Abstract
Bovine isolates of Mannheimia haemolytica, Pasteurella multocida, and Histophilus somni, collected from 2000 to 2009, were tested for in vitro susceptibility to ceftiofur, penicillin, danofloxacin, enrofloxacin, florfenicol, tetracycline, tilmicosin, and tulathromycin. Ceftiofur remained very active against all isolates. Penicillin retained good activity against P. multocida and H. somni isolates with no appreciable changes in susceptibility or minimal inhibitory concentration (MIC) distributions with time. While there was no obvious trend, the percent of M. haemolytica that were susceptible to penicillin ranged from 40.9% to 66.7%. Danofloxacin MIC50 and MIC90 values for M. haemolytica and P. multocida did not change beyond a single dilution over the 6 years it was included in the testing panel. The MIC90 for H. somni increased beyond 1 dilution. Enrofloxacin MIC50 values for the 3 pathogens also did not change over time, unlike the MIC90 values, which increased by at least 4-doubling dilutions. Ninety percent or more of M. haemolytica and H. somni isolates were susceptible to florfenicol, while susceptibility among P. multocida was 79% or greater. Less than 50% of the isolates tested as susceptible to tetracycline in many of the years. All 3 organisms showed declines in tilmicosin and tulathromycin MIC50 and MIC90 values over the years in which they were tested.
Introduction
Bovine respiratory disease (BRD) is a significant cause of morbidity and mortality among beef cattle in North America and elsewhere. The U.S. Department of Agriculture (USDA) survey of beef cattle operations in 1999 estimated that BRD was the most common cause of illness in U.S. feedlots, and 14.4% of cattle developed BRD after arrival at the feedlot (USDA: 2000, Feedlot ‘99. Part III: Health management and biosecurity in U.S. feedlots, 1999. Available at: http://www.aphis.usda.gov/animal_health/nahms/feedlot/downloads/feedlot99/Feedlot99_dr_PartIII.pdf. Accessed on July 2, 2012). In addition to animal welfare concerns, the economic impact of BRD is also substantial (USDA: 2000).19,20 Pasteurella multocida, Mannheimia haemolytica, and Histophilus somni have been identified as major bacterial etiologic agents for this disease complex.6,10
However, in spite of extensive study of cattle with BRD, there is still much that is not clear about the exact pathogenic role of these bacteria or the factors that contribute to development of disease. 21 Management of BRD includes the use of vaccines and antimicrobial drugs 7 and, in North America, there are a number of antimicrobial products that have approved labels for the treatment of this disease. At the beginning of the current study, the most recent data on antimicrobial usage on U.S. feedlots was the USDA Feedlot ’99 study (USDA: 2000). In that national survey, florfenicol was used most frequently (54%) by U.S. feedlots as part of the initial treatment of BRD, followed by tilmicosin, tetracyclines, cephalosporins, fluoroquinolones, and penicillins. Continued susceptibility of the BRD pathogens to these drugs was therefore important.
Exposure of bacteria to antimicrobial drugs has the potential to exert pressure for selection of resistant organisms and, in order to retain efficacy, veterinarians and producers are advised to practice good antimicrobial stewardship. The U.S. Food and Drug Administration (FDA), along with the American Veterinary Medical Association, have developed guidelines for beef cattle veterinarians on the judicious use of antimicrobial drugs (FDA: 2002: Judicious use of antimicrobials for beef cattle veterinarians. Available at: http://www.fda.gov/downloads/AnimalVeterinary/SafetyHealth/AntimicrobialResistance/JudiciousUseofAntimicrobials/UCM095568.pdf. Accessed on July 2, 2012). Similar guidelines have been published in Canada. 3 As a component of good antimicrobial stewardship, antimicrobial susceptibility trends should be monitored over time (FDA: 2002); however, to the authors’ knowledge, there have been no published, national surveys of the antimicrobial susceptibility of bovine respiratory disease pathogens most commonly found in North America since a 1988–1992 survey published in 1994. 25
The current report aims to fill the need for published data on the antimicrobial susceptibility of BRD pathogens in North America. The objective of the present study was to detect any changes of in vitro susceptibility among major BRD pathogens, isolated from pre- and postmortem bovine specimens across the United States and Canada, between 2000 and 2009.
