Abstract
Endocrine tumors are rarely observed in pigs, and pheochromocytomas have been only punctually described. The current report describes a white and firm, 15-cm in diameter, neoplastic mass located in the adrenal gland with metastasis to regional lymph nodes in a 2.5-year-old sow. The masses had marked desmoplasia that supported a population of polygonal-to-spindle–shaped neoplastic cells arranged into cords and packets within a delicate fibrovascular stroma. Immunohistochemical staining of the tumor was positive for chromogranin and negative for neurofilament protein in adrenal and lymph node masses, which was characteristic of a malignant pheochromocytoma.
Pheochromocytomas (PHCs) are functional tumors of the chromaffin cells (pheocromocytes) of the adrenal medulla. These cells are derived from neural crest cells (neuroectoderm), which can differentiate into either secretory cells (pheocromocytes) or sympathetic ganglion cells. Within this process, different stages of cellular differentiation can lead to the appearance of various types of adrenal medullary tumors: neuroblastoma, PHC, and ganglioneuroma. 2 The term “malignant PHC” is used to designate infiltrative or metastatic medullary tumor. 3 In human beings, these dispersed collections of specialized neural crest cells that arise in association with the segmental or collateral autonomic ganglia throughout the body are called paraganglia. Hence, paragangliomas include tumors arising from adrenal medulla, chemoreceptors, vagal body, and small groups of cells associated with the thoracic, intra-abdominal, and retroperitoneal ganglia. With this classification, PHCs are considered within the group of retroperitoneal paragangliomas and are usually denominated adrenal paragangliomas. 11
In domestic species, PHCs develop most often in cattle and dogs and infrequently in other species; however, they also occur frequently in certain strains of commonly used laboratory rats, mice, nonhuman primates, and other exotic species like African elephants or coatis (Nasua nasua).1,3,5,7,8,11 Retroperitoneal paragangliomas in human beings with clinical signs related to catecholamine (norepinephrine or epinephrine) production are relatively frequent (25–60% of patients); 12 however, functional PHCs have been infrequently demonstrated in animals. 2
Reports describing PHCs in suids are extremely rare. There is a 1968 report describing a PHC as an incidental finding in a slaughtered pig, 10 and a functional PHC has been reported in an African warthog (Phacochoerus aethiopicus). 4 In the present work, a malignant PHC with metastasis to a regional lymph node in a slaughtered sow is described and characterized.
A 2.5 year-old female pig was slaughtered in an officially inspected abattoir in Catalonia, Spain. During the post-slaughter inspection, bilateral, white, and firm masses of 15–20 cm in diameter were observed in the area of adrenal glands. Moreover, the lumbar lymph nodes were increased in size (5–10 cm in diameter), and the cut section revealed a multilobular, brown to white mass of intermediate consistency, invading the totality of the parenchyma (Fig. 1). The kidneys also had multifocal and irregular whitish areas of 1–3 cm in diameter located in the renal surface and cortex. The masses and regional lymph nodes were rejected, and different portions of the adrenal masses, lumbar lymph nodes, and kidneys were separately collected, fixed in 10% buffered formalin, and routinely processed for histopathology. Sections were stained with hematoxylin and eosin and with Masson trichrome.
