Abstract
Salmonella enterica subspecies enterica serovar Choleraesuis is rarely detected in Europe, but the clinical disease has been reported in wild boars. We describe here the clinical findings, pathologic changes, and microbiologic features of swine salmonellosis caused by S. enterica serovar Choleraesuis in weaned piglets in Serbia. In April 2019, on a large farrow-to-finish pig farm, increased mortality was reported in weaned piglets, marked by lethargy, anorexia, pyrexia, and respiratory distress. Gross pathology revealed dermal cyanosis, mesenteric lymphadenopathy, splenomegaly, hepatomegaly, interstitial pneumonia, and colitis. By direct culturing of lung, liver, spleen, and lymph nodes, S. enterica ser. Choleraesuis variant Kunzendorf was isolated after years of absence of the disease in pig farms in Europe. The source of this salmonellosis outbreak caused by S. enterica ser. Choleraesuis remains unknown.
Host-adapted Salmonella enterica subspecies enterica serovar Choleraesuis is isolated almost exclusively from diseased swine and is usually manifested by septicemia. 10 S. enterica ser. Choleraesuis variant Kunzendorf is highly prevalent in North America8,10 and Asia, 13 but is rare in the European Union (EU) and Australia.4-8 In the EU, S. enterica ser. Choleraesuis is not considered a dominant serovar in slaughter pigs and breeding herds, and only a few outbreaks have been reported among pig herds within the last few decades. Cases have been reported in 1999–2000 and 2012–2013 in Denmark,1,16 with occasional reports in a few other EU countries.4-7 As well, several outbreaks of the septicemic form, caused by S. enterica ser. Choleraesuis var. Kunzendorf, have been reported in the EU in wild boars.9,12,14,15 In Serbia, the most common S. enterica serovars in pigs are Typhimurium, Derby, Infantis, and Enteritidis. 11 Although these serovars can cause clinical salmonellosis in pigs, the extent of clinical salmonellosis in pigs in Serbia is unknown. Here, we describe the clinical signs, pathologic changes, and microbiologic features of salmonellosis caused by S. enterica ser. Choleraesuis in weaned pigs on a farrow-to-finish pig farm in Serbia.
In April 2019, an increase in mortality from 2.3% to 5% in weaned piglets was reported on a farrow-to-finish pig farm in southwest Vojvodina province in the northern region of Serbia. The herd had an inventory of 1,300 sows on a single site, and the farm was a continuous flow operation. Three nursery buildings on the farm housed ~2,000 weaned piglets per building. The farm vaccinated against Escherichia coli, Clostridium spp., Mycoplasma hyopneumoniae, Erysipelothrix rhusiopathiae, classical swine fever virus (Pestivirus C), pseudorabies virus (Suid alphaherpesvirus 1), porcine parvovirus (Ungulate protoparvovirus 1), porcine circovirus 2 (PCV2), and porcine reproductive and respiratory syndrome virus (PRRSV, genus Betaarterivirus; by vaccination of sows at weaning). In the previous 8 mo, the farm had experienced a severe outbreak of porcine epidemic diarrhea virus (PEDV) infection. Also, in the previous 18 mo, mycotoxicoses had occurred in different phases of feeding on the farm. The history of salmonellosis on the farm was unknown. During the previous 2 y, the farm had imported ~200 gilts and 7 boars from Hungary, Germany, the Netherlands, and Denmark, and the farm also regularly traded pigs with other swine farms within Serbia.
