Abstract
A multiplex enzyme-linked immunosorbent assay (ELISA) to detect antibodies to Bovine viral diarrhea virus (BVDV), Bovine herpesvirus 1 (BHV-1), Bovine parainfluenza virus 3 (BPV-3), and Bovine respiratory syncytial virus (BRSV) was developed on the commercially available Meso Scale Discovery (MSD) platform. The multiplex ELISA used the same antigens (relatively crude lysates of cultured virus) that are used in the current in-house separate ELISAs. Testing a panel of field samples demonstrated the multiplex assay could simultaneously detect the presence of several antibodies in a single assay and that there was good agreement between the multiplex ELISA and the in-house ELISAs. The use of a multiplex ELISA could offer significant benefits for routine serological testing through reduced staff time, reduced reagent cost, and streamlined laboratory operation.
Keywords
One of the most costly health problems faced by beef and dairy producers is bovine respiratory disease (BRD). 7 Many viruses and bacteria play a role in BRD syndrome. Several viruses have been associated with BRD such as Bovine herpesvirus 1 (BHV-1), Bovine parainfluenza virus 3 (BPV-3), and Bovine respiratory syncytial virus (BRSV). In addition, Bovine viral diarrhea virus (BVDV) has been identified in BRD outbreaks and is usually associated with other pathogens. 6 Even a mild case of BRD can set the stage for infection by other pathogens by weakening the immune system leaving the animal more susceptible to secondary infections. Symptoms of BRD include nasal and ocular discharges, fever, coughing, pneumonia, and respiratory tract lesions. 7 Mixed infections in cattle may be more severe when BVDV is present because BVDV is considered to act as an immunosuppressive by causing diminished leukocyte and lung macrophage function. 2
Serological assays, including enzyme-linked immunosorbent assays (ELISAs), provide a useful tool to screen animals for the presence of antibodies against a wide range of infectious agents (including viruses that cause respiratory disease in cattle) and are widely used in veterinary medicine to assist in the control and monitoring of disease. However, conventional ELISAs generally permit the measurement of only a single biomarker. Samples submitted to veterinary diagnostic laboratories will often be tested for a panel of disease-causing agents using several separate ELISAs, which is time-consuming, expensive, and sometimes require large quantities of sample. Multiplex tests, which allow samples to be screened for multiple targets in a single assay, could offer significant advantages for routine serological testing. Benefits include a reduction in the time and labor required and streamlined laboratory organization and would provide the basis of lower cost assays that could be used to underpin herd health, eradication, and disease surveillance efforts.
In the present study, the potential of the Meso Scale Discovery (MSD) platform to allow a multiplex ELISA to detect antibodies to BVDV, BPV-3, BRSV, and BHV-1 was examined. To assess the performance of the multiplex assay, a panel of samples, for which results from separate ELISAs were available, were tested. The results from these ELISAs and the multiplex ELISA were compared. The separate ELISAs are well-established assays3,4,9 used routinely at the Animal Health and Veterinary Laboratories Agency (AHVLA) as previously described 1 and use relatively crude viral antigen produced from lysates of virus-infected cell lines. The multiplex assay utilizes the same antigens as the parent ELISAs.
The MSD platform combines electrochemiluminescence (ECL) and microarray-style spatial gridding technology to allow the monitoring and detection of up to 10 separate assays within a single well of a 96-well microtiter plate. 5 The MSD 96-well plates are fabricated to contain separate carbon electrodes (from 1 to 10) within each well, which can be coated with different biological molecules. For the current study, the electrodes were coated with the different antigens used in the individual ELISAs. The fundamental basis of the assay is the same as a typical serology ELISA in that a series of incubation and wash steps in 96-well plate format result in the capture of antibodies in a serum sample and subsequent detection by addition of a labeled anti-species secondary antibody. In this case, the label is a proprietary MSD electrochemiluminescent molecule, SULFO-Tag™. a Generation and measurement of the electrochemiluminescent signal are achieved through electrical stimulation of the SULFO-Taga–labeled antibodies, where only labels in close proximity to the carbon electrodes are detected. Measurement of the resulting light signal from around the individual electrodes occurs in the MSD Sector Imager instrument, which effectively performs the same function as a microtiter plate reader. 8
Initial assay development and optimization of parameters such as antigen concentration, serum dilution, and washing conditions were carried out at the AHVLA laboratories in a single-plex format on the MSD platform, where the wells of the MSD 96-well plates contained a single carbon electrode. This provided the opportunity to optimize each assay individually before combining in a multiplex format.
