Abstract
Tyzzer disease (TD) is a highly fatal condition of animals caused by Clostridium piliforme and characterized pathologically by enteritis, hepatitis, myocarditis, and occasionally encephalitis. Cutaneous lesions have been reported only rarely in animals with TD, and infection of the nervous system has not been described in cats, to our knowledge. We describe here neurologic and cutaneous infection by C. piliforme in a shelter kitten with systemic manifestations of TD and coinfection with feline panleukopenia virus. Systemic lesions included necrotizing typhlocolitis, hepatitis, myocarditis, and myeloencephalitis. The cutaneous lesions consisted of intraepidermal pustular dermatitis and folliculitis, with necrosis of keratinocytes and ulceration. Clostridial bacilli were identified within the cytoplasm of keratinocytes by fluorescence in situ hybridization, and a PCR assay was positive for C. piliforme. C. piliforme can infect keratinocytes leading to cutaneous lesions in cats with the location suggesting direct contact with contaminated feces as a route of infection.
Tyzzer disease (TD) is caused by Clostridium piliforme, an anaerobic, filamentous, pleomorphic, spore-forming, and obligate intracellular bacterium that cannot be cultured by routine methods. 12 The condition is frequent in laboratory animals including mice, rats, gerbils, hamsters, guinea pigs, rabbits, and primates. 14 TD affects young domestic animals occasionally, especially foals,2,6,18 and kittens and puppies1,7,9,13; it has been reported rarely in deer fawns, 3 wild rabbits, rodents, and birds.10,11 Although TD is not considered an important zoonosis, cutaneous infection by C. piliforme has been described in an immunosuppressed patient infected by human immunodeficiency virus 1. 16
Predisposing factors for TD in animals include young age, poor hygiene, and immunosuppression related to overcrowding, concomitant viral infection, and treatment with corticosteroids.12,14 In cats with TD, coinfection by feline panleukopenia virus (FPV; Carnivore protoparvovirus 1), felid herpesvirus 1 (FHV1; Felid alphaherpesvirus 1), and feline leukemia virus (FeLV; Gammaretrovirus) is reported frequently.1,9,13 Transmission of C. piliforme is through ingestion of spores from a contaminated environment or feces. 17 The pathogenesis is poorly understood, but it is believed that ingested spores germinate in the intestine, colonize intestinal epithelial cells, and may reach the liver through invasion of capillaries in the intestinal lamina propria and draining into the portal system or within circulating infected macrophages. 17 Once in the liver, bacteria replicate within sinusoidal endothelial cells, Kupffer cells, and hepatocytes leading to lytic hepatocellular necrosis 17 and may disseminate to other organs such as the heart. 19 The characteristic triad of lesions of TD in laboratory animals includes necrotizing hepatitis, enteritis, and myocarditis. 14 Cutaneous infection by C. piliforme has been reported only rarely in animals. 5
An ~45-d-old female domestic shorthair kitten was presented to the referring veterinarian because of paraparesis of unknown duration. This kitten was rescued in Belo Horizonte, Minas Gerais, Brazil and fostered after birth by a Good Samaritan along with its stray dam and 4 kittens from the same litter. The dam and kittens were kept in a foster home with 15 other adult cats. On physical examination, the kitten had a tender abdomen on palpation with decreased pelvic limb proprioception. A complete neurologic exam was not performed. No abnormalities were detected on thoracic radiographs or abdominal ultrasound. A CBC and chemistry profile were not performed. Supportive treatment was initiated, but clinical signs rapidly progressed to paraplegia and diarrhea on day 2 after presentation. On day 3, the kitten had anorexia, nasal discharge, episodes of opisthotonos, and died during the night. After the death of this kitten, the dam and 2 siblings developed respiratory signs that responded to treatment with doxycycline and were forwarded to adoption. Subsequently, 4 unrelated kittens fostered in the same facility developed similar clinical signs of diarrhea and fever, with or without paraplegia, that lasted for ~30 d. Those kittens were euthanized because of lack of response to antimicrobial therapy and were not submitted for autopsy.
