Abstract
Three subgroups of the Finnish cat population underwent investigation for different aspects of feline toxoplasmosis. Blood samples of 445 purebred pet cats and 45 shelter cats were screened for Toxoplasma gondii–specific immunoglobulin G antibodies with a direct agglutination test. The overall seroprevalence was 48.4%; older cats and cats receiving raw meat in their diet were more often seropositive. Fecal samples were obtained from 131 shelters cats; 2 of the cats were found shedding T. gondii–like oocysts, and the oocysts shed by 1 of the 2 were confirmed as T. gondii with polymerase chain reaction. Among 193 cats submitted for necropsy during a 3.5-year period, 6 (3.1%) had been diagnosed with generalized toxoplasmosis and were retrospectively further investigated. The main pathological lesions included acute interstitial pneumonia, acute necrotizing hepatitis, and nonsuppurative meningoencephalitis with glial granulomas. Immunohistochemical staining demonstrated a mild to massive parasite burden in tissues with pathological lesions as well as in unaffected tissues. The results of the direct multilocus genotyping of T. gondii parasites detected were consistent with endemic genotype II, and the causative parasite strains were isolated from 2 of the generalized toxoplasmosis cases. The results indicate that cats in Finland commonly encounter T. gondii and contribute to the environmental oocyst burden, while the endemic genotype II can also prove fatal to the parasite’s definitive host. Preventing feline T. gondii infections is not only of public health importance but also a welfare issue for the cats themselves.
Introduction
Toxoplasma gondii is a cosmopolitan zoonotic parasite of a wide variety of host species. 7 Felids are the only definitive hosts, capable of excreting oocysts in their feces, and cats are considered the most important hosts from an epidemiological point of view. For example, although the major risk factor for human T. gondii infections is eating undercooked meat of infected animals, and the dreaded infection route is transplacental, the infection can also result from ingesting water, food, or soil contaminated with oocysts. 4 Moreover, the environmental oocyst burden forms an important source of infection for many domestic and game animals intended for human consumption. Felids are therefore at the very center of the epidemiology of toxoplasmosis.
Toxoplasma gondii is able to propagate without a sexual cycle in felids.15,23 Nevertheless, the opportunity for genetic recombination and the potential of shedding an environmental reservoir of oocysts make felids valuable hosts that ensure the parasite’s successful existence. Cats usually encounter T. gondii by carnivorism and require only 1 bradyzoite to shed millions of oocysts, which sporulate in 1–5 days to become infectious and environmentally resistant.5,6,12 Most cats shed only after their first infection, once, and for a limited patent period, but reinfection and reshedding are possible. 7 Seropositive cats are considered previous shedders. 7 In 1994, 63 (45%) of 141 Finnish cats were T. gondii seropositive, 20 and numerous similar epidemiological studies from different parts of the world 7 demonstrate a global contamination with T. gondii.
Although cats appear to be suitable hosts and most feline infections are subclinical, feline toxoplasmosis may cause clinical signs, such as fever, dyspnea, tachypnea, icterus, and ophthalmic manifestations, or be fatal.7,8,13,22 Toxoplasmosis in cats, human beings, and other hosts can be a devastating disease, but T. gondii infections are preventable. For risk assessment and control, knowledge of feline infections and their effect on the oocyst burden of the environment is essential, but such large-scale data has been lacking in Finland. Thus, the current study on toxoplasmosis in 3 subgroups of the Finnish cat population aimed to 1) estimate the current prevalence of toxoplasmosis; 2) identify risk factors for feline T. gondii infections; 3) estimate the proportional mortality rate of cats from generalized toxoplasmosis, and describe such feline cases in detail; and 4) identify the T. gondii genotypes that cats shed into the environment and from which they die.
Materials and methods
Three subgroups of the Finnish cat population underwent investigation. A cross-sectional epidemiological study included primarily serology to reveal the proportion of cats that had previously encountered T. gondii. The majority of the serology samples came from purebred pet cats whose owners completed a questionnaire for the risk factor analyses. Shelter cats were sampled for serology and for copromicroscopical examination for the presence of oocysts for genotyping of the parasite strains shed. Cats selected for the case series study, diagnosed postmortem with generalized toxoplasmosis by the veterinary pathologists at the Faculty of Veterinary Medicine, University of Helsinki (Helsinki, Finland), represented the naturally acquired, severe clinical cases. The parasites detected were confirmed as T. gondii and further characterized.
Epidemiological study
Serology
The minimal sample size required for the desired precision of the estimate of seroprevalence was calculated (www.openepi.com), with an a priori expected seroprevalence of 45%, 20 and determined to be 381 cats. The study included samples from 490 cats, collected from September 2008 to August 2009: plasma from 445 purebred cats from across Finland, and plasma or serum from 45 shelter cats from a cat shelter located in Helsinki. The purebred pet cats were sampled for research use with the owners’ consent as part of an ongoing feline DNA bank effort (ethical permit ESAVI-2010-0392/Ym-23). Samples from the shelter cats were subsamples of routine samples taken by shelter veterinarians or taken during necropsy. No blood was drawn solely for the current study.
The plasma and sera, stored at −20°C until analyzed, were screened for T. gondii–specific immunoglobulin G (IgG) antibodies with a direct agglutination test, a following the manufacturer’s instructions and including the positive and negative controls provided in the kit in all plates. The samples from the purebred cats were diluted to 1:40 and 1:4,000. The samples that tested positive at a dilution of 1:40 (cutoff) and samples with a prozone phenomenon, which tested positive only at 1:4,000, were defined as seropositive. In addition, a subsample of 80 purebred cat samples was further diluted to 1:60, 1:180, 1:540, 1:1,620, 1:6,000, 1:18,000, 1:54,000, and 1:162,000 to determine titers. Due to the limited sample quantities from many shelter cats, their samples were diluted to 1:40 only, and samples that tested positive at this cutoff dilution were defined as seropositive.
Questionnaire
The owners of the purebred cats completed a questionnaire b covering the cat’s signalment (e.g., date born, sex, altered or intact, breed), lifestyle (e.g., outdoor or indoor, diet), and disease history. Of the shelter cats, only limited background information was available.
