows, a fin or gill sample tested alone or in parallel was relatively more sensitive than a pooled organ sample by qRT-PCR. Specificity was 100% for all sample types in both species. The results suggest that fin and gill biopsies are useful tools to test for VHSV in live fish.
Introduction
Nonlethal sampling methods for virological testing in fish have several advantages including testing of individuals that might be too rare to allow for removal of large numbers from the population, permitting samples to be taken from a single individual over many time points, and benefiting animal welfare by reducing the number of animals euthanized in research. Many researchers have recognized the utility of molecular methods for use in nonlethal or minimally invasive sampling for virological testing of fish and some have begun to compare sensitivities of nonlethal and traditional sampling methods. In rainbow trout (Oncorhynchus mykiss; Walbaum, 1792) infected with Infectious pancreatic necrosis virus, virus was detected from a nonlethal pectoral fin biopsy as early as 24 hr postexposure. 5 Testing blood for Infectious salmon anemia virus using reverse transcription polymerase chain reaction (RT-PCR) was equally as sensitive as testing a kidney sample when Atlantic salmon (Salmo salar; Linnaeus, 1758) were symptomatic and more sensitive when fish were asymptomatic. 13 There was no difference in results between nonlethal (leukocytes or gill swab) and lethal samples (gills or kidney) in carp (Cyprinus carpio carpio; Linnaeus, 1758) tested for Koi herpesvirus (Cyprinid herpesvirus 3) by multiple PCR assays. 4 A nonlethal anterior kidney biopsy was the best method to detect persistent infection of Atlantic Cod (Gadus morhua; Linnaeus, 1758) with Nervous necrosis virus by quantitative RT-PCR (qRT-PCR). 18 Testing a gill biopsy by PCR was equally as effective as lethal sampling methods at detecting White sturgeon iridovirus 4–8 weeks postexposure. 10 For European genotypes of Viral hemorrhagic septicemia virus (VHSV), the use of a blood sample in a nested RT-PCR assay was a more sensitive method than using internal organs 19 ; however, the use of nonlethal sampling in the Great Lakes genotype of VHSV has not been studied.
The rhabdovirus VHSV causes substantial losses of fish worldwide. Its geographic range has expanded dramatically in the past 20 years; most recently, VHSV has been reported in the Laurentian Great Lakes of northeastern North America where it can affect at least 28 species of freshwater fish. 17
Substantial testing has occurred in aquacultured and wild fish to monitor for the presence of VHSV. This testing generally involves lethal sampling of the fish and collection of coelomic organs. Considerable surveillance of wild fish occurred after the detection of VHSV in the Laurentian Great Lakes.3,7,8,12 While the surveillance efforts have shown that the species and geographic range of VHSV in the Great Lakes is wider than previously thought, efforts have been limited in the species tested in part because of the problems associated with removing sufficient numbers of some species due to their importance as a fisheries resource or status as a protected species.
The type of nonlethal sample that is most effective tends to depend on the infection strategy of the pathogen of interest. For example, the greater sensitivity observed in mucus samples when testing for Tenacibaculum maritimum was explained by the observation that the bacterium enters via the mucus. 2 The fish gill provides a large concentration of endothelial cells, and the ability of VHSV to infect the gill has been shown to correlate with virulence. 6 In addition, testing a pooled sample of gill and brain has been shown to be more sensitive than coelomic organs in survivors of laboratory-induced infection with VHSV-Ia in rainbow trout. 21 The fin has been shown to be a route of entry for VHSV and therefore might also be a tissue where VHSV could be detected early in infection. 20 In early studies of the European genotype of VHSV, fin and gill were suggested as promising sample types for VHSV detection. 23 Therefore, in the current study, the sensitivity of fin and gill samples to pooled internal organ samples in 2 experimentally infected species was compared. In addition, experimental infection with generation of clinical signs and mortality of golden shiners (Notemigonus crysoleucas; Mitchill, 1814) exposed to VHSV-IVb was performed as well as a median lethal dose (LD50) dose for exposure of fathead minnows (Pimephales promelas; Rafinesque, 1820) to VHSV-IVb by intraperitoneal injection.
