Abstract
Highly pathogenic porcine reproductive and respiratory syndrome (HP-PRRS) characterized by high fever, high morbidity, and high mortality in pigs of all ages emerged in China in 2006 and spread rapidly throughout Southeast Asia. In July 2010, a highly contagious swine disease with clinical signs similar to those of HP-PRRS was observed in the Lao People’s Democratic Republic (Lao PDR). A field investigation covering 8 pig farms and 1 slaughterhouse in 7 different districts in the capital city of Vientiane was conducted to identify the disease. Total mortality rates ranged from 6.02% in boars to 91.28% in piglets (mean 54.15%) across the farms investigated. Emergence of the HP-PRRS virus (HP-PRRSV) in Lao PDR was confirmed using real-time reverse transcription polymerase chain reaction as well as virus isolation and identification. An animal inoculation study was performed to characterize the HP-PRRSV responsible for this outbreak. Isolate Laos 1.13 was inoculated into 70-day-old specific pathogen–free pigs to study pathogenicity. Clinical signs of high fever, rubefaction, respiratory distress, nervous symptoms, and diarrhea were observed in inoculated pigs, as well as pathological hemorrhagic lesions consolidated in the lungs. Morbidity and mortality were 100% and 60%, respectively, in inoculated pigs. HP-PRRSV was re-isolated from the inoculated pigs. Results suggested that the newly emerged HP-PRRSV was responsible for recent outbreaks of the swine disease in Lao PDR. The current report highlights the importance of continuous surveillance in neighboring countries to prevent introduction of PRRS to new regions.
Keywords
Porcine reproductive and respiratory syndrome (PRRS) was first reported in the United States in 1987. The disease causes reproductive failure during late-term gestation in sows and respiratory disease in pigs of all ages.1,3,8 In 2006, a new, highly pathogenic PRRS (HP-PRRS) strain emerged in the People’s Republic of China and Vietnam. This strain was characterized by high fever (40–42°C), skin discoloration or reddening, high incidence of illness (50–100%), and high death rate (20–100%) in pigs of all ages.2,6,9 The agent of HP-PRRS was identified as a highly pathogenic variant of the Porcine reproductive and respiratory syndrome virus (HP-PRRSV), which has a unique discontinuous 30 amino-acid deletion in one of its nonstructural proteins (NSP)2.6,9 Highly pathogenic PRRSV has spread to pigs in many parts of Asia. The affected countries include Cambodia, Myanmar, The Philippines, Russia, and Thailand (Roongroje T: 2011, It’s time for the taming of PRRSV. Proceedings of the 5th Asian Pig Veterinary Society Congress, pp. S16–S18. March 7–9, Pattaya, Thailand).2,6 In June 2010, the China Animal Disease Control Center (Beijing, China) and National Animal Health Centre (Lao People’s Democratic Republic [Lao PDR]) confirmed an outbreak of HP-PRRS in Lao PDR (Bounkhouang K: 2010, Porcine reproductive and respiratory syndrome, Laos. World Organization for Animal Health (OIE) World Animal Health Information Database interface. Available at: http://web.oie.int/wahis/public.php?page=single_report&pop=1&reportid=9484). The present report describes the emergence of HP-PRRS in Lao PDR and a study of animal inoculation tests with the HP-PRRSV Laos 1.13 strain.
From June 2010 through August 2010, an outbreak suspected to involve HP-PRRS occurred in the capital city of Vientiane in Lao PDR. Over a period of 69 days, 31 pig farms in 7 districts (housing a total of 13,977 pigs) reported the disease. The condition was characterized by high fever, abortion, and a high proportion of deaths in pigs of all ages. A total of 3,546 pigs died of the disease. The average mortality rate was 25.37% (Table 1). A summary of the PRRS outbreak is shown in Table 1, and a distribution of PRRS outbreaks is shown in Figure 1.
Porcine reproductive and respiratory syndrome outbreaks in the capital city of Vientiane, Lao People’s Democratic Republic from June 16 to August 25, 2010.