On postmortem examination performed by a veterinary pathologist at Universidade Federal de Minas Gerais, the female kitten was in poor body condition with pale mucous membranes. There was mucoid ocular and nasal discharge. Brown-to-green fecal staining was on the ventral aspect of the tail, perianal area, and along the haircoat of the pelvic limbs. A 0.5-cm alopecic area with crusts and erosions was on the medial aspect of the right tarsus. The intestinal contents were diffusely fluid and brown. The epicardium and myocardium contained multifocal-to-coalescing, 2–7-mm diameter, tan to pale-yellow, slightly raised areas. On cut surface of the brain, a 3 × 2 × 2-cm pale-yellow soft area was within the white and gray matter of the right frontal cerebrum, and a similar 1 × 1 × 2-cm area was in the right occipital lobe.
Histologically, the skin lesion on the right tarsal area consisted of pustular dermatitis and folliculitis. Panepidermal pustules containing degenerate neutrophils along with intense neutrophilic exocytosis were seen throughout the epidermis (Fig. 1A, 1B). The stratum spinosum was acanthotic, and the stratum corneum was expanded by moderate orthokeratotic hyperkeratosis. The epidermis was eroded multifocally and ulcerated focally. The lumen and the wall of hair follicles often contained intact and degenerate neutrophils (Fig. 1C) with rare furunculosis. Occasionally, keratinocytes had undergone hydropic degeneration or were hypereosinophilic and shrunken with karyorrhectic nuclei. On Warthin–Starry stain, numerous bundles of argyrophilic bacilli were seen in the cytoplasm of keratinocytes of the epidermis and hair follicles (Fig. 1D). Fluorescence