Materials and methods
Animals
Golden shiners a and fathead minnows a were shipped overnight to Cornell University (Ithaca, New York). All fish were certified free of VHSV, Infectious pancreatic necrosis virus, Spring viraemia of carp virus, Aeromonas salmonicida, Yersinia ruckeri, and heterosporosis. In addition, upon arrival, 10 fish of each species were euthanized and necropsied. No significant findings were noted on any fish. and no pathogens were detected. Both species were acclimated for at least 2 weeks to 10 ± 1°C in flow-through tanks. b
Fish were randomly allocated to experimental groups using a computer-generated randomized list (www.random.org). For golden shiners, fish were randomized into 2 experimental groups of 60 fish each and 1 control group consisting of 59 fish. For fathead minnow, fish were randomized into 5 experimental groups and 1 control group consisting of approximately 20 ± 1 fish each. Throughout the experiments, strict biosecurity protocols were maintained to ensure virus was not transferred between tanks of fish or outside of experimental rooms. All use of animals was approved by the Cornell University Institutional Animal Care and Use Committee.
Exposure to VHSV
The MI03 isolate of VHSV was used for all experiments. This isolate falls within genotype IVb, was propagated in epithelioma papulosum cyprini (EPC) cells11,22 stored in HMEM-10 media, 15 and was quantified using standard plaque assay protocols at the time of exposure to fish. Golden shiners received a 0.1-ml intraperitoneal injection of either sterile HMEM-10 cell culture media (controls), 104 plaque-forming unit (PFU)/ml of VHSV (low dose), or 107 PFU/ml of VHSV (high dose). Fathead minnows received a 0.1-ml intraperitoneal injection of either sterile HMEM-10 cell culture media (controls), or 1 of 5 ten-fold dilutions of VHSV ranging from 103 PFU/ml to 107 PFU/ml.
All fish were monitored at least twice daily for mortalities or moribund fish. Upon observation of a mortality or a moribund fish (complete loss of equilibrium and unresponsive to stimuli), the fish was euthanized with an overdose of tricaine methanesulfonate c buffered 1:1 (w:w) with sodium bicarbonate d in water. After the acute phase of mortality ended for golden shiners (49 days), 8 fish from each group were removed, euthanized, and necropsied for VHSV testing every 14 days until the experiment was ended at 89 days postexposure. For fathead minnows, there was not a clear acute phase in all groups, so only moribund or dead fish were removed until the experiment was ended 104 days postexposure.
At necropsy, fish were weighed, and total length was measured. Any gross clinical signs observed externally were recorded, then a small portion of gill and left pectoral fin were removed and immediately placed in a sterile bead-beater tube containing 200 µl of an RNA-stabilizing solution e and a 1.3-mm chrome steel bead f for RNA isolation using instruments disinfected with 20% household bleach (3–6% sodium hypochlorite, final solution approximately 10,000 mg/l active chlorine, contact time at least 30 sec). The fin and gill samples were stored at −80°C until RNA extraction. The fish were then dissected and any grossly visible internal clinical signs recorded. A portion of heart, liver, spleen, and anterior and posterior kidneys were removed and placed in a sterile bead-beater tube containing a 1.3-mm chrome steel bead. The tube was weighed to determine the weight of tissue and a volume of sterile HMEM-10 added to obtain a 1:9 dilution to allow virus isolation in cell culture. The samples were homogenized using a bead beater f for 1 min, immediately used for virus isolation in cell culture as described below, and frozen at −80°C for later RNA extraction.
Virus isolation in cell culture to confirm infectivity of VHSV
Homogenates of pooled organs were centrifuged for 2 min at 500 × g then diluted to 1:49 with HMEM-5 15 and filtered through a sterile 0.45-µm biological filter. g A portion of the filtered homogenate was further diluted to 1:249, and both the 1:49 and 1:249 were plated in triplicate onto 48-well cell culture plates containing confluent monolayers of EPC cells less than 48 hr old for a final in-well dilution of 1:99 and 1:499, respectively. All plates were stored in a 15°C incubator and monitored at least twice a week for cytopathic effects. All plates were passaged at least twice to determine the presence of virus.