The disease outbreak was observed in a nursery building in which there were 900 piglets 8–10 wk old. Approximately 120 of 900 piglets had clinical signs of lethargy, inappetence, reluctance to move, and cyanosis of ears, extremities, and abdomen. Approximately 50 piglets with these clinical signs were also febrile, with temperatures of 39.5–41.5°C, and had a shallow cough and hard breathing (thumping), indicating severe pneumonia. The same clinical manifestations were also observed in the other 2 nursery buildings a day later, and ~460 of the 6,000 weaned piglets at the farm had similar clinical signs. To control the outbreak, sick piglets were isolated in quarantine pens to reduce infection and contamination within nurseries, and antimicrobial treatment was initiated (enrofloxacin IM or PO). The disease outbreak in nurseries lasted for 3 wk. Morbidity reached 10% among weaned piglets, and mortality increased to ~5%. Of the 460 diseased piglets, 152 died, resulting in a case fatality rate of 33%.
Postmortem examinations of 47 pigs revealed emaciated carcasses and rough hair. Most carcasses had cutaneous cyanosis, especially of ears, nose, ventral neck, feet, and belly. In piglets that had survived for a few days, the ear tips were dry and dark-red, with sloughed portions (Fig. 1). Lymph nodes, especially mesenteric nodes, were enlarged, dark-purple, and moist (Fig. 2). In most cases, the spleen was enlarged, dark-purple, and pulpy; the liver was moderately enlarged, frequently with scattered ≈1.5-mm white necrotic foci (Fig. 2). In all cases, the lungs were resilient, failed to collapse, were firm, and red fluid separated lobules; lesions affected the cranial lobes. Colitis was present in some piglets; the colonic mucosa was slightly thickened and covered by patchy adherent gray-yellow fibrinous membranes (Fig. 3). Samples of lungs, liver, spleen, and lymph nodes from 6 animals were sent to the National Reference Laboratory for Salmonella (Institute of Veterinary Medicine, Belgrade) for Salmonella spp. identification and serotyping. In addition, samples of these organs were fixed in 10% neutral-buffered formalin and processed routinely for histologic examination.
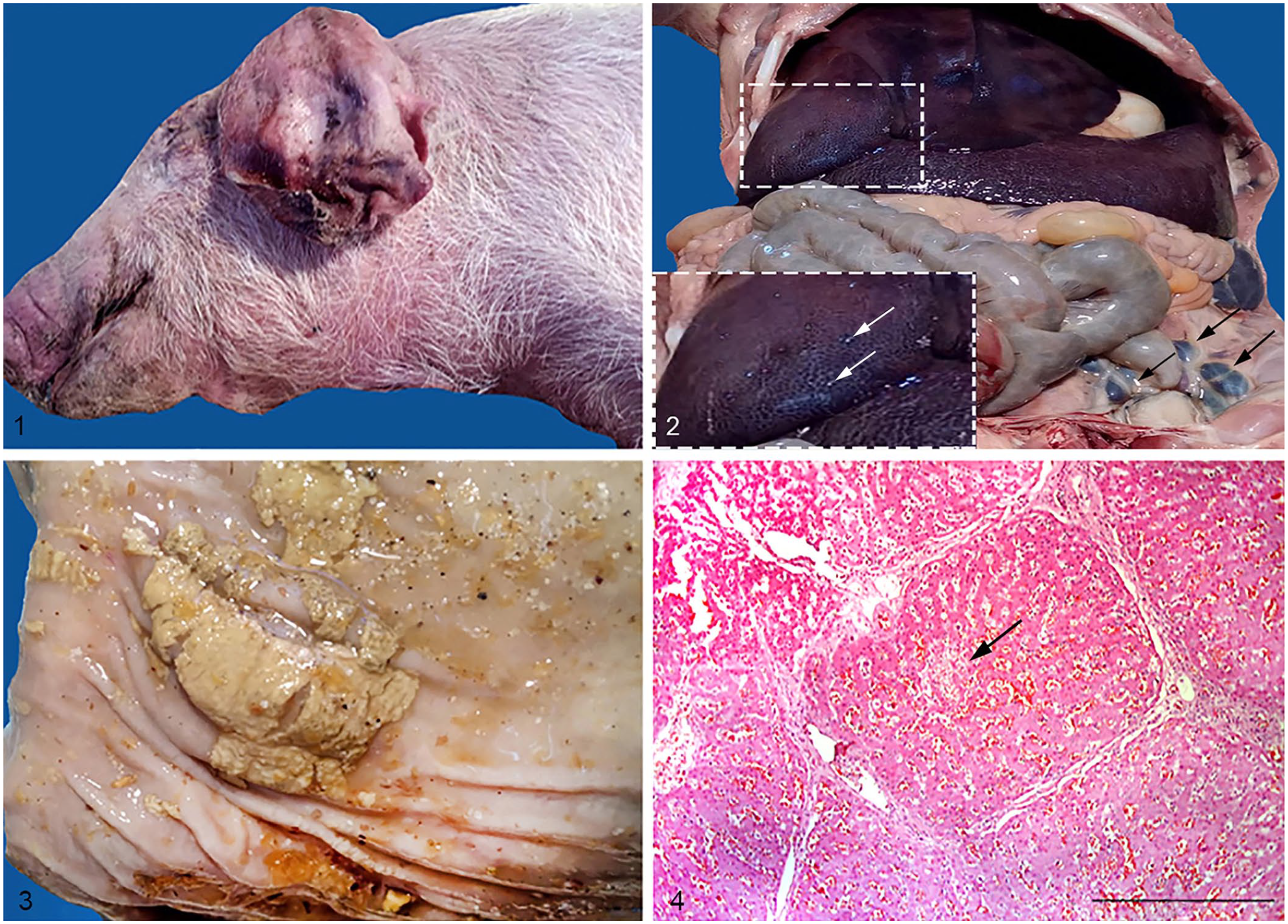
Pathologic findings in piglets infected by Salmonella enterica subsp. enterica serovar Choleraesuis.
