Abstract
Twenty-eight horses in southern Mozambique died after exhibiting severe respiratory distress. At necropsy, the overinflated lungs did not collapse, had prominent rib impressions, and were fibrotic and emphysematous. Microscopically, prominent proliferation of nonciliated epithelial (Clara) cells in the terminal bronchioles and pulmonary fibrosis were observed, indicative of chronic pneumotoxicity. Transmission electron microscopy demonstrated hyperplasia, desquamation, and apical bulging of Clara cells into the bronchiolar lumen. The outbreak was attributed to ingestion of Crotalaria dura J.M. Wood & M.S. Evans subsp. mozambica Polhill and Crotalaria monteiroi Taub. ex Baker f. var. monteiroi. Semiquantitative gas chromatography–mass spectrometry revealed the presence of pyrrolizidine alkaloids in these 2 Crotalaria species.
Crotalaria species poisoning with respiratory lesions in horses (crotalariosis equorum), also referred to as equine “jaagsiekte” in South Africa, is a chronic disease of horses and mules caused by the ingestion of certain species of Crotalaria.7,13 Jaagsiekte is the Afrikaans vernacular that refers to the rapid respiratory rate. The disease occurs, after an extended latent period, following ingestion of large quantities of 2 Crotalaria species, namely C. dura and C. globifera, over a number of days.7,13 These 2 species occur predominantly in the KwaZulu-Natal Province of South Africa and retain their toxicity on desiccation. Both can induce jaagsiekte when contaminated hay is fed to horses. 13 A pyrrolizidine alkaloid (PA), dicrotaline, has been isolated from both C. dura and C. globifera. 9 Crotalaria species often have trifoliate leaves, typical pea-like flowers, and swollen pods containing loose seeds (commonly known as rattlepods).7,8 The genus name Crotalaria is derived from the Greek crotalon, which means castanet and refers to the rattling sound the seeds produce when the dry, inflated pods are shaken. 8
Jaagsiekte is considered of academic or historical interest as, at present, horses are often stabled and are fed commercial rations and good quality hay. 7 However, occasional incidents of poisoning are still reported. The last recorded outbreak in South Africa occurred in 1999 (Last RD, Vorster JH, Burgoyne P, et al.: 2000, Two outbreaks of Crotalaria poisoning “jaagsiekte” in horses in KwaZulu-Natal. Congress of the South African Veterinary Association, Sept 20–22, Durban, South Africa). Clinically, the disease is characterized by fever, an increased respiratory rate (respiration is very superficial and is either costoabdominal or abdominal in character), dyspnea, subcutaneous emphysema, and tachycardia.7,13 At necropsy, severe pulmonary emphysema, characterized by overinflated lungs that do not collapse on opening the thoracic cavity and that have obvious rib impression marks on the pleural surfaces, is observed. Usually the lungs have a mottled appearance due to multifocally juxtaposed areas of atelectasis and interstitial pneumonia. Emphysematous bullae are also noticeable macroscopically in areas such as the peritracheal and periesophageal regions and subcutaneous tissues. In addition, fibrosis or cirrhosis of the liver has also been reported in some cases.7,13 Microscopy of the lungs reveals desquamation of the epithelial cells of the smaller bronchioles, and alveoli and their lumina are filled with plugs consisting of mucus, desquamated epithelial cells, and edema fluid. Proliferation of the epithelium of the bronchioles, interstitial fibrosis, and pronounced thickening of pulmonary blood vessel walls due to mural edema are also consistent findings.7,13 The present report describes an outbreak of equine jaagsiekte in southern Mozambique and the laboratory workup required to confirm this rare form of PA poisoning.
