Abstract
Cutaneous tumors are not usually observed in pigs, and mast cell tumors, in particular, have been rarely described. The current report describes numerous, pink to red, elevated, 1–5-cm nodules in the skin of a 6-month-old slaughter pig. The histopathology of the nodules revealed well-demarcated, but poorly differentiated, populations of round cells that extended from the dermis into subcutaneous tissues. The presence of eosinophils and the observation, at high magnification, of cells with cytoplasmic metachromatic granules, after toluidine blue staining, confirmed the diagnosis of multiple cutaneous mast cell tumors.
Keywords
Skin tumors are rarely reported in swine. However, a variety of cutaneous neoplasms have been sporadically described, including melanoma, lymphangioma, rhabdomyoma, rhabdomyosarcoma, papilloma, sweat gland adenoma, fibroma, and hemangioma. 3 In domestic animals, mast cell tumors (mastocytomas) are usually described in dogs, less frequently in cats and cattle, and more rarely in horses.8,11,16 Therefore, most of the literature has focused on the tumors of dogs and cats, where several classifications and grading systems have been proposed. 10 There are few reports describing mast cell proliferations in pigs, and the majority refer to systemic mastocytosis with associated cutaneous tumors; more rarely, spontaneous neoplastic proliferations confined to the skin have been reported.1,13 Moreover, mast cell leukemia with systemic mastocytosis has been described in miniature pigs.1,18 Sometimes, it may be difficult to determine if mast cell aggregations represent a neoplastic or an inflammatory process because mast cells can accumulate during chronic inflammation and parasitic diseases. In pigs, the distinctive metachromatic granules present in mast cells can be difficult to demonstrate with the usual histochemical procedures.5,13,17 Additionally, scalding of slaughtered pigs may also degranulate superficial mast cells, thus making the histopathological diagnosis difficult. In the present work, multiple, poorly differentiated mast cell tumors are described in the skin of a slaughter pig.
