Abstract
The morphology of ovarian tumors is characterized by a variety of histological features causing diagnostic difficulties. In human medicine, HBME-1 (Hector Battifora mesothelial epitope)-1 is one of the immunohistochemical markers employed in the diagnosis of ovarian epithelial tumors. The aim of the current study was to investigate the reliability of the marker HBME-1 in canine ovaries, granulosa cell tumors, and epithelial ovarian neoplasms to determine whether this marker could be included in an immunohistochemical panel for differential diagnoses of canine ovarian tumors. Samples were obtained from 4 normal ovaries, 10 granulosa cell tumors, and 18 epithelial ovarian tumors. After formalin fixation and paraffin embedding, tissue sections were stained with hematoxylin and eosin and probed immunohistochemically for the HBME-1 marker. Granulosa cells and related tumors were consistently negative for HBME-1. Normal ovarian surface epithelium and 17 out of 18 ovarian epithelial tumors were positive for HBME-1. The results suggested that HBME-1 would be a useful marker for the differential diagnosis of ovarian tumors in the dog.
Among domestic animals, ovarian tumors are most commonly observed in female dogs and cows. 4 In dogs, the reported incidence of ovarian neoplasm ranges from 0.5% to 6%. 6 Epithelial tumors are derived from the ovarian surface epithelium, a single layer of flat to cuboidal mesothelial cells that cover the ovary. 1 Epithelial tumors are the most frequently reported ovarian neoplasms, both in human and veterinary medicine.8,9,14 In women, as in female dogs, adenocarcinomas are more common than adenomas 6 and are characterized by early metastatic spread and poor clinical course; they extensively implant on the peritoneum and metastasize to the lymph nodes.10,18 Granulosa cell tumors (GCTs), classified as sex cord stromal tumors, are characterized by an indolent course, with a 10-year survival rate of 90% in woman. 18 In human and canine species, GCTs are considered uncommon, low-grade neoplasms that rarely metastasize.9,18 Histologically, GCTs exhibit a great variety of growth patterns (microfollicular, macrofollicular, trabecular, diffuse, Sertoli-like), which is recognized both in human beings and dogs.16,18 A mixture of patterns is often reported in a single tumor, 17 and GCTs frequently share histological patterns with epithelial ovarian neoplasms.15,19 The latter is particularly true for undifferentiated carcinomas, which may resemble a diffuse GCT. 18 Ovarian epithelial tumors are frequently malignant, and they progress rapidly; in contrast, GCTs are mostly benign. 18 Therefore, the differential diagnosis between these 2 conditions has great prognostic significance and represents a diagnostic challenge. Immunohistochemical studies focused on this topic have typically employed panels of antibodies to distinguish epithelial tumors from sex cord stromal tumors.
Inhibin-α (INHα) is a heterodimeric polypeptide hormone produced by granulosa cells and is considered a specific marker that can confirm the diagnosis of GCT in human beings and dogs.15,16 Cytokeratin 7 (CK7) is selectively expressed by normal and neoplastic ovarian surface epithelium 16 and is a marker for ovarian epithelial cell tumors. Other immunohistochemical antibodies, such as the mesothelial marker HBME (Hector Battifora mesothelial epitope)-1 raised against mesothelioma cells from human patients with malignant epithelial mesothelioma, have also been investigated for identifying ovarian epithelial cell tumors. HBME-1 reacts with undetermined epitopes on the microvilli surface of mesothelial cells, 12 and it is largely expressed by mesotheliomas 12 and ovarian adenocarcinomas that originate from the ovarian surface epithelium, a modified pelvic mesothelium. 13 HBME-1 has been rarely used in veterinary medicine but, recently, it was successfully employed for the diagnosis of feline mesothelioma. 2 To the authors’ knowledge, HBME-1 has never been applied to canine tissue specimens, and it has never been tested on ovarian samples.