Statistical analyses
Cross-tabulations and test statistics (chi-square and mid-P exact; www.openepi.com) were used to compare the results and to evaluate associations prior to the logistic regression analyses. c Confidence intervals were computed (www .openepi.com) using mid-P exact. Differences with P values < 0.05 were considered statistically significant.
Case series study
Selection and reexamination of cases
The database of the Veterinary Pathology Department, Faculty of Veterinary Medicine, University of Helsinki was searched, and all feline cases diagnosed postmortem with toxoplasmosis between January 2008 and June 2011 were selected for the case series study and reexamined by a veterinary pathologist. The clinical history and findings of the cases were recorded, and the original necropsy reports and histological tissue sections were evaluated. Tissue samples had been routinely fixed in 10% neutral buffered formalin and embedded in paraffin, and sections had been stained with hematoxylin and eosin (HE).
Immunohistochemical confirmation of the cases
Immunohistochemistry (IHC) for T. gondii was performed on formalin-fixed, paraffin-embedded (FFPE) tissues available from all the cases of the case series study to confirm the diagnoses and to evaluate the spread of the parasites in different tissues. The tissue sections (4 µm) were processed d for automated d IHC staining alongside sections without the primary antibody as well as known T. gondii–positive and –negative liver tissue sections serving as controls. Citrate buffer e (pH 6.0) was used for antigen retrieval, and T. gondii epitope-specific rabbit antibody f diluted to 1:500 was the primary antibody; other reagents were from a streptavidin–biotin immunoenzymatic antigen detection system. d
Characterization of T. gondii parasites
The T. gondii parasites detected in the studies, both the T. gondii–like oocysts detected in the fecal samples and the IHC-confirmed T. gondii parasites in the tissues of the cats of the case series study, were further characterized with molecular methods. In addition, isolation of the strains was attempted.
Detection, concentration, and confirmation of T. gondii–like oocysts from fecal samples
Fecal samples from 131 shelter cats, including 76 samples from individual cats and 21 pooled samples from 55 cats kept in groups, were screened for the presence of T. gondii–like oocysts with the aim to genotype the parasite strains being shed. Most of the samples came from cat shelters located in Helsinki, southern Finland; 15 of the samples came from shelters in western and northern Finland. The cats were of different ages, but their exact ages were unknown.
The fecal samples were collected in 2009 and kept in +4°C until examined within 7 days. Of each fecal sample, 10 g (2 parallel 5-g samples) were examined for the presence of T. gondii–like oocysts using a flotation technique g that is based on flotation with saturated MgSO4 solution, h centrifugation, and translation of the top portion of the suspension, as described in the instructions of the manufacturer of the apparatus. g After 3 subsequent freeze–thaw cycles, DNA was extracted i from the top portion of those fecal samples that had tested positive for T. gondii–like oocysts. For diagnostic polymerase chain reaction (PCR) targeting the 529-bp fragment, 14 a total volume of 50 µl comprised commercial master mix j in a final concentration of 1×, 0.5 µM of the primers 14 TOX4 and TOX5, 7 µl of the extracted DNA, and nuclease-free water. j The PCR conditions k were 94°C for 7 min followed by 35 cycles of 94°C for 1 min, 55°C for 1 min, and 72°C for 1 min, and a final extension of 72°C for 10 min. DNA extracted from reference T. gondii strains grown in cell cultures, Neospora caninum DNA, and laboratory water served as controls.
Isolation of T. gondii parasites from acute fatal infections
Fresh tissue samples were collected for isolating T. gondii during necropsy of the cats of the case series study, which had been suspected to have generalized toxoplasmosis based on clinical history or gross pathology findings. Samples of different tissues (brain, heart, liver, lung, and lymph node) were cut into small pieces with sterile instruments and further homogenized with a syringe and a needle, and poured onto zero- to 5-day-old African green monkey kidney epithelial (Vero) cell l cultures grown in Dulbecco modified Eagle medium m and 10% fetal bovine serum n in T25 flasks o at 37°C and 5% CO2. p Penicillin and streptomycin m were added for the first 2 passages. The cultures were regularly monitored by microscopy q for signs of T. gondii growth.
Direct genetic characterization of T. gondii parasites
DNA was extracted j from FFPE liver tissue, frozen tissues, and cell cultures (if available) of the toxoplasmosis cases of the case series study, into an elution volume of 30 µl. In addition, the DNA extractions i from the PCR-confirmed T. gondii oocyst-positive fecal samples were analyzed. The genotyping method is based on an analysis of the length polymorphism at known microsatellite markers. 1 In addition to 6 microsatellite markers (B18, TUB2, TgM-A, W35, B17, and M33) for genotyping, a seventh marker (M48) was included for further characterization.1,2,16 The forward primers r were 5′-end-labeled with either 6-carboxyfluorescein (B18, TUB2, and M48), hexachlorofluorescein (TgM-A, W35, and B17), or 2,7′,8′-benzo-5′-fluoro-2′,4,7-trichloro-5-carboxyfluorescein (M33). For the multiplex PCR, the total volume of 25 µl comprised commercial master mix i in a final concentration of 1×, a 0.04-µM concentration of each primer, r 4 µl of DNA extract, and 5 µl of nuclease-free water. j The PCR conditions k were 95°C for 15 min followed by 40 cycles of 94°C for 30 sec, 61°C for 4 min, and 72°C for 1 min, and a final extension of 60°C for 30 min. After electrophoresis r of PCR products, the results were analyzed with specific software. r DNA from T. gondii parasites RH (genotype I), s GIL (type I), PSP-2007-TON (type II), PRU (type II), TOU-2004-FEU (type III), and NED (type III) t served as positive controls and references. The negative controls included laboratory water, Neospora caninum strain NC1, u and FFPE liver tissue samples from a cat euthanized due to Pasteurella multocida meningitis.
