Abstract
Canine distemper is uncommon in the pet trade in the United States, in large part due to effective vaccines against Canine distemper virus (CDV). This is a report of CDV affecting 24 young dogs of multiple breeds shortly after sale by 2 pet stores in Wyoming during August–October 2010. Cases were diagnosed over 37 days. Diagnosis was established by a combination of fluorescent antibody staining, reverse transcription polymerase chain reaction, negative stain electron microscopy, and necropsy with histopathology. Viral hemagglutinin gene sequences were analyzed from 2 affected dogs and were identical (GenBank accession no. JF283477). Sequences were distinct from those in a contemporaneous unrelated case of CDV in a Wyoming dog (JF283476) that had no contact with the pet store dogs. The breeding property from which the puppies originated was quarantined by the Kansas Animal Health Department. Puppies intended for sale were tested for CDV. Canine distemper was diagnosed on site in November 2010. At that point 1,466 dogs were euthanized to eliminate dispersal of the disease through commercial channels. The investigation underscores the risks inherent in large-scale dog breeding when vaccination and biosecurity practices are suboptimal.
Canine distemper is generally considered the most contagious disease of domestic dogs.8,9 The disease has become rare in dogs in the United States due largely to effective vaccines. 12 The disease persists because of persisting wildlife reservoirs of Canine distemper virus (CDV; order Mononegavirales, family Paramyxoviridae, subfamily Paramyxovirinae, genus Morbillivirus), particularly in raccoons and skunks. 13 Canine distemper is most likely to affect dogs when they are crowded, stressed, immunologically susceptible, and directly or indirectly exposed to infected wildlife (Hurley, KF: 2005, Canine distemper in the shelter: lessons learned from a Chicago outbreak. Animal Sheltering, Sept/Oct: 39–43). 13 Dogs are most at risk when not routinely vaccinated in accordance with established guidelines, 12 and are vulnerable in shelters and dog rescue organizations.6,12 The occurrence of highly contagious diseases such as canine distemper in wholesale and retail commercial establishments is poorly documented in the veterinary literature. 5 The current study documents canine distemper in puppies purchased from 2 pet stores in Wyoming. Most or all affected dogs originated from a single large-scale, out-of-state breeding operation where canine distemper was subsequently diagnosed.
Over 37 days (August 31–October 6, 2010), canine distemper was confirmed in 24 young dogs of multiple breeds from 4 Wyoming towns (Table 1). Affected puppies originated from 2 Wyoming pet stores approximately 180 miles apart; the stores had 1 owner. All but 2 dogs came from a single breeding facility in Kansas and were imported into Wyoming without veterinary health certificates. The source of the 2 remaining dogs was unknown. The Wyoming state veterinarian was informed of a large cluster of canine distemper cases involving pet stores. It was unusual to see a large cluster of laboratory-confirmed cases over a short period in Wyoming. A review of archival cases of canine distemper in the Wyoming State Veterinary Laboratory (WSVL; Department of Veterinary Sciences, University of Wyoming, Laramie, Wyoming) over the previous 11 years (2000–2010) found an average of 5.4 cases annually (range: 0–11/year). Diagnosed cases of canine distemper tend to affect unvaccinated or poorly vaccinated rural dogs, most likely in contact with wildlife, and many originating in the Wind River Indian Reservation (west central Wyoming).