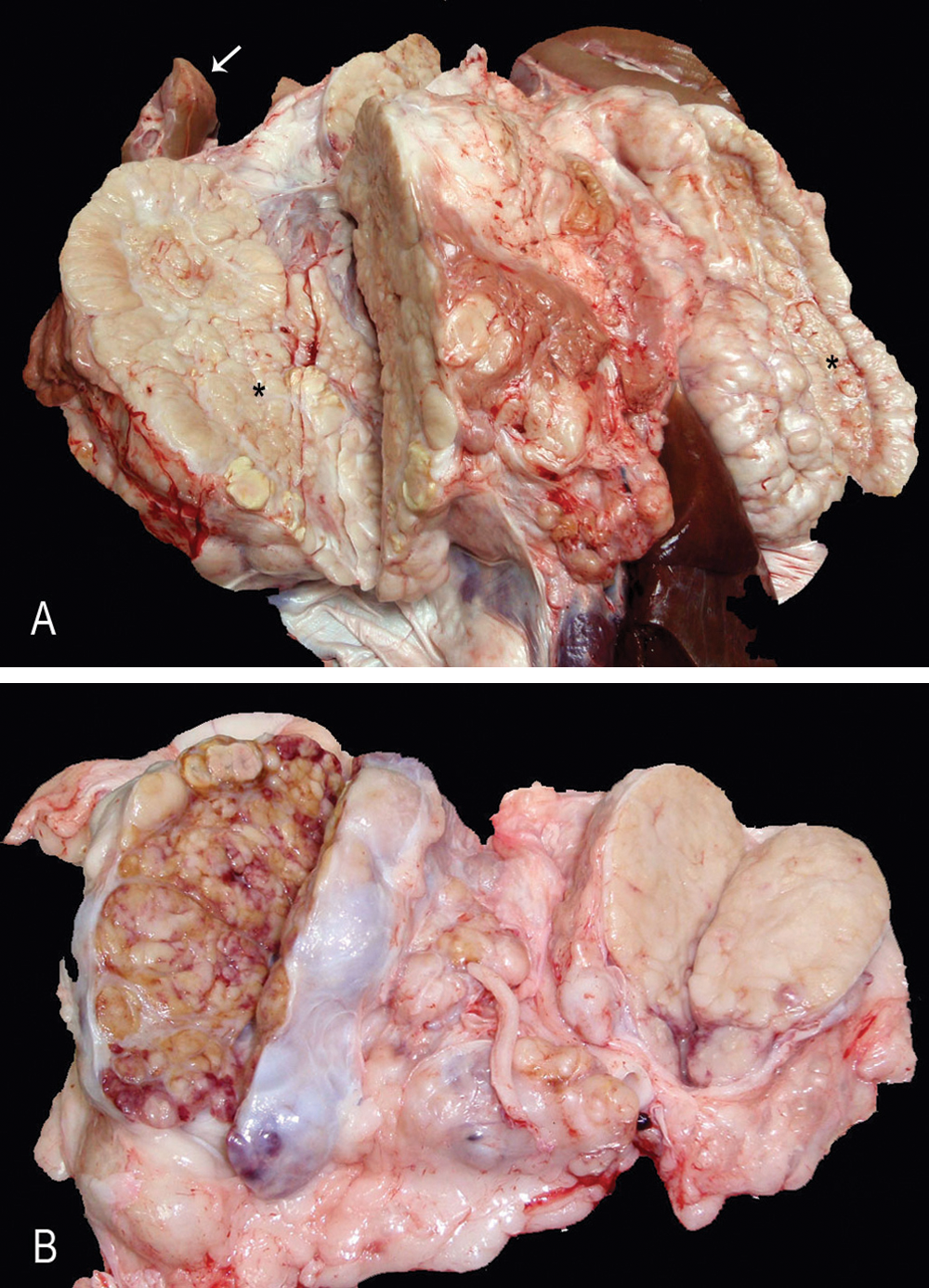
Pig.
Serial sections from the neoplasm and regional lymph node were immunohistochemically examined for 3 markers. Briefly, slides were pretreated with heat-induced epitope retrieval performed at 95ºC with 0.1 M citrate buffer (pH 6) for 20 min, before application of primary antibodies for chromogranin A (rabbit antiserum, 1:400), a synaptophysin (rabbit antiserum, 1:100) b and neurofilament NF-200kD (mouse antiserum, 1:20,000). c A non–avidin-biotin immunoperoxidase-diaminobenzidine detection method was used for all markers. d For immunohistochemical control purposes, adrenal for chromogranin A and cerebellum tissues from a healthy pig were used for synaptophysin and total neurofilaments; omission of the primary antibody resulted in the absence of specific reaction.