Results of the epidemiological study
Serology and questionnaire: seroprevalence and risk factors for seropositivity
Toxoplasma gondii–specific IgG antibodies were detected in 237 (48.4%) of the 490 cats (Table 1). Prozone phenomenon was observed in 6 samples. The titer was 40 in 11 samples, and 60 in 5, 180 in 2, 1,620 in 2, 4,000 in 5, 6,000 in 20, 18,000 in 31, and 54,000 in 4 samples. Thus 60 (75.0%) of the 80 samples diluted further had high titers (4,000 or higher). Of the 445 purebred cats, 221 (49.7%) tested seropositive. By contrast, 16 (35.6%) of the 45 shelter cats tested seropositive, but this difference was not statistically significant.
Toxoplasma gondii seroprevalence in Finnish cats, by selected categories.
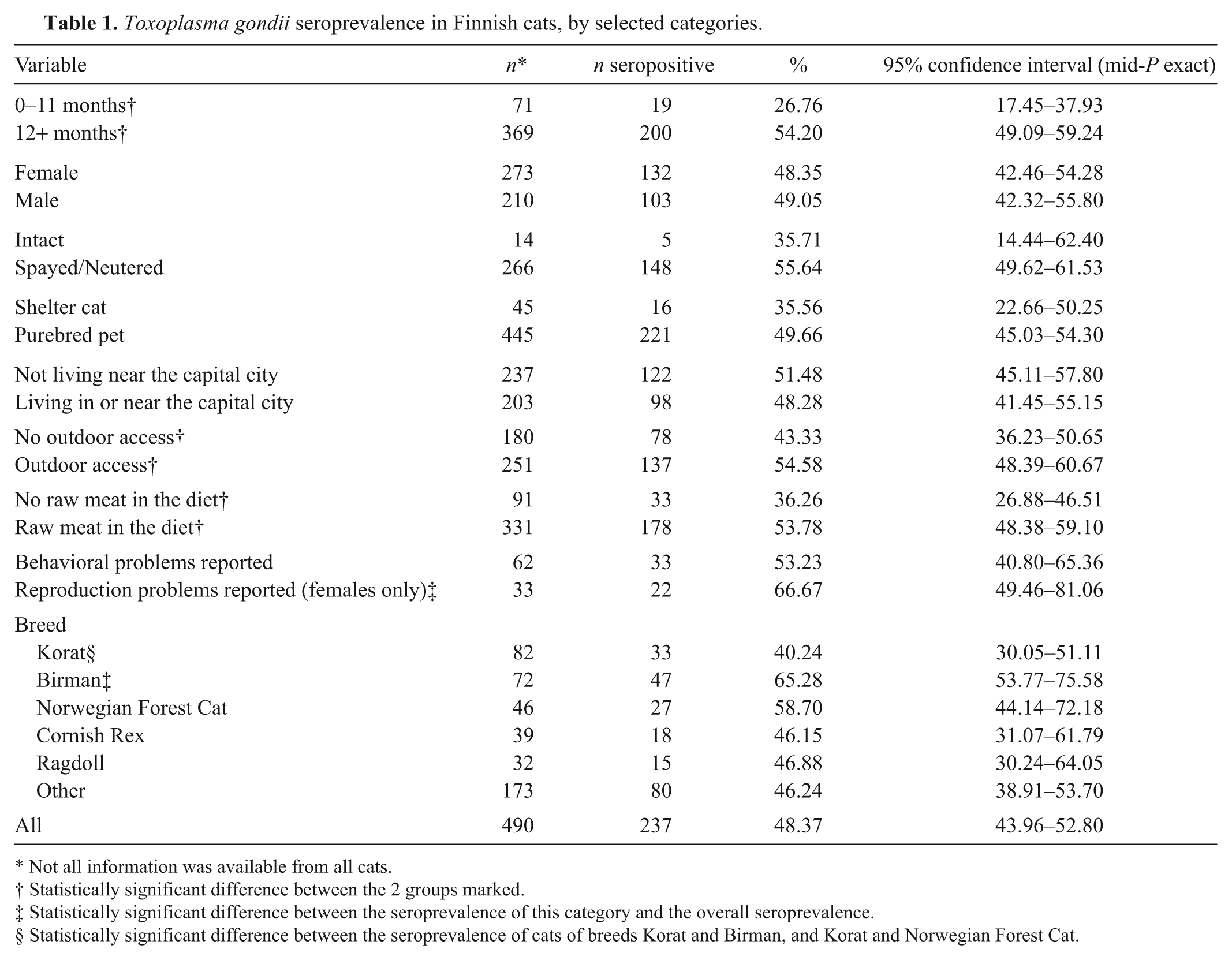
Not all information was available from all cats.
Statistically significant difference between the 2 groups marked.
Statistically significant difference between the seroprevalence of this category and the overall seroprevalence.
Statistically significant difference between the seroprevalence of cats of breeds Korat and Birman, and Korat and Norwegian Forest Cat.
Predictably, adult cats (>12 months old) were more often seropositive than young cats (P < 0.0001). In the different logistic regression models built, the adults had 3.2–3.5 times (95% confidence interval [CI]: 1.8–5.7 and 1.9–6.3, respectively) higher odds to test seropositive than the young cats, and when the exact age of the cats was used as a continuous variable, the odds of testing seropositive increased by 20% (95% CI: 14–27%) for every 1 unit (year) increase in age.
In the univariable analysis and simple logistic regression analysis, access to the outdoors was a significant (P < 0.05) risk factor for seropositivity with an odds ratio of 1.6 (95% CI: 1.1–2.3), but this factor did not fit into the multivariable models. Raw meat in the diet was an important and significant (P < 0.001) risk factor with an odds ratio of 2.0 in the univariable analysis as well as in the two-variable model with age group (95% CI: 1.3–3.3 and 1.2–3.2, respectively).