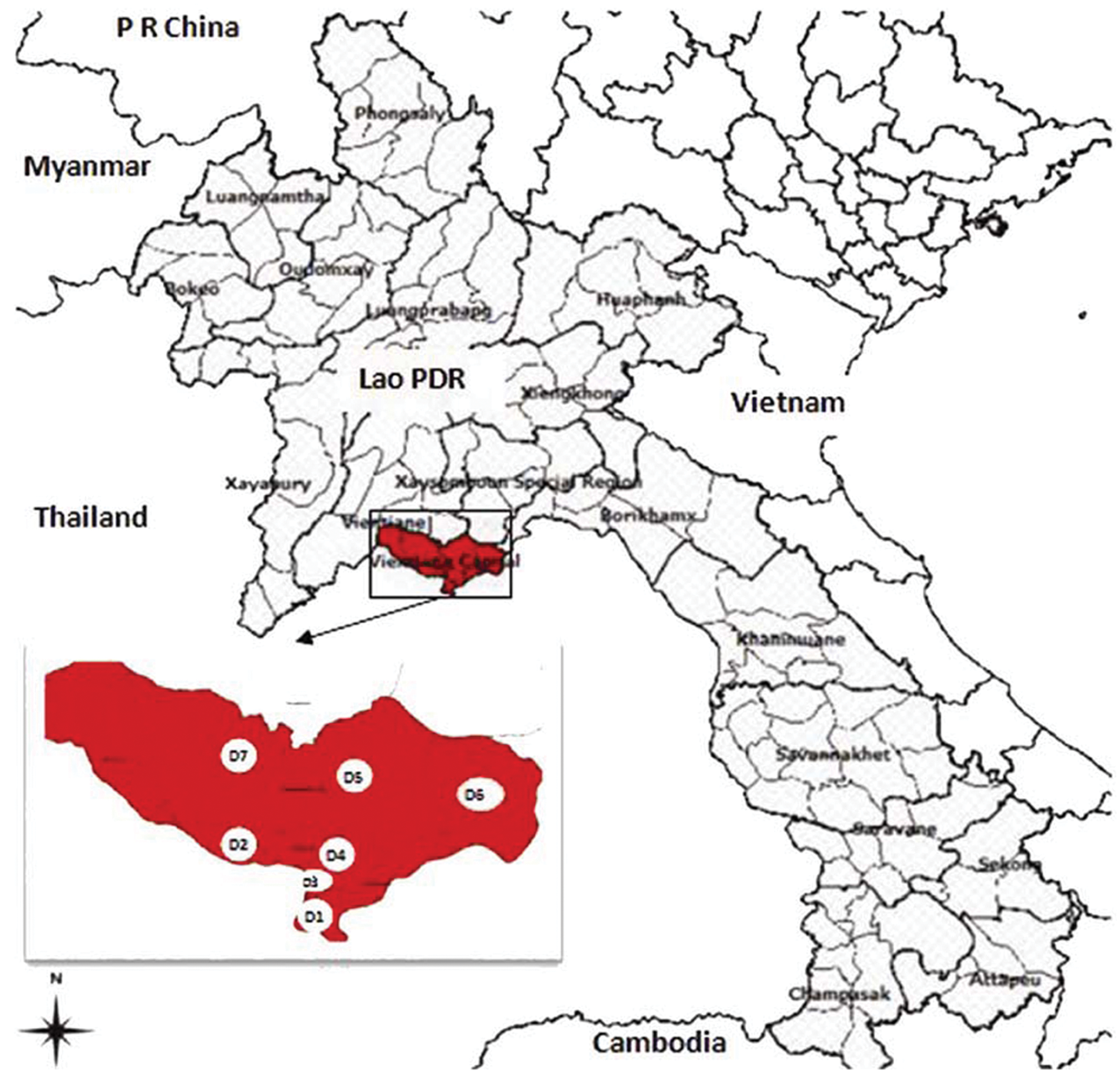
Porcine reproductive and respiratory syndrome outbreaks in Vientiane, Lao People’s Democratic Republic.
The first case was reported in Banphao village, Hadsayphong District, Vientiane, Lao PDR, in June 2010. A field investigation was carried out on 8 farms, including backyard farms and large-scale operations (Table 2). The average mortality rate varied by type of pig: 27.97% (0–53.33%) in sows, 48.48% (9.33–64.46%) in finishing pigs, 91.28% (57.50–100.00%) in piglets, and 6.02% (0–50.00%) in boars. The average mortality rate was 54.15% across all farms investigated (Table 2). The disease was characterized by the following symptoms: high and continuous fever (40–42°C), petechiae, coughing, nervous symptoms, hind limb paralysis, waggling, and reproductive failures including abortion and stillbirth at all semesters of gestation. Other common symptoms included inappetence, conjunctivitis, diarrhea, constipation, and dyspnea. Gross necropsy findings were comprised mainly of interstitial pneumonia with hemorrhage and consolidation (especially common in lobus apicalis and septal leaflet) and enlargement and hemorrhage in submaxillary lymph nodes and pulmonary hilar lymph nodes.
