Abstract
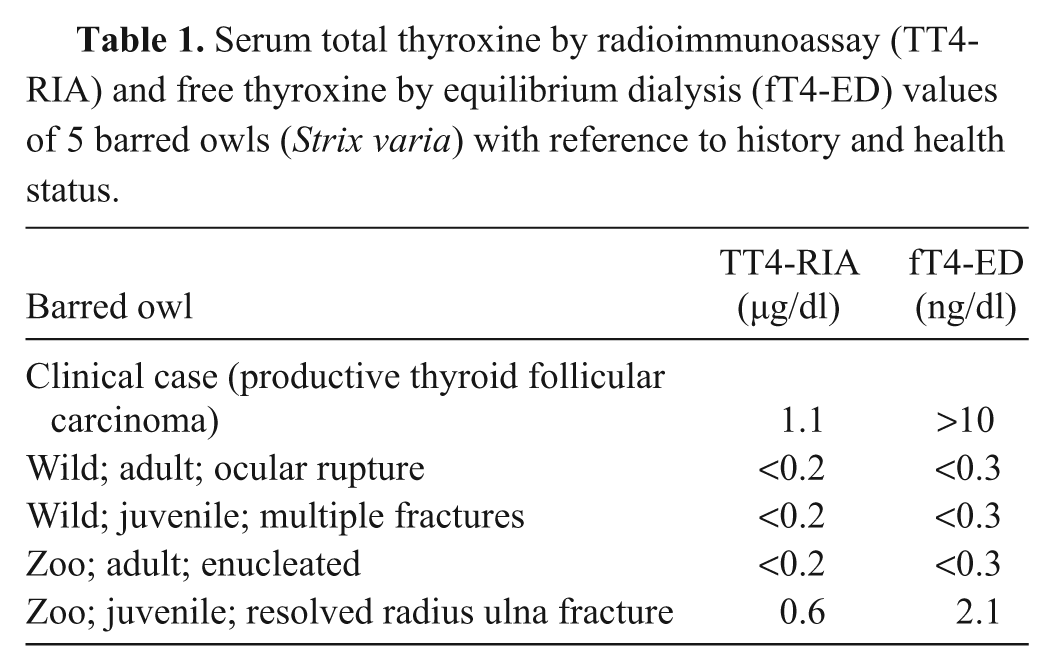
An adult male barred owl (Strix varia) was found unable to fly on a pasture during the day. On presentation, several lacerations were noted on the left wing. The animal was anesthetized for radiographic examination, which revealed mild swelling and irregularity of the soft tissues of the left wing. Over the plane of the syrinx and great vessels, ill-defined soft tissue opacity was present. The anesthetic recovery was unsuccessful, and the patient died. On gross necropsy, a 1 cm in diameter, round, soft, red-tan nodule, with scattered light tan to white foci was noticed between the right subclavian artery and the syrinx. The histopathology of this structure was characteristic of a thyroid follicular carcinoma. Neoplastic cells were immunoreactive to thyroglobulin and pancytokeratin proteins. A blood sample, taken antemortem, was analyzed for total and free thyroxine. Due to the lack of reference intervals for the current species, 4 blood samples from other barred owls were taken, 2 of which were clinically normal and 2 with an unhealthy status. The thyroid values were higher than the controls (total thyroxine by radioimmunoassay [µg/dl] 1.1 vs. <0.2, <0.2, 0.6, <0.2; free thyroxine by equilibrium dialysis [ng/dl] >10 vs. <0.3, <0.3, 2.1, <0.3). Although the other 4 birds are not intended to serve as a reference interval because of the low number and unhealthy status, findings are indicative of a productive thyroid follicular carcinoma.