The adrenal mass consisted of a densely cellular, infiltrative, poorly demarcated, and encapsulated neoplasm without any rest of normal adrenal tissue. Cells were arranged in poorly defined cords within moderate amount of fibrous stroma identified as collagen with the Masson trichromic stain. Cells were polygonal to spindled, 15–20 μm in size, with indistinct borders and moderately abundant pale eosinophilic cytoplasm, sometimes with clear vacuoles. In general, nuclei were round, centrally located, with finely stippled chromatin and single basophilic nucleoli. There was marked anisocytosis and anisokaryosis, with occasional karyomegalic nuclei and 2–3 mitotic figures in 10 random fields at 400×. Mature eosinophils were scattered among the tumor cells. In contrast, neoplastic cells in the lymph node were mostly polygonal, with moderated amount of slightly basophilic cytoplasm and arranged in packets of variable size separated by delicate fibrovascular stroma (Fig. 2). Tumor emboli were observed in the lumen of afferent lymphatic vessels. In the kidney, a moderate and multifocal interstitial lymphoplasmacytic infiltrate with fibrosis was observed, accompanied by tubular lytic necrosis with neutrophilic infiltration, in either the cortex or medulla. These changes were consistent with chronic interstitial nephritis and bacterial-like pyelonephritis, presumably not related to the tumor.
A large number of neoplastic cells in the primary neoplasm and in the lymph node metastasis were strongly positive for chromogranin A (Fig. 3). However, the cells were negative for neurofilaments. For synaptophysin, an extracellular nonspecific reaction was observed, and cells were considered negative for this marker.
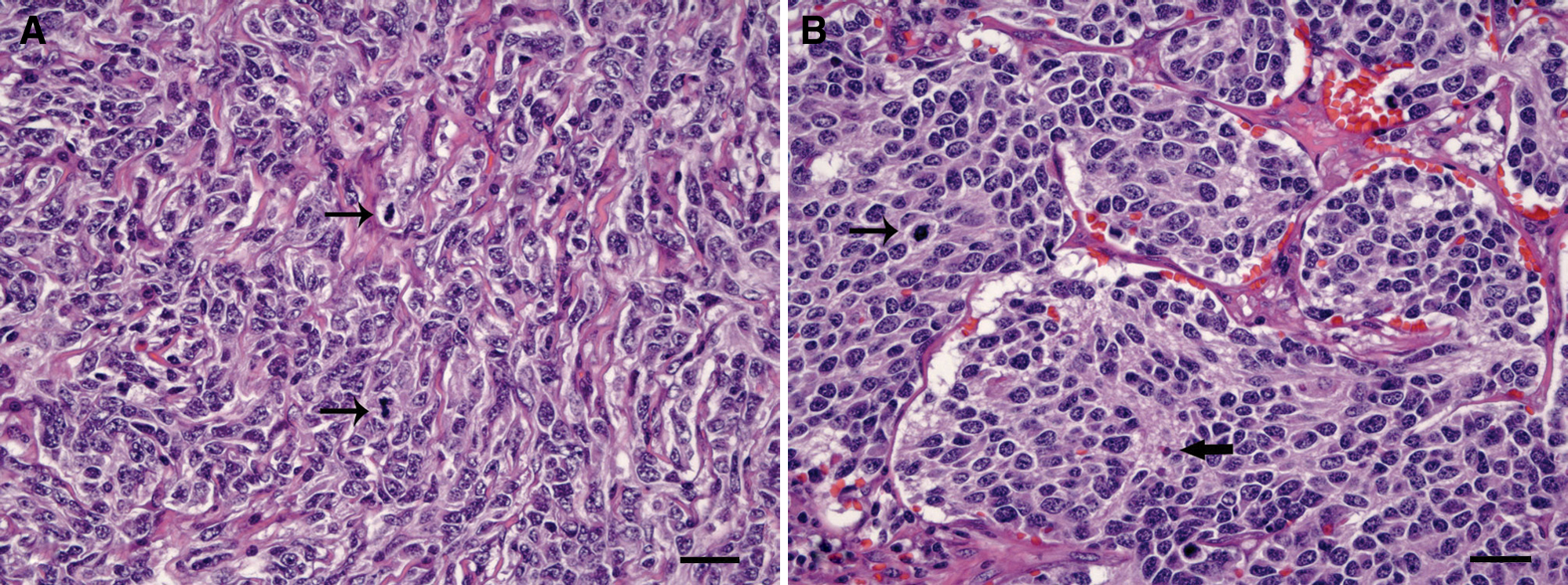
Pig.
