Abstract
The objective of the present study was to determine whether offspring of dams infected with Mycobacterium avium subsp. paratuberculosis (Map) have an increased risk for Map infection. Antemortem and postmortem disease surveillance data were used to identify positive and test-negative ruminants born at the Zoological Society of San Diego (ZSSD) between 1991 and 2007 and to estimate cumulative lifetime incidence. A matched case-control study, nested within the population, was conducted and conditional logistic regression analyses were used to quantify the association between infection status of offspring and their dams. Cases (infected ruminants, n = 47) were matched to controls (test-negative ruminants, n = 152) by species, birth date, birth enclosure, and follow-up time to control for confounding factors. The overall cumulative lifetime incidence of infection was estimated at 2.2%, but it decreased over time and varied by species. There was a significant association between infection status of offspring and their dams (odds ratio [OR] = 6.8, P < 0.01), which is consistent with studies in domestic livestock species. The association was stronger for animals whose dam was diagnosed within 2 years of their birth (OR = 9.0, P < 0.01) than for animals whose dam was diagnosed more than 2 years after their birth (OR = 6.0, P < 0.01) compared to animals with test-negative dams. For positive animals born to a positive dam, 85.3% of the Map infections were attributable to having a positive dam. For the entire population of ZSSD ruminants, 36.8% of the cases were attributable to having a positive dam. Findings will help guide future management of Map infection in zoo ruminant populations.
Introduction
Mycobacterium avium subsp. paratuberculosis (Map) causes a contagious, bacterial infection of the intestinal tract in ruminant animals. Commonly called “Johne's disease,” this enteric infection manifests as a chronic wasting disease characterized by weight loss, diarrhea, loss of productivity, and, ultimately, death. 23 In addition to having devastating effects on the livestock industry, Johne's disease is a significant cause of morbidity and mortality in captive and endangered wildlife species (Manning EJB, Ziccardi M: 2000, Johne's disease and captive nondomestic hoofstock: prevalence and prevention. In: Proceedings of the American Association of Zoo Veterinarians and International Association for Aquatic Animal Medicine joint conference, pp. 432–434. AAZV, Media, PA). 6,15 Since 1991, Map has been either directly or indirectly responsible for deaths of numerous hoofed mammals housed at the Zoological Society of San Diego (ZSSD; San Diego and Escondido, CA).
The potentially calamitous effects of Johne's disease on captive wildlife health and conservation make it imperative that effective infection surveillance and control policies are identified (Wildlife Conservation Society, Zoological Society of San Diego, and White Oak Conservation Center: 1998, Diagnosis, prevention, and control of Johne's disease in non-domestic hoofstock. In: Proceedings of the workshop on diagnosis, prevention, and control of Johne's disease in non-domestic hoofstock, pp. 1–63. White Oak Conservation Center, Yulee, FL. Accessed Nov. 3, 2008. Available at: http://www.johnes.org/handouts/files/whiteoak.pdf). 13 Control of Johne's disease in nondomestic ruminant species has been largely based on methods used in the domestic livestock industry. 13 In cattle, the recommendation is that infected herd members be identified by diagnostic testing and immediately culled from the herd. 6 An additional key control point focuses on the substantial increase in risk of disease transmission from dam to offspring that has been documented in multiple studies. 1,2 Young animals, which are considered far more susceptible than adults, 8,10,25 are thought to acquire the infection from their mothers through suckling on manure stained teats, ingesting contaminated material in the calving pen, or ingesting bacteria shed directly in the mothers' milk or colostrum. 20 In utero transmission can also occur. 26 For these reasons, Johne's disease control policies for infected herds recommend that offspring be promptly removed from their dams and bottle-fed milk replacer, pasteurized milk, or milk from test-negative animals after birth. 6
Many of the methods used for domestic livestock, such as culling and herd depopulation, may not be appropriate for exotic hoofstock, particularly endangered species, because they adversely affect population sustainability and breeding efforts. Bottle feeding of young is not always possible for species with specialized dietary needs and can impede conservation efforts. Furthermore, the risk of transmission between dam and offspring has not been quantified for nondomestic species housed in zoos. There is currently a need to evaluate the epidemiology of Johne's disease in zoo animals to understand underlying patterns of infection transmission and to develop appropriate control methods to help mitigate risk of transmission.
The objective of the current study was to quantify the relationship between infection status of offspring and their dams in a variety of nondomestic wildlife species housed in a zoo environment. The ZSSD has an intensive Johne's surveillance program that began following an outbreak in 1991. The resulting longitudinal data set contains over 17 years of data, including serial fecal culture results and postmortem diagnostic tests. These data offer a unique opportunity to investigate the epidemiology of this slow-progressing disease in multiple, exotic species and to determine to what extent transmission between dam and offspring may occur. The management implications of this disease are important to the ZSSD collection and to zoological institutions throughout the world.