Collection of water samples to determine shedding
For golden shiners, water samples were generated by randomly selecting 8 fish from each group weekly for the first 7 weeks and every 14 days thereafter. Each selected fish was placed in an individual container with 525 ml of dechlorinated city water for 1.5 hr to measure viral shedding. After 7 weeks, the golden shiners that were periodically sampled were the same fish used to collect water samples. For fathead minnow, 5 fish from each tank were placed in individual containers as described for golden shiners every 7 days for the duration of the experiment. Approximately 10 ml of water was collected from each container, filtered with a sterile 0.45-µm biological filter, g plated in triplicate by adding 250 µl of water to 48-well plates h with confluent monolayers of EPC cells, which were stored in a 15°C incubator, and monitored at least twice a week for cytopathic effects. The remaining filtered water was stored at −80°C for RNA extraction.
RNA extraction and qRT-PCR testing
Samples were thawed and stored on ice at all times. HMEM-10 was added to fin and gill samples for a 1:9 (w:v) dilution, and homogenization was carried out as described with the internal organs samples above. All samples were centrifuged at 500 × g for 2 min prior to RNA extraction. Extraction of nucleic acids was performed using a magnetic bead extraction system i and the viral RNA isolation kit i using the protocols described in the kit and extraction program AM1836_DW_50_V2. Eluted RNA was immediately placed in sterile microcentrifuge tubes following extraction and frozen at −80°C until use in the qRT-PCR assay.
For VHSV nucleoprotein (N) gene detection by qRT-PCR, samples, a no template control, and at least 3 standards were loaded onto a 96-well PCR microplate j in duplicate. A previously described qRT-PCR was used. 16 Nucleic acid quantity and quality was assessed just prior or just after samples were loaded onto the qRT-PCR plate using a spectrophotometer. k Copy numbers in unknown samples were determined by calculation of a linear regression from the standards and then standardized to 50 ng RNA.
Statistical analysis
Kaplan–Meier survival analysis for both species and LD50 calculations for fathead minnow were carried out in R version 2.10.1 (http://www.R-project.org). McNemar χ2 test was used to determine relative sensitivity of sample types, and Cohen kappa was used to measure concordance between the sample types. Both tests were conducted in OpenEpi (www.OpenEpi.com). Because all fish were exposed to VHSV by intraperitoneal injection, exposure was used as the true infection status.
Results
Golden shiners infected with VHSV started dying on day 2 (high dose) and day 7 (low dose) postexposure. Gross lesions observed in infected fish included petechial hemorrhages on the abdomen, operculae, eyes, flanks, vent, the pectoral fin bases, and within the cranium; pale gill lamellae; exophthalmia; erythema; abdominal distention; inflammation and mottling of the posterior and anterior kidneys, spleen, and liver; hepatic and cardiac pallor; serosanguinous ascites; and lepidorthosis (Fig. 1). Additionally, behavioral changes including erratic swimming, lethargy, and loss of equilibrium were often seen prior to death. Overall mortality in the high and low doses reached 52% and 38%, respectively, by day 49 when periodic sampling was started (Fig. 2). No mortalities were observed in the controls. While there was a significant difference in mortality between all 3 groups (χ2 = 41, degrees of freedom [df] = 2, P < 0.0001), there was no significant difference in overall mortality between the high and low doses (χ2 = 2.6, df = 1, P = 0.108).