Neoplasias affecting the thyroid gland have been described in avian species and are generally diagnosed at necropsy. Although the prevalence of thyroid neoplasia in avian species is undetermined, it appears to be low. In a retrospective study from the Philadelphia Zoo, among 1,811 birds submitted for necropsy between 1901 and 1965, no thyroid neoplasias were detected. 25 In poultry, thyroid neoplasias are suggested to be extremely rare; however, thyroid adenomas and adenocarcinomas have been reported in chickens.12,19 Among thyroid neoplasms, follicular adenoma has been suggested to be the most common neoplasm in pet birds. 1 Other tumor types affecting the thyroid gland have also been described, including a thyroid cystic fibroadenoma in a black-chested buzzard-eagle (Geranoaetus melanoleucus), and a thyroid follicular cystadenoma in a crested caracara (Caracara sp.).6,23
Although histopathological features may be characteristic of a thyroid neoplasm, it does not assess how productive the gland is. The use of immunohistochemistry for the diagnosis and prognosis of thyroid neoplasias has been described both for human and veterinary medicine.3,16-18
Among avian species, other thyroid pathologies like hypothyroidism and goiter may occur. To the authors’ knowledge, hyperthyroidism with or without gland hyperplasia has not been reported in avian species. Goiter is suggested to be the only well-documented thyroid disorder in birds, which may be a consequence of neoplasia or iodine deficiency. 12 Goiter appears to be overrepresented in certain breeds of pigeons and budgerigars. 12 Thyroid disorders in raptors are rare although hypothyroidism (due to dietary iodine deficiency) has been suspected in the white-backed vulture (Gyps africanus) and southern crested caracara (Caracara plancus). 11
Several laboratory methods to measure thyroid hormones are available: radioimmunoassay (RIA), equilibrium dialysis (ED), modified equilibrium dialysis, and tandem mass spectrometry.22,26 Due to the low physiological values of total thyroxine (TT4) and lack of laboratory testing sensitivity to low values, diagnosis of hypothyroidism based on TT4 is of questionable value.7,9 A TT4 measurement method using a modified free thyroxine (fT4) RIA has recently been validated.9 The sensitivity of this test is reported to be 3 times greater than the solid-phase total thyroxine kit, which allows accurate measurement of low TT4 values.9 The test, to the authors’ knowledge, is no longer commercially available. It is suggested that fT4 has higher plasma concentrations in birds than in mammals when measured by ED or by column chromatography.13 When measured by RIA, fT4 and free triiodothyronine values were similar to reference intervals in mammals.13,14 Free thyroxine levels are less affected by protein concentrations and concomitant diseases. Such testing, in conjunction with TT4, allows identification of euthyroid sick syndrome.24 The current gold standard for fT4 measurement is ED because it is less affected by thyroxine-binding protein.15,26 The current report describes a case of a productive thyroid follicular carcinoma in a wild barred owl (Strix varia), and the use of TT4 and fT4 as well as immunohistochemistry for diagnosis.
An adult male barred owl was found on the ground and unable to fly. On presentation, the patient was quiet but responsive. The body condition score was good (5/9), with a body weight of 540 g. Cardiac and respiratory auscultation was unremarkable. On initial assessment, multiple deep lacerations surrounded by hematoma were noticed on the left patagium, humerus, and radius-ulna area. The underlying bones were visible through the lacerations but no orthopedic fractures were noticed. The remainder of the physical exam, including ophthalmological evaluation, was unremarkable.
Whole body radiographs were performed with the patient under general anesthesia. The patient was physically restrained for anesthetic induction via face mask with isoflurane. Once anesthetic induction was achieved, the patient was intubated with a non-cuffed endotracheal tube (3.0 Fr) and maintained with isoflurane. Radiographically, there was mild swelling and irregularity of the soft tissues of the left brachium and antebrachium. These findings were consistent with edema, hemorrhage, and/or cellulitis, and consistent with the grossly identified wounds. Additionally, over the plane of the syrinx and great vessels, an ill-defined, 1.2 cm × 0.9 cm, angular, soft tissue opacity was present on the right side (Fig. 1). Due to axial rotation present on the radiographs, the primary differential for this finding was an atypical appearance of the great vessels secondary to obliquity. A soft tissue nodule such as a granuloma or neoplasm was considered less likely.