All samples were cultured on blood agar, MacConkey agar, and xylose–lysine–deoxycholate agar (XLD) under aerobic conditions for 24 h at 37°C. Compatible colonies were confirmed by detection of invE and invA genes using PCR 18 and a commercial kit (BBL crystal identification systems enteric/nonfermenter; Becton Dickinson). Serogroup identification was performed by using the Salmonella sero-quick group kit (SSI Diagnostica). The identification of the serovar was conducted according to the Kauffmann–White scheme. 17 Salmonella spp. isolates that belonged to C1 group, serovar 6,7:c:1,5 were further sequenced for the fliC gene using flinC-F and flinC-R primers as described previously. 3 The sequences were queried in GenBank to identify best matches with the fliC gene from Salmonella. The 871-bp sequence of the fliC flagellin gene of Salmonella from our study was submitted to GenBank (MN563751).
A tentative diagnosis of salmonellosis caused by S. enterica ser. Choleraesuis was based on clinical signs and postmortem lesions. Lesions detected by autopsy of piglets that died indicated septicemia, which was confirmed by the isolation of S. enterica ser. Choleraesuis from lung, spleen, and liver in the autopsied animals. The hallmarks of the histology findings were as follows: hepatic congestion and focal necrosis (Fig. 4), with interstitial infiltration by mononuclear cells; diffuse interstitial pneumonia with septal thickening accompanied by suppurative bronchopneumonia, edema, and congestion; splenic congestion, increased white pulp, and focal necrosis; and hyperemia, with areas of necrosis, in lymph nodes.
Six bacterial isolates, obtained directly without pre-enrichment, were classified as S. enterica ser. Choleraesuis var. Kunzendorf by phenotypic serotyping and biochemical tests. All isolates were assigned to the antigenic formula 6,7:c:1,5 and were Vi capsular polysaccharide (Vi antigen) negative, with the following biochemical features: dulcitol (–), H2S (+), and mucate (–). All isolates generated the 963-bp PCR product of the fliC gene. The reverse primer FlinC-R amplified a unique sequence specific for S. enterica ser. Choleraesuis and S. enterica ser. Paratyphi C. Because S. enterica ser. Paratyphi C infects only humans and not swine, 2 and given that isolates were negative for Vi antigen, the positive signal of the fliC gene from these isolates is conclusive for S. enterica ser. Choleraesuis.