The present study aimed to improve the immunohistochemical panel recently suggested for the differential diagnosis of canine ovarian tumors 15 by investigating HBME-1 in 4 normal ovaries and 28 ovarian neoplasms from adult female dogs. The neoplasms were sampled from dogs of different breeds, ranging in age from 6 to 15 years old; 10 of the neoplasms were GCTs and 18 were primary ovarian epithelial tumors. The specimen collection was realized as part of an ongoing multicenter study on canine ovarian cancer involving the Veterinary Schools at the Universities of Milan, Naples, and Sassari. All samples were fixed in 10% neutral buffered formalin, processed by conventional methods, and paraffin embedded. For each sample, 5-µm thick sections were obtained and stained with hematoxylin and eosin. The growth patterns of canine GCTs were described according to the morphological descriptions of human GCTs, 18 as follows: microfollicular, macrofollicular, trabecular, and solid. The presence of Sertoli cell differentiation was also recorded. Immunohistochemical staining of both normal and neoplastic ovaries was performed on deparaffinized sections with the avidin–biotin–peroxidase (ABC) complex a procedure. 7 The antibodies included the monoclonal antibody against INHα b (dilution 1:40) and HBME-1 c (dilution 1:1-000). For 14 samples (8 GCTs and 6 epithelial tumors), HBME-1 staining was performed with and without heat-induced antigen retrieval (10 min microwave in pH 6 citrate buffer) in order to test whether this procedure could improve the immunohistochemical reaction. For the remaining samples, only heat-induced antigen retrieval was performed. Negative controls were performed under identical conditions, but the sections were incubated with normal horse serum instead of specific antibodies. Diaminobenzidine tetrahydrochloride a or 3-amino-9-ethilcarbazol a was used as chromogen. Finally, the sections were counterstained with Mayer hematoxylin. According to the previous report, 15 tumors were graded based on the proportion of cells stained by each antibody: 4+ indicated 60–100% of neoplastic cells were stained, 3+ indicated 30–60% of cells were stained, 2+ indicated 10–30% of cells were stained, and 1+ indicated that less than 10% of neoplastic cells were stained.
Histological and immunohistochemical results are shown in Table 1. Normal ovaries included oocytes, follicles at different stages of development, and corpora lutea, embedded within a dense fibrovascular stroma. No lesions were observed. The 10 GCTs examined were characterized by different patterns of growth, including follicular, trabecular, Sertoli-like, diffuse (solid), and sarcomatoid patterns (Table 1). Microfollicular and diffuse (solid) were the prevailing types, but in 9 out of 10 cases, more than 1 histological pattern coexisted. Neoplastic cells were moderately pleomorphic, small, round to oval in shape, with indistinct cell borders, and scant to moderate faintly eosinophilic cytoplasm. Nuclei were generally round to oval and hyperchromatic, with dense chromatin. In areas that showed solid growth, neoplastic cells were polygonal, with abundant cytoplasm, round to oval nuclei, and marginated chromatin. The 18 epithelial tumors were generally composed of irregular cystic cavities and tubular or papillary structures, and often projected into the cystic cavities (Fig. 1A). The papillae consisted of thin, connective tissue stalks lined with single or multiple layers of cuboidal to columnar epithelial cells. Neoplastic cells, often ciliated, had variably distinct cell borders, medium nucleus-to-cytoplasm ratio, moderate amount of homogeneous, eosinophilic cytoplasm, and round to oval nuclei, basally located, with 1 or 2 central nucleoli. Anisokaryosis and anisocytosis were generally moderate; mitotic figures ranged from 0 to 1 per high power field. Four out of 18 epithelial neoplasms were characterized by multifocal areas of “diffuse/solid” growth, in which solid sheets of polygonal cells were closely opposed and separated by scant fibrovascular stroma. In 1 case (case no. 16), the diffuse (solid) pattern of growth was predominant (Fig. 1B). Only 1 tumor was diagnosed as adenoma; the other 17 cases were classified as adenocarcinomas, characterized by stromal invasion (Fig. 1C), large areas of necrosis, and hemorrhages (Table 1).