Gross pathology observed in golden shiners (Pimephales promelas; Rafinesque, 1820;
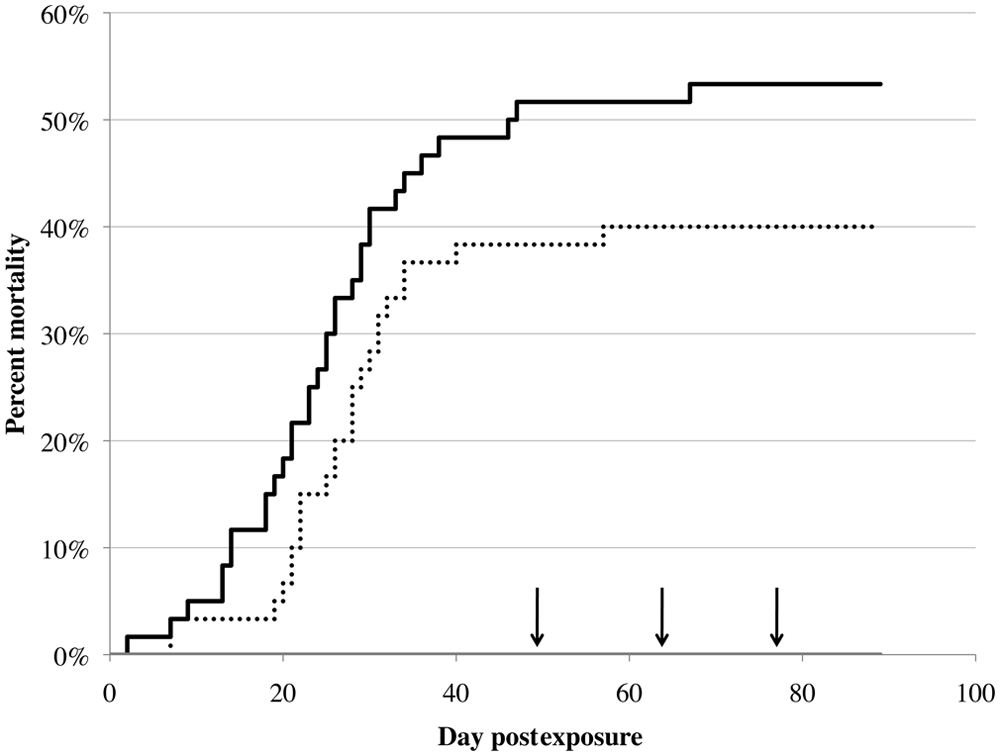
Mortality in golden shiners (Pimephales promelas; Rafinesque, 1820) infected via intraperitoneal injection with Viral hemorrhagic septicemia virus genotype IVb isolate MI03. Sixty fish received 106 plaque-forming units (PFU; solid black line), 60 fish received 103 PFU (dotted black line), and 59 fish received a placebo control (solid gray line). After days 49, 64, and 77, 8 fish from each group were sampled (indicated by arrowheads on the figure). The experiment was ended at day 89 postexposure.
Virus isolation in cell culture was successful in 33 out of 60 golden shiners in the high-dose group and in 32 out of 60 fish in the low-dose group. No virus was isolated from any of the control fish. Virus was detected in at least some water samples by virus isolation in cell culture until day 42, and by qRT-PCR until day 28, and on days 77 and 84 postexposure. When virus was detected by virus isolation in water samples, the proportion of fish shedding ranged from 0.125 to 0.625 and from 0.125 to 0.375 when virus was detected by qRT-PCR.
The relative sensitivity of the nonlethal samples tested compared with the pooled organ samples was significantly lower in golden shiners (Table 1). However, there was significant concordance between the different sample types tested, with fair to moderate agreement seen (Table 1). Specificity for all 3 sample types was 100%.