A 6-month-old female pig, belonging to a batch of 170 pigs, was slaughtered in an officially inspected abattoir in Catalonia (Spain). During the post-slaughter inspection, multifocal and very numerous cutaneous nodules, 1–5 cm in diameter, were noticed in the skin of the body and the head. The nodules were slightly elevated and solid with a characteristic pink to red color (Fig. 1). The carcass was automatically rejected, and no internal organs were inspected. Three of the nodules were randomly collected, fixed in 10% buffered formalin, and routinely processed for histopathology. Sections were stained with hematoxylin and eosin, toluidine blue, and Giemsa. In order to eliminate the presence of a lymphoma, immunohistochemistry with 2 primary antibodies (anti–cluster of differentiation [CD]3 and anti-CD79α) a was performed with a standard avidin–biotin–peroxidase method, as previously described. 4
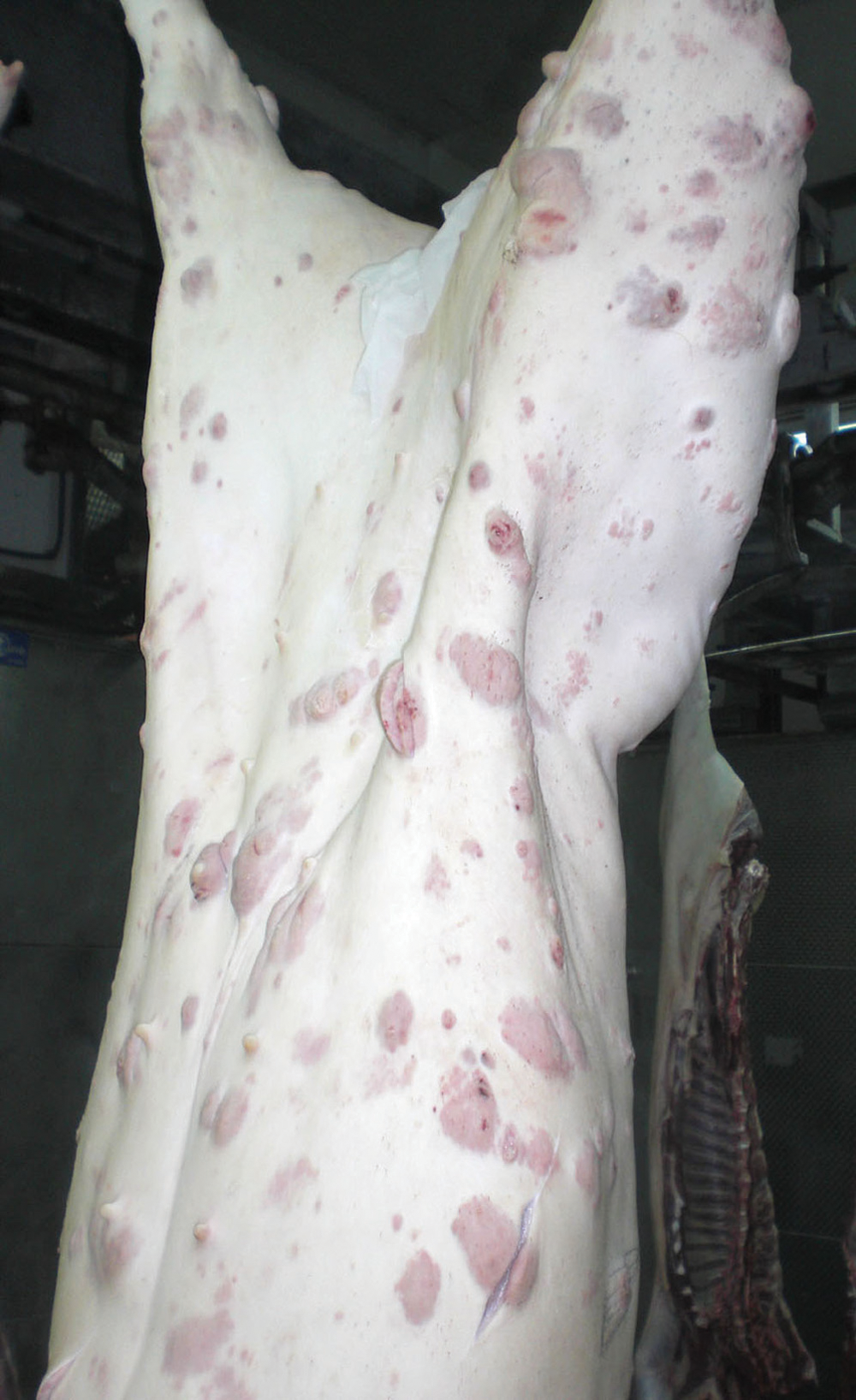
Multiple cutaneous, 1–5-cm, pink to red nodules (mast cell tumors) in the skin of a pig.
All nodules were histologically similar and consisted of a neoplastic proliferation of mast cells within the dermis and subcutaneous tissue, with ulcerations of the epidermis observed in some areas. The tumors were nodular, densely cellular, well demarcated, and nonencapsulated. Cells were arranged in sheets within a scant preexisting collagenous stroma. Cells were round to polygonal, 10 µm in diameter, with distinct borders and a moderate amount of basophilic cytoplasm; in some cells, there were small and poorly defined cytoplasmic granules identifiable only at high magnification. In general, nuclei were centrally located, oval with finely stippled chromatin, and no visible nucleoli. There was moderate anisocytosis and anisokaryosis with few mitoses. Mature eosinophils were scattered diffusely among the tumor cells (Fig. 2).
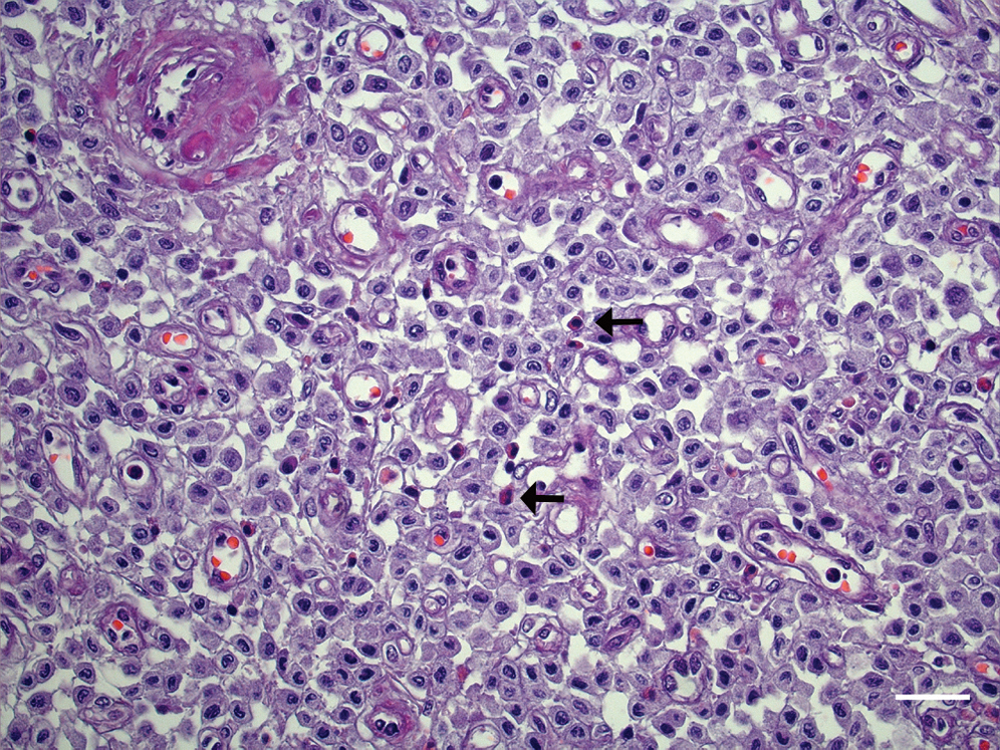
Cutaneous mast cell tumor, pig. Round neoplastic cells accompanied by scattered eosinophils (arrows). Hematoxylin and eosin. Bar = 25 µm.