Materials and methods
Study population
The ZSSD consists of 2 facilities: the San Diego Zoo on 120 acres of land near downtown San Diego, CA, and the San Diego Zoo's Wild Animal Park (SDZWAP) on 1,500 acres of land in a rural setting near Escondido, CA. The ruminant collection at the ZSSD is dynamic, and animals are moved between the two ZSSD campuses and other zoological intuitions for breeding, conservation, and collection management. Animals are housed in a variety of single- and mixed-species enclosures that range in size from small holding pens to field exhibits over 60 acres. All animals are closely monitored and cared for on a daily basis by keepers and veterinary staff.
Johne's disease in the study population
Johne's disease was recognized in ruminants in field exhibits at the SDZWAP in 1991 and an infection surveillance program was initiated at both ZSSD institutions. Beginning in 1992, individual fecal samples were collected from ruminants at the ZSSD and sent to the Johne's Testing Center at the University of Wisconsin (Madison, WI) for testing. Liquid culture followed by genetic (polymerase chain reaction) identification of mycobacteria isolates was used for all samples throughout the study period. 4
The goal of the ZSSD surveillance program was to collect and test samples from all ruminants over the age of 10 months at least 1 or more times per year, although sampling was not always possible because of animal imports and exports, individual and species behavior, health-related events, and management-related reasons. If a fecal culture was contaminated by other organisms that prevented the isolation of mycobacteria, results from the sample were not used and efforts were made to submit another sample for that animal. The frequency of testing and the reason for testing animals varied over time, but in general, the majority of animals present in the collection in a given year were screened for Map infection.
When Map was isolated from a fecal sample, the animal was culled from the population. Intensive fecal sampling of familial contacts (parents, siblings, and offspring) and herd mates of the culture-positive animal was initiated with the occasional culling of these animals (depending on the species, outbreak severity, and suspected prevalence in the population). From January 1992 through July 2004, investigations focused on familial contact as well as enclosure mates. After July 2004, the intense infection investigation included all enclosure mates, regardless of lineage.
Postmortem histology and tissue cultures were also used to screen for infection. All animals that died during the study period underwent a postmortem examination, including complete histologic evaluation by board-certified staff pathologists unless advanced autolysis precluded histopathology. In addition to routine hematoxylin and eosin staining of all tissues, Ziehl-Neelsen acid-fast staining was performed on sections of small intestine, mesenteric lymph nodes, and intestinal fecal contents of clinical suspects, animals with postmortem lesions suggestive of Johne's disease, and herd mates and relatives of Johne's-positive animals when exposure history and pedigree information was available. Tissues were sent to the Johne's Testing Center for mycobacterial culture for cases where confirmation of Map was important for diagnosis or management. Data on postmortem findings were stored in a searchable, proprietary pathology database that included text reports and systematized nomenclature of medicine (SNOMED) codes for final diagnoses.
Case definition and control selection
A nested case-control study 18 was conducted in the population-based cohort of ruminants born at the ZSSD from 1991 through 2007 to identify associations between offspring and dam infection status. Eligible ruminants were identified from the ZSSD's computerized database of accessioned animals. Ruminants were eligible for the study if they were born at the ZSSD between 1991 and 2007 and if they lived to be at least 1 day old.
Case animals were defined as infected animals from which Map was ever isolated from feces or tissues or from which acid-fast organisms were identified in the lamina propria of the small intestine during histologic evaluations. Cases were identified using archived records of fecal culture results and the ZSSD pathology database with keyword searches and SNOMED code searches. Presence of infection was assumed to be a lifetime indicator of infection status, including the perinatal period. The age at diagnosis was determined from the date the first positive sample was collected and was used to match cases to appropriate controls.
Controls were selected from among test-negative animals present in the ZSSD cohort; they must had been evaluated for Map infection (via antemortem fecal screening or postmortem diagnostics) at least once (but often included multiple evaluations) over the age of 2 years and always tested negative. Up to 6 randomly selected controls were matched to each case on species, date of birth (± 1 year), and birth enclosure. To ensure equal follow-up time, all controls must have been evaluated for infection at an age that was equal to or greater than their corresponding cases' age at diagnosis. Regardless of a case's age, all controls must have been evaluated for infection at a minimum age of 2 years, because of the increased probability of detecting infection by fecal culture that has been reported to occur beyond the age of 2 years. 16,25 Some animals could not be classified as cases or potential controls. Infection status was classified as “unknown” for animals that were never tested or that were only tested at <2 years old (but always tested negative at this young age).
Dam status and other confounders and effect modifiers
The primary exposure of interest was the infection status of the dam. Dams were identified for all cases and controls from pedigree records, and the dam's infection status was based on the same criteria described previously for classifying the offspring as cases or potential controls. Case and control offspring were only eligible for inclusion in the case-control study if the infection status of their dam was known. The association between case and control status and the lifelong infection status of the dam was first evaluated as a bilevel predictor of risk. Test-negative dams were used as the baseline group for comparison. Then, as a second analysis, subjects whose test-positive dams were diagnosed within 2 years of the case or control offspring's birth and subjects whose dams were diagnosed more than 2 years after their birth were separately compared to subjects with test-negative dams.