Intriguingly, differences in seroprevalence between cat breeds were detected. Korats had significantly lower seroprevalence than did Birmans or Norwegian Forest Cats (P < 0.01 and P < 0.05, respectively). Birmans had the highest seroprevalence (>65%), which is significantly higher (P < 0.05) than the estimated overall prevalence in the current study. The different age distribution among the samples from different breeds failed to explain why Birmans had such a clearly higher seroprevalence than did Korats. In contrast, the mean age of Birmans was 4.6 years, whereas that of Korats was 5.8 years. Raw meat was included in the diet of 85.7% of Birmans, whereas only 44.3% of Korats were fed diets with raw meat (P < 0.001); 81.9% of Birmans had outdoor access, whereas only 19.8% of Korats were not exclusively indoors (P < 0.001).
Another interesting finding was the high seroprevalence (66.7%) among female cats with reported reproduction problems, such as abortions and stillbirths: significantly higher (P < 0.05) than the estimated overall prevalence. The seroprevalence in cats with reported behavioral problems showed no significant difference from the overall prevalence estimate.
Results of the case series study
Number of cases: proportional mortality rate
From 2008 to 2011, 193 cats were submitted to the Faculty of Veterinary Medicine, University of Helsinki for postmortem examination. Six of these cats were diagnosed with generalized toxoplasmosis, which had resulted in death or euthanasia. Thus, the proportional mortality rate from toxoplasmosis was 3.1% (95% CI: 1.3–6.4%).
Reexamination of cases: clinical histories and pathologic findings
Table 2 summarizes the breed, age, clinical signs, and selected laboratory results of the 6 cats of the case series study. The cats all originated from southern Finland. Three cats had been euthanized because of their rapidly deteriorating general condition, and 3 cats had died. Cat 2 had been vaccinated 1.5 weeks earlier, cat 4 had traveled abroad 2 weeks before falling ill, and cat 5 was a shelter cat originally included in the study for fecal investigations.
The clinical history and selected laboratory results of the 6 cats with generalized toxoplasmosis.*
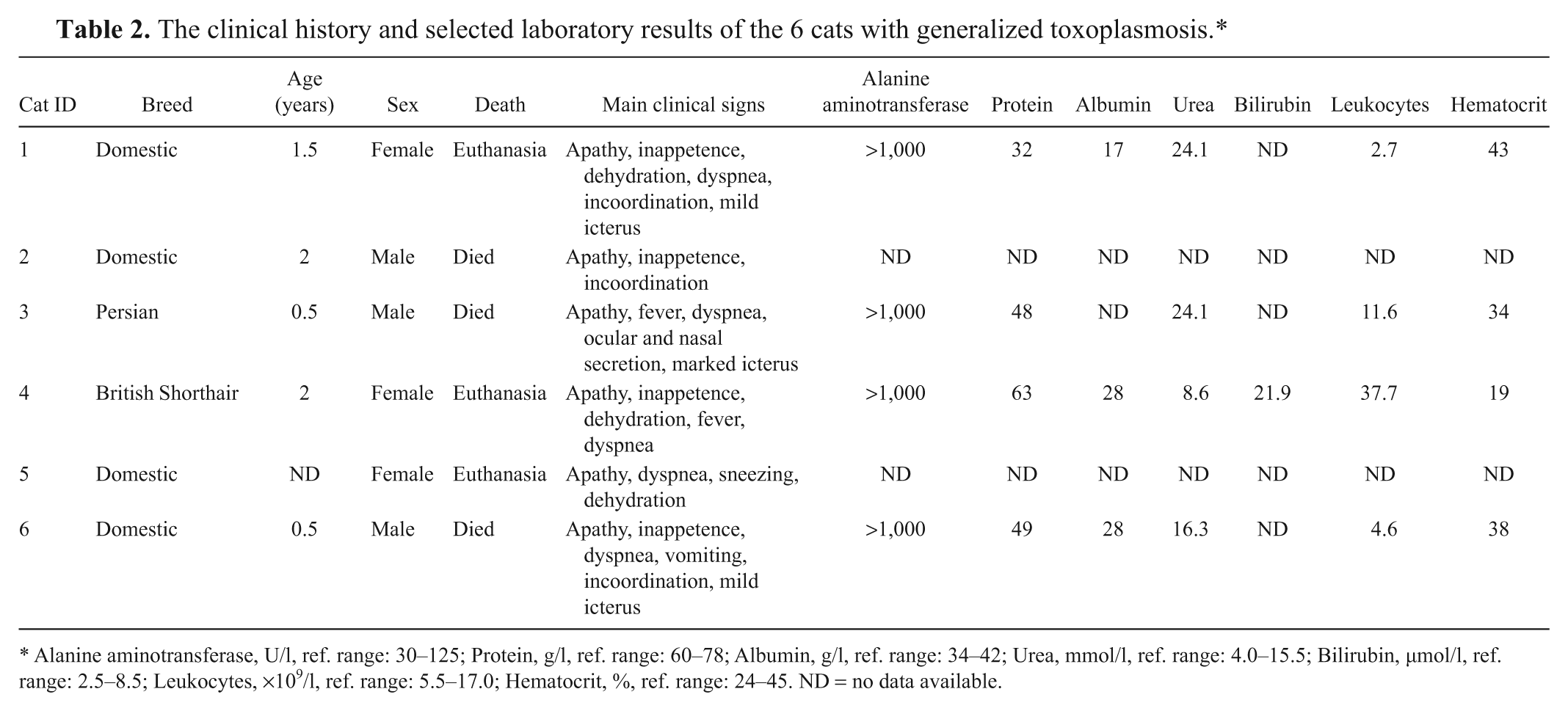
Alanine aminotransferase, U/l, ref. range: 30–125; Protein, g/l, ref. range: 60–78; Albumin, g/l, ref. range: 34–42; Urea, mmol/l, ref. range: 4.0–15.5; Bilirubin, µmol/l, ref. range: 2.5–8.5; Leukocytes, ×109/l, ref. range: 5.5–17.0; Hematocrit, %, ref. range: 24–45. ND = no data available.
All cats had a history of acute illness lasting approximately 1 week. The clinical signs were unspecific: the most common signs were apathy, inappetence, dehydration, fever, and respiratory signs, such as dyspnea, sneezing, and nasal secretion. Incoordination was observed in 3 cats, and 3 cats had icteric mucous membranes.