Materials and methods
Participating laboratories
Twenty-four veterinary diagnostic laboratories across the United States and Canada participated in the surveillance program, although not all submitted isolates every year. Laboratories from the major beef cattle–producing areas of the United States and Canada are included in the program, and the regions from which isolates were obtained are shown in Table 1.
Isolate origin and number of isolates per year by region for a 10-year (2000–2009) study of antimicrobial susceptibility of bacteria that cause bovine respiratory disease complex—Mannheimia haemolytica, Pasteurella multocida, and Histophilus somni—in the United States and Canada.*
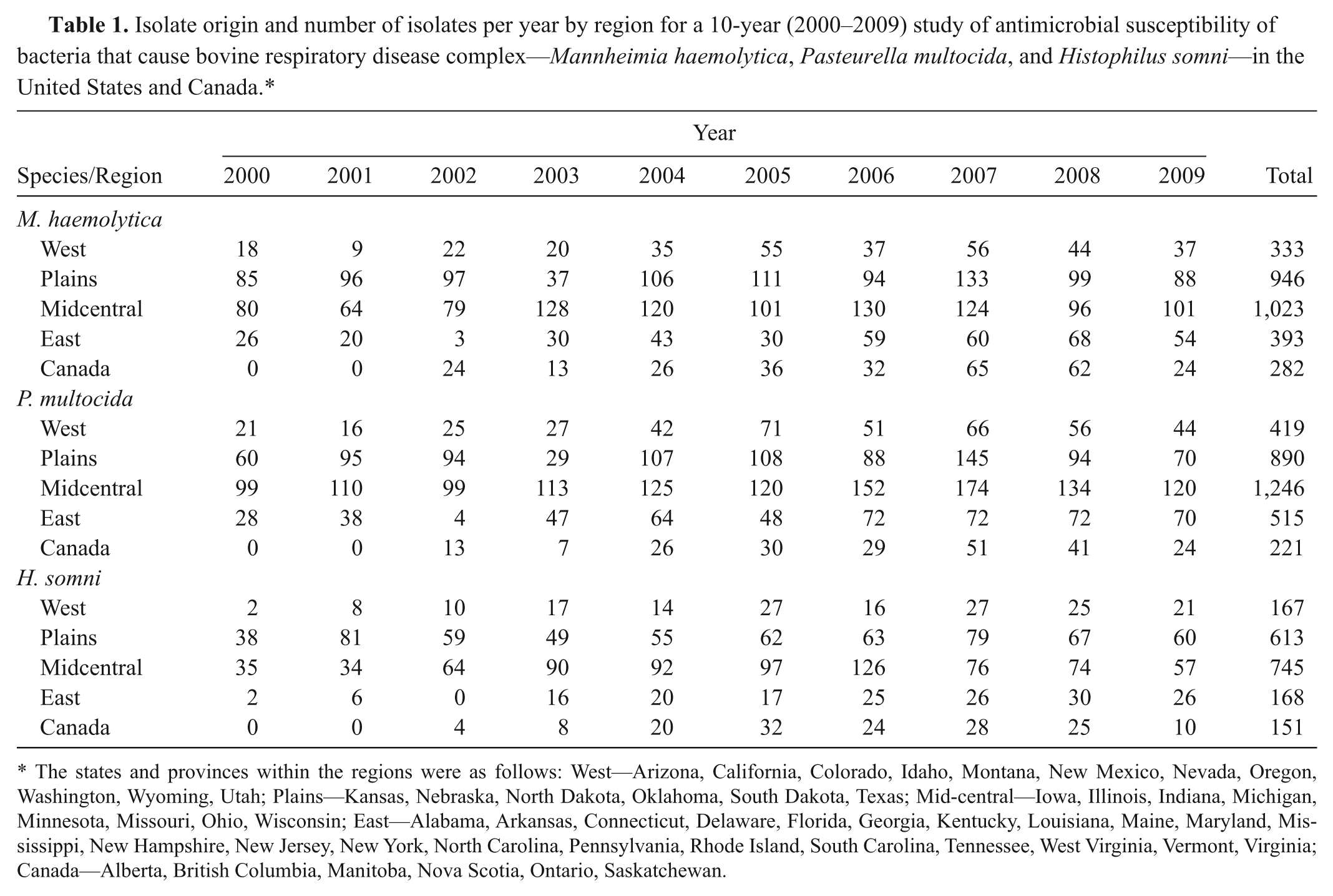
The states and provinces within the regions were as follows: West—Arizona, California, Colorado, Idaho, Montana, New Mexico, Nevada, Oregon, Washington, Wyoming, Utah; Plains—Kansas, Nebraska, North Dakota, Oklahoma, South Dakota, Texas; Mid-central—Iowa, Illinois, Indiana, Michigan, Minnesota, Missouri, Ohio, Wisconsin; East—Alabama, Arkansas, Connecticut, Delaware, Florida, Georgia, Kentucky, Louisiana, Maine, Maryland, Mississippi, New Hampshire, New Jersey, New York, North Carolina, Pennsylvania, Rhode Island, South Carolina, Tennessee, West Virginia, Vermont, Virginia; Canada—Alberta, British Columbia, Manitoba, Nova Scotia, Ontario, Saskatchewan.
Bacterial isolates
Mannheimia haemolytica, P. multocida, and H. somni were requested from veterinary diagnostic laboratories for this program. All bacteria had been isolated from diseased or deceased animals, although there was no information available on either the age or any recent antimicrobial treatment administered to the animals from which the bacteria were isolated. The isolates were selected by the laboratories, but the laboratories were requested not to use susceptibility as a criterion for submission. In order to limit overrepresentation from any one geographic area, the participating laboratories were asked to submit no more than a maximum number each year. While this number changed slightly during the 10-year period, the number was ≤40 isolates of