Abstract
Human pathogens have evolved to infect vertebrate hosts other than human beings without causing symptoms of the disease, thus permitting them to complete their life cycle and to develop into infectious forms. The identification and management of infected animals are alternatives to control dissemination of the disease and to prevent human illness. In the current study, the potential use of staphylococcal A or streptococcal G proteins was evaluated with enzyme-linked immunosorbent assays (ELISAs) for seroepidemiological studies. Sera were collected from animals that were representative of 23 different Brazilian wild mammals. A high protein A binding rate was observed in all animals, except for the orders Didelphimorphia, Artiodactyla, and Rodentia, in which affinity was medium or low. Affinity for streptococcal G protein was higher in animals of the order Artiodactyla, whereas no streptococcal G protein binding was observed in samples obtained from felines (order Carnivora). Bacterial protein binding to mammalian immunoglobulins was confirmed by immunoblotting. The results suggest that secondary detection systems should be better investigated in ELISA protocols before their implementation in seroepidemiological studies involving wild mammals.
The identification and management of infected animals are alternatives to control the dissemination of diseases and to prevent human illness. Direct isolation and characterization of the pathogen in samples obtained from infected reservoirs continues to be the gold standard for diagnosis. 11 However, ethical and operational concerns make it impossible in some cases to directly search for the microorganism because wild animals need to be maintained in captivity to complete some experimental procedures. 1 In these cases, indirect techniques are helpful to track present or past infections in the animal to a particular pathogen and to draw a possible map of disease distribution at a particular site. In this respect, immune-based assays are valuable tools because they require less expensive equipment and less laboratory expertise than molecular techniques. Enzyme-linked immunosorbent assay (ELISA) is one of the techniques used to identify a past infection by evaluating immunoglobulin titers in host sera. One option is the use of bacterial secreted proteins, such as staphylococcal A and streptococcal G proteins, which are able to recognize and bind to the Fc portion of circulating immunoglobulins. 9 Little is known about the reactivity of circulating immunoglobulins from wild Brazilian mammals to the bacterial proteins cited. In the current study, the potential use of staphylococcal A or streptococcal G proteins was evaluated with ELISA procedures, which may be useful in detecting antigen-specific immunoglobulins in Brazilian wild mammals for the diagnosis of tropical diseases.