Clinical chemistry and hematological parameters were examined in 4 cats (cats 1, 3, 4, and 6), and the main abnormality detected was markedly elevated liver enzyme alanine aminotransferase (Table 2). In addition, fecal examination for oocyst shedding was performed from cat 5, and oocysts consistent with T. gondii were detected 10 days before clinical signs appeared.
The spread and frequency of detected gross and histopathological findings are shown in Table 3. The gross lesions observed in postmortem examination were sparse and unspecific. All of the cats had marked pulmonary edema with heavy, fluid-filled, uncollapsed lung lobes. Cats 1 and 6 also had mild pleural effusion. Cats 1 and 3 had livers with small (1–2 mm), irregular, pale or reddish areas on the surface, and cats 4 and 6 had pale, yellowish, and fragile livers. Five cats (cats 2–6) had mildly or moderately enlarged mesenteric lymph nodes, and 3 cats (cats 3, 4, and 6) had mildly enlarged pulmonary lymph nodes. Splenomegaly was evident in cats 2 and 4. The mucous membranes were pale or icteric in 5 cats (cats 2–6). Cat 3 had yellowish nasal and ocular secretion. No gross lesions were detected in the brains.
The spread of lesions and Toxoplasma gondii parasites in the 6 cats with generalized toxoplasmosis.*
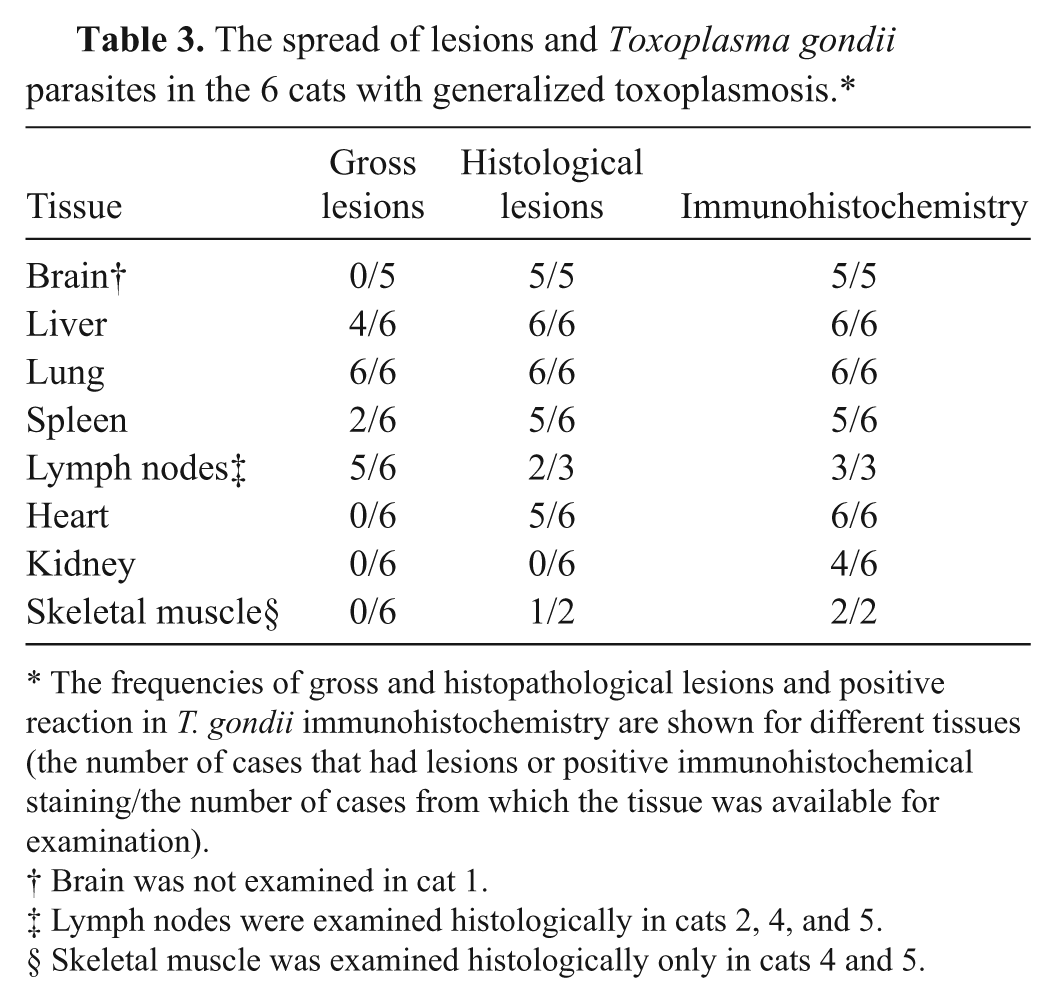
The frequencies of gross and histopathological lesions and positive reaction in T. gondii immunohistochemistry are shown for different tissues (the number of cases that had lesions or positive immunohistochemical staining/the number of cases from which the tissue was available for examination).
Brain was not examined in cat 1.
Lymph nodes were examined histologically in cats 2, 4, and 5.
Skeletal muscle was examined histologically only in cats 4 and 5.
In all the cats, histopathological lesions were consistently present in the liver and lungs. In the liver, the lesions consisted of acute necrotizing hepatitis characterized by variably sized, irregular foci of hepatocellular necrosis with hemorrhage and mild infiltration of neutrophilic granulocytes and macrophages (Fig. 1). The distribution was random throughout the parenchyma, and the lesions were multiple. The portal areas contained multifocally mild to moderate infiltrates of macrophages and lymphocytes as well as occasional neutrophilic granulocytes and plasma cells. Parasites were only occasionally seen near necrotic foci and were challenging to detect in the HE-stained sections.