each bacterial species per laboratory per year. Starting in 2003, Pfizer Animal Health (PAH) requested that participating laboratories send no more than 1 isolate of each bacterial species from a herd or ranch each quarter-year to reduce the risk of overrepresentation of clones from localized outbreaks. Isolates were identified to the genus and species level by the submitting laboratory before shipment to PAH. Standard biochemical tests and commercially available identification systems were used to confirm or further characterize the isolates when necessary.a,b
Minimal inhibitory concentration determinations
The use of multiple laboratories to conduct susceptibility testing can introduce bias into a surveillance study.2,12 Therefore, over the 10 years, all susceptibility testing was performed in only 2 laboratories (PAH, Kalamazoo, Michigan; Microbial Research Inc., Fort Collins, Colorado) to minimize potential testing bias. There was adherence to Clinical and Laboratory Standards Institute (CLSI) standardized methods in both laboratories, 5 as well as quality control during susceptibility testing (Table 2). Minimal inhibitory concentrations (MICs) for all isolates were determined using a broth microdilution system. c At the beginning of the current study, in 2000, the custom 96-well microtiter panels included serial dilutions of the following antimicrobial agents: ceftiofur, enrofloxacin, florfenicol, penicillin, tetracycline, and tilmicosin. Two additional drugs, danofloxacin and tulathromycin, which are also indicated for the treatment of BRD, became available after the survey began and were added to the panel in 2004.
Summary of methods used for determination of minimal inhibitory concentrations in a 10-year (2000–2009) study of antimicrobial susceptibility of bacteria that cause bovine respiratory disease complex—Mannheimia haemolytica, Pasteurella multocida, and Histophilus somni—in the United States and Canada.*

CAMHB = cation-adjusted Mueller–Hinton broth; VFM = veterinary fastidious medium; ATCC = American Type Culture Collection. Methodology obtained from Clinical and Laboratory Standards Institute document M31-A3.