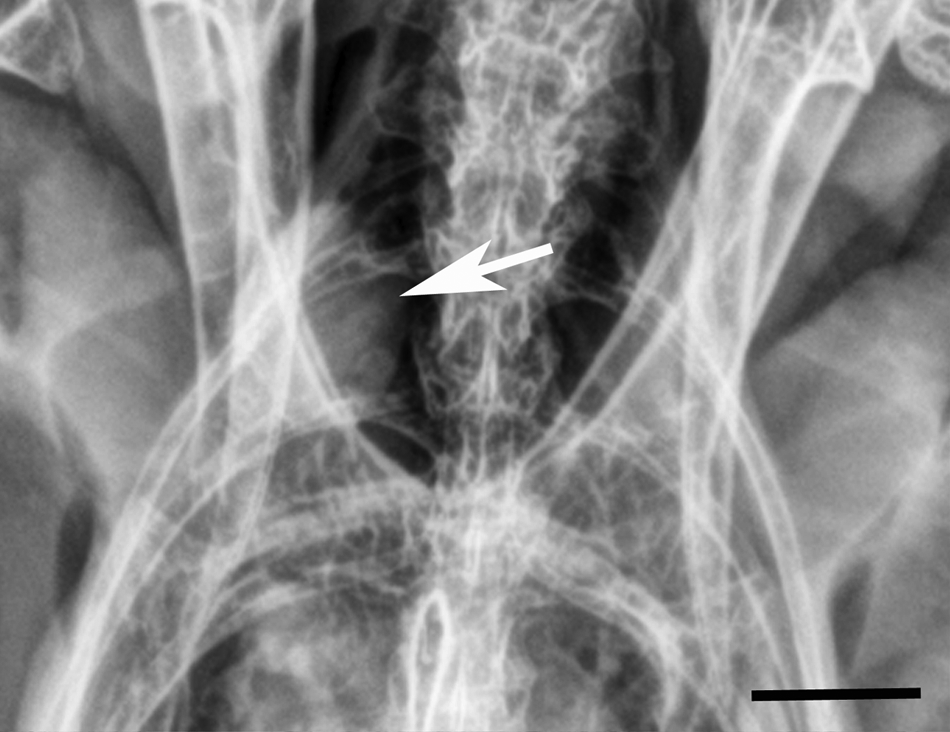
Thyroid gland; barred owl (Strix varia). Radiograph illustrating poorly defined, soft tissue opacity in the area of the right thyroid gland (arrow). Bar = 1 cm.
Following the radiological exam, the lesions were cleaned and a figure “8” bandage was applied to the wing. During the entire anesthetic episode, the patient demonstrated spontaneous respiration, and capnography measurement remained between 20 and 30 mmHg. Once treatments were concluded, a 25-ml subcutaneous bolus of lactate ringer solution (LRS) was given, and isoflurane was discontinued. Oxygen supplementation was continued, but over the course of approximately 5 min the patient ceased to exhibit spontaneous ventilation. Emergency procedures included intermittent positive pressure ventilation (IPPV), an intraosseous catheter, and continuous rate infusion of LRS and hetastarch (4.5 ml/hr and 5.4 ml/hr, respectively). Based on the history and abnormal anesthetic recovery, traumatic brain injury was suspected, and empirical treatment with mannitol (1 mg/kg, intraosseous infusion, slow bolus) was administered. A blood sample was collected from the right jugular vein. Blood glucose concentration, measured with a glucometer a was 550 mg/dl (normal: 297 ± 84), and packed cell volume was 33% (normal: 40.3 ± 4.5). 10 Remaining blood was placed in a lithium heparin tube b without separator. Sample was centrifuged within 5 min of collection, and plasma was preserved at −80°C for future diagnostic tests. The patient did not respond to resuscitative efforts, and IPPV was discontinued after 45 min. The patient went into cardiac arrest and died. Necropsy was requested shortly after and was performed on the same day.
On gross necropsy, the patient nutritional status was fair with mild postmortem autolysis. The skin and soft tissues of the left wing were bruised with multiple puncture wounds on the patagium. The humerus, radius, and ulna were exposed with hematoma on the underlying muscles. The beak was covered with a small amount of blood (<10 ml). Between the right subclavian artery and the syrinx, and associated with the right internal carotid artery, there was a 1 cm in diameter nodule in the region of the right thyroid. The nodule was round, soft, reddened, with widely scattered light tan-white foci (<1 mm in diameter; Fig. 2). Left thyroid was not recognized. Although not considered significant, additional findings included few hepatic granulomas, severe testicular tubular degeneration and necrosis, and an epididymal granuloma with intralesional nematode. The remaining necropsy was unremarkable.