Multiplex plates, where different antigens were adsorbed onto the individual carbon electrodes within each well, were printed by MSD using proprietary robotic printing systems. In the current study, 7-plex plates were printed, which consisted of a carbon electrode coated in each of the 4 positive antigens, and 3 electrodes coated in negative antigen (lysates of uninfected cell lines; calf kidney cells for BHV-1, BPV-3, and BRSV, and calf testes cells for BVDV). The antigens were printed following dilution of stock antigens (produced as described 1 ) in 0.01 M phosphate buffered saline buffer (pH 7.2) containing 1% octyl β-D-glucopyranoside. The dilutions used were 1/10, 1/40, 1/64, and 1/10 for BVDV, BHV-1, BPV-3, and BRSV antigens, respectively. These antigens were more concentrated than those used for coating the single-plex ELISA plate wells. 1 The printed plates were shipped to the AHVLA laboratory in a dry state for further use.
To assess the diagnostic sensitivity and specificity of the MSD multiplex assay, a panel of 148 field samples were randomly selected from samples previously submitted and tested for BVDV, BHV-1, BRSV, and BPV-3. The samples were from a range of cattle breeds farmed within the United Kingdom and included animals of both sexes and different ages.
To perform the MSD multiplex assay, 150 µl of serum/conjugate diluent (0.5 M phosphate buffer with 0.5 M NaCl, 1 mM ethylenediamine tetra-acetic acid, 1% polyvinyl pyrolidone 40, and 0.05% Tween-20, at pH 7.2) was added to each well and incubated for 1 hr at room temperature on a shaker at 400 rpm. Serum samples were diluted 1/100 in serum/conjugate diluent and 25 µl was transferred to each well and incubated for 2 hr at room temperature on a shaker at 400 rpm. The plate was washed 3 times by adding 200 µl of MSD wash buffer a and tapping onto absorbent towels to remove any excess. Polyclonal rabbit anti-bovine immunoglobulin b was diluted 1/10,000 in serum/conjugate diluent and 25 µl was added to each well and incubated for 1 hr at room temperature on a shaker at 400 rpm. Following the incubation, the plate was washed 3 times as described above, and 25 µl of goat anti-rabbit immunoglobulin, conjugated to MSD SULFO-Tag a and diluted at 1/500 in serum/conjugate diluent, was added to each well. The plate was incubated for 1 hr at room temperature on a shaker at 400 rpm. Finally, 150 µl of MSD read buffer T a (diluted 1/2 in distilled water) was added to each well immediately before reading the plate using an automated imager. a
The raw ECL data were corrected for background levels by subtracting the appropriate negative antigen signal from that of the positive antigen signal. Receiver operator curve (ROC) analysis was performed to determine the optimal positive/negative cut-off values for the MSD multiplex assay using GraphPad Prism software version 5. c The areas under the curve (AUCs) for the ROC curves were 0.9498, 1.0, 0.9734, and 0.9315 for the BVDV, BHV-1, BPV-3, and BRSV components of the multiplex, respectively (Fig. 1). The optimal cutoffs (1,975 for BVDV; 21,670 BHV-1; 5,015 BPV-3; and 6,273 BRSV; all values in relative ECL units) were used to generate 2 × 2 boxes and calculate the diagnostic sensitivity and specificity of the MSD multiplex assay compared to the parent ELISA (Table 1).