Results of field investigation and identification of etiological agent of outbreak of swine disease in Vientiane, Lao People’s Democratic Republic.*
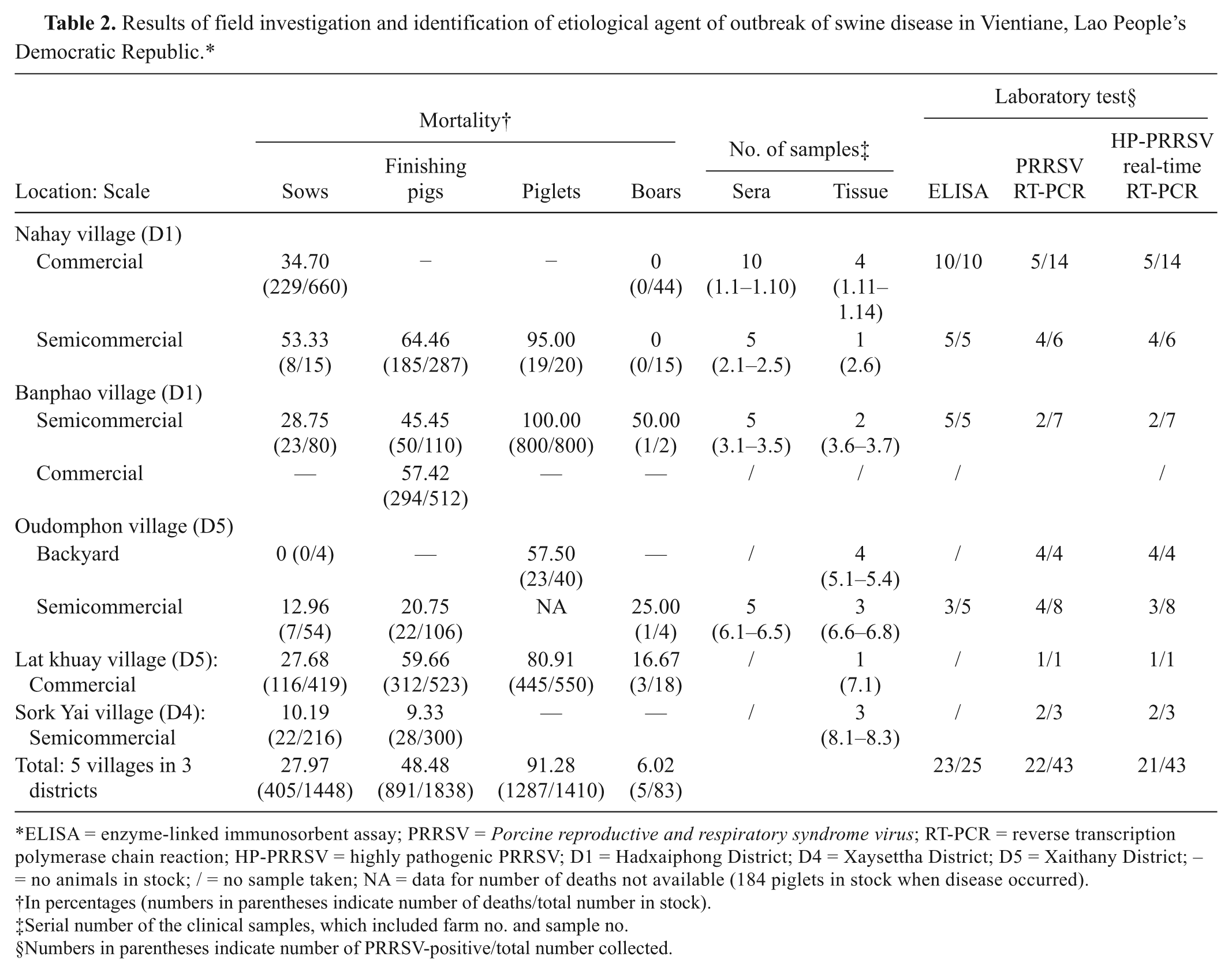
ELISA = enzyme-linked immunosorbent assay; PRRSV = Porcine reproductive and respiratory syndrome virus; RT-PCR = reverse transcription polymerase chain reaction; HP-PRRSV = highly pathogenic PRRSV; D1 = Hadxaiphong District; D4 = Xaysettha District; D5 = Xaithany District; – = no animals in stock; / = no sample taken; NA = data for number of deaths not available (184 piglets in stock when disease occurred).