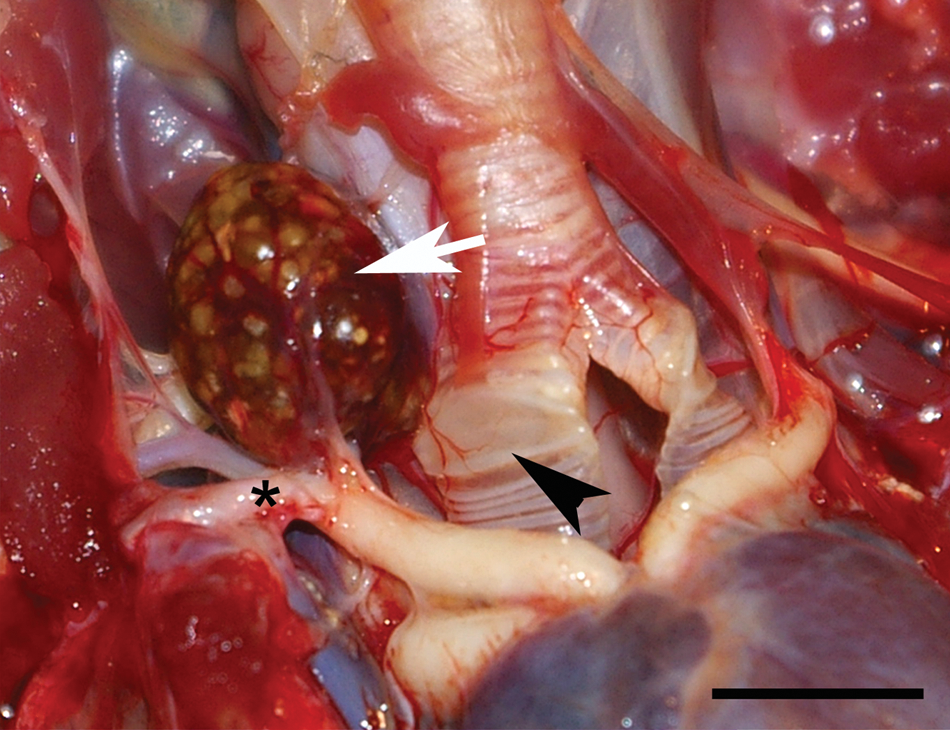
Thyroid gland; barred owl (Strix varia). Necropsy image of the nodule radiographically depicted in Figure 1 (arrow). The reddened, 1 cm in diameter nodule has widely scattered light tan-white foci and is located between the right subclavian artery (*) and the syrinx (arrowhead). Bar = 1 cm.
Tissue samples were fixed in neutral buffered, 10% formalin solution. Trimmed tissue sections were routinely processed, embedded in paraffin, and sectioned at approximately 5 µm. All tissue sections were stained with hematoxylin and eosin.
Immunohistochemical staining for thyroglobulin and pancytokeratin was performed. For thyroglobulin, the primary antibody used was rabbit polyclonal anti-human thyroglobulin. c The protein concentration of antibody was 8.1 g/l in phosphate buffered saline (PBS) and preserved with sodium azide. Antibody dilutions had previously been carried out on positive controls to find the greatest dilution of the antibody, which yielded optimal staining. The optimal dilution of the primary antibody was determined with a final concentration of 1:6,000 with staining time of 60 min. The antibody was diluted, c and the antigen retrieval performed was enzymatic in nature and consisted of using proteinase K c for 5 min. Endogenous peroxidase was blocked using 3% hydrogen peroxide. d All other blocking was completed with blocking reagent. e Positive tissue controls consisted of formalin-fixed, paraffin-embedded canine thyroid. As a negative control, the antibody was eliminated and substituted with universal negative control provided as the immunoglobulin fraction of serum from nonimmunized rabbits. The secondary antibody used was biotinylated anti-rabbit immunoglobulin G (IgG), made in goat. f As the tertiary antibody, streptavidin conjugated to horseradish peroxidase in PBS was used. c The substrate chromogen system used was 3,3′-diaminobenzidine (DAB). c The tissue sections were counterstained with Gill II hematoxylin and bluing, dehydrated in alcohol levels of 70%, 95%, and 100%, cleared in xylene, and mounted with xylene-based mounting medium.