Comparison of nonlethal and lethal sampling techniques in exposed golden shiners (Notemigonus crysoleucas; Mitchill, 1814).*
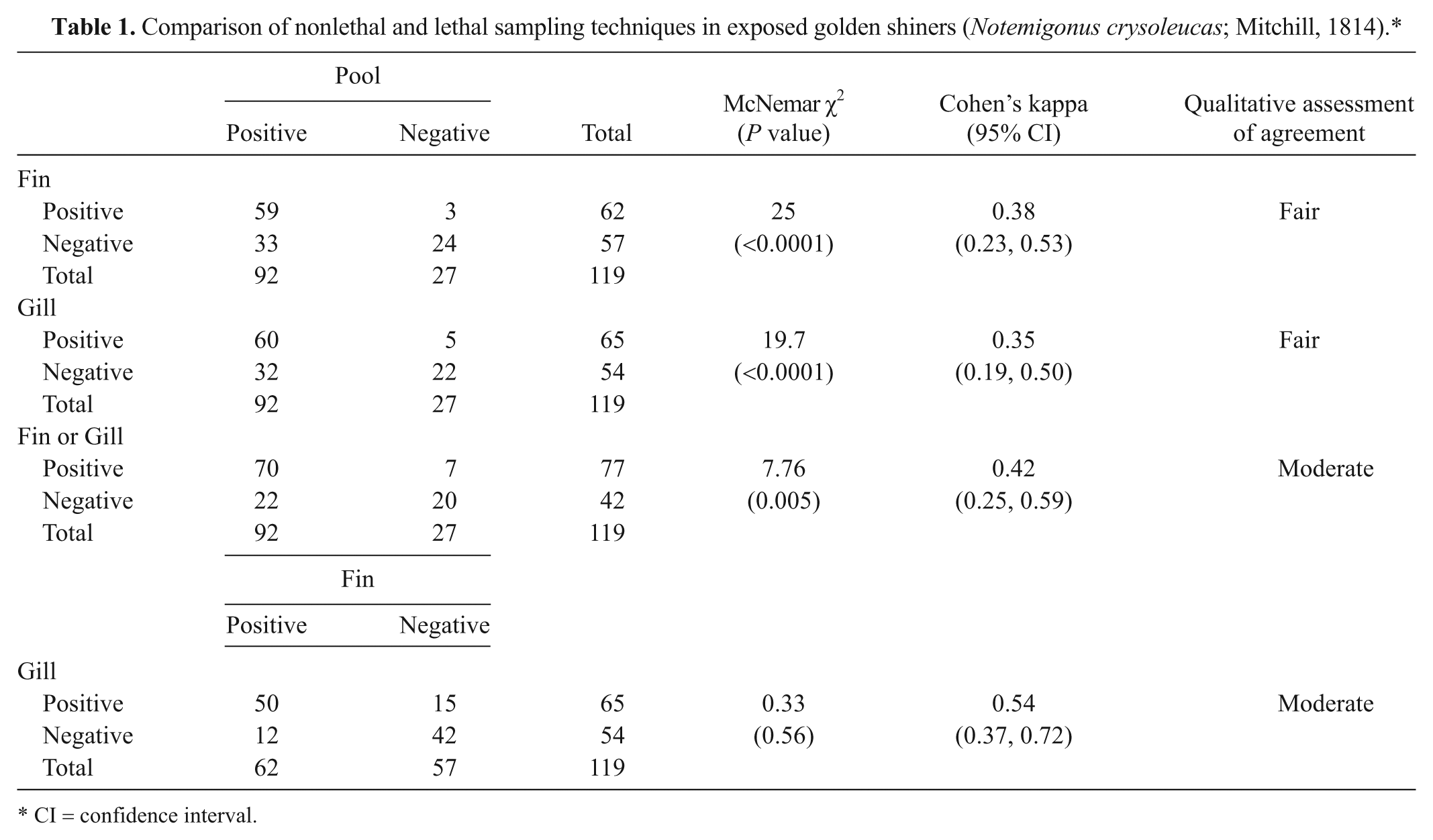
CI = confidence interval.
In fathead minnows, clinical signs observed included petechial hemorrhages or erythema on the flanks, ventrum, all fins, mouth, cranium, operculae, and ovaries; distended abdomen; exophthalmia; enlarged spleen and posterior kidney; hepatic and renal pallor; lepidorthosis; and serous to serosanguinous ascites (Fig. 1). Fathead minnow also displayed behavioral changes such as loss of equilibrium, erratic swimming, and lethargy prior to dying. Mortalities started on day 13 in the highest dose and on day 29 in the lowest dose (Fig. 3). No mortalities were observed in the controls. Overall mortality was dose-dependent and ranged from 35% in the lowest dose to 95% in the highest dose. There was a significant difference in mortality between all groups (χ2 = 93.8, df = 5, P < 0.0001). There was also a significant difference in mortality between the exposed groups (χ2 = 56.2, df = 4, P < 0.0001). The LD50 from this experiment was 553 PFU.