In percentages (numbers in parentheses indicate number of deaths/total number in stock).
Serial number of the clinical samples, which included farm no. and sample no.
Numbers in parentheses indicate number of PRRSV-positive/total number collected.
Tissue (lungs, lymph nodes, and fetal lungs) and sera samples from sick and dead pigs were collected and sent to the China Animal Disease Control Center and National Animal Health Center. The clinical diagnosis was confirmed by detection of HP-PRRSV RNA using real-time reverse transcription polymerase chain reaction (RT-PCR) and virus isolation and identification.
Forty-three samples (25 serum and 18 tissue) from 8 farms were tested for suspicious agents including PRRSV, Porcine circovirus-2 (PCV2), Foot-and-mouth disease virus (FMDV), Classical swine fever virus (CSFV), Swine influenza virus (SIV), and Streptococcus suis serotype 2 using RT-PCR and PCR. Of these, 7 serum and 15 tissue samples were found to be PRRSV-positive using PRRSV RT-PCR. Out of these 22 PRRSV-positive samples, 21, covering all the farms investigated tested HP-PRRSV–positive using a commercial real-time RT-PCR kit.a,4 All samples from these farms were found to be negative for FMDV, CSFV, SIV, and S. suis. However, samples 3.7 and 7.1, collected from farms 3 and 7, respectively, were found to be PCV-2–positive. Twenty-three sera samples tested positive for anti-PRRSV antibody by enzyme-linked immunosorbent assay. b Results indicated that the farms investigated were exposed to PRRSV (Table 2).
Seven lung tissue samples (samples 1.13, 2.6, 3.7, 5.1, 6.6, 7.1, and 8.1) from 7 pig farms were used for isolation of the virus. Porcine alveolar macrophages (PAMs) were inoculated with the lung homogenates at a dose of 500 μl/cm 2 and then incubated at 37°C and 5% CO2 for 6–8 days postinoculation. Two consecutive passages were performed. The cytopathic effect was observed daily. Cell cultures were harvested and tested using a commercial real-time RT-PCR kit. a The PAM cells on the coverslip were inoculated and examined by immunohistochemistry 72 hr postinoculation for the presence of PRRS antigen. This process involved monoclonal antibody against the Northern American type of PRRSV (the hybridoma cell line, 15A c ).11,12 All 7 samples tested positive by virus isolation with an obvious cytopathological effect, characterized by cell aggregation, contraction, nucleus condensation, and brushing off at 2 days postinoculation. The isolates were identified as HP-PRRSV–positive by real-time RT-PCR and immunohistochemical staining.
An animal inoculation study was performed using the PRRSV Laos 1.13 strain isolated from the Kaisone Phouvong farm. The strain was continuously passaged for 5 generations on a PAM cell monolayer. The fifth-generation cell culture was harvested, and the viral titer was determined (105.0 TCID50/ml). Five specific pathogen–free pigs were individually inoculated with 3 ml 105.0 TCID50/ml PRRSV Laos 1.13 strain via muscular injection. The animals were kept in separate rooms throughout the experiment in a biosafety level 3 facility. This procedure was approved by the China Animal Disease Control Center ethics committee. 9 The clinical manifestations observed in the inoculated pigs were similar to those of diseased pigs from the Kaisone Phouvong farm. Signs included persistent high fever (Fig. 2), anorexia, red discolorations on the bodies, and blue ears. Diarrhea and nervous symptoms such as tremors and convulsions were observed on 7 days postinoculation (DPI). The gross necropsy findings of the inoculated pigs showed congestion hemorrhage, consolidation, and interstitial hyperplasia in the lungs (Fig. 3). Findings also showed obvious hemorrhagic spots in the lymph nodes, liver, kidneys, and spleen, and purple skin lesions progressing to multifocal red scabs. Three of the 5 pigs inoculated with the PRRSV Laos 1.13 strain died within 12 DPI (either 8 or 12 DPI). This indicates that the tested PRRSV strains were highly virulent.9,13 All inoculated pigs were found to be HP-PRRSV–positive by real-time RT-PCR. The PRRSV antibodies were detected in the 2 surviving pigs at 14 DPI (3 pigs died before seroconversion). Histopathological lesions were observed in the lungs, brain, and lymph nodes. Immunohistochemical evaluations were conducted on parafilm-embedded sections from the necropsy tissue specimens using monoclonal antibodies against PRRSV. Specific PRRSV antigen was detected in cytoplasm of macrophages in the alveolar wall, alveolar space, spleen, lymph node, and tonsils (data not shown). Viruses were re-isolated from all inoculated pigs and identified as HP-PRRSV by duplex real-time RT-PCR. Five sham-inoculated control pigs failed to show any clinical signs and remained healthy throughout the experiment. The results indicate that the outbreak of swine disease in Lao PDR in July 2010 was caused by HP-PRRSV infection.