For pancytokeratin, the primary antibody used was mouse monoclonal anti-pancytokeratin (Lu-5). e The antibody was provided as ascites, diluted in PBS, and preserved with sodium azide. Antibody dilutions had previously been carried out on positive controls to find the greatest dilution of the antibody, which yielded optimal staining. The optimal dilution of the mouse monoclonal anti-pancytokeratin antibody was determined with a final dilution of 1:100 with staining time of 45 min. The primary antibody was diluted, as mentioned previously, c and antigen retrieval, endogenous peroxidase, and all other blocking were performed as described above. Positive tissue controls consisted of formalin-fixed, paraffin-embedded canine skin. As a negative control, the primary antibody was eliminated and substituted with universal negative control that is provided as a tissue culture supernatant containing equal concentrations of monoclonal mouse IgG1, IgG2a, IgG2b, IgG3, and IgM. c The secondary antibody used was biotinylated anti-mouse IgG, rat absorbed, made in horse. e As the tertiary antibody, streptavidin conjugated to horseradish peroxidase in PBS was used, as mentioned previously. c The substrate chromogen system used, counterstaining, and subsequent steps were performed as described above.
Based on the suspicion of thyroid neoplasia, a blood sample collected antemortem was sent for measurement of TT4 by RIA, and fT4 by ED. 5 Due to the lack of reference intervals for barred owls, samples from 4 other individuals of the same species were collected and submitted to the same laboratory as before. Of the 4 control samples, 2 animals were wildlife casualties presented to the Zoological Medicine Service at the University of Georgia (Athens, Georgia) with trauma-related injuries and 2 animals were non-releasable wild animals kept at a local zoo (Table 1).
Serum total thyroxine by radioimmunoassay (TT4-RIA) and free thyroxine by equilibrium dialysis (fT4-ED) values of 5 barred owls (Strix varia) with reference to history and health status.
Microscopically, the thoracic nodule was characteristic of a thyroid neoplasm (Figs. 3–4). The neoplasm was composed of moderately pleomorphic polygonal cells arranged into closely apposed follicular structures, several of which had branched, papillary luminal projections, filled with a homogenous eosinophilic material and sloughed cells. The scant fibrovascular stroma had focal perivascular collections of lymphocytes. Polygonal cells had moderately abundant eosinophilic cytoplasm with cytoplasmic clear vacuoles, golden pigment granules, and distinct cells margins over a basement membrane. The hyperchromatic nuclei varied from round to oval with moderate anisokaryosis and distinct nucleoli. Mitoses in 10 random fields at 400× were 1, with no detected extension into vascular channels. No other significant findings were reported. While immunodetection of thyroglobulin was widespread throughout the neoplasm, detection of pancytokeratin was patchy (Fig. 4). No other significant findings were reported.
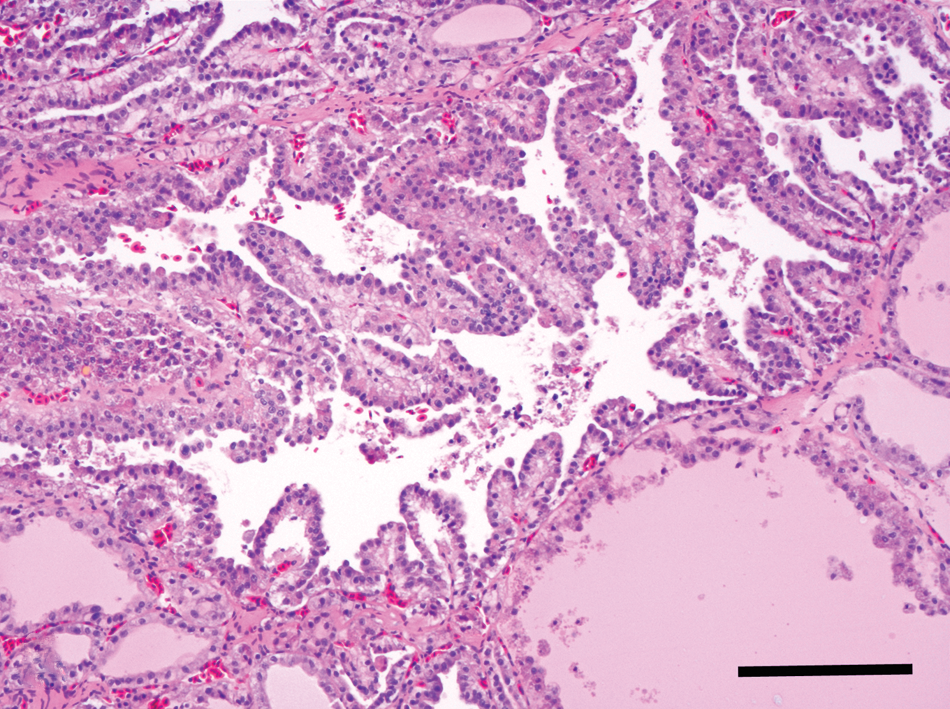
Thyroid gland; barred owl (Strix varia). Representative section of the thyroid follicular carcinoma, in which neoplastic polygonal cells are arranged into branched papillary luminal projections in the center of the image. Bar = 50 µm.