Three other variables were assessed as potential confounders or effect modifiers: sex, whether the study subject nursed or was bottle fed, and age of the dam at calving. The dam's age was assessed as a bilevel predictor of risk. Subjects were classified as having an “old dam” or “young dam” relative to that particular species' median age at birth (the species median age at birth was determined using data from the entire cohort to calculate the median age at which offspring were born, separately for each species).
Statistical analyses
Cumulative lifetime incidence was estimated for each birth year as the number of ruminant animals born at the ZSSD in a given year that ever tested positive compared with all ruminants born that same year whose lifetime infection status was classified either test-positive or test-negative. Infection was presumed to occur around birth, as reported for other species, but age at diagnosis would be expected to differ even in the same animal if based on periodic surveillance cultures, clinical disease, or postmortem findings. Therefore, time from infection to diagnosis was not used in calculating incidence estimates. Animals with an unknown infection status were not included in incidence calculations. Cumulative lifetime incidence across all birth years, both pooled and within species, was also determined. Fleiss 95% confidence intervals (CIs) were calculated for species while variance inflation was incorporated through the intracluster correlation coefficient for pooled estimates. 3,19
Association of the infection status of the dam and her offspring was first assessed with univariate conditional logistic regression analyses. a Multivariable conditional logistic regression was used to consider 3 potential confounder and effect modifier variables separately, in conjunction with dam infection status. The Wald statistics for these variables and the effects of adding these variables on the odds ratios (ORs) for dam infection status were evaluated. Variation in the ORs of more than 15% between models fit with and without the potential confounder variable was interpreted to indicate significant confounding. 18 Effect modification was evaluated by comparing models with only main effects to those with main effects and their first-order interaction terms. 9
The attributable proportion among the exposed (i.e., the proportion of incidence in animals with positive dams attributable to having positive dams 22 ) was estimated from the equation (OR − 1)/OR, where the OR estimate from the univariate conditional logistic regression model containing the dams' infection status was used to approximate the relative risk. 22 The population attributable proportion (i.e., the proportion of the incidence in the entire population attributable to exposure to positive dams 22 ), was estimated as [(OR − 1)/OR] * f, where f was the proportion of infected individuals in the source population that had positive dams (f = 0.3378; 25 of 74 infected offspring had positive dams). Statistical analyses were performed using SAS. a Statistical significance was defined at the P < 0.05 level.
Results
A total of 9,435 ruminant animals representing 107 different species were born at ZSSD from 1991 to 2007, with 3,461 remaining in the population until they were at least 2 years old. Ruminants that left the population before the age of 2 years were either censored (i.e., the study ended before their reaching 2 years old) or they were removed for health, breeding, or management-related reasons, although many of these young animals had been tested for Map infection. In the targeted population, 74 animals from 30 different species were found to be infected with Map (34 animals were diagnosed at ≥2 years old, 40 animals were diagnosed <2 years old), and 3,224 animals from 101 species were test-negative at ≥2 years old. Through surveillance, 71 other ruminants at ZSSD were also found to be infected with Johne's disease that were born before 1991 or were imported from other institutions after birth.