Canine distemper outbreak linked to 2 Wyoming pet stores (August–October 2010).*
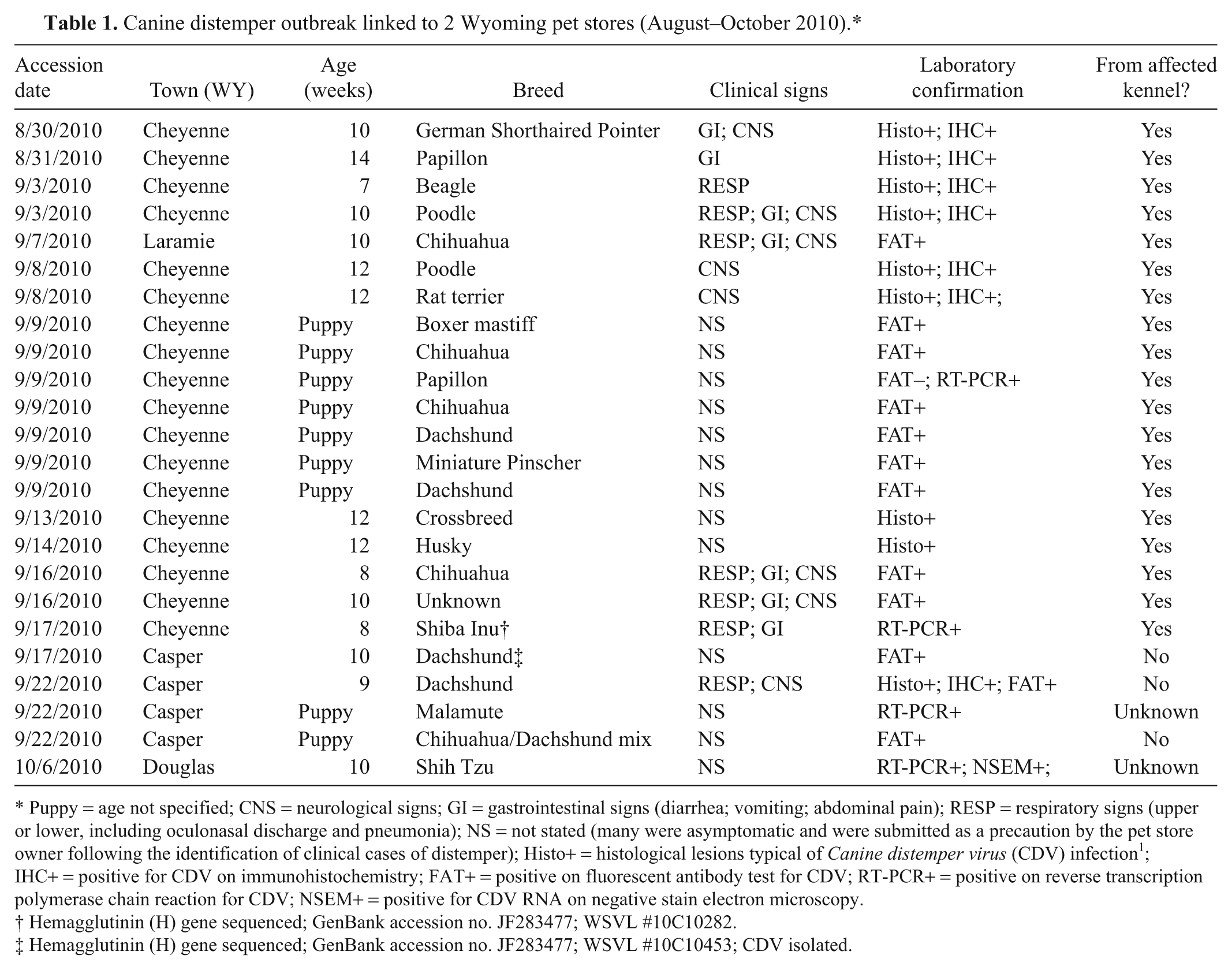
Puppy = age not specified; CNS = neurological signs; GI = gastrointestinal signs (diarrhea; vomiting; abdominal pain); RESP = respiratory signs (upper or lower, including oculonasal discharge and pneumonia); NS = not stated (many were asymptomatic and were submitted as a precaution by the pet store owner following the identification of clinical cases of distemper); Histo+ = histological lesions typical of Canine distemper virus (CDV) infection 1 ; IHC+ = positive for CDV on immunohistochemistry; FAT+ = positive on fluorescent antibody test for CDV; RT-PCR+ = positive on reverse transcription polymerase chain reaction for CDV; NSEM+ = positive for CDV RNA on negative stain electron microscopy.
Hemagglutinin (H) gene sequenced; GenBank accession no. JF283477; WSVL #10C10282.
Hemagglutinin (H) gene sequenced; GenBank accession no. JF283477; WSVL #10C10453; CDV isolated.