in situ hybridization utilizing a probe targeting Clostridium spp. 8 resulted in positive labeling in the cytoplasm of keratinocytes (Fig. 1D inset). A Gram stain revealed a few gram-positive cocci in the stratum corneum. Periodic acid–Schiff stain was negative, and mites or other parasites were not identified.
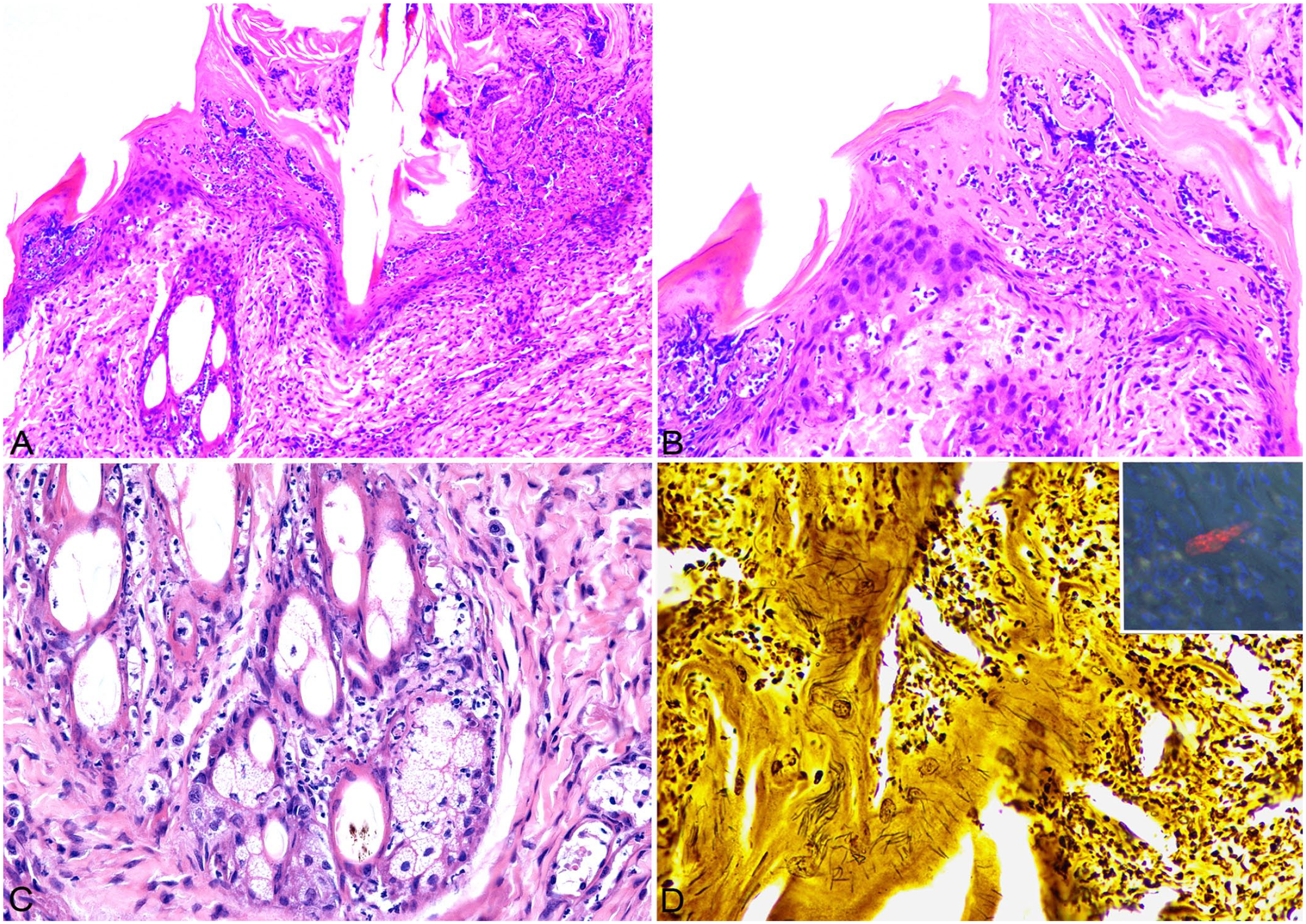
Pustular dermatitis associated with Clostridium piliforme infection in a kitten.
Intestinal lesions consisted of necrotizing typhlocolitis. The mucosa of the cecum and colon had erosions and occasional ulcers extending into the submucosa characterized by necrotic cellular debris and degenerate neutrophils. Multifocally, crypts were dilated and contained neutrophils and necrotic cells. The submucosa and muscularis layers were infiltrated by macrophages, neutrophils, and fewer lymphocytes and plasma cells. Severe lymphoid depletion was noted in the gastrointestinal-associated lymphoid tissue of the ileum and in lymphoid follicles of the spleen and mesenteric lymph nodes. The duodenum and jejunum were unaffected.
The lumbar segment of the spinal cord (Fig. 2) and the telencephalon (Fig. 3) had multifocal-to-coalescing necrotizing and neutrophilic myeloencephalitis. Lesions were characterized by subacute necrosis and neutrophilic-to-lymphoplasmacytic inflammation affecting both the white and gray matter, with a predominance of gray matter lesions in the spinal cord. The liver had random foci of coagulative-to-liquefactive necrosis with occasional neutrophilic inflammation (Fig. 4). In the heart, severe lymphoplasmacytic, histiocytic, and neutrophilic inflammation effaced most of the myocardium and extended into the endocardium and pericardium (Fig. 5). Remaining cardiomyocytes were often degenerate or necrotic.