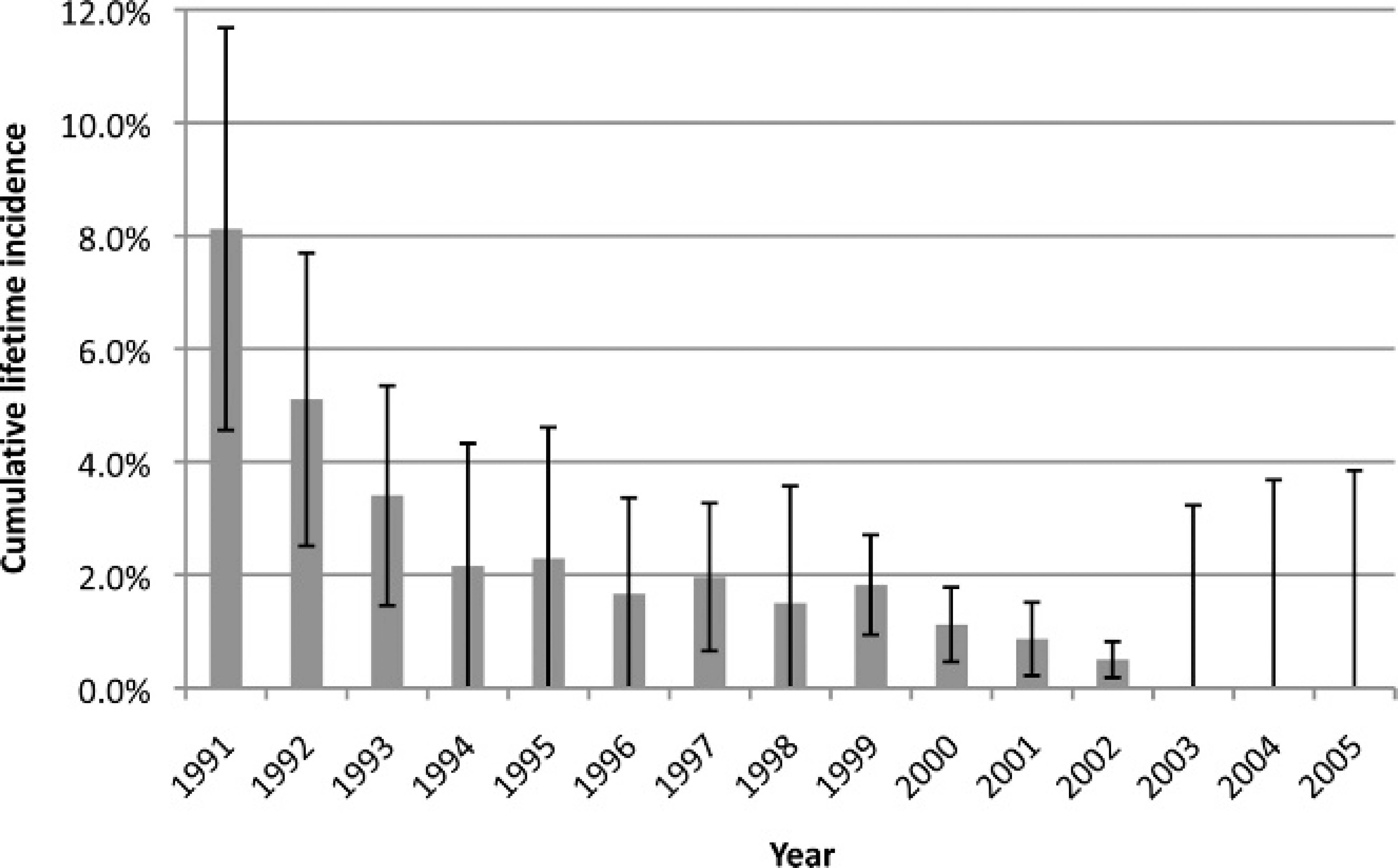
Estimated cumulative lifetime incidence of Johne's disease, by year of birth, at the Zoological Society of San Diego (ZSSD), 1991–2005. Birth cohorts from 2006 and 2007 are not included because of the lack of insufficient follow-up time. Cumulative lifetime incidence was defined as the proportion of animals that were born at the ZSSD that ever tested positive, compared with all animals in the same birth cohort whose lifetime infection status was classified either test-positive or test-negative.
Animals diagnosed as infected with Map ranged in age from 4 days to >13 years; the median age at diagnosis was 1.7 years. The median number of antemortem fecal samples was slightly higher for animals that were classified as negative (4 per animal, minimum = 0, maximum = 28) than for those that were found to be infected (3 per animal, minimum = 0, maximum = 13). Fecal samples were collected from animals that ranged from 7 days old to more than 16 years old, with a median spread of 2.8 years between collection of the initial and most recent sample. Forty-eight animals were diagnosed via antemortem fecal culture, whereas the remaining 26 animals were diagnosed during postmortem evaluations.
The overall cumulative lifetime incidence was 2.2% (74/3,298; 95% CI = 1.5–2.9%). Cumulative lifetime incidence by birth year was highest in 1991 (8.1%; 22/271 births; 95% CI = 4.6–11.7%) and decreased to 0% among animals born after 2003 (Fig. 1), although animals from later birth cohorts had less follow-up time. Total numbers of animals and cumulative lifetime incidence estimates by species are shown in Table 1.
The infection status of the dam could be determined for 98% (3,235/3,298) of the ruminants that were classified as infected or test-negative. Among the offspring that had infected dams, 25.5% (25/98) were positive for Map infection while only 1.4% (43/3,137) of offspring from test-negative dams were positive for Map infection. However, these 2 groups represented different species and time-period compositions.
In the case-control study, 47 cases and 152 controls matched on species, birth date, birth enclosure, and follow-up time were further evaluated. Eligible controls were not available for the 27 remaining cases. Cases subselected for the case-control study were diagnosed between the ages of 27 days to 8.2 years, and the median age at diagnosis was 1.8 years. All but 1 case had antemortem fecal cultures, ranging from 1–14 samples, with a median of 3 fecal samples collected per animal. Map was isolated from the feces or tissues in 45 of the ruminants; the remaining 2 did not have tissues available for culture and were diagnosed based on histopathology findings. The controls' age of evaluation ranged from 2 years to more than 14 years, and the average age was 6 years. All 152 controls had between 2 and 18 antemortem fecal cultures, and a median of 6 cultured samples per animal. Fifty-nine of the controls had died by the end of the study period and received postmortem evaluations, showing no evidence of Map. Eight of these animals also had negative postmortem tissue culture results.
Cases were more likely to have had infected dams than controls (OR = 6.8, P < 0.01, 95% CI = 2.122.5). In the multivariable model, the time lapse between when offspring were born and when the dams were diagnosed (≤2 years or >2 years) affected the odds of Map infection (OR = 9.0, P < 0.01, 95% CI = 1.4–57.9 and OR = 6.0, P < 0.01, 95% CI = 1.6–23.1, respectively). Neither sex of the offspring, nor bottle feeding, nor the age of the dam when the offspring was born modified the relationship between dam and offspring infection status. No significant interaction or additional confounders were identified. The 2 models with significant terms are shown in Table 2.