Histological and immunohistochemical findings in 28 canine ovarian tumors tested in the current study.*
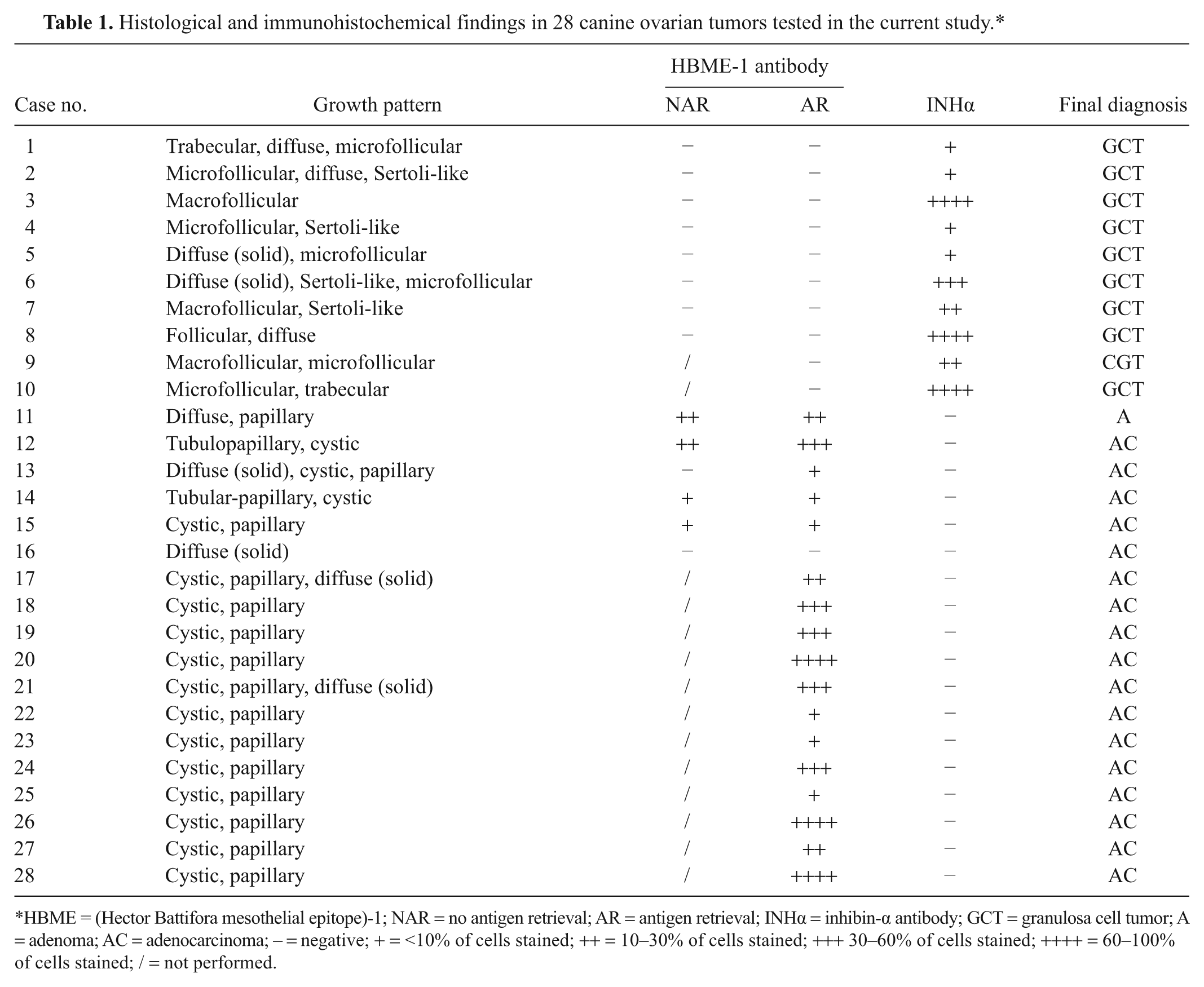
HBME = (Hector Battifora mesothelial epitope)-1; NAR = no antigen retrieval; AR = antigen retrieval; INHα = inhibin-α antibody; GCT = granulosa cell tumor; A = adenoma; AC = adenocarcinoma; – = negative; + = <10% of cells stained; ++ = 10–30% of cells stained; +++ 30–60% of cells stained; ++++ = 60–100% of cells stained; / = not performed.