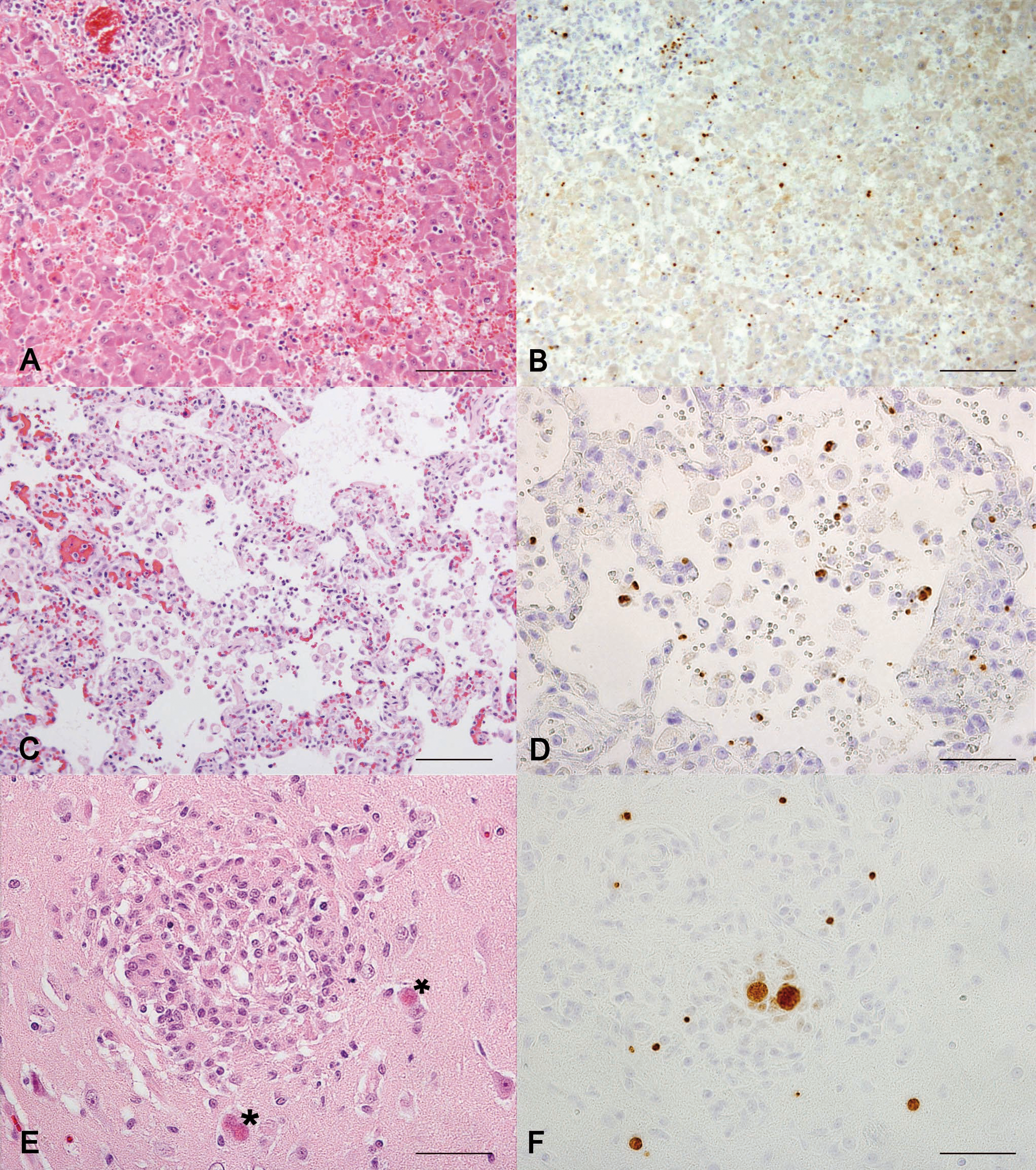
Major histological lesions and Toxoplasma gondii parasites visualized with immunohistochemistry (IHC) in the main tissues affected in feline generalized toxoplasmosis.
Cats 1, 3, 4, and 6 had severe diffuse acute interstitial pneumonia associated with thickened and edematic alveolar septae and multifocal septal necrosis with mild infiltration of neutrophilic granulocytes and mononuclear cells (Fig. 1). The bronchiolar epithelium was also multifocally necrotic. The alveolar spaces contained edema fluid, an elevated number of large foamy macrophages, and a scant amount of fibrin. Cats 2 and 5 had similar but milder and more focal pulmonary lesions. Cat 4 showed marked regenerative changes indicated by marked hyperplasia of the alveolar lining type II pneumocytes and mild interstitial fibrosis. In the lungs, only occasional parasitic structures were found in the HE-stained sections.
Of the 6 cats, 5 cats had brain lesions (the brain of cat 1 was unexamined). The major lesion was variably sized glial granulomas observed multifocally throughout the neural parenchyma (Fig. 1). Granulomas were found in the cerebral cortex, cerebellum, pons, and medulla with no obvious predilection site. The granulomas were often associated with parasitic cyst structures, but parasites were also detected distant to the lesions. In addition, mild multifocal nonsuppurative meningitis was detected in all 5 examined cats, and mild perivascular cuffing was detected in 3 cats. The spinal cords were not examined.
Minor lesions associated with toxoplasmosis were seen in multiple organs. The spleen appeared affected in 5 cats (cats 1–4 and 6). The mesenteric lymph nodes of cats 2 and 4 showed acute necrotizing lymphadenitis with irregular areas of necrosis mainly in the cortex. Cats 1, 2, and 4–6 had mild multifocal lymphohistiocytic myocarditis, and cats 4 and 6 had mild acute necrotizing adrenalitis. Mild lymphohistiocytic myositis in the skeletal muscle was detected in cat 5, and cat 4 had mild acute necrotizing pancreatitis. No other concurrent diseases were diagnosed.
Immunohistochemistry: parasite burden and spread in tissues
The diagnoses of the 6 cats of the case series study were confirmed with IHC: strong positive staining was detected in all the cats and parasitic structures were easily visible (Table 3). Large numbers of parasites were seen in the liver, lungs, spleen, and lymph nodes (Fig. 1). Parasites were present in different types of cells, including epithelial cells, macrophages, and smooth muscle cells. In the brain, the parasite burden varied from mild to moderate. In addition, scattered positive staining was found in multiple tissues, including the myocardium, thyroid gland, skeletal muscle, tongue, renal glomeruli and tubular cells, intestinal mucosa and lymphoid tissue, and bone marrow.