Among animals that had a positive dam, 85.3% of the cases were attributable to having a positive dam, assuming that having a positive dam was causal. Likewise, when considering the amount of exposure in the total population, 28.8% of cases were attributable to having a positive dam.
Discussion
Johne's disease surveillance data from the ZSSD program provide a well-documented, longitudinal source of test results and animal information for thousands of individual animals from a wide variety of species. Through the surveillance that began in 1991, infection status was determined for 94% (3,258/3,461) of all animals born into the population that were still cohort members at the age of 2 years (i.e., the minimum age at which they were classified as negative) and 98% (3,395/3,461) of their dams. This near-complete sampling and evaluation provided a unique and comprehensive dataset for estimating lifetime infection risk and evaluating relationships between dam and offspring infection status.
. Estimated cumulative lifetime incidence of Mycobacterium avium subspecies paratuberculosis (Map) infection among animals born at the Zoological Society of San Diego (ZSSD), 1991–2007, by species.
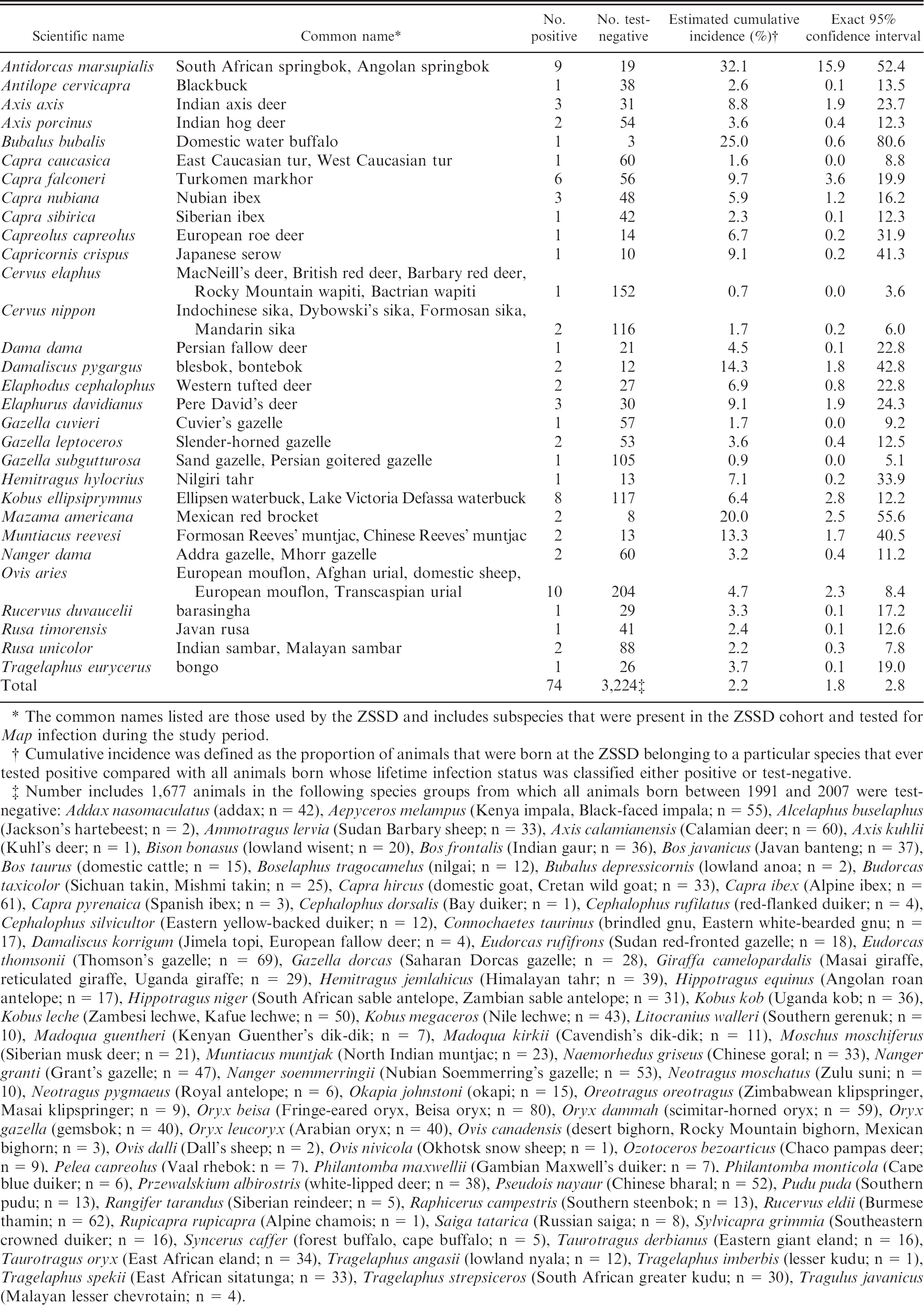
The common names listed are those used by the ZSSD and includes subspecies that were present in the ZSSD cohort and tested for Map infection during the study period.