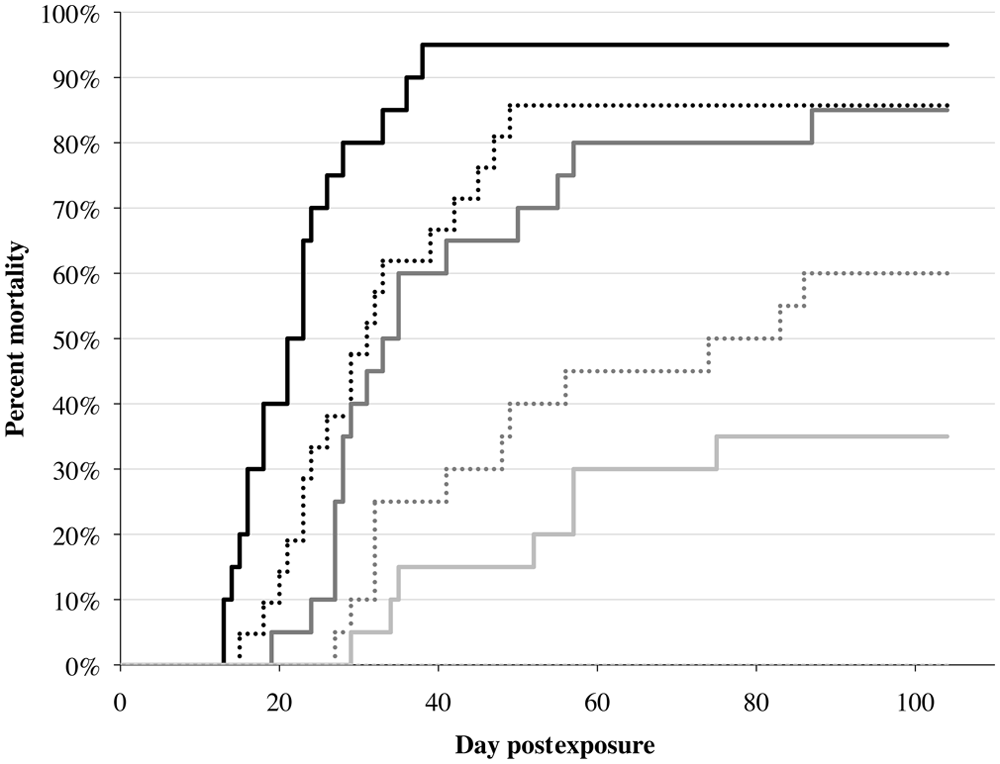
Dose-dependent mortality of fathead minnows (Pimephales promelas; Rafinesque, 1820) infected with Viral hemorrhagic septicemia virus genotype IVb isolate MI03. Fish were exposed to one of 6 doses: 106 (black solid line), 105 (black dotted line), 104 (dark gray solid line), 103 (dark grey dotted line), 102 (light gray solid line), and placebo control (light gray dotted line; no mortalities were seen in the control).
Virus isolation in cell culture for tissues from infected fathead minnow was successful in 89 out of 98 exposed fish. No virus was isolated from any of the control fish. Virus isolation from water samples occurred on days 28–63 and on day 77. Virus was detected in at least 1 water sample by qRT-PCR until day 63, and then also on day 104.
In fathead minnow, the relative sensitivity of a fin sample, a gill sample, and a fin and gill sample tested in parallel was higher than that of a pooled organ sample (Table 2). Concordance between sample types was poor and not significant in most cases except between fin and gill, where it showed significant, fair concordance (Table 2). There was no difference in relative sensitivity between a fin and gill sample. Specificity was 100% for all sample types.
Comparison of nonlethal and lethal sampling techniques in exposed fathead minnows (Pimephales promelas; Rafinesque, 1820).