This disease outbreak occurred in the nurseries (i.e., in piglets of the age most susceptible to salmonellosis).8,10 Many studies2,9,16 emphasize the importance of stress factors as activators of various types of infections in weaned piglets, including factors such as the weaning and re-grouping animals, the triggering effect of immunosuppressive viruses (e.g., PCV2, PRRSV), and the fact that piglets 2–3 mo old have the lowest level of antibodies since birth. 19 Activation by stress factors also applies to Salmonella spp., particularly in the case of strains of S. enterica ser. Choleraesuis that can persist in asymptomatic carriers. In our case, the farm used vaccination against major and specific pig pathogens, and there had been an outbreak of severe PEDV infection only a couple of months before the S. enterica ser. Choleraesuis outbreak. However, we do not know the history of salmonellosis on the farm, and we can only speculate whether PEDV infection contributed to the severity of the S. enterica ser. Choleraesuis outbreak in weaned piglets. If asymptomatic S. enterica ser. Choleraesuis infection was already present in weaned piglets, then activation of this infection and the subsequent acute outbreak may have been triggered by underlying diseases and conditions (e.g., mycotoxicoses). As well, decreased antibody levels, especially the level of antibodies against PRRSV given that only sows are vaccinated, and therefore the onset of active PRRSV infection in piglets 20 may have caused stress-induced immunosuppression. Of course, S. enterica ser. Choleraesuis does not need any previous immunosuppressant infections, as described in outbreaks of S. enterica ser. Choleraesuis in wild boars in Spain and Italy.9,12
The salmonellosis outbreak in our report was characterized by a mortality rate of 33%, which is the same high rate reported in outbreaks of salmonellosis caused by S. enterica ser. Choleraesuis in Danish pig farms in 2012–2013,1,16 with the exception that in our case there were no obvious concomitant infections during the disease. The clinical signs and pathologic findings that we observed were fully compatible with those described for farm-raised weaned pigs, 8 and comparable to those described in similar processes affecting wild boars.9,15 As well, we noted colitis in several cases, an uncommon pathologic feature of septicemic salmonellosis caused by S. enterica ser. Choleraesuis.8,10
The most likely source of infection in our case was live carrier pigs, although other sources, such as feed, wild boars, or even humans as passive vectors, cannot be ruled out. The farm had continuously imported a large number of gilts and boars in the previous 2 y from the EU and had also purchased pigs from farms in Serbia. Therefore, it is possible that S. enterica ser. Choleraesuis was introduced to the farm by undetected carrier animals. However, to our knowledge, S. enterica ser. Choleraesuis has not been detected previously in Serbia and has not been reported recently from the EU countries from which pigs had been imported to this farm. However, owing to intensive importation of pigs into Serbia for the last 20 y from many EU countries, we cannot exclude the possibility that S. enterica ser. Choleraesuis could have been present undetected in asymptomatic pigs on certain farms in the country, or in the wild boar population, and even the pig feed chain. However, it is generally accepted that S. enterica ser. Choleraesuis is rarely found in pig feed,8,10 and in Serbia there is no stock of wild boars in the area (https://www.stat.gov.rs/en-US/search). The farm in our case is well managed and has good biosecurity, therefore, we ruled out these potential reservoirs of S. enterica ser. Choleraesuis as sources or vectors of the infection on the farm.
Footnotes
Acknowledgements
We thank the veterinary practitioners for the farm information and assistance during autopsy and sampling. Also, we thank the technical staff from the Department of Bacteriology and Pathology of the Institute of Veterinary Medicine Belgrade, Serbia for their help.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Our work was partially funded by Serbian Ministry of Education, Science and Technological Development (contract 451-03-68/2020-14/200030).