Concentration ranges for each antimicrobial agent were chosen to encompass appropriate quality control ranges and clinical breakpoints, wherever possible. In some years, the range of drug concentrations was altered or drugs were removed from the panel, as in 2004, when enrofloxacin was not included in order to accommodate additional antimicrobial agents in the 96-well microtiter plates. Direct colony suspensions were used when testing all organisms, and suspensions were prepared to yield a final concentration of bacteria of approximately 5 × 105 colony-forming units/ml. The CLSI-recommended quality control organisms were included with each day of testing (Table 2).
The CLSI has published veterinary-specific clinical breakpoints for ceftiofur, enrofloxacin, florfenicol, tetracycline, and tulathromycin and tentatively approved veterinary breakpoints for penicillin (Mark Papich, personal communication, 2011) for M. haemolytica, P. multocida, and H. somni, 5 and these are shown as bold vertical lines in Tables 3-5. The CLSI has established clinical breakpoints for tilmicosin in vitro activity against M. haemolytica, but not for P. multocida or H. somni. The drug sponsor has published a susceptible breakpoint for tilmicosin against P. multocida of ≤8 µg/ml, 18 which is the same tilmicosin breakpoint as has been published by CLSI for M. haemolytica. There are also CLSI-published breakpoints for susceptibility, but not resistance, for danofloxacin against M. haemolytica and P. multocida.
The minimal inhibitory concentration (MIC) distribution frequencies of Mannheimia haemolytica isolates from the United States and Canada, 2000–2009.*

No. = the number of isolates tested; %S = the percentage of isolates that are susceptible to the drug using Clinical and Laboratory Standards Institute (CLSI) criteria; – = no isolates with an MIC at that concentration. % MIC frequency rows may not add to 100% due to rounding. Vertical lines indicate the CLSI-approved breakpoints for susceptibility and resistance in bovine respiratory disease pathogens. The unshaded areas show the range of drug concentrations used in the microtiter panel. Numbers in the lowest concentration of the tested range represent the percentage of isolates that had MICs less than or equal to the lowest drug concentration tested that year. Numbers above the highest concentration in the tested range represent the percentage of isolates that had MICs greater than the highest drug concentration tested that year.
The minimal inhibitory concentration (MIC) distribution frequencies of Pasteurella multocida isolates from the United States and Canada, 2000–2009.*

No. = the number of isolates tested; %S = the percentage of isolates that are susceptible to the drug using Clinical and Laboratory Standards Institute (CLSI) criteria; – = no isolates with an MIC at that concentration; NA = not applicable because there are no veterinary-specific breakpoints approved by CLSI. % MIC frequency rows may not add to 100% due to rounding. Vertical lines indicate the CLSI-approved breakpoints for susceptibility and resistance in bovine respiratory disease pathogens. The unshaded areas show the range of drug concentrations used in the microtiter panel. Numbers in the lowest concentration of the tested range represent the percentage of isolates that had MICs less than or equal to the lowest drug concentration tested that year. Numbers above the highest concentration in the tested range represent the percentage of isolates that had MICs greater than the highest drug concentration tested that year.
The minimal inhibitory concentration (MIC) distribution frequencies of Histophilus somni isolates from the United States and Canada, 2000–2009.*

No. = the number of isolates tested; %S = the percentage of isolates that are susceptible to the drug using Clinical and Laboratory Standards Institute (CLSI) criteria; – = no isolates with an MIC at that concentration; NA = not applicable because there are no veterinary-specific breakpoints approved by CLSI. % MIC frequency rows may not add to 100% due to rounding. Vertical lines indicate the CLSI approved breakpoints for susceptibility and resistance in Bovine Respiratory Disease pathogens. The unshaded areas show the range of drug concentrations used in the microtiter panel. Numbers in the lowest concentration of the tested range represent the percentage of isolates that had MICs less than or equal to the lowest drug concentration tested that year. Numbers above the highest concentration in the tested range represent the percentage of isolates that had MICs greater than the highest drug concentration tested that year.