Mammals kept in captivity were obtained from the Uberaba City Zoo, Minas Gerais State, Brazil (19°45’1.55” S; 47°55’58.85” W), or captured during a fauna rescue mission performed before implementation of the Manso Hydroelectric Power Plant, FURNAS Central Electric S.A, Manso, Mato Grosso State, Brazil (14°52’7.72” S; 55°47’2.18” W). Animals representative of 23 different species were identified and classified as belonging to the phylogenetic orders Carnivora, Didelphimorphia, Xenarthra, Primates, Artiodactyla, and Rodentia (Table 1). Animals that could serve as potential reservoirs of tropical disease agents, such as Paracoccidioides brasiliensis, Trypanosoma sp., Leishmania sp., and several bacteria and viruses, were selected.2,3,10
Differential reactivity of serum immunoglobulins from Brazilian mammals to staphylococcal A and streptococcal G proteins in immunoenzymatic assay, according to taxonomic order of the animal.
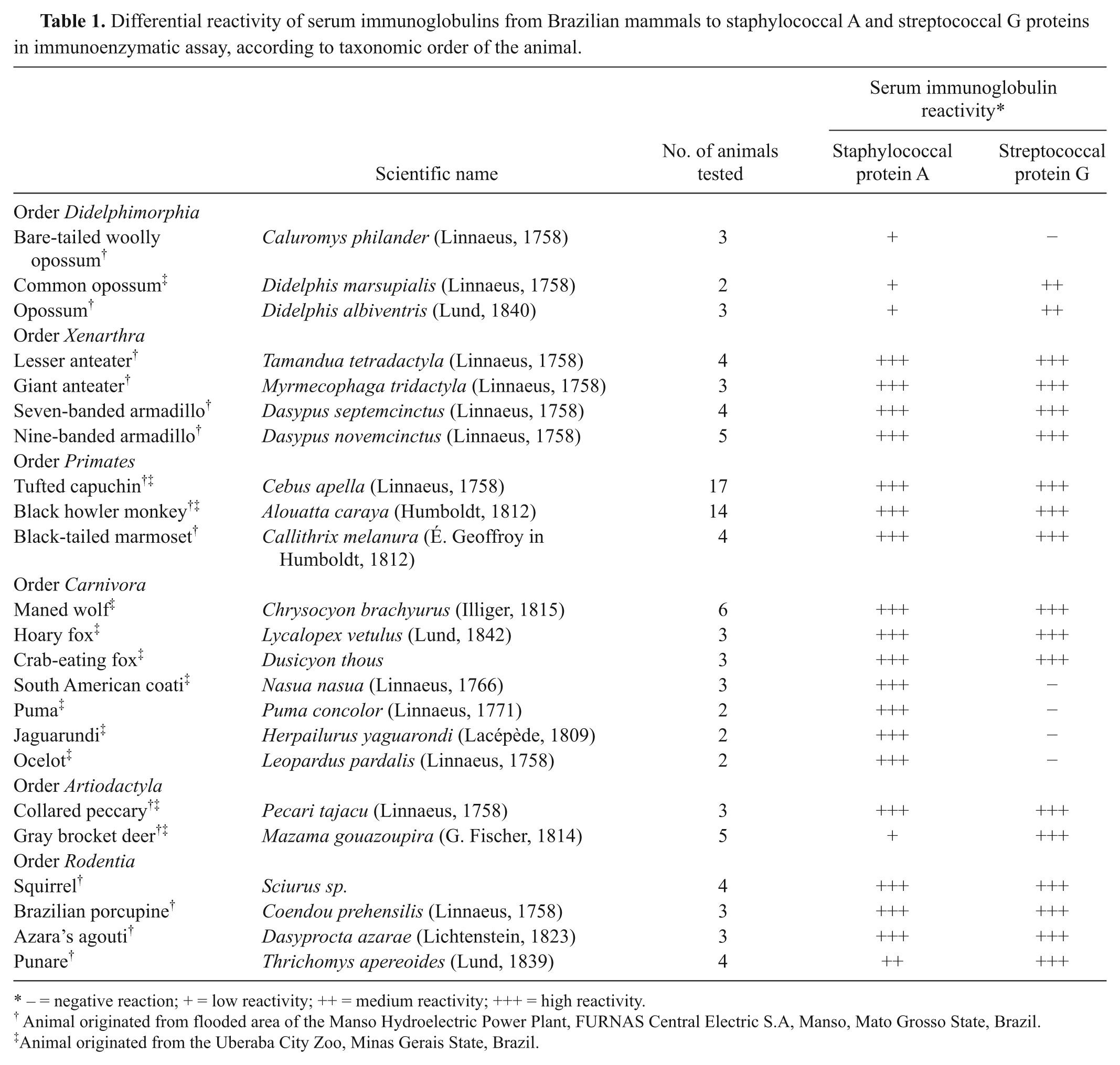
− = negative reaction; + = low reactivity; ++ = medium reactivity; +++ = high reactivity.
Animal originated from flooded area of the Manso Hydroelectric Power Plant, FURNAS Central Electric S.A, Manso, Mato Grosso State, Brazil.
Animal originated from the Uberaba City Zoo, Minas Gerais State, Brazil.
The animals were anesthetized with 1.0 mg/kg of midazolam. a Venous blood was collected with tripotassium–ethylenediamine tetra-acetic acid (K3-EDTA) vacuum tubes. The blood samples were centrifuged at 350 × g for 10 min, and plasma was stored at −70ºC until the time of use.
Captured animals were handled according to the regulations of the Uberaba City Zoo. The Brazilian Institute of Environment and Renewable Natural Resources (Instituto Brasileiro do Meio Ambiente e dos Recursos Naturais Renováveis) approved the protocols.
Polystyrene plates b were sensitized with 100 μl of the animal sera diluted at sequential ratios of 1:50, 1:250, 1:1,250, 1:6,250, 1:31,250, 1:156,250, 1:781,250, and 1:3,906,250 (v/v) in carbonate/bicarbonate buffer (pH 9.4), for 18 hr at 4°C. The plates were then washed 4 times with phosphate buffered saline (PBS) containing 0.05% Tween 20. c Nonspecific binding was prevented by incubation with 2% bovine serum albumin (BSA) c in PBS for 4 hr at room temperature. Next, the plates were washed and incubated with 2.5 μg/ml of peroxidase-conjugated staphylococcal A or streptococcal G proteins c diluted in PBS for 1 hr at room temperature. The plates were washed 4 times in PBS–Tween 20. Enzymatic activity was developed by incubation with orthophenyldiamine. c The reaction was interrupted by the addition of 1 N sulfuric acid solution. Absorbance was read at 490 nm in a microplate reader. d Samples were run in duplicates, and the mean optical density value was used for data presentation. The results are expressed as negative, low-, medium-, or high-reactive sera according to maximum dilution detected over blank controls in the ELISA, as follows: low (+: 1:50–1:250), medium (++: 1:1,250–1:156,250), and high (+++: 1:781,250–1:3,906,250).