Twenty-eight horses died of a respiratory disease between mid-October 2010 and January 2011 on a farm in the northern part of the Province of Inhambane, Mozambique. The property is situated inland (22°02′13.21′′S 35°12′56.46′′E), approximately 30 km southwest of the coastal town of Vilankulos in southern Mozambique. The farmer reported that, due to urbanization in the coastal areas, he was forced to move inland to access grazing for his horses. Clinically, the horses exhibited severe tachypnea and dyspnea with flaring nostrils and a prominent heave-line during respiration. The ocular mucus membranes were severely congested. One animal was euthanized by intravenous administration of an overdose of pentobarbital, and a necropsy was performed. Macroscopically, the lungs did not collapse on opening the thoracic cavity, and prominent rib impressions were visible on the pleural surfaces (Fig. 1A). Severe lung edema, fibrosis and emphysema (Fig. 1B), and moderate hydrothorax were also observed. Samples of the lung, liver, spleen, cerebrum, cerebellum, colon, and mesenteric lymph node were collected, fixed in 10% buffered formalin, and submitted for histopathology. Another set of formalin-fixed tissue samples (lung, liver, heart, spleen, and cerebrum), collected from another horse in this same herd that was necropsied in December 2010, was also available for histopathological examination. The tissue samples were routinely processed, sectioned, and stained with hematoxylin and eosin. In addition, sections of lung were stained with Masson trichrome to identify collagen fibers. Lung tissue, retrieved from the wax blocks, was infiltrated with 1% osmium tetroxide in xylene and a xylene–epoxy resin mixture before embedding in epoxy resin. 14 Ultrathin resin sections were examined with a transmission electron microscope a operated at 80 kV.
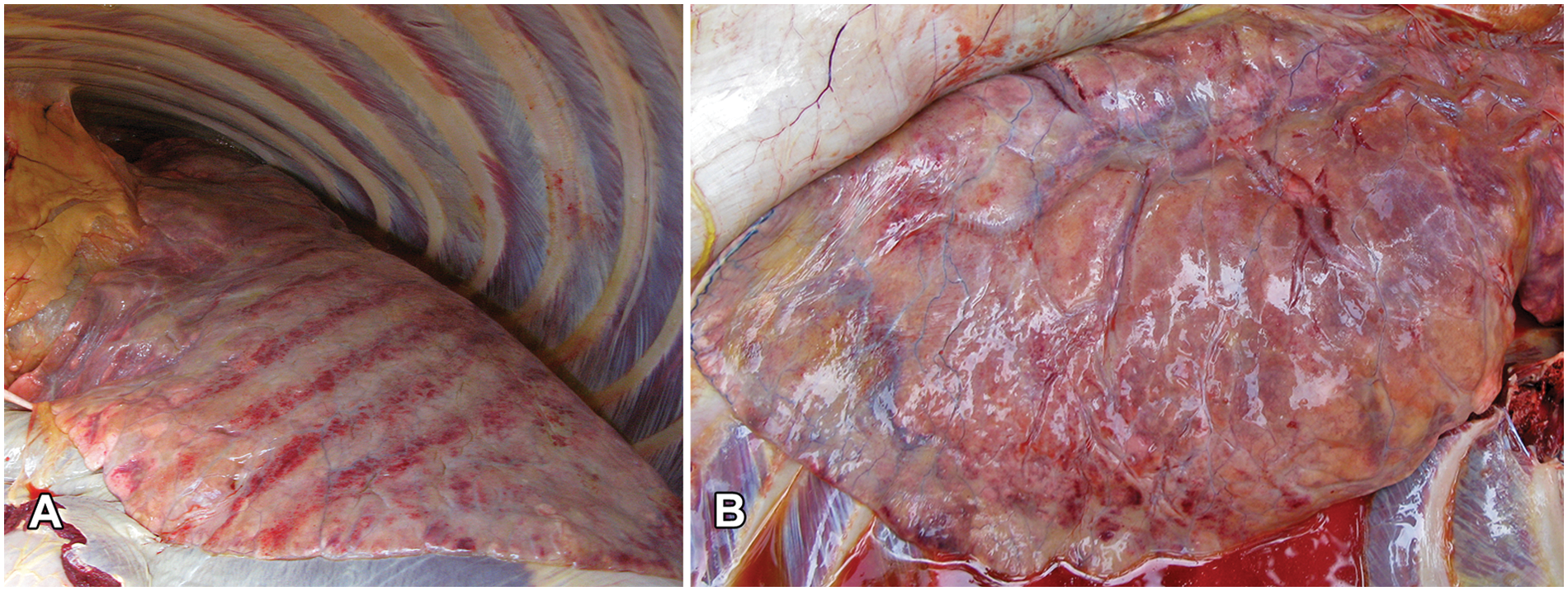
Crotalariosis equorum or equine “jaagsiekte”:
A provisional diagnosis of jaagsiekte was made, and the paddock was inspected for Crotalaria species. Two plants, believed to be Crotalaria species, were collected, pressed, and submitted for botanical identification. When the suspect plants were pointed out to the farmer, he indicated that the horses were very fond of and actually competed with each other to gain access to these plants and even dug up the roots to consume these as well. The plants were identified by the HGWJ Schweickertd Herbarium (University of Pretoria, South Africa) as Crotalaria dura J.M. Wood & M.S. Evans subsp. mozambica Polhill (Fig. 2A) and Crotalaria monteiroi Taub. ex Baker f. var. monteiroi (Fig. 2B).