Results
Mannheimia haemolytica
The in vitro susceptibility of 2,977 M. haemolytica isolates was evaluated using CLSI-approved methods (Table 3). There were no apparent changes in MIC distribution over time of the 2 β-lactams, penicillin and ceftiofur, tested against M. haemolytica. All M. haemolytica isolates demonstrated in vitro susceptibility (MIC ≤ 2 μg/ml) to ceftiofur over the 10 years of the survey. The percentage of isolates that had penicillin MIC values at or below the susceptible breakpoint (MIC ≤ 0.25 μg/ml) varied quite widely each year, but there was no clearly identifiable movement towards higher or lower MICs over this 10-year period.
There was a decline in the percentage of M. haemolytica isolates that were susceptible to danofloxacin (Table 3). Between 2000–2003 and 2008–2009, the enrofloxacin MIC frequency distributions showed shifts towards higher MICs over time, and the MIC90 increased (Table 3). Enrofloxacin was not included in the testing panel in the years 2004–2007.
Among the M. haemolytica isolates, the annual florfenicol MIC50 values during the period 2000–2009 were 1 µg/ml, except for 2001, when the MIC50 was 1 dilution lower. The percentage of isolates that were susceptible to florfenicol declined by about 10%. Approximately 50% of all M. haemolytica isolates demonstrated in vitro susceptibility to tetracycline (MIC ≤ 2 µg/ml; Table 3).
The percentage of M. haemolytica isolates that were susceptible to the 2 macrolides, tilmicosin and tulathromycin, declined over time (Table 3). Tilmicosin and tulathromycin MICs showed appreciable shifts toward higher concentrations over the years they were tested in the current survey, and the MIC90 values reflected these changes.
Pasteurella multocida
A total of 3,291 bovine P. multocida isolates were tested between 2000 and 2009. Over the 10-year survey, there was little or no change in the MIC frequency distributions, MIC50, or the MIC90 of either penicillin or ceftiofur (Table 4). All of the P. multocida isolates demonstrated in vitro susceptibility to ceftiofur during the 10-year period.
Danofloxacin testing began in 2004 and, at that time, 88.2% of the P. multocida isolates were susceptible. Over the following 5 years, there was little change in the danofloxacin MIC50 or MIC90 (Table 4). In 2000, the enrofloxacin MIC90 was ≤0.03 µg/ml, and all of the P. multocida isolates showed in vitro susceptibility to enrofloxacin; over the next 3 years, a few resistant isolates were observed (Table 4). In 2008, when enrofloxacin was included in the testing panel again, the MIC90 values had increased to 0.5 µg/ml.
In vitro susceptibility of the P. multocida isolates to florfenicol showed minor fluctuations during the 10-year period of the survey (Table 4). There was no discernible trend, either up or down, in the MIC50 or MIC90 for tetracycline between 2000 and 2009 although the proportion of isolates that were susceptible (MIC ≤ 2 μg/ml) declined.
There was a shift over time in the frequency distribution of tilmicosin MICs among the P. multocida isolates toward the higher MICs (Table 4), and the percentage of isolates that were susceptible to tilmicosin, using the sponsor-proposed breakpoint of ≤8 µg/ml, decreased to 59.7% (percentage susceptible data not shown in Table 4). Ninety percent or more of P. multocida isolates were susceptible to tulathromycin over the 6 years that it was tested (Table 4), and the MIC90 increased 3 fold.
Histophilus somni
A total of 1,844 H. somni isolates were tested between 2000 and 2009. In vitro susceptibility to the β-lactams remained high, with no resistance to ceftiofur detected over the years, and more than 90% of isolates demonstrated susceptibility to penicillin (Table 5). The data does not indicate any consistent change in the MIC frequency distributions of ceftiofur or penicillin MIC values over the 10 years.