In toluidine blue–stained sections, at high magnification (1,000×), abundant densely packed purple cytoplasmic granules were observed in a moderate number of tumor cells (Fig. 3). Immunohistochemistry with anti-CD3 and anti-CD79 as well as Giemsa staining were negative in the 3 nodules.
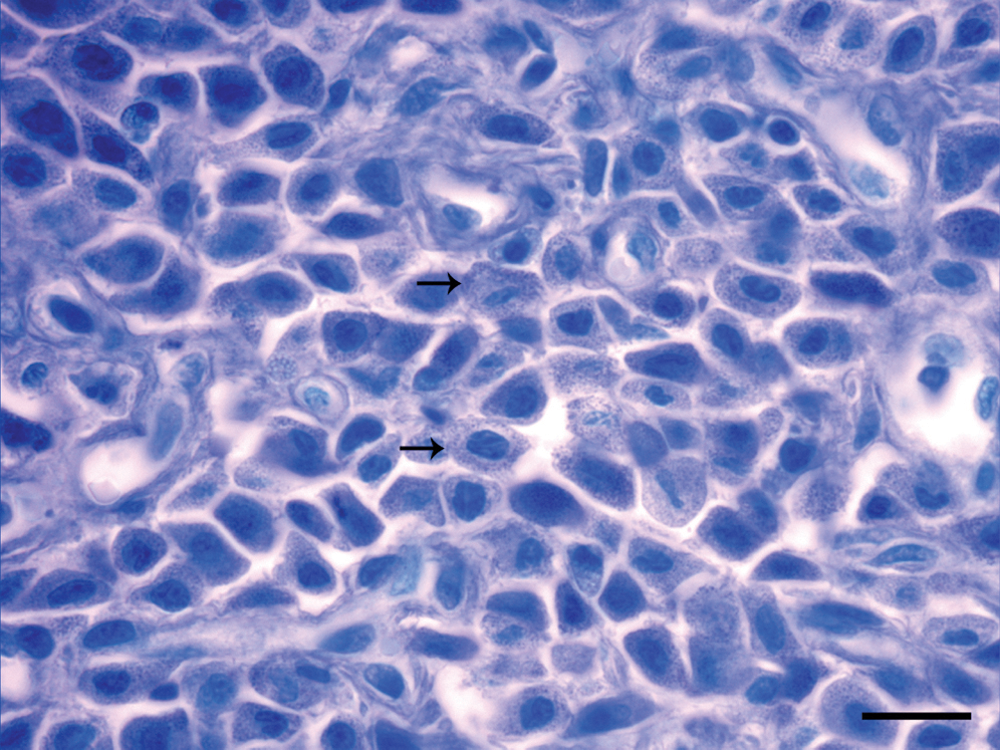
Cutaneous mast cell tumor, pig. Neoplastic mast cells with abundant metachromatic cytoplasmic granules (arrows). Oil immersion. Toluidine blue. Bar = 15 µm.
Mast cell tumors are common in many animal species. Within the skin, the neoplasm can be focal or multicentric and may occasionally involve internal viscera such as the spleen, the liver, or the intestine. 7 Although relatively frequent in some species such as dogs and cats, only few cutaneous mast cell tumors in pigs have been described.12,13 As in the present case, metachromatic granules of mast cells were difficult to demonstrate with hematoxylin and eosin in these descriptions; hence, oil immersion with a toluidine blue stain was necessary to identify a few cytoplasmic metachromatic granules in a relatively small population of neoplastic mast cells. The presence of these granules eliminates other cutaneous round cell tumors such as lymphomas, plasmacytomas, histiocytomas, histiocytic sarcomas, Merkel cell tumors, and malignant melanomas. Furthermore, the negative results obtained with the anti-CD3 and anti-CD79α immunohistochemistry precludes the presence of a granular lymphoma. Therefore, the results lead to a diagnosis of cutaneous mastocytoma.