Crotalaria species collected on the farm in Inhambane Province, southern Mozambique:
A semiquantitative PA analysis on the 2 incriminated Crotalaria species was performed. Positive (Senecio latifolius and Senecio inaequidens) and negative control samples (nontoxic Crotalaria agatiflora and Crotalaria distans) were included in the analytical investigations in order to compare PA concentrations. The positive control samples and the C. distans sample were obtained as dried, milled plant material from the Division of Toxicology, ARC-Onderstepoort Veterinary Institute (South Africa) where they had been stored in a freezer (–8°C) for an unspecified period of time. Crotalaria agatiflora was freshly collected from the Douw Steyn Poisonous Plant Garden (Faculty of Veterinary Science, University of Pretoria, South Africa) and air-dried before being milled.
The extraction procedures followed the method described previously. 4 Briefly, the dried, milled plant material (0.5–2 g) was weighed into tubes, homogenized for 10 min with 20 ml of 90% ethanol, b and left on a mechanical shaker for approximately 2 hr. The solids were allowed to settle, and the samples were centrifuged at 2,200 × g for 15 min. The clear solution was divided into equal parts (reduced fraction and free basic alkaloid fraction) and evaporated at 50°C under a mild stream of nitrogen. The extracts were reconstituted in 2 ml of dilute hydrochloric acid (0.1 M). b Chlorophyll and waxes were extracted with 2 × 5 ml hexane, b and the hexane layer discarded. The N-oxide fraction was reduced by adding approximately 0.5 g of zinc powder b and stirring overnight. Both fractions were then made basic (pH >9) by the addition of approximately 0.5 ml of 25% ammonia solution. b The alkaloids were extracted using 3 × 5 ml of ethyl acetate. b The ethyl acetate was evaporated and the PAs stored dry at −20°C until analysis. Gas chromatography–mass spectrometry (GC-MS) c was performed (GC-MS in electron impact [EI] mode with a 30 m × 0.25 mm × 0.25 μm column installed). d The extracts were reconstituted in 0.5 ml of ethyl acetate before injection. Extracted ion chromatograms (m/z 120 + m/z 136) were used to identify possible PAs. Quantification was estimated by comparison with retrorsine-spiked e control samples and reported as µg/g retrorsine equivalents (Table 1).