The danofloxacin MIC50 remained fairly constant, although the MIC90 increased over time with the MIC90 equal to 2 µg/ml in 2009. The enrofloxacin MIC50 was 0.06 µg/ml, and the MIC90 was 1.0 µg/ml in 2009. The percentage of isolates that had an MIC at or below the enrofloxacin susceptible breakpoint (0.25 µg/ml) declined from 100% in 2000 to 86.2% in 2009.
While there was little change in the florfenicol MIC50, H. somni did show a shift in the distribution of MIC values over the 10-year period (Table 5), and the proportion of isolates that were susceptible (MIC ≤ 2 µg/ml) to this drug decreased. There was also a decrease over time in the proportion of H. somni isolates that were susceptible to tetracycline, with the MIC50 increasing multiple dilutions to 4 µg/ml in 2009 (Table 5). Between 2000 and 2009, H. somni showed a shift in MICs distributions for the macrolides (Table 5), with a decline over time in the proportion of isolates that have tilmicosin and tulathromycin MICs less than or equal to 16 µg/ml (Table 5).
Discussion
Given the welfare and economic concerns associated with 14–24% of North American cattle becoming sick or dying from BRD each year,9,20 it is of considerable importance that the etiological agents of BRD remain susceptible to the antimicrobial drugs that are used to treat this disease. It is surprising then that there are so few widespread studies that have been conducted in the United States or Canada that look for changes in the susceptibility of pathogens to these antimicrobial drugs over time. 24 Indeed, there are few national large-scale studies anywhere that monitor antimicrobial susceptibility of the BRD pathogens over time. The German Federal Office of Consumer Protection and Food Safety (Bundesamt für Verbraucherschutz und Lebensmittelsicherheit [BVL]) conducts one of the few national surveillance programs for antimicrobial susceptibility among bovine respiratory pathogens22,23 and, in 2008 and 2011, published the summaries of the most recent findings (BVL: 2008, GERMAP 2008: Antibiotika-Resistenz und -Verbrauch [Antibiotic resistance and consumption]. Available at: http://www.p-e-g.org/econtext/germap. In German. Accessed on July 2, 2012; BVL: 2010, GERMAP 2010: Antibiotika-Resistenz und -Verbrauch [Antibiotic resistance and consumption]. Available at: http://www.p-e-g.org/econtext/germap. In German. Accessed on July 2, 2012). The BVL reported that no M. haemolytica isolates tested in 2004/2005 showed resistance, using CLSI interpretive criteria, to florfenicol and tulathromycin, but by 2006/2007, 2% of the 55 isolates tested were resistant to these 2 drugs (BVL: 2010). Tetracycline resistance was found in approximately 20% of M. haemolytica isolates in Germany in both 2004/2005 and 2006/2007. While this was lower than the levels that were observed in North American isolates, there was little change in the proportion of isolates that were resistant to tetracycline over the time period 2004–2007 in both surveys. The Antibiotic Resistance in Bacteria of Animal Origin (ARBAO-II) project of the European Union collected data on the antimicrobial susceptibilities of cattle pathogens 11 and has published data on M. haemolytica and P. multocida isolates collected from cattle between 2002 and 2004 from E.U. countries. The 529 M. haemolytica isolates from Denmark, France, and The Netherlands that were included in the ARBAO-II report were all susceptible to ceftiofur, while some of the isolates from France and The Netherlands showed resistance to fluoroquinolones. Tetracycline resistance was high, especially in France and The Netherlands, where greater than 20% of the M. haemolytica isolates were resistant in at least 2 years of the survey. Pasteurella multocida resistance was low for all antimicrobial drugs with the exception of tetracycline. The sampling schemes in these 2 European reports were different from each other and different to the sampling conducted in the current North American study. In addition, the E.U. studies covered different time periods, and susceptibility testing of the isolates in the ARBOA-II study was conducted in different laboratories using different methodologies. Comparisons between the results of the different surveys are therefore challenging.