Results of characterization of T. gondii parasites
PCR-confirmed T. gondii oocyst shedding
Toxoplasma gondii–like oocysts were detected in the fecal samples in 2 (1.5%, 95% CI: 0.26–4.95%) out of 131 shelter cats. Both of these cats were from southern Finland. One (cat 5) of the 2 fecal samples that tested positive for T. gondii–like oocyst was confirmed to be T. gondii with PCR. The PCR-confirmed T. gondii oocyst shedding prevalence was thus 0.76% (95% CI: 0.04–3.70%).
Isolation of T. gondii and growth patterns in cell cultures
Isolation of the parasite strains directly to cell cultures was performed from fresh tissue samples of cats 5 and 6 of the case series study. In the other 4 cases, toxoplasmosis had not been suspected sooner than upon evaluation of the histological samples (i.e., when fresh samples were no longer stored).
Rampant T. gondii infection was established by the third passage from the brain tissue and heart muscle of cat 5, and from the brain and lung tissue of cat 6. The growth pattern was a patchy, multifocal coalescent disruption of the cell layer resembling that of genotype II reference strain PSP-2007-TON in both cases. The strains obtained were maintained by serial passages to fresh cell flasks, and cryopreserved.m,v Attempts to isolate parasites from liver and lymph node samples were unsuccessful.
Genetic characterization: T. gondii genotypes
The direct genetic characterization was successful at all 7 markers from the FFPE liver tissue of cats 1–4; from the frozen brain tissue, heart muscle, tongue, and skeletal muscle of cat 5; from the frozen liver tissue, brain tissue, and lung tissue of cat 6; and from cell cultures of isolated strains from cats 5 and 6. The results at the 6 genotyping markers were all fully consistent with genotype II. In addition, the same but partial results were obtained from the frozen lymph node and a mixture of lung and liver tissue of cat 5. Full results fitting genotype II were also obtained from the oocyst-positive fecal sample from cat 5. The length of the PCR product at the seventh marker was 223, 215, 231, 235, 229, and 215 base pairs in cats 1–6, respectively, revealing some polymorphism.
Discussion
The estimate of seroprevalence (48.4%) indicates that the cats of the study population often encounter T. gondii. The seropositive cats had presumably shed oocysts earlier: the oocyst contamination of the environment in an area with this high a seroprevalence in cats is surely not negligible. The prevalence of oocyst shedding that was ongoing at the time of sampling was expectedly low, as cats only shed oocysts for a limited period: 2 cats were detected to be contributing to the environmental contamination with T. gondii–like oocysts, 1 of which confirmedly with oocysts of endemic T. gondii genotype II.
Seropositivity means that the cat has encountered T. gondii previously and mounted a measurable antibody response against it. In diagnosing clinical feline toxoplasmosis, serology is of limited value, and a single seronegative result does not rule out toxoplasmosis. 7 Examination of feces for the presence of T. gondii oocysts has even more limited value in diagnosing feline toxoplasmosis. Cats shed these oocysts for a short time, and there are no confirmed reports of shedding by clinically ill cats 7 : most shedding cats are apparently healthy, and most feline T. gondii infections remain subclinical. In the present study, cat 5 was found to be shedding PCR-confirmed T. gondii oocysts 10 days before developing clinical signs of toxoplasmosis.
For epidemiological toxoplasmosis studies, serology provides valuable information of the presence, prevalence, and spread of the infection, whereas examining feces of cats gives nothing but a snapshot (i.e., the proportion of cats that were shedding at the time of sampling). However, it is the oocyst shedding that makes cats important hosts of T. gondii from a public health point of view, and confirmatory information of the dynamics of this shedding is important. Moreover, direct detection of the parasites, not only the response of the host, enables the confirmation of the diagnosis by PCR and further genetic characterization of the parasite strains. Toxoplasma gondii oocysts can be detected with any standard fecal flotation technique. 7 The method chosen for the current study is laborious but served as a practical enrichment step for the molecular methods because the translation of the top portion of the suspension allows for easy collection.
The seroprevalence had not changed significantly from the previous estimate 20 available from Finland. However, the study populations are incomparable, as the previous study focused on shelter cats, and the majority of samples in the current study were from pet cats with their owners’ voluntary participation, which affects the breed distribution. The exact ages of the shelter cats of this study were unknown, which is unfortunate, as their lower seroprevalence might be partly due to their age distribution.
The questionnaire distributed to the owners of the purebred pet cats in the epidemiological study revealed a striking risk factor: raw meat given to the cat by the owner. Over 78% of the owners reported feeding their cat raw meat, such as pork or minced meat. The cats that received raw meat had a significantly higher seroprevalence than did the cats that had no raw meat in their diet, with an odds ratio of 2.0. Cats do need meat in their diet, but this result emphasizes that the meat should not be served raw, but cooked or frozen to protect the cats from T. gondii infection. The differences in seroprevalence among cat breeds were also, in fact, most likely due to different lifestyles. A majority of the Korats, the breed with the lower seroprevalence, lived indoors only and received no raw meat.
The proportional fatal morbidity rate from generalized toxoplasmosis was 3.1% in cats submitted for postmortem examination. This measure of disease incidence is subject to variation in the material included; however, the result from the current study does not differ significantly from that of a previous published report, 3.2% (5/155), from Denmark. 13
The 6 cats diagnosed with generalized toxoplasmosis were 2 years old or younger, and systemic infections are most common in young animals.3,9–11 As seen in these cases and described previously, the clinical signs of generalized toxoplasmosis are usually unspecific, generalized, and dependent on affected organ systems.3,10,12,24 The abnormal clinical chemistry and hematologic parameters were related mainly to acute liver injury and inflammatory response.