Cumulative incidence was defined as the proportion of animals that were born at the ZSSD belonging to a particular species that ever tested positive compared with all animals born whose lifetime infection status was classified either positive or test-negative.
Number includes 1,677 animals in the following species groups from which all animals born between 1991 and 2007 were test-negative: Addax nasomaculatus (addax; n = 42), Aepyceros melampus (Kenya impala, Black-faced impala; n = 55), Alcelaphus buselaphus (Jackson's hartebeest; n = 2), Ammotragus lervia (Sudan Barbary sheep; n = 33), Axis calamianensis (Calamian deer; n = 60), Axis kuhlii (Kuhl's deer; n = 1), Bison bonasus (lowland wisent; n = 20), Bos frontalis (Indian gaur; n = 36), Bos javanicus (Javan banteng; n = 37), Bos taurus (domestic cattle; n = 15), Boselaphus tragocamelus (nilgai; n = 12), Bubalus depressicornis (lowland anoa; n = 2), Budorcas taxicolor (Sichuan takin, Mishmi takin; n = 25), Capra hircus (domestic goat, Cretan wild goat; n = 33), Capra ibex (Alpine ibex; n = 61), Capra pyrenaica (Spanish ibex; n = 3), Cephalophus dorsalis (Bay duiker; n = 1), Cephalophus rufilatus (red-flanked duiker; n = 4), Cephalophus silvicultor (Eastern yellow-backed duiker; n = 12), Connochaetes taurinus (brindled gnu, Eastern white-bearded gnu; n = 17), Damaliscus korrigum (Jimela topi, European fallow deer; n = 4), Eudorcas rufifrons (Sudan red-fronted gazelle; n = 18), Eudorcas thomsonii (Thomson's gazelle; n = 69), Gazella dorcas (Saharan Dorcas gazelle; n = 28), Giraffa camelopardalis (Masai giraffe, reticulated giraffe, Uganda giraffe; n = 29), Hemitragus jemlahicus (Himalayan tahr; n = 39), Hippotragus equinus (Angolan roan antelope; n = 17), Hippotragus niger (South African sable antelope, Zambian sable antelope; n = 31), Kobus kob (Uganda kob; n = 36), Kobus leche (Zambesi lechwe, Kafue lechwe; n = 50), Kobus megaceros (Nile lechwe; n = 43), Litocranius walleri (Southern gerenuk; n = 10), Madoqua guentheri (Kenyan Guenther's dik-dik; n = 7), Madoqua kirkii (Cavendish's dik-dik; n = 11), Moschus moschiferus (Siberian musk deer; n = 21), Muntiacus muntjak (North Indian muntjac; n = 23), Naemorhedus griseus (Chinese goral; n = 33), Nanger granti (Grant's gazelle; n = 47), Nanger soemmerringii (Nubian Soemmerring's gazelle; n = 53), Neotragus moschatus (Zulu suni; n = 10), Neotragus pygmaeus (Royal antelope; n = 6), Okapia johnstoni (okapi; n = 15), Oreotragus oreotragus (Zimbabwean klipspringer, Masai klipspringer; n = 9), Oryx beisa (Fringe-eared oryx, Beisa oryx; n = 80), Oryx dammah (scimitar-horned oryx; n = 59), Oryx gazella (gemsbok; n = 40), Oryx leucoryx (Arabian oryx; n = 40), Ovis canadensis (desert bighorn, Rocky Mountain bighorn, Mexican bighorn; n = 3), Ovis dalli (Dall's sheep; n = 2), Ovis nivicola (Okhotsk snow sheep; n = 1), Ozotoceros bezoarticus (Chaco pampas deer; n = 9), Pelea capreolus (Vaal rhebok; n = 7), Philantomba maxwellii (Gambian Maxwell's duiker; n = 7), Philantomba monticola (Cape blue duiker; n = 6), Przewalskium albirostris (white-lipped deer; n = 38), Pseudois nayaur (Chinese bharal; n = 52), Pudu puda (Southern pudu; n = 13), Rangifer tarandus (Siberian reindeer; n = 5), Raphicerus campestris (Southern steenbok; n = 13), Rucervus eldii (Burmese thamin; n = 62), Rupicapra rupicapra (Alpine chamois; n = 1), Saiga tatarica (Russian saiga; n = 8), Sylvicapra grimmia (Southeastern crowned duiker; n = 16), Syncerus caffer (forest buffalo, cape buffalo; n = 5), Taurotragus derbianus (Eastern giant eland; n = 16), Taurotragus oryx (East African eland; n = 34), Tragelaphus angasii (lowland nyala; n = 12), Tragelaphus imberbis (lesser kudu; n = 1), Tragelaphus spekii (East African sitatunga; n = 33), Tragelaphus strepsiceros (South African greater kudu; n = 30), Tragulus javanicus (Malayan lesser chevrotain; n = 4).