Affected puppies were of multiple breeds and 8–14 weeks old (Table 1). Clinical signs were reported on accession forms for 11 of the 24 dogs, and involved respiratory (8 dogs), gastrointestinal (6), and neurological (8) systems. No clinical history was provided for the remaining 13 puppies. Some of the latter were asymptomatic at the time of sampling and were submitted by the pet store owner as a precautionary measure. In an effort to contain testing costs, samples from live dogs were screened initially by fluorescent antibody test (FAT) of conjunctival smears. a Buffy coat leukocytes from FAT-negative puppies were then tested by reverse transcription polymerase chain reaction (RT-PCR). Some diagnoses were corroborated by virus isolation or negative stain electron microscopy (examination for viral nucleocapsids). Postmortem diagnosis was made on the basis of characteristic lesions 1 and a combination of FAT, RT-PCR, histopathology, and immunohistochemical staining for CDV antigen using commercially available polyclonal antiserum b and a commercial detection kit. c Nucleotide sequence analysis of the CDV hemagglutinin (H) gene was performed using brain from 2 puppies (1 from each of 2 stores). The H gene was chosen for sequencing due to its value in identifying sublineages for geographical grouping. 7 Sequences were compared to those in a conjunctival swab that was taken in late 2010 from an adult Wyoming dog. The animal was 1 year old, came from the Wind River Indian Reservation, had typical clinical signs of canine distemper, and had no known contact with the pet store cases described herein. RNA was extracted in accordance with manufacturers’ instructions from tissue, d blood, e or ocular swabs. f RNA was eluted in nuclease-free water, and stored at −80°C until used. The H gene was amplified in 3 overlapping segments from 5 µl of RNA template by RT-PCR using a commercial kit. g Primer sequences are listed (Table 2) and yielded product bands of the expected size. The PCR products purified using a commercial clean up kit h were submitted for sequencing to Clemson University Genomics Institute (Clemson, South Carolina) using forward and reverse PCR primers for each amplicon. Nucleotide sequences were aligned with the help of commercial software. i The H gene sequences of the 2010 Wyoming CDV strains were compared phylogenetically to the Onderstepoort and Snyder Hill strains, as well as 8 recent U.S. viral isolates. Alignment analysis was performed using nucleotide and deduced amino acid sequences.
Oligonucleotide primers used for amplification of the Canine distemper virus (CDV) H gene.
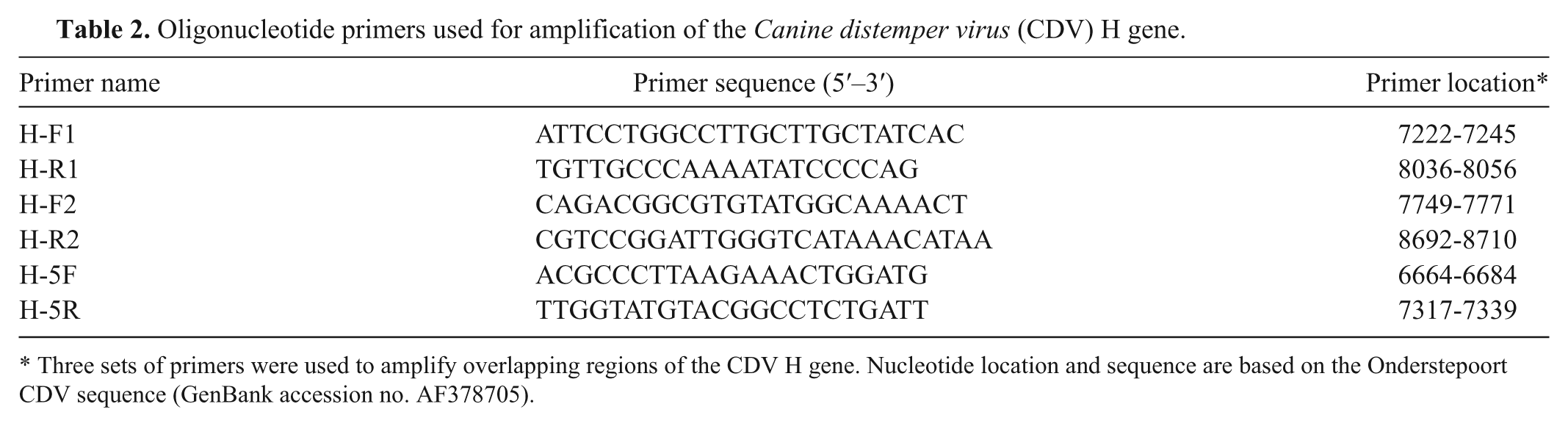
Three sets of primers were used to amplify overlapping regions of the CDV H gene. Nucleotide location and sequence are based on the Onderstepoort CDV sequence (GenBank accession no. AF378705).