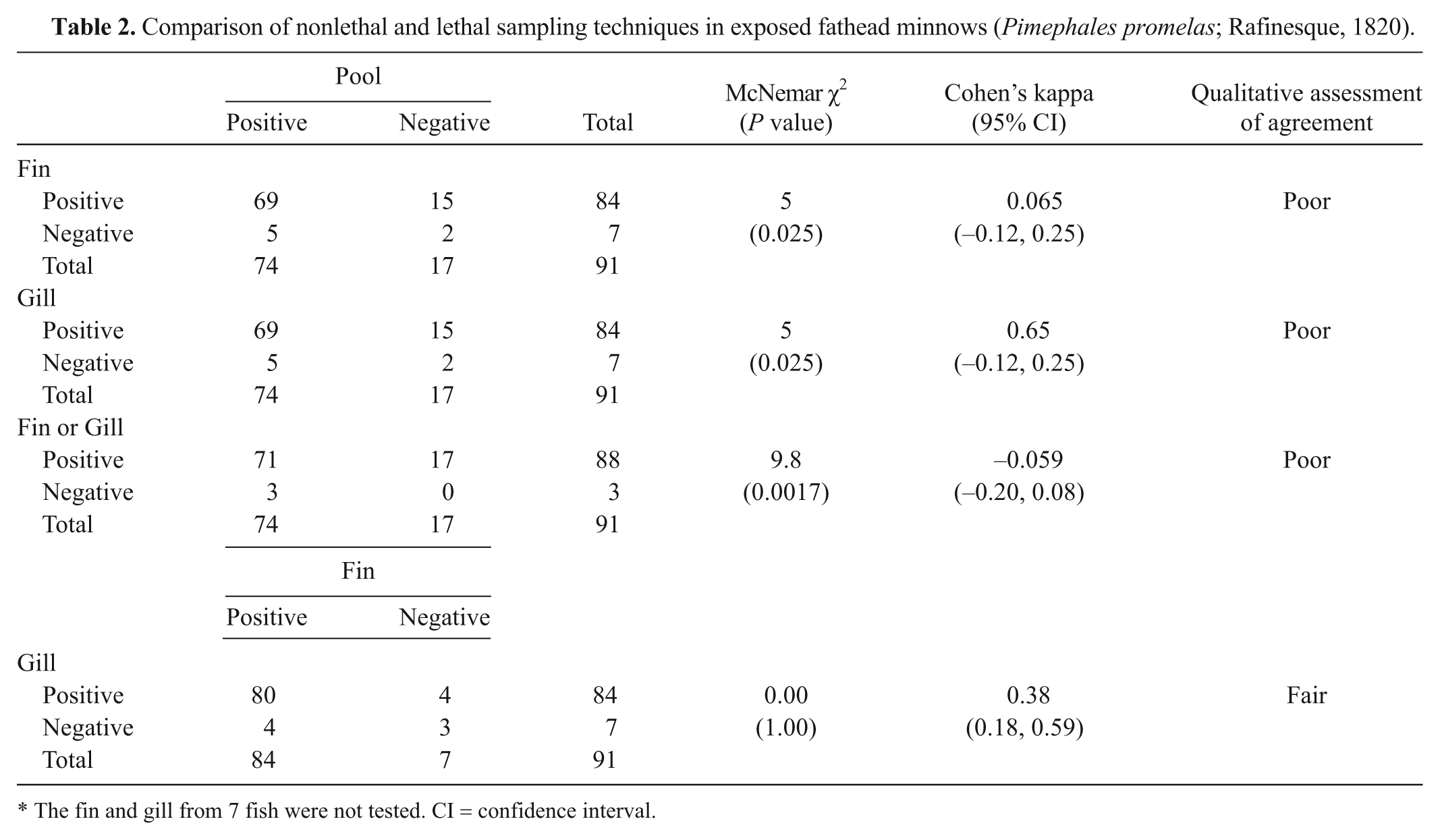
The fin and gill from 7 fish were not tested. CI = confidence interval.
Discussion
Both golden shiners and fathead minnows are susceptible to VHSV-IVb by intraperitoneal injection. Fathead minnows have previously been shown to be susceptible to VHSV-IVb. 1 Both species experienced significant mortality during the trials (Fig. 2, golden shiners; Fig. 3, fathead minnows), showed clinical signs consistent with VHSV infection, and tested positive for VHSV by virus isolation in cell culture and qRT-PCR. Infected fish shed virus at multiple time-points, suggesting the potential for infected fish of both species to spread the virus to other fish.