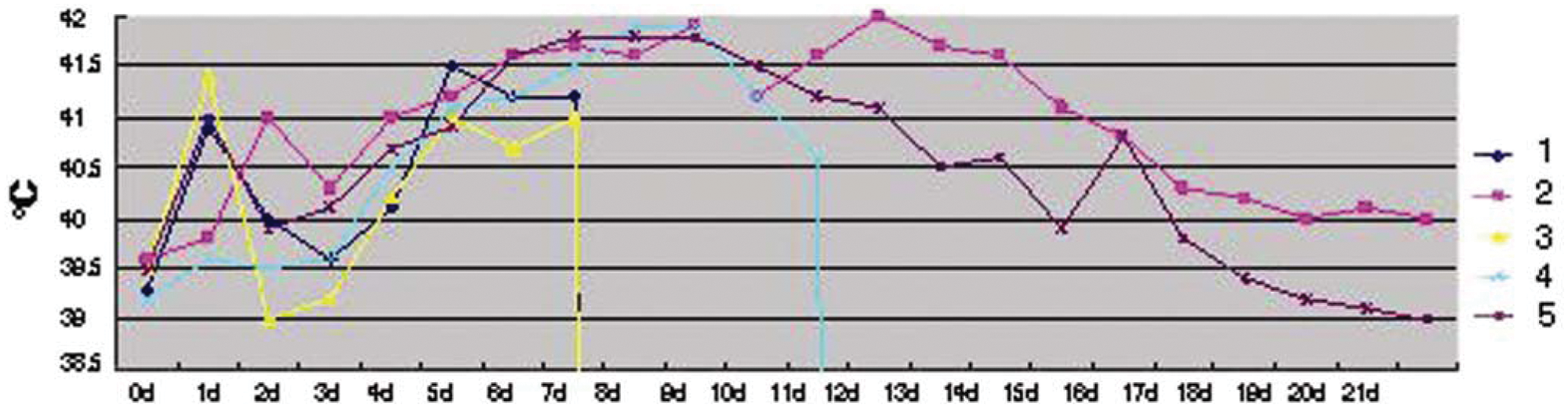
Curve graph of body temperatures of 5 experimental pigs inoculated with Porcine reproductive and respiratory syndrome virus Laos 1.13 strain. Note: Pigs 1 and 3 died 8 days postinoculation (DPI), pig 4 died 12 DPI, and pigs 2 and 5 were euthanized for necropsy 21 DPI.
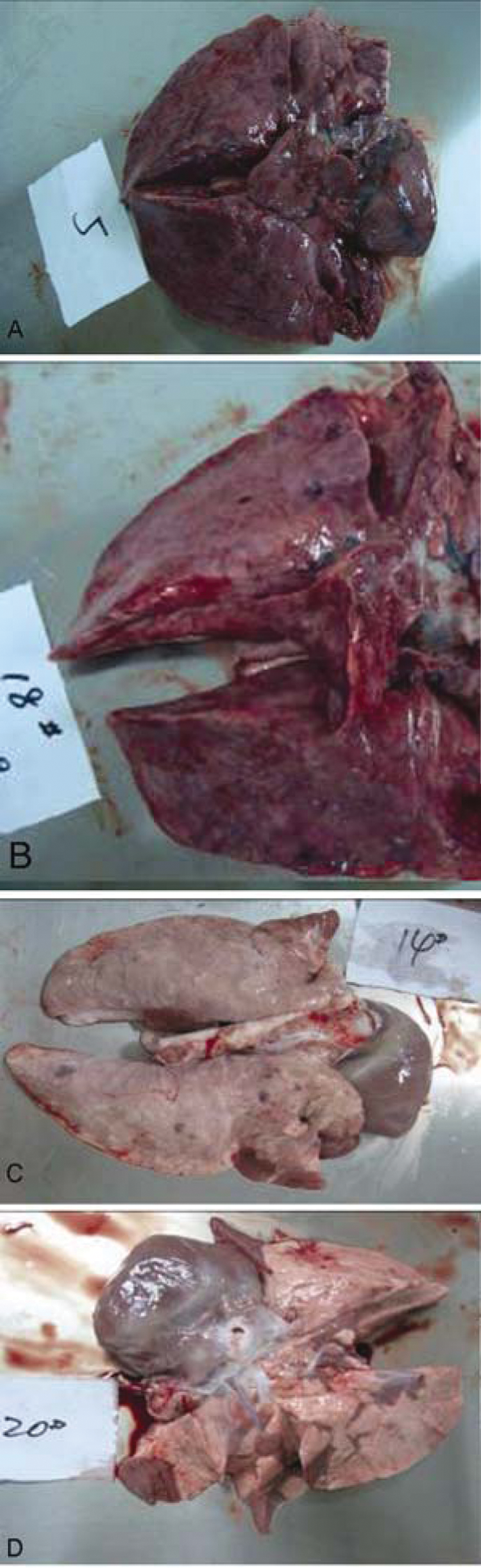
Gross lesions in lungs of pigs inoculated with Porcine reproductive and respiratory syndrome virus Laos 1.13 strain:
In summary, 31 pig farms in 7 districts reported the disease, which was characterized by high fever (40–42°C), abortion, and a high death rate in pigs of all ages. Clinical diagnosis was confirmed by detection of HP-PRRSV RNA using real-time RT-PCR and virus isolation and identification.
Conventional PCR and RT-PCR were used to rule out other suspected agents, including CSFV, PCV-2, FMDV, SIV, and S. suis. Porcine circovirus-2 was detected in farms 3 and 7, but, other than this virus, HP-PRRSV was the only agent found in any of the investigated farms. Salmonella choleraesuis, Erysipelothrix rhusiopathiae, Actinobacillus pleuropneumoniae, and Actinobacillus suis, the primary causes of hemorrhagic disease in pigs, are all sensitive to some kind of antibiotic. However, in the present reported outbreak, the application of antibiotics had almost no effect. Therefore, it was deemed unnecessary to search for these agents in the current study.7,14
Though coinfections and secondary infections could not be ruled out, which may have increased the severity of the clinical symptoms and the mortality rate, it was concluded that HP-PRRSV was a major factor in the July 2010 outbreak. The discovery was accomplished through analysis of the clinical features, through real-time RT-PCR detection, and through virus isolation and identification. Analysis of genetic characterization of the virus strain is vital to confirming a specific pathogen as the cause of an incident and to tracing possible sources of infection. 10 Further studies are needed to determine the genetic relationship between the Lao PDR PRRSV isolates and prototype HP-PRRSV (HP-PRRSV isolates from China and Vietnam).