Map infection was detected in 30 of the 101 exotic ruminant species tested. Others have also reported that Map can affect a wide variety of zoo and wildlife species, 11,12,15 and some consider breed and species to play only minor roles in Johne's risk. 14 The variance in cumulative lifetime incidence between species could be due to differences in levels and timing of exposure, intrinsic variability in susceptibility to infection or disease, behavior, or other factors that covary with species. Much larger studies with careful control of confounders are needed to interpret the processes underlying species differences.
The observed decrease in cumulative lifetime incidence over time is a goal of a successful Johne's disease surveillance and management program. In the current study, cumulative lifetime incidence may have been underestimated if infected animals have not yet been detected, especially in more recent birth cohorts. Even when surveillance programs are present, true cases may go unnoticed for many years because of the long incubation period and intermittent shedding. 7,25 For example, in 2007, 2 Pere David's deer (Elaphurus davidianus), born in 1994 and 1997 to negative dams, were diagnosed with Map infection. Although tested multiple times, these animals did not have positive fecal cultures until more than 10 years after they were born. The source of their infection is not known, but such data demonstrate the importance of long-term follow-up and continued testing for Map infection in a herd with a history of Johne's disease. Based on the overall cumulative lifetime incidence estimate of 2.2% (1.5–2.9%), approximately 6 (4–9) animals per year would be expected to become infected in a population where 300 animals are born, assuming that new infections were acquired by young animals shortly after birth.
The strong association between infection status of offspring and their dams suggests that transmission of Map infection from mothers to their offspring is important in captive wildlife, as is recognized for dairy cows. 1,2 Numbers of animals were too small to determine if this relationship differed across species in the present study, although the OR for pooled cervid species was similar to that for combined bovid species (7.2 and 6.8, respectively). In domestic livestock, the most likely source of mother-to-offspring transmission is considered to be fecal contamination of the birthing pen. 20 Births at the ZSSD most often occur in large field exhibits of several acres (as opposed to small pens); the environments are relatively clean, and fecal matter is usually removed on a daily basis. Transmission in utero or through Map directly shed in milk or colostrum 20,26 remain intriguing alternative hypotheses for infection maintenance in the ZSSD population.
The odds for infection were highest for animals whose dams were diagnosed within 2 years of their birth. A previous report 21 suggested that Map transmission to fetal offspring was most likely to occur among dams that were heavy fecal shedders. Another study 2 found that transmission was not higher for calves of dams born within 2 years of diagnosis in their study herd, but contamination of calving pens by other cows was a major alternative source of transmission. At ZSSD, screening was conducted at intervals and generally not during the perinatal period. With Map shedding recognized to be intermittent, 25 timing of the relationship between Map shedding and infection of the offspring could not be more precisely characterized with existing data. The results of the current study support initiating fecal surveillance to target dams after parturition and during lactation to help find shedders and to identify offspring being exposed during a critically susceptible period.
. Conditional logistic regression models* describing the relationship between offspring and dam infection status.
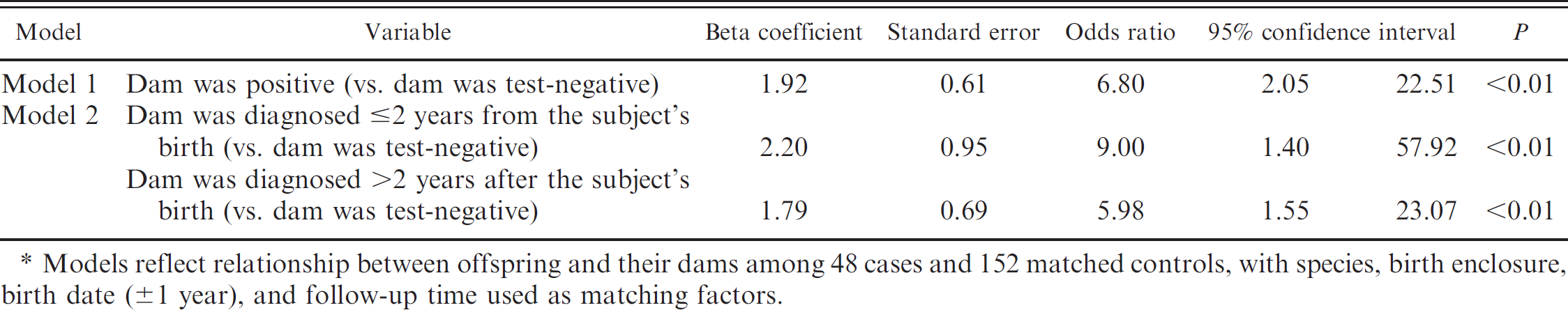
Models reflect relationship between offspring and their dams among 48 cases and 152 matched controls, with species, birth enclosure, birth date (±1 year), and follow-up time used as matching factors.