Comparative quantities of pyrrolizidine alkaloids (PAs) in the dried, milled plant material analyzed in the current study.*
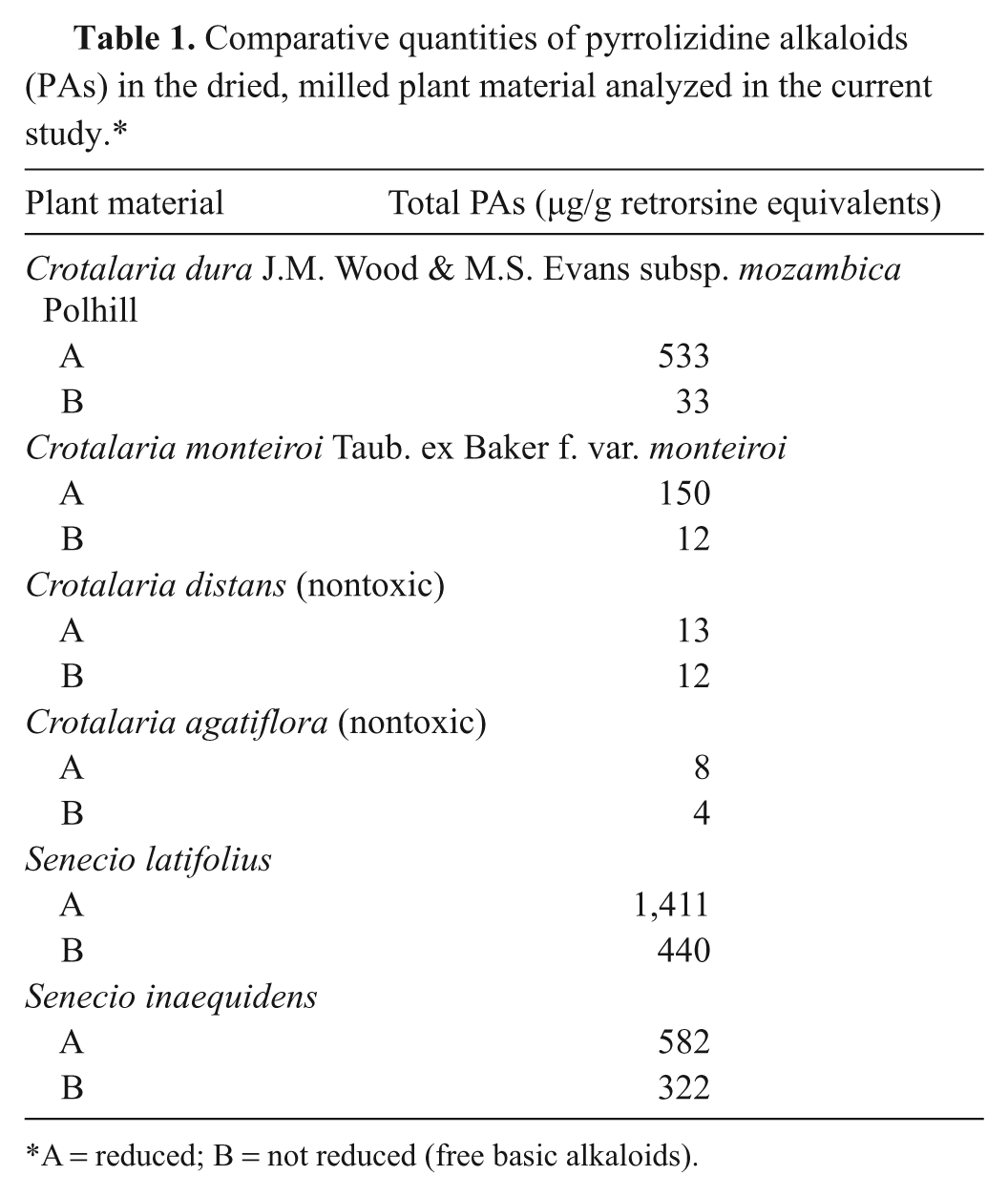
A = reduced; B = not reduced (free basic alkaloids).