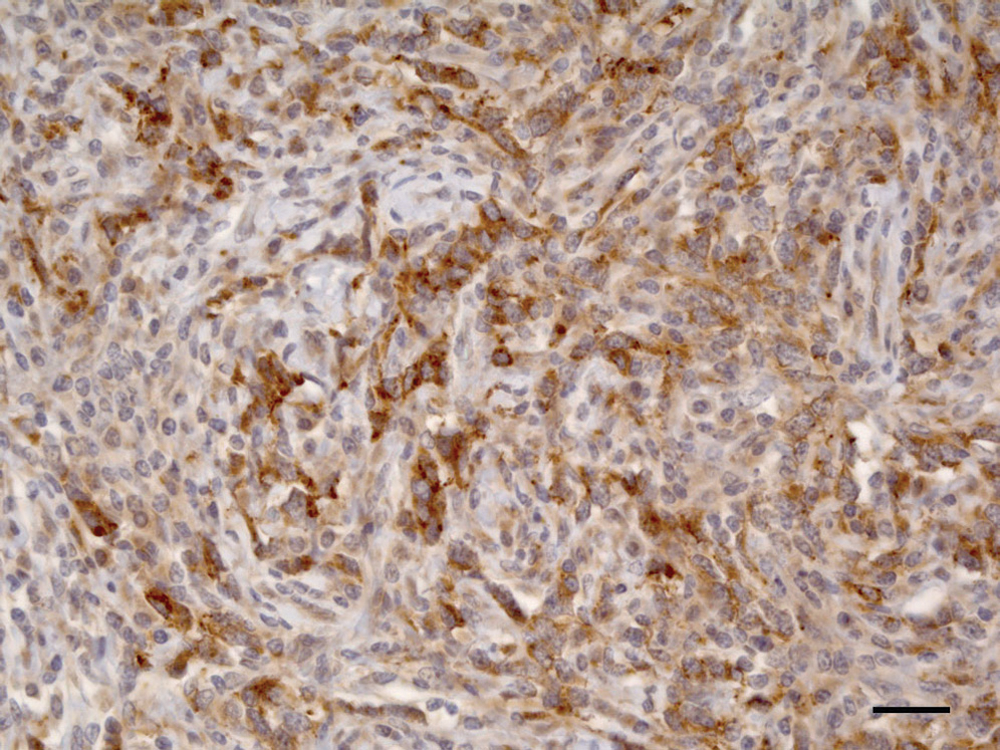
Pig, adrenal gland. Between 25–50% of neoplastic cells were positive in the cytoplasm for chromogranin A immunohistochemical staining. Bar = 25 µm.