The mammalian sera were subjected to sodium dodecyl sulfate–polyacrylamide gel electrophoresis on a 7.5% polyacrylamide gel in an electrophoresis cell apparatus. d Sera were used without prior heating or urea treatment. Samples were electrophoretically transferred to nitrocellulose membranese in buffer containing 20 mM of Tris–hydrogen chloride, 190 mM of glycine, and 20% (v/v) methanol at 100 V/cm for 60 min in an electrophoretic transfer cell apparatus. d The membranes were then blocked for 4 hr at room temperature in PBS (pH 7.8), containing 1% BSA. c
For the immunoblotting assay, the membranes were washed once in washing buffer (PBS–0.05% Tween 20, pH 7.8). Then, membranes were incubated with peroxidase-conjugated staphylococcal A or streptococcal G proteins c diluted 1:2,000 in PBS for 4 hr at room temperature under constant stirring. The membranes were washed twice and developed by incubation with 3,3’-diaminobenzidine tetrahydrochloride substrate c in PBS (0.05% DAB, 0.015% hydrogen peroxide) for 2 min. Rinsing in double-distilled water interrupted the reaction. Human serum was included as a positive control of bacterial protein binding to immunoglobulin G (IgG) and served as a reference for the electrophoretic migration pattern (approximately 150 kDa).
An ELISA evaluated the detection of immunoglobulins from Brazilian mammals by bacterial proteins, and the results are shown in Table 1. Serum immunoglobulins from 23 different species of wild mammals, either captured during a fauna rescue or kept in captivity at a city zoo, presented strong binding (+++) to staphylococcal A protein, except for the orders Didelphimorphia, Artiodactyla, and Rodentia, in which some animals had medium (++) or low (+) binding rates. However, affinity for streptococcal G protein varied among animals of these orders, with higher binding rates observed for samples from the order Artiodactyla. In contrast, no streptococcal G protein binding (–) was observed in samples from felines (order Carnivora), whereas high optical densities for this protein (+++) were detected in all other animals.
Immunoblot analysis using randomly selected animals was performed to determine whether the bacterial proteins really bound to mammalian circulating immunoglobulins as observed in ELISA.5,8 The results indicated that an antibody protein binding relatively similar to that observed for human IgG (Fig. 1) exists. In addition, animals with higher ELISA optical density (+++) had a stronger immunoblot band than animals with a weak or absent ELISA reaction (+ and –, respectively).
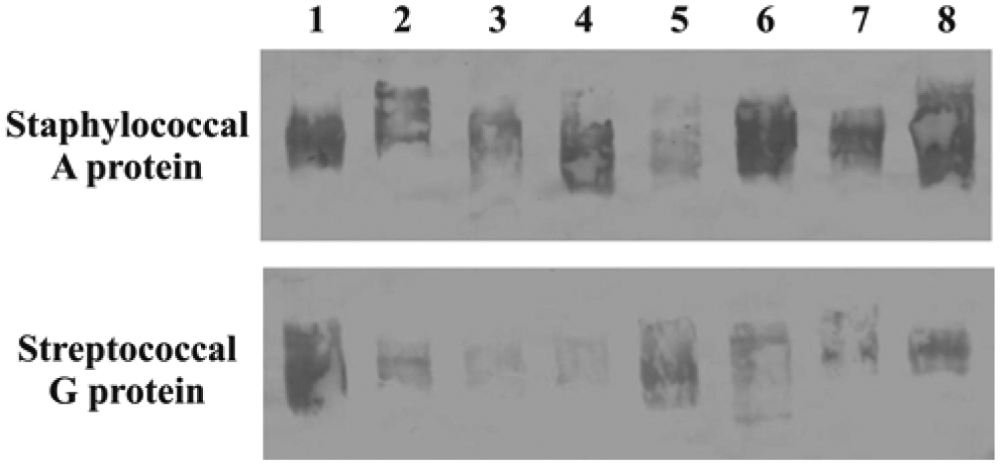
Evaluation of the binding of mammalian serum immunoglobulins to bacterial proteins. The immunoblotting assay, performed as described in the materials and methods section, was used to determine the binding of staphylococcal A (upper) and streptococcal G proteins (bottom) to circulating immunoglobulins. Sera were collected from 1 healthy adult human (lane 2) used as control and from 7 randomly selected mammals belonging to the species Myrmecophaga tridactyla (lane 1), Chrysocyon brachyurus (lane 3), Puma concolor (lane 4), Mazama gouazoupira (lane 5), Alouatta caraya (lane 6), Tamandua tetradactyla (lane 7), and Dasyprocta azarae (lane 8).