Microscopic examination of the pulmonary parenchyma revealed multifocal to coalescing areas of moderate to severe proliferation of nonciliated bronchiolar epithelial (Clara) cells in the terminal bronchioles and associated alveolar ducts (Fig. 3A). Some of the bronchiolar lumina were occluded by the Clara cell hyperplasia and desquamation of these epithelial cells. There was also mild to severe fibrosis within the interstitium, perivascularly, as well as in alveolar walls and in pulmonary septae (Fig. 3B). Foci of fibrosis consisted of spindle-shaped to stellate cells often haphazardly arranged within a collagen matrix. Fibrotic areas were also often associated with septal and alveolar edema, and some areas had mild alveolar emphysema. In particular, alveoli exhibited a marked accumulation of proteinaceous and fibrin-rich edema, associated with scattered to numerous alveolar macrophages. In addition, there was prominent diffuse pulmonary congestion in all sections of lung examined. A few areas within the lung parenchyma also revealed mild purulent inflammation. There was also evidence of mural edema within pulmonary blood vessels. In the liver sections, mild portal lymphoplasmacytic inflammation and moderate bile stasis in the periacinar hepatocytes were observed. There was also moderate karyomegaly of hepatocytes (Fig. 4). No additional notable lesions were visible in the other organs available for examination.
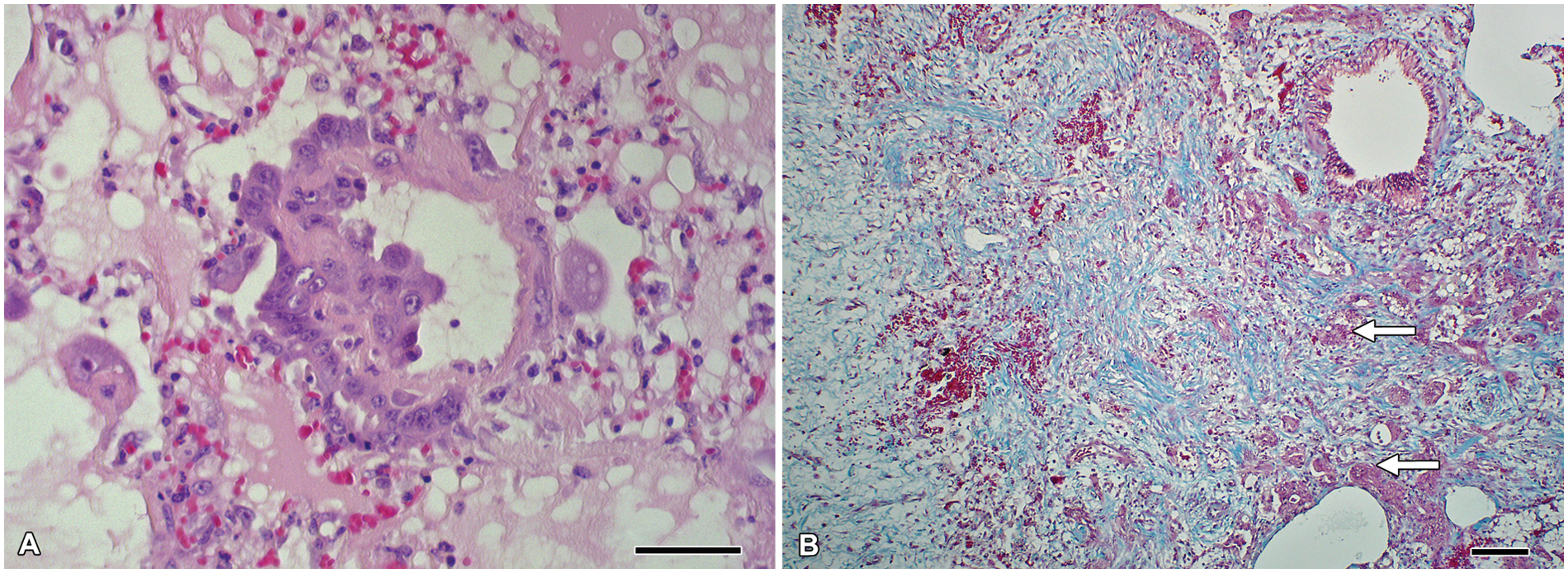
Micrograph; lung; horse.