Overlapping amplification products were of the expected size and were sequenced directly. Assembly of segments gave a 1,615-bp sequence representing 85% of the H gene, including the protein coding start site. Sequences from the 2 pet store puppies were identical and were deposited in the National Center for Biotechnology Information database as one accession (GenBank JF283477). The sequence from the geographically separate Wyoming dog (JF283476) had 94.8% nucleotide and 95.5% amino acid identity with the pet store sequence. Phylogenetic analysis indicated that the pet store CDV H gene sequence had the highest nucleotide identity (98.2%) with a CDV isolated in 1992 from a Chinese leopard (Panthera pardus japonensis) in a California zoological collection 4 (Z54156; Fig. 1). The ranch dog CDV H gene nucleotide sequence was most closely related (99% identity) to a 2004 isolate from a dog in Missouri (AY964110; Fig. 1). 10 The Wyoming CDV sequences were not closely related to Onderstepoort (AF378705) or Snyder Hill strains (AF259552), both of which are commonly used in modified live vaccines (Fig. 1). Similar levels of identity were obtained by phylogenic analysis of the predicted amino acid sequences.
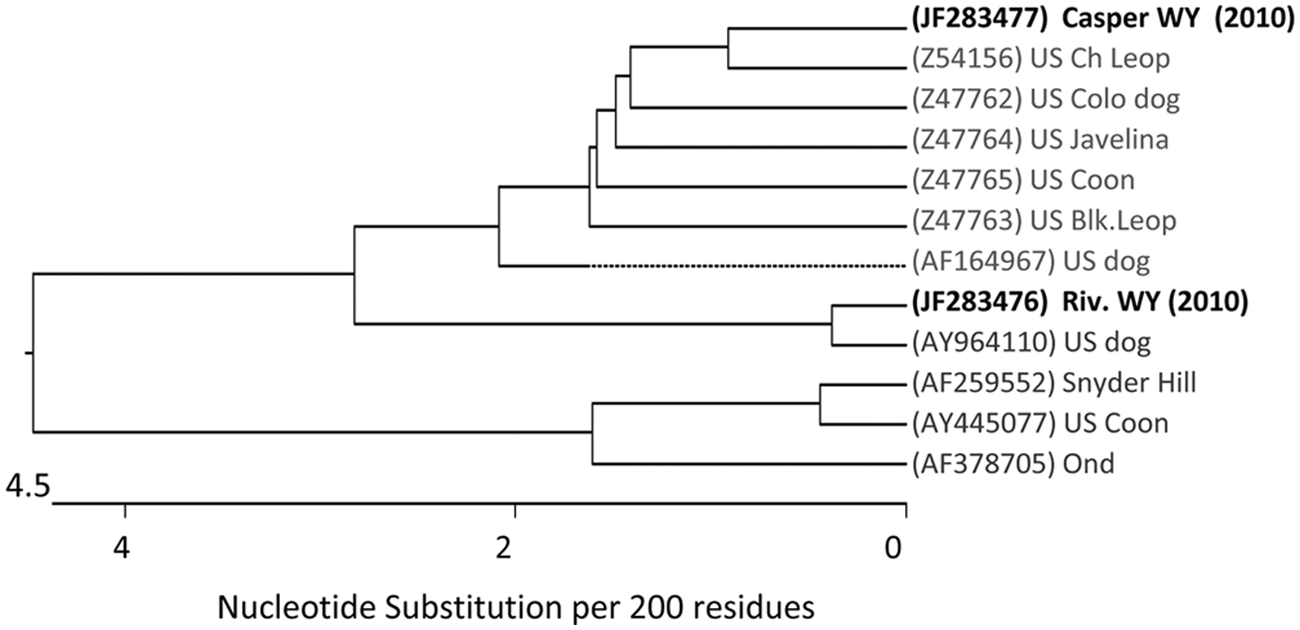
Phylogenic tree showing relationship of 2010 Wyoming Canine distemper virus isolates and other U.S. isolates, based on H gene nucleotide sequence. Distance values were calculated using MegAlign software i by the ClustalW method, and using the multiple alignment parameters of gap penalty; 15, delay divergent sequences; 30%, and DNA transition weight; 0.50. Sequences comparisons are from the public database and are identified by GenBank accession numbers in parentheses.