Variations in the interaction between staphylococcal A protein and mammalian immunoglobulins have been reported in a pioneering study whose authors showed that precipitation or inhibition reactions presented extremely variable results and proportions among animal orders and also among animals of the same mammalian order. 8 Divergent data regarding the interaction between bacterial proteins and animal sera have also been reported in some studies5,8 investigating wild animals and infectious diseases. By using a radioimmunoassay for the measurement of antibodies against alphaviruses, it was observed that serum from the American common opossum (Didelphis marsupialis) failed to react, whereas sera from other species, including sheep, goat, and deer, reacted poorly to the staphylococcal A protein preparation. 5 In the present study, order Didelphimorphia sera presented lower affinity for staphylococcal A protein when compared to streptococcal G protein, suggesting that the latter protein is more suitable for the immune-based diagnosis of diseases that occur in this order.
Another study comparing IgG subclasses among human beings, baboons, and macaques using anti-human IgG subclass reagents showed that macaques differ from baboons in terms of the IgG3 subclass and of the capacity of serum immunoglobulins to bind to bacterial A and G proteins. 12 The possible inability of IgG3 to bind to staphylococcal A protein has been previously described using human gG myeloma globulins. 9
Seroepidemiological studies using commercial ELISA kits have reported different positivity rates for the species evaluated. An extensive serosurvey of carnivores in the Bolivian Chaco using various commercial serological tests, 4 obtained divergent results for the animals captured; all jaguarundi species were seronegative on all tests for Canine adenovirus, Canine coronavirus, Canine distemper virus, Canid herpesvirus 1, Canine parvovirus, Feline calicivirus, Feline coronavirus, Felid herpesvirus 1, Feline immunodeficiency virus, Feline leukemia virus, Feline panleukopenia virus, Leptospira interrogans, and Brucella canis. Such finding suggests either that the animals were not exposed to a previous infection or that these kits fail to detect circulating jaguarundi immunoglobulins. 4
A serological survey of Mycobacterium leprae infection in nine-banded armadillos using an ELISA showed a correlation with other immune-based techniques, such as the immunochromatographic flow test, indicating that either method might be used as a first screening test of M. leprae sensitization in these animals. 3 However, false-negative results obtained by ELISA in comparison to polymerase chain reaction indicate that further studies are needed to improve immunoassays for the study of armadillos or other species. 6
Interpretation of results present in Table 1 suggests that in some particular cases, prior evaluation of bacterial protein reaction to serum from each wildlife species is needed before the development of ELISA kits. In fact, this observation seems correct if the adopted streptococcal protein G protocol is used for detecting immunoglobulins in animals from the orders Didelphimorphia and Carnivora, and if the staphylococcal protein A protocol is used for detection of immunoglobulins in animals from the order Artiodactyla. In this case, some minor modifications on the current ELISA protocol or even the adoption of a sample preparation step prior to its incubation (e.g., a sample buffer specially delivered in the kit to be used only with some mammalian species) may be considered as an alternative to address the discrepancies observed. On the other hand, the evaluation of the results obtained in samples from the orders Xenarthra, Primates, and Rodentia indicates that this individual species evaluation is not needed for both bacterial proteins. The same statement could be applied for samples from orders Didelphimorphia and Carnivora using staphylococcal A protein and order Artiodactyla using the streptococcal G protein.
Comparison of IgG and Fc receptor binding between human beings and other animals of diverse orders induced by Human herpesvirus 1 antigen domains and Staphylococcus aureus A protein showed that the binding pattern of many antigens depends on the origin of the antigen and on the Fc receptor type. 7 Further studies involving wild populations from rural and urban areas are needed to improve understanding of pathogen transmission between human beings and domestic and wild animals. The present results suggest that species-specific variations should be considered when serological tests are performed in seroepidemiological studies.
Footnotes
Acknowledgements
The authors thank The Brazilian Institute of Environment and Renewable Natural Resources (IBAMA; Instituto Brasileiro do Meio Ambiente e dos Recursos Naturais Renováveis), FURNAS Centrais Elétricas, Companhia Energética de Minas Gerais, and the Municipal County of Uberaba for helpful logistic and technical assistance. The current work was performed as part of the Tapiti operation conducted by IBAMA within a larger IBAMA project (protocol nos. 084/APM Manso; 060/APM-Manso; 31/2001; 075/APM Manso). Afonso Pelli and Lucio R. Castellano made equal contributions to the study.
a.
Dormonid®, Roche Brasil, Rio de Janeiro, Brazil.
b.
Nunc® MaxiSorp™, Sigma-Aldrich Denmark A/S, Copenhagen, Denmark.
c.
Sigma-Aldrich, St. Louis, MO.
d.
Mini-Protean® II, Transblot®; Bio-Rad Laboratories, Hercules, CA.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support was obtained in part by a grant from FUNEPU/UFTM and Research Support Foundation of Minas Gerais. Lúcio R. Castellano was the recipient of a postgraduate fellowship from Coordination for the Improvement of Higher Education (CAPES, Brazil).