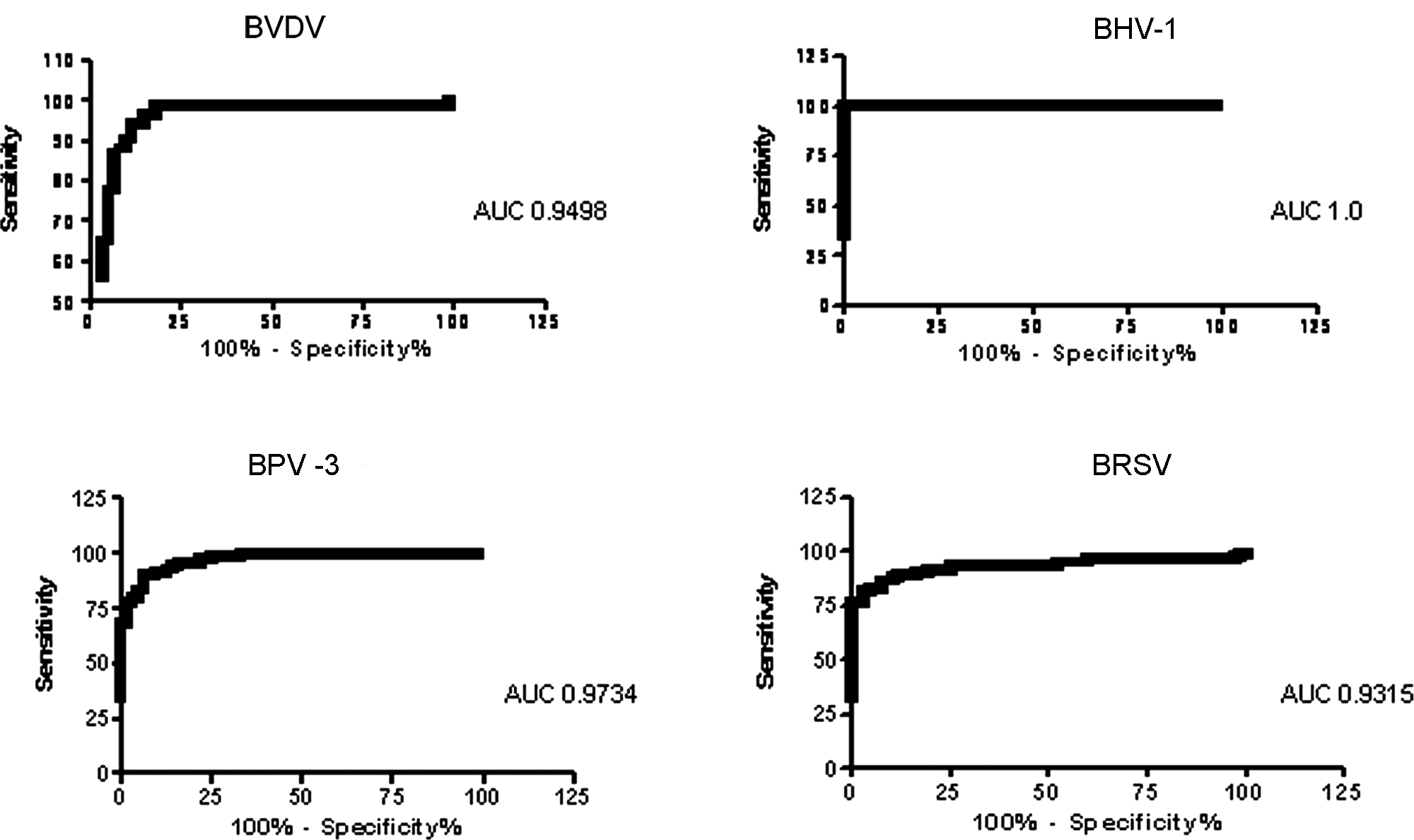
Receiver operator curve analysis of the 4 assays in the Meso Scale Discovery multiplex assay using the established enzyme-linked immunosorbent assays as the gold standard. The area under the curve is shown with the graphs. BVDV = Bovine viral diarrhea virus; BHV-1 = Bovine herpesvirus 1; BPV-3 = Bovine parainfluenza virus 3; BRSV = Bovine respiratory syncytial virus.