Pheochromocytomas are the most common neoplasm arising in the adrenal medulla of animals, and develop most often in cattle and dogs, infrequently in other domestic animals, and are extremely rare in swine.3,4 In a 1968 abattoir survey of 3.7 million swine in Great Britain, only one 2.5-year-old female was diagnosed with an adrenal PHC. This neoplasm was, however, not immunohistochemically characterized. The extremely low prevalence of primary tumors of the endocrine glands in pigs is probably due to the fact that many animals are slaughtered before reaching maturity. Moreover, slaughter inspection may not reveal the true prevalence of these lesions because anatomical location of endocrine glands is such that only tumors of considerable size are likely to be found at meat inspections. 10
Symptoms related to production of norepinephrine or epinephrine, namely hypertension or tachyarrhythmias, occur in 25–60% of human patients with retroperitoneal paragangliomas. 12 In contrast, functional PHCs have been demonstrated infrequently in animals. 2 Tachycardia, edema, and cardiac hypertrophy observed in several dogs and horses with this type of tumor were attributed to excessive catecholamine secretion. Arteriolar sclerosis and widespread medial hyperplasia of arterioles have been reported in dogs with PHCs, which were associated with clinical signs suggestive of paroxysmal hypertension. 2 In suids, only 1 case of a functional pheochromocytoma was reported in a captive African warthog. 4 The warthog had epistaxis and elevated arterial pressure and serum norepinephrine levels; however, arterial sclerosis or medial hyperplasia of arterioles was not reported. In the present case, neither gross or microscopical lesions nor clinical signs were observed at slaughter suggestive of the existence of a functional PHC.
Multiple factors are involved in the pathogenesis of PHCs. In rats, factors include genetic background, chronic high levels of growth hormone or prolactin associated with pituitary tumors, dietary factors, and stimulation of the autonomic nervous system. In human beings, adrenal medullary hyperplasia appears to precede the development of PHC; however, 10% of sporadic adrenal PHCs are bilateral.3,6 In the present case, pituitary alterations could not be investigated because the cranial cavity was not inspected. Hence, the possible cause of the bilateral presentation of this tumor remains undetermined.
Differential diagnoses of adrenal medullary tumors in animals include neuroblastoma and ganglioneuroma. Neuroblastomas arise from primitive neuroectodermal cells, often in younger animals, and are differentiated from PHCs by the composition of small tumor cells resembling lymphocytes and the tendency to form pseudorosettes; neurofibrils or unmyelinated nerve fibers can be demonstrated. Ganglioneuromas are benign tumors arising in the medulla, composed of multipolar ganglion cells and neurofibrils with a prominent fibrous connective tissue stroma. 3 In human beings, nonhuman primates, and dogs, immunohistochemical evaluation against various neuropeptides is useful for confirmation of chromaffin cell origin of neoplasms, namely chromogranin A and synaptophysin among others.7,9 In the present case, a large number of cells in the primary neoplasm and some neoplastic cells in the metastasis were strongly positive for chromogranin A, but negative for total neurofilaments and synaptophysin. Histological differences between adrenal gland mass and lymph node metastasis are not surprising because different patterns of arrangement of neoplastic cells have been described in malignant PHCs. 2 Altogether, the findings confirmed the presence of a PHC and ruled out the possibility of neuroblastoma, ganglioneuroma, or other extra-adrenal paragangliomas. Based on available results, and even of very rare occurrence, PHC should be included in the differential diagnosis of neoplasms arising from the perirenal region in pigs.
Footnotes
Acknowledgements
The authors thank Dr. Martí Pumarola for his helpful comments on the diagnosis. The authors are grateful to Aida Neira, Ghizlane El Korchi, Blanca Pérez, Ester Blasco, and Lola Pérez for their technical assistance. The authors thank the logistic coordination of Dr. Enric Vidal from Servei de Suport a Escorxadors (Slaughterhouse Support Service; SESC-CReSA), financed by Agència de Protecció de la Salut (Health Protection Agency), Departament de Salut, Generalitat de Catalunya (Spain).
a.
Ref. A0430, Dako Diagnósticos SA, Barcelona, Spain.
b.
Ref. A0010, Dako Diagnósticos SA, Barcelona, Spain.
c.
Ref. N0142, Sigma-Aldrich Química SA, Madrid, Spain.
d.
EnVision+ System, Dako Diagnósticos SA, Barcelona, Spain.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