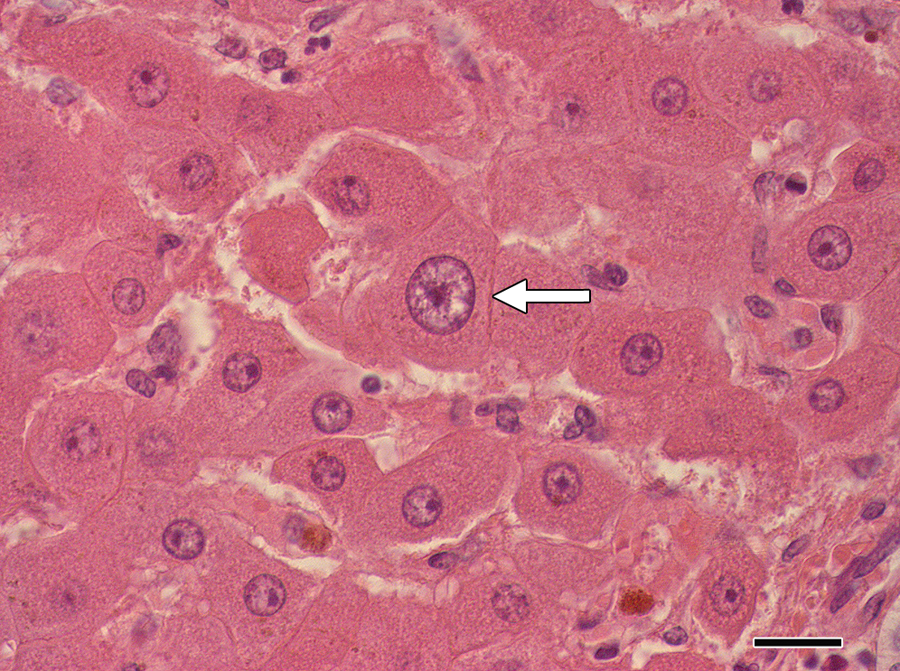
Micrograph; liver; horse. Karyomegaly of hepatocyte (arrow) and bile stasis in Kupffer cells. Hematoxylin and eosin. Bar = 20 µm.
Ultrastructurally, hyperplasia of the nonciliated epithelial cells (Clara cells) in the terminal bronchioles was the most outstanding change observed in the lungs (Fig. 5A). Clara cells with vacuolated cytoplasmic apices bulging into the lumen of the bronchioles and desquamation of Clara cells were evident (Fig. 5B). In addition, the Clara cells showed degenerative changes, such as cytoplasmic vacuolation. Some Clara cells contained a few electron-dense granules, whereas others were devoid of the granules.