There have been a few antimicrobial resistance surveys of BRD pathogens in North America, although most of the surveys tested isolates from a single state/province or region.8,26 A survey of BRD pathogens collected between 1994 and 2002 and submitted to the Oklahoma Diagnostic Laboratory reported there had been no significant changes in the susceptibility of M. haemolytica or P. multocida to ampicillin or ceftiofur, but declines were reported for susceptibility to florfenicol and tilmicosin. 26 In a report from Oklahoma on susceptibility of bacteria isolated at necropsy in 2007, 13 the isolates showed decreased susceptibility to those reported in the current study, but the isolates from that previous study came only from deceased animals and many had received 2 or more treatments for BRD.
The most recent, published, extensive survey of isolates in North America was published in 1994 25 and examined the in vitro susceptibility of BRD isolates collected from across the United States and Canada between 1988 and 1991. In that survey, 100% of the isolates of Pasteurella haemolytica (which were split, in 1999, into multiple Mannheimia species including Mannheimia haemolytica), P. multocida, and Haemophilus somnus (now Histophilus somni) were susceptible to ceftiofur. Penicillin was not included in that survey, although the survey did include ampicillin and found that 39.5% of P. haemolytica, 11.9% of P. multocida, and 9.9% of H. somnus isolates were resistant to this drug.
In the current study, ceftiofur remained very active against the BRD pathogens, and there was no appreciable change in penicillin MICs over the 10 years of the program. The fluoroquinolones, like the cephalosporins, are powerful and important antimicrobial agents, and the majority of BRD isolates that were tested between 2000 and 2009 demonstrated susceptibility to danofloxacin and enrofloxacin, although there was a slow decline in the percentage of isolates that were susceptible.
There were also decreases in susceptibility to florfenicol among all 3 pathogens, although the majority of isolates were susceptible to the drug. While there was quite a variation in the levels of susceptibility to tetracycline among the M. haemolytica and P. multocida isolates each year, there did not appear to be any substantial changes in the MIC distributions.
The percentage of M. haemolytica isolates that were susceptible to tilmicosin dropped from 89.4% in 2000 to 59.5% in 2009. As breakpoints have not been established by CLSI for tilmicosin against P. multocida and H. somni, it is not possible to determine any decrease in susceptibility, but there was a shift in the MIC frequency distributions towards higher MIC values suggesting that more P. multocida and H. somni isolates may become resistant to tilmicosin. An increase in the percentage of isolates that had tulathromycin MICs greater than the CLSI breakpoint for susceptibility occurred in the 6 years during which this antimicrobial drug was included in the survey. In 2004, the year before tulathromycin was approved for use in the United States, 2–6% of the BRD pathogens showed MICs that were greater than the susceptible breakpoint of 16 μg/ml and, by 2009, 6–20% of isolates had MICs greater than 16 μg/ml. Data from the current study shows that some BRD pathogens in North America were already resistant to tulathromycin at the time of its approval in the United States.
The FDA (http://www.fda.gov/downloads/AnimalVeterinary/SafetyHealth/AntimicrobialResistance/JudiciousUseofAntimicrobials/UCM095568.pdf) and Health Canada 7 have pointed out that there are many factors that can contribute to the occurrence and dissemination of antimicrobial resistant organisms. The data presented in the current study, especially the data that shows that there has been a loss of susceptibility to important antimicrobial agents, should serve to underscore the importance of prudent use of these drugs when treating BRD (and other infections). Careful antimicrobial drug stewardship is part of an overall strategy to retain effective use of these drugs for many years.