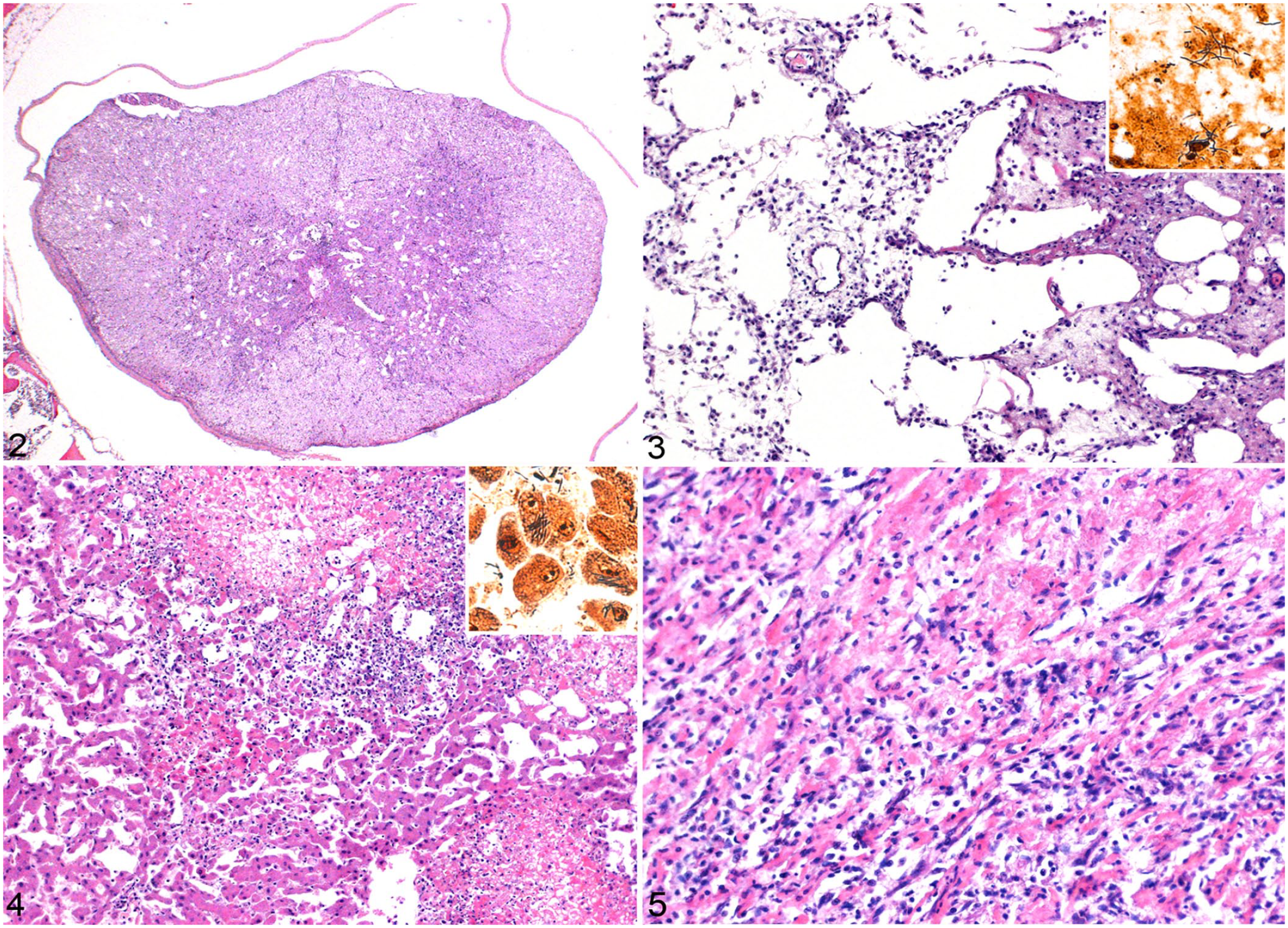
Tyzzer disease in a kitten.
On a Warthin–Starry stain, bundles of long argyrophilic bacilli were seen extracellularly and within the cytoplasm of colonocytes, neurons (Fig. 3 inset), hepatocytes (Fig. 4 inset), and cardiomyocytes. Gram stain results were variable. In neurons, hepatocytes, and colonocytes, most of the intracellular long bacilli detected with Warthin–Starry were gram-positive with occasional intracellular gram-negative bacilli observed. Bacteria were not detected in the heart with the Gram stain. Other infectious organisms were not identified on Giemsa or periodic acid–Schiff stains.
Frozen samples from ileum, colon, liver, and heart as well as formalin-fixed, paraffin-embedded sections from skin, brain, spinal cord, lung, and nasal cavity were submitted for DNA extraction. Conventional PCR for C. piliforme was performed on DNA samples extracted from skin, liver, heart, intestines, brain, and spinal cord, as described previously. 3 All of the samples, except for the spinal cord, were positive for C. piliforme. The PCR product was submitted for sequencing, and ~270 bp were aligned with sequences in GenBank. The search resulted in a match to C. piliforme with 100% cover and 100% identity. A sample of ileum tested positive for FPV by real-time PCR (rtPCR) performed at a reference laboratory (TECSA, Brazil). FHV1 was not detected by a rtPCR assay of samples from the nasal cavity and lung. FeLV proviral DNA and feline immunodeficiency virus were not detected in gut content by a rtPCR assay.