For infection management, it is important to estimate the magnitude of the contribution of dam-to-offspring transmission of Map in captive wildlife. The attribution of 85% of cases among offspring of positive dams and 29% of all cases in the population to dam-to-offspring transmission is remarkably similar to estimates from dairy cattle (85% and 34%, respectively). 1 The divergence in attributable proportion between the exposed population and the full cohort is because a small number of animals had positive dams. Many positive dams still had test-negative offspring (74%; 73/98) and, likewise, many positive offspring were born to test-negative dams (66%; 49/74). If some of these test-negative animals were false negatives, missed because of intermittent or sparse shedding of Map undetectable by culture, 24 the proportion of cases attributable to dam-to-offspring transmission would be higher than these present calculations. However, if a large portion of the cases in the population are not attributable to vertical transmission, culling offspring or dams of positive animals would not necessarily have an overall beneficial effect on Johne's incidence.
Ruminants born at the ZSSD were usually raised by their mothers, unless there were signs of maternal neglect, failure to nurse, health conditions that precluded parental rearing, or other management-related issues that prevented maternal care. This resulted in a small number of bottle-reared ruminants (n = 20). In the present study, bottle feeding had no effect on the relationship between infectious status of offspring and dam, although separation of dam and offspring is considered to reduce Map transmission among domestic livestock. 5,6 Many of the bottle-fed offspring were left with their mothers as long as possible (>1 day) to provide ample opportunity for colostral transfer and maternal bonding, and length of dam nursing was not recorded. Consequently, some animals classified as bottle fed may have nursed after birth and been exposed to Map during the susceptible open-gut period when nutrients, and perhaps infectious agents, are more readily absorbed in the intestinal lining. 20
The matched study nested within a cohort allowed for a focused investigation of the relationship between dam and offspring infection but limited comparisons to explore other potential transmission routes. Cases and controls were tightly matched on their birth enclosure and birth date (to account for environmental exposures to Map or herd stressors at the time of birth), species (to account for differences in species susceptibility or management), and follow-up time (for Johne's disease diagnosis, but allowing longer follow-up time for controls). Controls were required to be at least 2 years old, because an increased probability of detecting infection by fecal culture has been reported to occur beyond the age of 2 years. 16,25 The potential for misclassification errors among controls was further addressed by the use of serial testing strategies, using multiple types of diagnostic tests and selecting multiple controls per case when possible. The overall study design essentially matched animals on determinants of other transmission routes to control for many confounders during the design phase, ensure comparability of study subjects, and increase statistical efficiency for assessing vertical transmission. Further studies, perhaps controlling for dam infection status, are needed to assess the relative contribution of other types of transmission.
The assumption that Map infection was acquired by exotic ruminants near the time of birth was based on current hypotheses about transmission in livestock, where infected cows, goats, and sheep are generally presumed to have been infected as calves soon after birth. 20 Age-related susceptibility to Map infection is unknown for species represented in the current study. The time point of actual infection could not be determined in this observational study, and it is possible that animals were infected as adults. 17,20,25 This could account for some of the test-positive offspring with test-negative dams. Horizontal transmission would tend to mask the true magnitude of vertical transmission unless adult-to-adult spread was also more common between dams and their offspring.
Conversely, the association between dam and offspring status would be overestimated if directly related animals received more intense fecal screenings or were more scrutinized during postmortem examinations than other animals in the same pen. The extent to which screening varied among all ruminants in the present study is unknown. A requirement for fecal surveillance of all ruminants every year and thorough postmortem examinations as a part of normal operating procedures are necessary to counter these potential biases and are currently in place at the ZSSD. Additionally, the matching design (i.e., matching cases and controls by species and enclosure) often ensured that the matching animals remained enclosure mates throughout their lives, which helped balance the intensity of fecal surveillance between cases and controls.
Results from the current study extend the understanding of Map transmission to additional species and provide a useful model for other zoological institutions that implement Johne's disease control policies. The management and disease-control strategies used by the ZSSD over the study period were generally those recommended to manage the risk of Map transmission in zoos and are characteristic of many zoological institutions (Wildlife Conservation Society et al.: 1998, Diagnosis, prevention, and control of Johne's disease in nondomestic hoof-stock). 13 The association of infection status between dams and offspring suggests increased emphasis on testing dams for Map shedding when offspring susceptibility is high. However, the results also support efforts to decrease potential for transmission through other pathways apart from vertical transmission with research to determine the relative importance of alternative modes of transmission in zoo environments. Additional research is needed to understand the benefits and consequences of different disease-control strategies for controlling the spread of Johne's disease in captive wildlife.
Acknowledgements
The authors thank the Ellen Browning Scripps Foundation for their funding support; I. Stalis for data retrieval; E. Jezier, L. Keener, and personnel from the Zoological Society of San Diego (ZSSD) Clinical Pathology Laboratories for data tracking and maintenance; E. J. B. Manning and the University of Wisconsin's Johne's Testing Center for diagnostic testing, diagnostic data validation, and manuscript review; and personnel from the ZSSD Wildlife Disease Laboratories, veterinarians, animal keepers, and managers for their continued efforts on Johne's disease surveillance.
Footnotes
a.
SAS [statistical analysis software] procedure PHREG, version 9.2, SAS Institute Inc., Cary, NC.