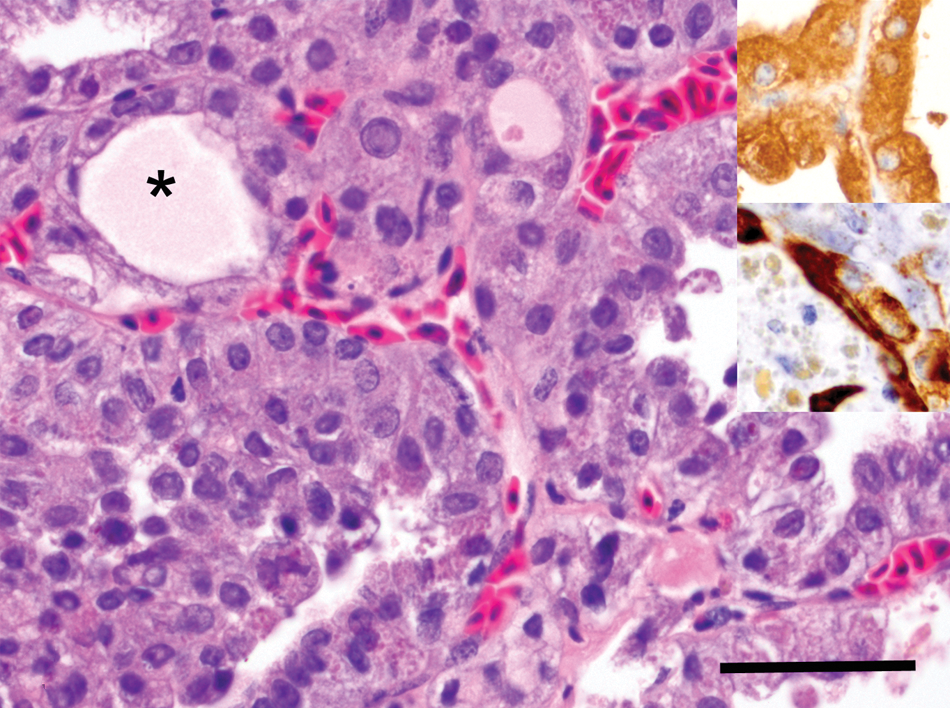
Thyroid gland; barred owl (Strix varia). Higher magnification of the thyroid follicular carcinoma composed of polygonal cell arranged into follicular structures filled with a homogenous eosinophilic material or colloid (*). Hematoxylin and eosin. Bar = 100 µm. Upper inset: immunohistochemical reactivity for rabbit polyclonal anti-human thyroglobulin antibody. Diaminobenzidine chromogen. Hematoxylin counterstain. Lower inset: immunohistochemical reactivity for mouse monoclonal anti-pancytokeratin. Diaminobenzidine chromogen. Hematoxylin counterstain.