Through the collaborative effort of the China Animal Disease Control Center and the National Animal Health Center, the etiological agent of this outbreak was identified promptly. Consequently, a large-scale outbreak in Lao PDR was contained by the combined efforts of efficient containment, slaughtering, and vaccine inoculation. 10 This outbreak highlights the importance of HP-PRRSV surveillance of pigs in neighboring countries to prevent introduction of PRRS to new regions. 5
Footnotes
Acknowledgements
The authors thank Dr. Jinxiang Li from the Ministry of Agriculture of PR China, Dr. Khambounheuang Bounkhouang, and Dr. Phouvong Phommachanh from the Ministry of Agriculture and Forestry of the Lao PDR for the kindness and cooperation that they showed in the field investigation. The authors also thank colleagues at Hangzhou Jianliang Animal Health Co. Ltd. for granting access to their ABSL-2 facility and Dr. Xue Han for kind help in editing the manuscript. Jianqiang Ni, Shibiao Yang, and Douangngeun Bounlom contributed equally to this work.
a.
Porcine Reproductive and Respiratory Syndrome North American type classical and highly pathogenic stain duplex real time RT-PCR kit, Beijing Anheal Laboratories Co. Ltd., China.
b.
Laboratoire Service International, Lissieu, France.
c.
Kindly provided by Dr. Liu Yang, Iowa State University, Ames, IA.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the National Scientific and Technical Supporting Programs in the 11th 5-year Plan (no. 2006BAD06A01, no. 2007BAD86B04, and no. 2007BAD86B05), the National Agricultural Programme for Public Welfare (200903037), Yunnan Province Science and Technology Innovation Project in Agriculture (2008LA019), and Yunnan Province Personnel Training Plan (20080C002).