There are many important reasons for presenting MIC frequency distributions in surveillance reports. 17 Comparisons between datasets that use different breakpoints/epidemiological cutoff points, or even where no breakpoints have been established, can be made when the MIC frequencies are published. Providing this data also allows for observation of shifts in MICs that are not reflected in such calculated values such as MIC90 or percentage susceptible. While adding substantially to the length of the current report, the authors believe that there is value in including the MIC frequency distributions and that these provide more details of the dynamics of change in antimicrobial susceptibility among BRD pathogens than would be available if just the summarized values were included.
The present study was initiated to detect loss of in vitro susceptibility to commonly used veterinary drugs among 3 BRD pathogens, but not to determine prevalence rates of resistance or determine efficacy of specific antimicrobial drugs in vivo. Although standardized susceptibility testing has been used as a guide by practitioners for over 50 years, the response among individuals to drug treatment is complicated by many factors that play a role in determining efficacy.1,14,24 It is also important to note that many of the isolates in this 10-year study have been collected and sent to state laboratories after treatment with antimicrobial drugs had failed and so this survey may be biased and may reflect a more resistant population than may be found in the broader pathogen population. In spite of these limitations, the data may, however, provide a warning of the occurrence of isolates that show loss of in vitro susceptibility, which may indicate the potential for loss of efficacy in certain populations. Furthermore, it is recognized that multidrug resistance is an increasingly important characteristic of bacterial pathogens, and there are some studies showing the emergence of multidrug resistance among bovine pathogens,4,16 but analysis was not performed to determine coresistance patterns among the isolates from this 10-year study.
As with any surveillance program, the present survey has its limitations and potential biases. The data in this program came from over 8,000 clinical bovine isolates and, while this is a substantial number, it is only a small representative sample of the BRD pathogen population in the United States and Canada.
A number of authors have highlighted the challenges of surveillance systems and the potential biases that may be encountered.12,15 As the isolates in the current study originated from many veterinary diagnostic laboratories, the methods of sample selection, collection, and submission varied among laboratories. The design of the survey, including limits on the number of isolates collected within a given time period from a single ranch or herd and from a single diagnostic laboratory, can help reduce, but not eliminate selection bias. The use of just 2 laboratories to perform the MIC testing minimized potential testing bias, and both laboratories adhered strictly to standard microbiological methods for susceptibility testing and quality control standards.
The current report provides an extensive survey of the antimicrobial susceptibility of major BRD pathogens isolated from cattle in the United States and Canada, during the years 2000–2009. The data shows that M. haemolytica, P. multocida, and H. somni exhibit in vitro resistance to some important antimicrobial drugs that are frequently used to treat this disease. In other instances, there was little change in the proportions of susceptible isolates or the MIC distributions with time. Continued surveillance of antimicrobial susceptibilities will remain an important tool for monitoring and evaluating interventions designed to ensure the efficacy of veterinary antimicrobial drugs.
Footnotes
Acknowledgements
The authors would like to thank the following veterinary diagnostic laboratories for their generous assistance by providing the bacterial isolates for this study: Cornell University, Iowa State University, Kansas State University, Manitoba Agriculture Services, Michigan State University, North Carolina Department of Agriculture, Ohio Department of Agriculture, Oklahoma State University, Pennsylvania State University, Purdue University, South Dakota State University, Texas A&M (Amarillo), Texas A&M (College Station), University of California, Davis (Davis), University of California Davis (Tulare), University of Guelph, University of Illinois, University of Minnesota, University of Nebraska, University of Saskatchewan, University of Wisconsin, Washington State University, and 2 additional veterinary diagnostic laboratories who wish to not be acknowledged.
a.
API Microbial Identification kits, bioMérieux, Durham, NC.
b.
Biolog Microbial Identification System, Hayward, CA.
c.
Sensititre Division, Trek Diagnostic Systems Inc., Thermo Scientific, Cleveland, OH.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Ellen Portis, Cynthia Lindeman, and Lacie Johansen are employed by Pfizer Animal Health. Gillian Stoltman receives funding from a contract between Pfizer Animal Health and Waterwood Consulting LLC.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Pfizer Animal Health.