Thyroid hormone measurement results for TT4-RIA and fT4-ED were 1.1 µg/dl and >10 ng/dl, respectively. The results obtained from the control animals were <0.2, <0.2, 0.6, <0.2, and <0.3, <0.3, 2.1, <0.3 for TT4-RIA (µg/dl) and fT4-ED (ng/dl), respectively.
Thyroid carcinomas have been previously reported in avian species. Three cases (species not specified) of thyroid adenocarcinomas with invasive behavior among 557 avian tumors have been reported.8 Thyroid adenocarcinomas have also been reported in a green pheasant (Phasianus versicolor) and in a bald eagle (Haliaeetus leucocephalus), while a thyroid cystadenocarcinoma was reported in a Saker falcon (Falco cherrug).2,4,20 The diagnosis of these conditions was based on histopathology. No reference to immunohistochemistry or productive status was reported.
The current report describes a case of a productive follicular thyroid carcinoma shown both by immunohistochemistry and thyroid hormone measurement. To the authors’ knowledge, productive thyroid follicular carcinomas and subsequent hyperthyroidism in avian species has been underreported. The specific cause of death in this patient was not determined; however; given the perimortem hormonal levels, it is suspected that the thyroid neoplasm may have contributed to the death of the animal due to overproduction of thyroid hormones. Thyrotoxic or thyroid storm (sudden overproduction of thyroid hormone) has been reported in cats and human beings.27,28 A thyrotoxic storm is a life-threatening condition related to an exacerbation of clinical thyrotoxicosis. 21 In human beings, a thyroid storm may be related to thyroid surgery, administration of iodine, discontinuation of antithyroid medication, decrease in the production of thyroxine-binding proteins, and production of inhibitors blocking binding of thyroid hormones (which can be related to euthyroid sick syndrome) or iatrogenic.21,29 The diagnosis of a thyroid storm is difficult because it has been shown that there may not be a significant difference between an uncomplicated thyrotoxicosis and thyrotoxic storm. 21 While thyroid storm may cause an acute exacerbation of clinical state, chronic hyperthyroidism can result in wasting and exhaustion. In avian species, this could result in impaired flight and decreased ability to escape predation. While chronic hyperthyroidism is also considered as a possible cause of morbidity in the current case, it would be expected that the body condition would be decreased. The body condition in the barred owl described herein was normal. Whether the owl’s thoracic limb wounds were a cause or effect of being unable to fly could not be determined.
In order to correctly assess thyroid function in avian species, validation of new tests is necessary as well as determination of reference intervals. In the current study, the measurement of thyroid values in suspected euthyroid animals was reported. Such values are not intended to be considered reference intervals for barred owls, due to the low number and the health status of these individuals. The 4 animals described were wildlife casualties that were suffering or had been suffering from unhealthy conditions (i.e., orthopedic fractures), which is not optimal but provided general information on suspected euthyroid animals. The thyroid values reported in the patient with thyroid carcinoma were much higher in both assays. Future studies are necessary to assess the normal values for both TT4 and fT4 in barred owls as well as test validation for avian species. Although not used in the current case, endogenous thyroid stimulating hormone may have also been useful, but validation of measurement methods is necessary, and significance of the results requires further investigation.
Footnotes
a.
AlphaTrak, Abbott Laboratories, Abbott Park, IL.
b.
BD Microtainer, BD, Franklin Lakes, NJ.
c.
Rabbit polyclonal anti-human thyroglobulin (A0251), antibody diluent (S0809), proteinase K (S3020), LSAB 2 Streptavidin (K1016), 3,3′-diaminobenzidine (K3466), universal negative control–mouse (N1698); Dako North America, Carpinteria, CA.
d.
3% hydrogen peroxide (H312-500), Fisher Scientific, Fair Lawn, NJ
e.
Power Block (HK085-5K), mouse monoclonal anti-pancytokeratin [Lu-5] (MU181-UC), biotinylated anti-mouse IgG, rat absorbed, made in horse (BA-2001); BioGenex Laboratories Inc., San Ramon, CA.
f.
Biotinylated anti-rabbit IgG, made in goat (BA-1000), Vector Laboratories Inc., Burlingame, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for the current report was provided by the Pamela de Journo Endowment Fund and by the Athens Veterinary Diagnostic Laboratory, College of Veterinary Medicine, University of Georgia, Athens, GA.