There was no difference in mortality between the 2 doses used to infect golden shiners; however, there was a significant difference in mortality in the doses used to infect fathead minnows. The calculated LD50 for fathead minnows by intraperitoneal injection was 553 PFU and suggests that this species is moderately to highly susceptible to VHSV. Intraperitoneal injection is likely not representative of the natural route of infection for VHSV in fish and may increase apparent susceptibility because it bypasses the skin, a major source of immunity in fish. 20 However, the use of intraperitoneal injection was necessary in the experiments in the current study so that infection status of each fish could enable calculation of sensitivity and specificity for the different sample types. Additionally, exposing fish by intraperitoneal injection allowed the researchers to be certain that virus traveled from the coelomic cavity to the fins and gills and did not just adsorb from the water onto the fin and gill tissues.
Fin and gill biopsies appear to be acceptable nonlethal samples for testing fathead minnows and golden shiners for VHSV-IVb. In fathead minnows, a fin or a gill biopsy alone or in parallel was more sensitive than testing a pooled organ sample (Table 2). In golden shiners, although a fin or a gill biopsy alone was less sensitive than a traditional pooled sample, the relative sensitivity increased when testing a fin and gill sample in parallel (Table 1). Specificity was high (100%) and equivalent for all sample types in both species. The results also suggest that there may be some differences in viral distribution between species. There did not appear to be a difference in the relative sensitivity of a fin sample versus a gill sample in either species. Because a fin sample is less traumatic than a gill sample, it appears that a fin sample is a better sample to take when using nonlethal sampling techniques.
Fin biopsies have been used to study other genotypes of VHSV with good success. Rainbow trout have been shown to replicate VHSV-I in fin tissues.9,23 Excised fin tissues of herring (Clupea pallasii pallasii; Valenciennes, 1847) can replicate VHSV-IVa. 14 Genotype I of VHSV infects gill tissue in rainbow trout, and the ability of the virus to translocate across the gill epithelium correlates with in vivo virulence. 6
Results of the current study strongly suggest that fin and gill biopsies can be used as an effective nonlethal sample type for VHSV-IVb testing. Because of the very small amount of tissue required for qRT-PCR (<0.05 g), a fish could be tested multiple times during an experiment through fin and or gill samples to track infection in individual fish over time. Although these nonlethal sample types are not a substitute for regulatory testing for VHSV, which has specific requirements for sample types, they will greatly increase the ability to study infection dynamics of VHSV-IVb in the field and laboratory. Additionally, such nonlethal sample types will allow determination of infection status in fish that cannot be euthanized for testing.
Footnotes
Acknowledgements
The authors thank Gregory Wooster for technical assistance.
a.
Anderson’s Minnow Farms, Lonoke, AR.
b.
Living Streams, Frigid Units Inc., Toledo, OH.
c.
Western Chemical, Ferndale, WA.
d.
Sigma-Aldrich, St. Louis, MO.
e.
Ambion RNALater, Life Technologies Corp., Carlsbad, CA.
f.
Minibeater-16, BioSpec Products, Bartlesville, OK.
g.
Acrodisc, Pall Life Sciences, Port Washington, NY.
h.
CoStar-3548, Corning Life Sciences, Tewksbury, MA.
i.
MagMax, Life Technologies, Carlsbad, CA.
j.
Axygen, Union City, CA.
k.
NanoVue spectrophotometer, GE Healthcare, Piscataway, NJ.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Katherine Hambury was supported by the New Visions Life Sciences Program at Cornell University. This project was supported in part by an Assistance Award to Cornell University from Grant 2007.883 made by the Great Lakes Fishery Trust to the Western Fisheries Research Center, United States Geological Survey, Seattle, WA. This report is a resulting product from project R/FTD-11 funded under award NA10OAR4170064 from the National Sea Grant College Program of the U.S. Department of Commerce’s National Oceanic and Atmospheric Administration, to the Research Foundation of State University of New York on behalf of New York Sea Grant. The statements, findings, conclusions, views, and recommendations are those of the author(s) and do not necessarily reflect the views of any of those organizations.