The histopathologic findings, PCR results, and intracellular identification of clostridial organisms by special stains and fluorescence in situ hybridization confirmed the diagnosis of systemic TD in this kitten. Perianal ulcerative dermatitis with intraepithelial Steiner-positive stacked rods in epidermal keratinocytes and follicular epithelium was seen in 2 of 19 kittens with TD. 5 Localized cutaneous infection by C. piliforme has been reported in an immunosuppressed human patient infected by human immunodeficiency virus. 16 The person had a group of verrucous, crusted, tender papules on the skin of the chest that resolved after surgical resection. 16 The lesions appeared ~2 wk after staying in an apartment with a rodent infestation. 16 The histologic findings were similar to those in our case and included epidermal acanthosis and hyperkeratosis with pustules and intracellular bundles of bacterial rods, identified as C. piliforme by PCR and sequencing. 16
The most likely portal of entry for the cutaneous infection in this kitten was direct contact with contaminated feces in the environment or via penetration through a cutaneous wound. The neurologic infection led to paraparesis and dragging of the limb, which could have resulted in abrasions in the hindlimbs that predisposed keratinocytes to infection by C. piliforme through direct contact with spores in the hair coat or environment. Unfortunately, the clinical evolution of the cutaneous lesion is unknown. Although the kitten had systemic lesions and C. piliforme is believed to disseminate through infected macrophages, 20 the hematogenous route of skin infection is considered unlikely in our case given the localized lesion and infection of epithelial cells. Superficial pyoderma caused by Staphylococcus spp. is the main differential diagnosis for our case. A few gram-positive cocci were seen in the superficial corneal layer. Although these might have contributed to the lesion, they were considered a secondary finding given the low abundance of superficial cocci in comparison with the numerous intracellular clostridial rods.
In cats and dogs, TD is often reported in immunosuppressed patients with concomitant or previous viral infection, including FHV1 or FPV in cats, and canine distemper virus in dogs,7,9,13 both of which viral infections result in lymphoid depletion, lymphopenia, and immunosuppression, predisposing to secondary infections.7,9 In experimental animal models, administration of corticosteroids can result in significant replication of C. piliforme and clinical disease in infected mice. 19 In our case, the severe lymphoid depletion in the spleen, mesenteric lymph nodes, and Peyer patches was associated with infection by FPV, confirmed by PCR. Both C. piliforme and FPV can result in necrotizing typhlocolitis as seen in our case. It is unclear if previous or concomitant infection by intestinal pathogens could predispose to intestinal colonization and invasion by C. piliforme. Additionally, overcrowding has been established as a predisposing factor for the infection in mice as a result of stress leading to immunosuppression, and an environment contaminated with high bacterial loads 15 that could have contributed to disease onset in our case. The other kittens from the foster home that died may also have had TD and/or feline panleukopenia. However, no testing was attempted on those kittens, thus other causes of death cannot be excluded.
The neurologic signs were attributed to the necrotizing and suppurative encephalomyelitis affecting mostly the telencephalon and lumbar segment of the spinal cord. Infection of the CNS is reported uncommonly in TD, except for gerbils, in which encephalitis is a frequent lesion. 20 C. piliforme has been also described as the cause of neurologic signs and death from encephalitis in free-ranging passerine birds 11 and in a newborn marmoset. 21 We retrieved no cases of neurologic TD in cats in a search of Google, PubMed, CAB Direct, Web of Science, and Scopus, suggesting that no descriptions of this lesion have been reported in cats.
Although C. piliforme is more often described as a gram-negative bacillus in tissues, this bacterium can be gram-positive, as seen in our case. 4 C. piliforme DNA was detected in all tissues tested, except for FFPE samples of spinal cord. In this kitten, the spinal cord was collected with intact vertebrae, fixed in formalin, and demineralized with formic acid solution for trimming and histologic processing. Demineralizing solutions are known to affect the integrity of DNA, and this is the most likely explanation for the negative PCR result in the spinal cord. The presumed route of infection of the CNS is suggested to be through infected macrophages and, in some cases, encephalitis is the only detected lesion.11,21 The hyperacute clinical course resulting in death by bacteremia, myocarditis, and hepatitis is the most likely explanation for the uncommon infection of the CNS in TD. 21 In our case, the myocardial lymphoplasmacytic infiltrate in the heart and gitter cells in the brain suggest a subacute-to-chronic clinical course. In foals with TD, neurologic signs have been associated with hepatic encephalopathy. 6
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