Several grading systems have been proposed to classify canine mast cell tumors. The system most commonly used classifies the tumor from grades I to III, depending on how well the mast cells are differentiated, mitotic activity, location within the skin, invasiveness, and the presence of inflammation or necrosis, with grade III being the most aggressive (characterized by the presence of undifferentiated, immature mast cells with a high potential for metastasis). 14 In the current case, the presence of multiple large nodules, the extension into the dermis, and the poorly differentiated mast cells could be comparable to grade III described for dogs. Recently, a new 2-tier histologic classification for canine cutaneous tumors has been proposed. 9 This grading system is based on the presence of mitotic figures, multinucleated cells, bizarre nuclei, and karyomegaly to predict biological behavior, likely time to metastasis, new tumor development, and survival time. In the present case, although the neoplasia did not show any of these histological parameters, the presence of multiple cutaneous tumors suggests a malignant behavior. Unfortunately, the low prevalence of mastocytomas in pigs makes a direct comparison with classification and prognosis of canine tumors difficult.
In rodents (mice and rats) and human beings, mast cells have been classified into connective tissue (typical) mast cells and mucosal (atypical) mast cells, based on the morphology of their granules, tinctorial properties, formaldehyde resistance, and granular components.15,19 However, in other species like the dog, mast cells cannot be classified according to this system. 10 In the present case, with the aim to further characterize the population of mast cells present in the tumors, immunohistochemistry with several antibodies used to identify mucosal and connective tissue mast cells in rodents was performed. To identify mucosal mast cells, antibodies against rat mast cell protease II and mouse mast cell protease I, considered specific markers of mucosal mast cells in rats and mice, respectively, were used. b Similarly, to identify connective tissue mast cells, an antibody against mast cell tryptase V-13, considered a specific marker of connective tissue mast cells in rodents, was used. c In all cases, the staining obtained was negative or considered to be nonspecific (but staining in rodent tissues was positive). Therefore, the antibodies against rodent mast cell proteases seem inappropriate for use in pigs. The results further confirm the existence of species-specific differences in mast cell tryptases and proteases, as previously shown in rodents and human beings and postulated in dogs, 10 thus making difficult mast cell classification and precise identification across species.
Heavy accumulations of mast cells may occur in a variety of parasitic, mycotic, allergic, and idiopathic inflammatory syndromes, in which cases the term mastocytosis is warranted. In this way, cutaneous and systemic mastocytosis have been described in several domestic species. Cutaneous mastocytosis (equivalent to urticaria pigmentosa in human beings), consisting of diffuse dermal and subcutaneous infiltrates of well-differentiated mast cells that occur over large areas of the body, have been rarely described in young dogs and cats.6,10 In a previous report, cutaneous and systemic mastocytosis was associated with Mycoplasma (Eperythrozoon) suis infection in 3 slaughter pigs from the same batch. 2 The authors observed intraerythrocytic organisms within the mast cell aggregates, thus suggesting an association between the infection and the mastocytosis. Nevertheless, mast cell leukemia with systemic mastocytosis was described in a miniature pigs not associated with M. suis infection.1,18 In the present case, the carcass was automatically rejected, and no internal organs or blood smears were investigated; hence, a systemic mastocytosis or mast cell metastasis cannot be ruled out. However, the absence of other affected pigs within the same batch, and the fact that no intraerythrocytic organisms were observed during the histopathological assessment following a Giemsa stain, makes an active infection with M. suis unlikely. Despite the multiple skin nodules, the poorly differentiated mast cells precluded the diagnosis of cutaneous mastocytosis and supported the diagnosis of multiple cutaneous mast cell tumors.
Footnotes
Acknowledgements
The authors thank Aida Neira, Ghizlane El Korchi, Blanca Pérez, Mónica Pérez, and Emma Martínez for their technical assistance, as well as Josefina Quintana and Nuria Erra (slaughterhouse veterinary inspectors) for macroscopic pictures and case description. The authors thank the logistic coordination of Dr. Enric Vidal from Servei de Suport a Escorxadors (SESC-CReSA), which is financed by Agencia de Protecció de la Salut (APS), Departament de Salut, Generalitat de Catalunya.