The gross and histological lesions were very similar to those described previously.3,8,11,13,18,21,22 Toxoplasmosis should be suspected if a young cat is diagnosed with acute interstitial pneumonia, acute necrotizing hepatitis, or nonsuppurative meningoencephalitis. Interstitial pneumonia was observed in all cases in the present study and is the most predominant lesion in naturally occurring fatal toxoplasmosis in cats.7,11 Toxoplasma gondii parasites are difficult to detect in HE-stained sections, and their numbers are likely underestimated, but the spread of the parasites to different tissues can easily be evaluated with IHC. Toxoplasma gondii is able to infect a wide range of cells and tissues,7,11 but the typical, specific tissue tropism for certain tissues, such as the lungs and liver, was seen in the present study as well.
The outcome of T. gondii infection depends largely on the immune status of the host. 19 A poor immune response after primary infection results in overwhelming tachyzoite replication and tissue necrosis, instead of a chronic form of T. gondii infection.3,10 A chronic infection may become activated when the immunity of the host is impaired, but this is considered rare in cats. 18 According to their clinical history, the cats described herein had received no immunosuppressive medications, but the presence and role of some other kinds of immunosuppression remain unclear. Enlargement of the mesenteric lymph nodes, observed in 5 cats, and the histopathological lesions observed within, examined and observed in 2 of the 5 cats, could indicate the spread of acute infection from the intestine rather than the reactivation of a chronic infection, whereas lesions in the central nervous system and skeletal muscle along with the concomitant presence of parasite cysts could indicate reactivation.8,18 However, both lymph node and brain lesions were found in the same cats, and 1 of the cats (cat 5) was shedding shortly before it developed clinical signs. Considering that tissue cysts are already forming within 2 weeks after primary infection, 3 caution should be taken when estimating the course and duration of the infection.
Little is known about the effect of the infecting T. gondii strain on the outcome of feline toxoplasmosis. Genotyping parasites that cause life-threatening and fatal infections is a way to investigate this. Using a direct genotyping technique, genetically characterizing the parasites directly from the sample material, is beneficial over techniques that include a bioassay step because that step might cause selection in the parasite strains finally genotyped. Interestingly, T. gondii parasite strains belonging to genotype II, which appear to be endemic in Finland16,17 and are typically nonvirulent in mice, 19 have killed European brown hares and mountain hares 16 as well as Eurasian red squirrels 17 in Finland in addition to the cats described in the current study.
Owners of pet cats could do more to protect their cats from T. gondii infection. Cats should not be allowed to hunt small prey, and owners should cook or freeze the meat they serve to their pet cats. Preventing T. gondii infections in cats would diminish the contamination of the environment with oocysts, which has direct and indirect implications for public health, as human beings, as well as animals intended for human consumption, may become infected by ingesting the oocysts. Preventive measures practiced by the owners of pet cats could make a difference, especially in an area like Finland, which has a practically nonexistent feral cat population. Moreover, preventing T. gondii infections is also a cat welfare issue because the cats themselves may develop clinical disease and even die from the infection.
Footnotes
Acknowledgements
The cat owners and Minna Virta from the feline genetic research group of the University of Helsinki are thanked for the serology samples, the cat shelters for the fecal samples, and Laura Rinaldi for kindly providing the FLOTAC. Veterinary pathologists Hanna-Kaisa Sihvo, Karoliina Hagner, and Anna Knuuttila performed the necropsies of some of the cats. SVA (Sweden) and the Toxoplasma Biological Resource Centre (France) provided the Toxoplasma gondii reference material, and Katarzyna Gozdzik (Witold Stefanski Institute of Parasitology, Warsaw, Poland) provided the Neospora caninum DNA.
a.
Toxo-Screen DA, bioMérieux SA, Marcy l’Etoile, France.
b.
The questionnaire is available from the corresponding author upon request.
c.
Stata 11.0 software, StataCorp LP, College Station, TX.
d.
PT Module, Lab Vision Autostainer 480, UltraVision Plus Detection System (anti-polyvalent HRP/DAB kit); Lab Vision Corp., Fremont, CA.
e.
Citric acid monohydrate, Merck KGaA, Darmstadt, Germany.
f.
Toxoplasma gondii epitope-specific rabbit antibody, Thermo Fisher Scientific, Runcorn, Cheshire, UK.
g.
FLOTAC, University of Naples Federico II, Naples, Italy.
h.
MgSO4, VWR International, Leuven, Belgium.
i.
QIAamp DNA Stool Mini Kit, 2x Multiplex PCR Master Mix; Qiagen GmBH, Hilden, Germany.
j.
2x PCR Master Mix, Nuclease-free water, Tissue and Hair Extraction Kit with the DNA IQ System; Promega Corp., Madison, WI.
k.
PTC-200 Peltier Thermal Cycler, MJ Research Inc., Waltham, MA.
l.
Vero cells, European Collection of Cell Cultures, Salisbury, UK.
m.
DMEM + GlutaMAX-I, PenStrepGlutamine, Recovery (cell culture freezing medium); Life Technologies Ltd., Paisley, UK.
n.
Fetal bovine serum, Thermo Fischer Scientific, Cramlington, Northumberland, UK.
o.
Easy Flask 25 V/C, NUNC A/S, Roskilde, Denmark.
p.
Serie CB Incubator, Binder GmbH, Tuttlingen, Germany.
q.
Leica DMIL, Leica DFC425; Leica Microsystems GmbH, Wetzlar, Germany.
r.
Applied Biosystems oligos, Applied Biosystems 3130xl Genetic Analyzer, Applied Biosystems Peak Scanner Software version 1.0; Life Technologies Ltd., Paisley, UK.
s.
National Veterinary Institute, Uppsala, Sweden.
t.
Toxoplasma Biological Resource Centre, Limoges, France.
u.
Witold Stefanski Institute of Parasitology, Warsaw, Poland.
v.
Sanyo Ultra Low (MDF-C2156VAN), Sanyo Electric Co. Ltd., Osaka, Japan.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partly supported by a grant from the Finnish Cat Association, Kissaliitto (a member of the Fédération Internationale Féline).