2 × 2 box analysis of the 4 assays in the Meso Scale Discovery (MSD) multiplex enzyme-linked immunosorbent assay (ELISA) compared to the results of the individual ELISAs.*
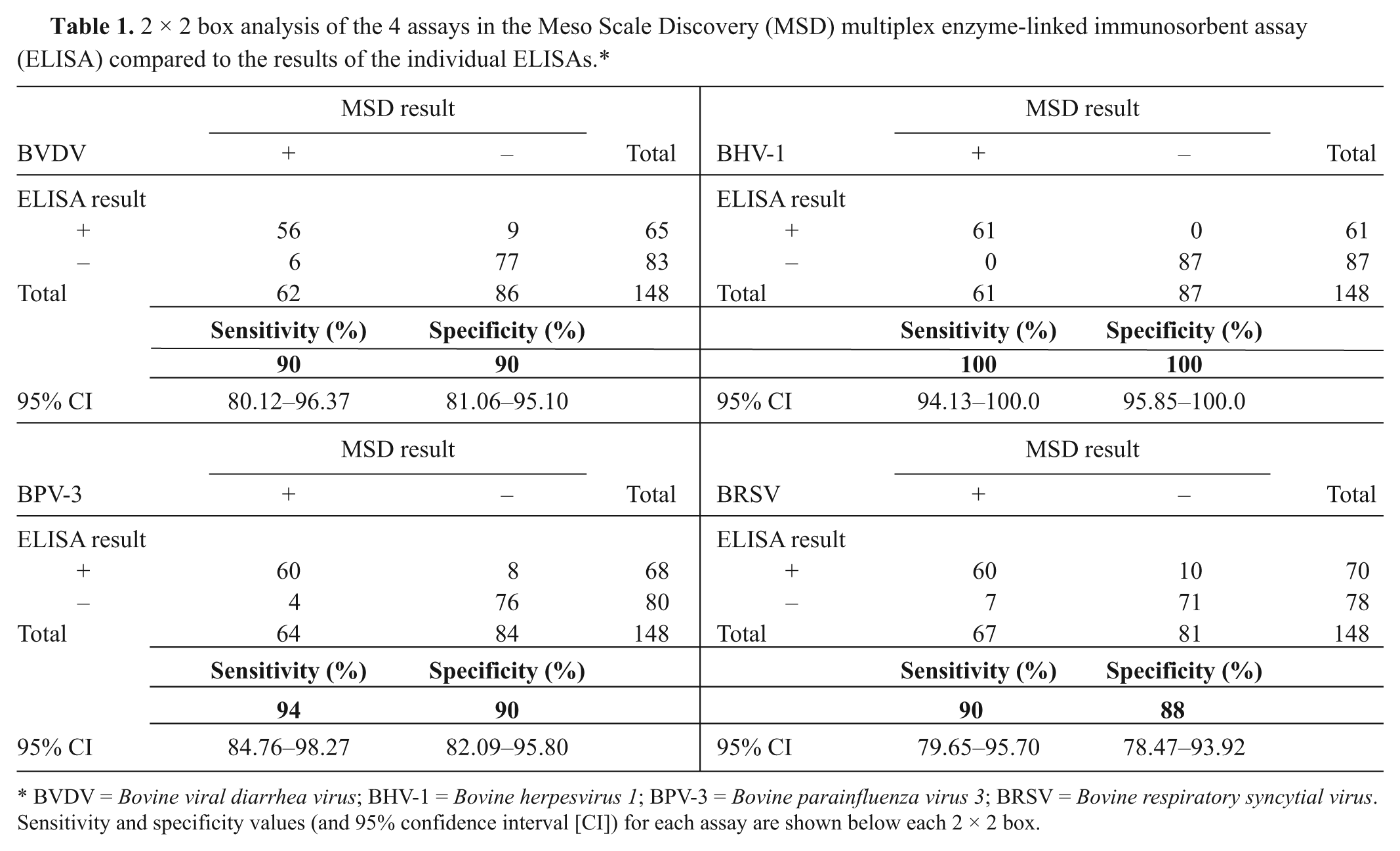
BVDV = Bovine viral diarrhea virus; BHV-1 = Bovine herpesvirus 1; BPV-3 = Bovine parainfluenza virus 3; BRSV = Bovine respiratory syncytial virus. Sensitivity and specificity values (and 95% confidence interval [CI]) for each assay are shown below each 2 × 2 box.