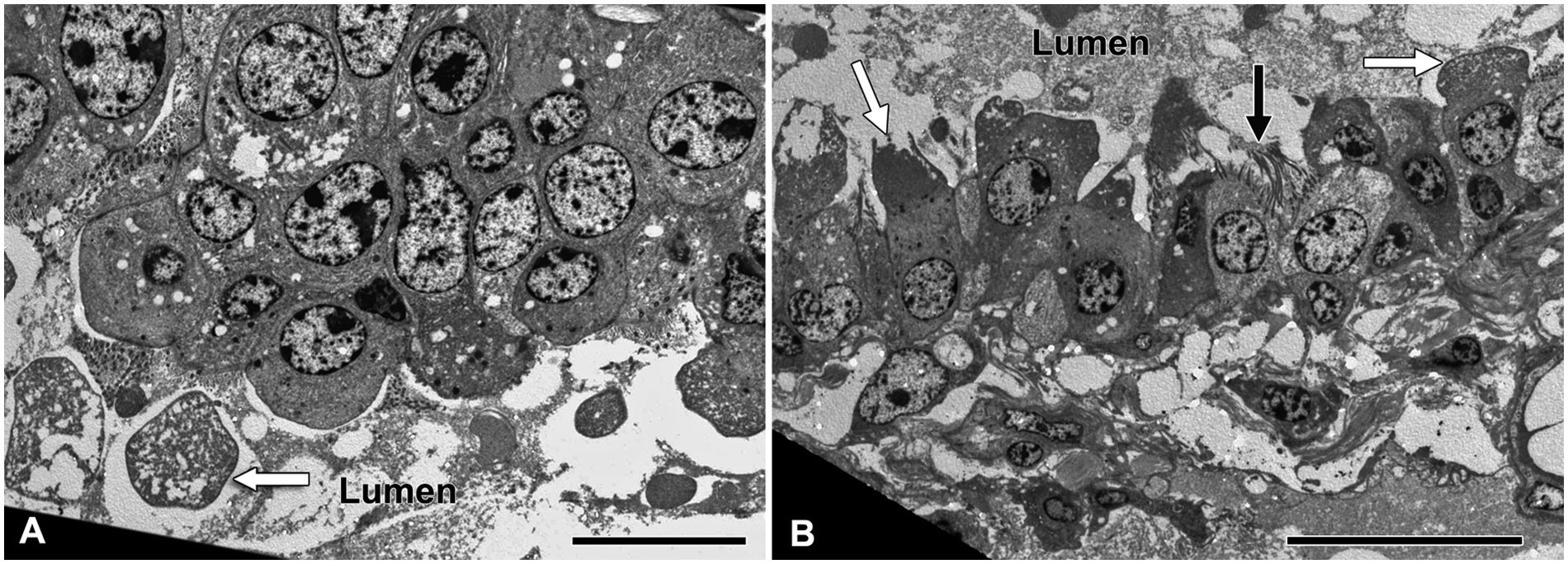
Transmission electron micrograph; lung; horse. Terminal bronchiole.
The microscopic appearance of the lung tissue, indicating chronic parenchymal injury, is suggestive of a chronic pneumotoxic condition. The proliferation of Clara cells is suggestive of intoxication by a xenobiotic metabolized via the cytochrome P-450 monooxygenase system, such as PAs and furanosesquiterpenoids.2,7,15
In southern Africa, the histological lesions observed in the lung are characteristic of equine jaagsiekte. In the current case, the presence of karyomegaly within the liver also supported exposure to PAs, which are known to induce an antimitotic effect.6,7 Thickening of the pulmonary blood vessel walls, due to mural edema, is also consistent with jaagsiekte in horses.7,13 The total PA concentration (Table 1) in Crotalaria dura mozambica appears to be high compared to the other 2 nontoxic Crotalaria species analyzed and is similar to S. inaequidens, previously reported to be toxic to cattle and rats. 4 Crotalaria dura mozambica also contains approximately 3 times more PAs compared to C. monteiroi monteiroi and is probably the main culprit causing jaagsiekte in the outbreak described herein. However, ingestion of C. monteiroi monteiroi could have had an additive effect. Based on the microscopic and ultrastructural lesions and the presence of Crotalaria species, which contained PAs, the provisional diagnosis of jaagsiekte was confirmed. The large number of horses that were reported to have died with similar clinical signs (without timely veterinary intervention) is ascribed to Mozambique being a developing country, where veterinary services are not optimal in the more remote areas.