The Wyoming state veterinarian informed his Kansas counterpart about canine distemper in dogs purchased from pet stores in Wyoming, and that most or all originated from a Kansas breeding facility. The federally licensed and inspected facility was established in 1991. Its 5-acre site contained approximately 1,400 dogs of multiple pure and mixed breeds. There was no documented history of canine distemper on site. The Kansas property was inspected by personnel with the Kansas Animal Health Department (KAHD) as a result of the regulatory contact. Dogs were housed in various types of outdoor pens with or without runs, and in prefabricated dog kennels and converted semi-trailers. Most stud dogs were co-housed in a single pen and were seldom moved. No complete perimeter fence existed to exclude wildlife. Multiple inspections by KAHD between September and December 2010 resulted in the identification of several problems, including limited written records of vaccine administration, illness, or treatment, limited quarantine space, dogs without food, no extra food in storage, watering units at the wrong height, inadequate pen space, introduction of purchased dogs of unknown health status from multiple sources including pet auctions, transportation of dogs in inadequately disinfected crates, and recurrent contact between facility dogs and local wildlife including raccoons and skunks. The owner’s immunization strategy involved vaccinating litters of puppies, as well as dams after their litters were weaned. This resulted in little or no booster vaccination of dams that did not whelp. Stud dogs were not vaccinated. The owner maintained no written records of the pregnancy, vaccination, or health status for individual dams. Similar problems were documented by USDA inspectors over a 4-year period (2006–2010). These included no health certificates for dogs shipped out of state, dog housing in disrepair, inappropriate living conditions, and dogs in need of veterinary care.
The facility was quarantined by KAHD due to a concern that canine distemper might be present. The KAHD required the facility’s current veterinarian to collect pooled nasal swabs to test for canine distemper from all consignments of puppies. Requirements for lifting the quarantine included provisions for veterinary care, and the development of written protocols for vaccination, recordkeeping, and medical treatment. No protocols were submitted to KAHD by an October 2010 deadline. Nasal swabs were tested for CDV by RT-PCR at the Nebraska Veterinary Diagnostic Center (Lincoln, Nebraska). All pools were test-negative for canine distemper until November 2010, at which time 3 of 6 pools comprising 28 dogs tested positive. The Nebraska laboratory attempted unsuccessfully to isolate CDV. In mid-November, the breeder’s veterinarian submitted a dead Vizsla puppy to the Nebraska laboratory. Canine distemper was diagnosed. In December 2010, KAHD collected individual samples from 28 dogs, 1 of which tested positive for CDV by RT-PCR at the Kansas State Veterinary Diagnostic Laboratory (Manhattan, Kansas). The KAHD decided to depopulate the breeding facility because the owner had no place in which sick dogs could be isolated, individual animal records were lacking, and it was not possible to safely sell or otherwise relinquish dogs exposed to CDV. Personnel with KADH euthanized 1,466 dogs in December 2010. The department successfully adopted out 50 nonpregnant canine distemper–vaccinated adult dogs after quarantine. No canine distemper developed subsequently in the adopted dogs.
Several features suggest the Wyoming canine distemper episode originated in Kansas. There was no documentation that puppies were healthy when purchased by the pet store owner, as they were introduced into Wyoming without a veterinary health certificate. Disease was identified in some puppies at the pet stores or shortly after retail sale. There was no obvious source of the disease in the stores, such as contact with wildlife or with other infected dogs. The hemagglutinin sequences obtained from 2 dogs during the outbreak were identical, and distinct from those in a canine distemper–infected Wyoming dog that had no contact with pet store animals. Canine distemper was subsequently diagnosed in the source property in Kansas. Several months of testing were required to identify the disease on the property, presumably because most litters were protected by vaccination. It was not possible to compare the pet store strain to those circulating in the breeding facility, as no sequencing was done using samples from the dogs with canine distemper in Kansas. In hindsight, it is likely that some puppies were incubating the disease when shipped to Wyoming, and that infection originated in the Kansas facility.