The high sensitivity and specificity of each of the separate assays in the multiplex relative to the established ELISAs (88% or above), and the AUC values of 0.9% and above, demonstrate that it was possible to carry out a multiplex serology assay on the MSD platform and obtain good agreement with the parent ELISAs. There was some variation in the performance of the individual tests within the multiplex ELISA. The best agreement was seen with the BHV-1 assay, while the BRSV assay performed least well when compared to the parent ELISA. In broad terms, these results are promising. They are better than those observed when using the same antigens in the Luminex multiplex platform, 1 where the performance of the BVDV and BRSV components of a Luminex multiplex assay were significantly worse. In the Luminex study, chemical conjugation of the antigens to Luminex beads may have affected the antigen and led to poorer assay performance. 1 The improved results observed in the current study may be because, in the MSD platform, the antigens are adsorbed onto the carbon electrodes, analogous to adsorption in standard ELISA plates. As a result, the antigens behave in a functionally more similar manner. If this is the case, conversion of existing ELISAs may be more straightforward with the MSD platform than with methods that require biochemical conjugation of antigens or reagents. The results from the current study illustrate that reagents used within standard ELISAs, including relatively crude, complex antigens, can be successfully transferred for use on the MSD platform.
Although promising, because of the relatively small number of samples tested in the present study, these results should be seen as an initial assessment of the potential of the MSD system for multiplex testing. To provide a more thorough assessment, a larger number of ELISA samples would need to be tested along with a panel of samples taken from animals of known infection status such as experimentally infected animals. In addition, the ELISAs used in the current study are well established at the AHVLA but are not considered gold standards. Therefore, the limitations of these assays should also be acknowledged, as the ELISA results may not always be correct.
The present work was primarily intended as an evaluation of the MSD platform using BRD as a model to evaluate the feasibility of performing a multiplex serological assay. In practice, this particular assay is likely to be of limited value for countries where widespread vaccination regimes are currently in place.
The MSD platform is a technology well suited to routine laboratory operation. The reader has no moving parts and has no requirement for regular maintenance or reagents. Data acquisition is rapid (plate read time similar to a conventional ELISA plate reader), and the software is user friendly. The MSD plates are also available in different configurations, which could be useful for specific applications; for example, a 4-plex 384-well plate could be useful for disease outbreak testing. In addition, it has been demonstrated that single-plex “no wash” ELISAs are possible on the MSD platform because of the proximity-based nature of signal generation, where only labels near the carbon electrode are stimulated, 8 which offers the intriguing prospect of multiplex “no wash” assays.
There are disadvantages to the MSD platform, such as relatively high reader and plate cost (compared to existing ELISA readers and plates) combined with specialist printing requirements of multiplex plates. At present, printing of multiplex plates can be performed only at MSD facilities in the United States. As a result, laboratories outside the United States have to obtain import permits for antigens and reagents, which can be a logistical burden and restricts the ability of researchers to construct their own in-house multiplex assays. These factors may make this technology more suited to high-throughput laboratories with well-defined testing requirements. In addition, the maximum number of assays that can be multiplexed in a 96-well plate format at this time is 10, which may be limiting for some applications.
In summary, the results of the present study demonstrated that established antibody ELISAs can be transferred to the MSD platform in a multiplex format using the same antigens. The MSD platform is a promising approach that offers the potential for reducing assay test costs and streamlining laboratory operation.
Footnotes
Acknowledgements
The authors would like to thank the Animal Health and Veterinary Laboratories Agency Laboratories Services Department for providing test samples and the Reagent Production Unit for preparing and supplying antigens for this work. At Meso Scale Discovery, thanks go to Tuc Ahmad and Yvonne Clements in the United Kingdom and the U.S. team who provided the multiplex plates. © Crown copyright 2012. Reproduced with the permission of the Controller of Her Majesty’s Stationery Office/Queen’s Printer for Scotland and Animal Health and Veterinary Laboratories Agency.
a.
MSD wash buffer, SULFO-Tag™, read buffer T, Sector Imager 6000 reader; Meso Scale Discovery, Gaithersburg, MD.
b.
Dako Denmark A/S, Glostrup, Denmark.
c.
GraphPad Software Inc., La Jolla, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Animal Health and Veterinary Laboratories Agency Test Development Programme.