There is a wide variation in susceptibility to PA poisoning between animal species, and the target organ affected may also differ between species. 6 Generally, with PA poisoning, the liver is primarily affected, but other organs, such as the lungs and kidneys, are also occasionally involved.6,10 Crotalaria species are known to be pneumotoxic to horses, but also to other domestic animal species such as pigs and occasionally ruminants. 6 The explanation provided for the pneumotoxic effect is that following PA metabolism and subsequent bio-activation by the liver cytochrome P-450 monooxygenase system, the pyrroles escape (“spillover”) from the liver and induce the pulmonary vascular effects and thus pneumotoxicity.6,10 In the outbreak in the current study, the liver lesions were very mild compared to the lung lesions and this begs the question of whether extrahepatic metabolic activation of the PAs could also occur (e.g., in the Clara cells located in the terminal bronchioles). 2 Thus, a more feasible explanation would be that there is spillover of unmetabolized PAs with subsequent cytochrome P-450–catalysed activation in the Clara cells located in the terminal bronchioles, leading to pulmonary injury. Although, the general consensus is that PAs are not biotransformed to toxic metabolites in the lung, 10 experiments evaluating pulmonary metabolism of PAs have been conducted in nonequid animal models.
In equine jaagsiekte, the primary injury to the Clara cells in the terminal bronchioles appears to be important in the genesis of emphysema and thereby the clinical sign of dyspnea. The multilayered proliferation, apical bulging, and desquamation of the Clara cells lead to partial obstruction of the terminal bronchioles. Inspired air may be able to pass through the narrowed bronchiolar lumen into the alveoli more readily than in the opposite direction, consequently creating a “valve effect” and overinflation of the alveoli.
Severe lung lesions in horses have also been reported following ingestion of other Crotalaria species. Crotalaria crispata, also implicated as a cause of Kimberley horse disease in northern Australia, induces severe pulmonary lesions, notably emphysema, desquamation of epithelium, and interstitial fibrosis. 5 In Brazil, 20 horses died following ingestion of a ration containing 40% C. juncea seeds. 12 Microscopically, a diffuse fibrosing alveolitis, alveolar epithelialization, areas of emphysema, and hyperplastic bronchiolar epithelium with excessive mucus secretion were observed. 12
Another rare plant-induced respiratory intoxication in horses in southern Africa has been reported from Zimbabwe. Ingestion of Ageratina adenophora (crofton weed) caused frequent coughing and signs of chronic emphysema. On postmortem examination, marked pulmonary fibrosis was observed. 1
In southern Africa, severe respiratory signs can also be caused by certain viruses and bacteria, notably by African horse sickness virus, which should always be excluded from the list of differential diagnoses, taking into account epidemiological data as well as macro- and microscopic pathology. 3 Horses that are regularly stabled could contract a condition referred to as recurrent airway obstruction (heaves or broken wind). In this disease, affected horses develop bronchoconstriction, due to inhalation of airborne allergens such as fungal spores and dust mites, and present with severe respiratory distress. 11
After due consideration of the other potential respiratory diseases of horses in southern Africa, the diagnosis of Crotalaria species poisoning in the herd of horses was based on the presence of the plants in the field, the observation of severe respiratory distress, and typical macroscopic and histological findings.
Footnotes
Acknowledgements
The authors thank Dr. Magda Rösemann, National Horse Racing Authority, Johannesburg for the pyrrolizidine alkaloid analysis; Prof. Braam van Wyk, University of Pretoria, and Dr. Marianne le Roux, University of Johannesburg, for plant identification; Prof. Ben-Erik van Wyk, University of Johannesburg, for the photographs of the 2 Crotalaria species; Mrs. Erna van Wilpe, Faculty of Veterinary Science, University of Pretoria, for the electron microscopy; and Mrs. Charmaine Vermeulen, Faculty of Veterinary Science, University of Pretoria, for reformatting the images.
a.
CM10 TEM, Philips Healthcare, Best, The Netherlands.
b.
Merck (Pty) Ltd, Johannesburg, South Africa.
c.
HP 6890, Agilent Technologies, Johannesburg, South Africa.
d.
HP-5MS, J&W Scientific supplied by Agilent Technologies, Johannesburg, South Africa.
e.
Sigma-Aldrich Chemie GmbH, Steinheim, Germany.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation, South Africa.