The WSVL used a two-step method to screen samples from suspect and in-contact dogs (FAT, followed by RT-PCR). This was an effective way to keep testing costs low for the pet store owner, veterinarians, and animal owners. Multiple assays are available to detect canine distemper in dogs,1,2,6,8,9 but few recommendations exist about which assay makes the most diagnostic and economic sense when multiple dogs are exposed. Reverse transcription PCR on buffy coat samples is used in some outbreaks as the primary diagnostic method (Hurley KF: 2005, Canine distemper in the shelter). 9 In the current episode, it is likely that the owner of the pet store as well as some private owners would have declined testing due to the price of performing RT-PCR. Fluorescent antibody testing detected most, but not all, live infected dogs, with the remainder detected by RT-PCR. Although less sensitive, FAT has the advantages of being fast and inexpensive. Unlike RT-PCR, 9 FAT carries little risk of false-positive results in recently vaccinated puppies.
Little peer-reviewed literature exists to guide veterinarians managing contagious diseases in large-scale dog-breeding facilities. As of 2008, there were 4,604 federally licensed pet breeders and 1,116 pet brokers. 11 Such numbers understate the size of the industry due to the minimum number of dogs or breeder income required for licensure under the 1970 Animal Welfare Act. Published recommendations for vaccination and control of canine distemper outbreaks in public and private animal shelters can be adapted for outbreaks in breeding facilities.6,12 Large commercial operations pose particular challenges to the veterinary profession and regulatory personnel. Many operate in rural areas with facilities that are less than ideal.3,6,11 The constant turnover of animals in breeding operations makes resident dogs vulnerable to contagion, particularly when no policy exists to isolate and vaccinate animals on entry. Facilities at the source property in Kansas were used to maximum capacity. This provided little leeway to separate unexposed dogs from infected, exposed, and at-risk dogs. Such separation helps control canine distemper in animal shelters. 6
Federal reports document multiple disease and welfare problems associated with some large-scale dog-breeding facilities in the United States. 11 A strategy used by many shelters when confronted with outbreaks of canine distemper is to move at-risk dogs to adopters with rescue organizations or with other shelters. 6 This is difficult to apply when private breeders are involved, given the desire of the latter for animals to be returned at the end of outbreaks, and the controversial nature of large-scale dog breeding.3.6,11 The cost of testing was a major consideration in deciding the fate of >1,400 at-risk dogs. Continuous disease surveillance using diagnostic laboratories is less likely in commercial breeding facilities than in municipal animal shelters. There may be a tendency for some breeders to miss, ignore, or hide evidence of outbreaks of diseases like canine distemper. 11 Signs of the disease, particularly when endemic in partly vaccinated populations, can present as nonspecific upper respiratory disease, or as death in young puppies (Hurley KF: 2005, Canine distemper in the shelter).1,2,9 An estimated 25–75% of dogs with canine distemper do not show clinical signs of disease. 9 The principal vaccination strategy at the Kansas facility involved administering vaccines to dams after litters were weaned, and of vaccinating litters. The absence of an effective means to exclude wildlife from the Kansas facility put resident animals at risk of infectious diseases, including canine distemper.
Canine distemper was once the most common and serious infectious disease of dogs.8,9 The disease became uncommon in much of the United States due to commercial vaccines that generate long-lived immunity. 12 The episode described herein underscores the ease with which pet stores and commercial breeding establishments may disperse canine distemper during outbreaks.
Footnotes
Acknowledgements
The authors acknowledge Marce Vasquez for help with virology, Paula Jaeger and Mercedes Thelen for histology preparations, and Carol Hearne for electron microscopy.
a.
Canine distemper virus (CDV) FITC FA Conjugate, VMRD Inc., Pullman, WA.
b.
Canine distemper virus (CDV) antisera (caprine origin), VMRD Inc., Pullman, WA.
c.
LSAB+ kit, Dako North America Inc., Carpinteria, CA.
d.
Qiagen RNeasy mini kit, Qiagen GmBH, Hilden, Germany.
e.
QIAamp RNA Blood Mini Kit, Qiagen GmBH, Hilden, Germany.
f.
TRIzol LS Reagent, Invitrogen Corp., Carlsbad, CA.
g.
Superscript One-Step RT-PCR Kit, Invitrogen Corp., Carlsbad, CA.
h.
ExoSAP-IT, GE Healthcare, USB Corp., Cleveland, OH.
i.
MegAlign software, Lasergene 8; DNAStar Inc., Madison, WI.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Part of this work was supported by the USDA Animal Health program WYO-453-10.