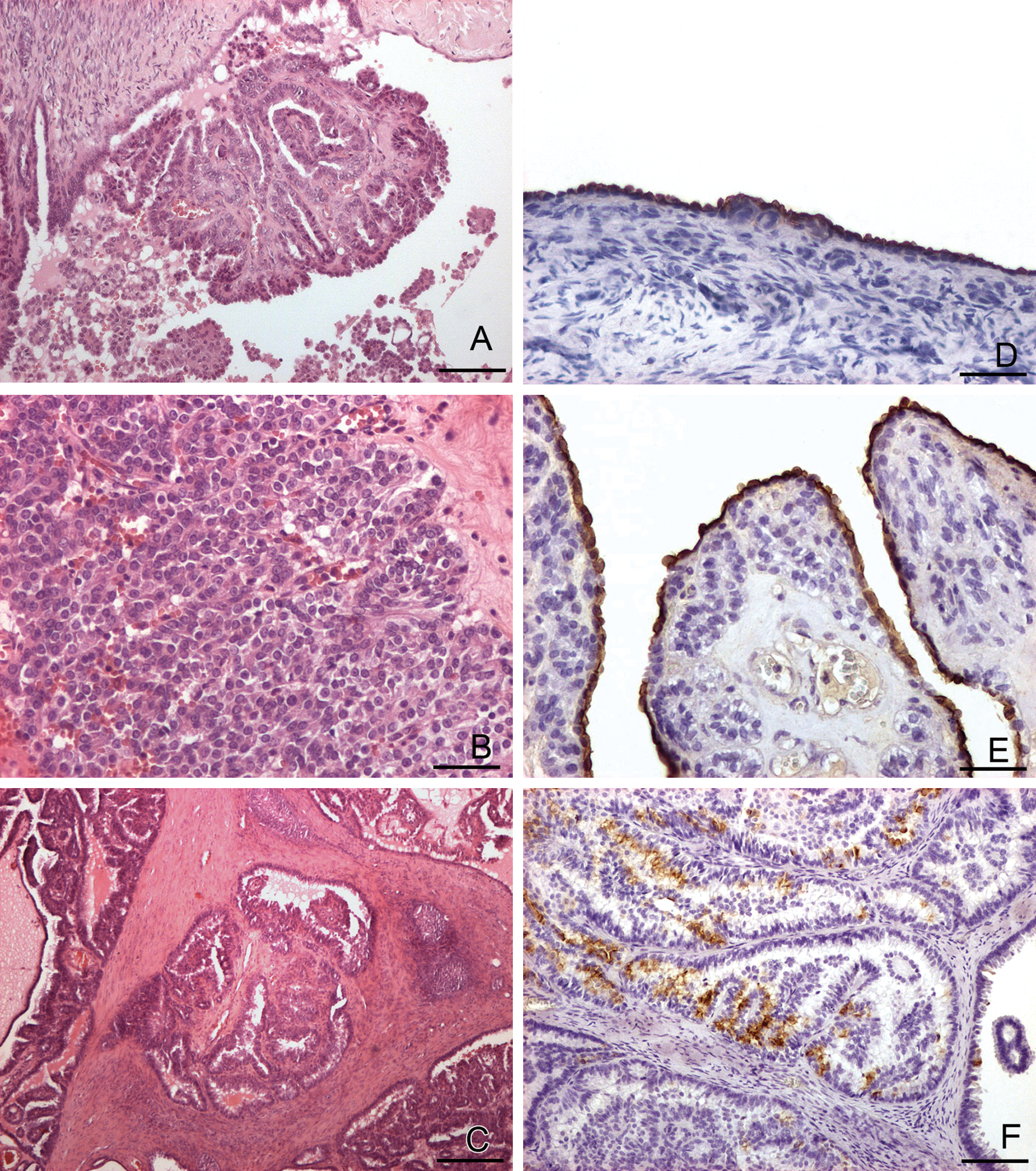
Histological features of canine ovarian epithelial tumors (A–C) and immunohistochemical expression of HBME-1 (D) in normal and neoplastic canine ovaries (E, F).
Immunohistochemically, in normal ovaries, the surface epithelium strongly reacted for HBME-1, and immunostaining was restricted to the apical region of the cytoplasm (Fig. 1D). Granulosa cells were negative immunolabeled with HBME-1 but stained positively for INHα. Corpora lutea were negative for all the markers employed.
All 10 GCTs were negative for HBME-1 but consistently expressed INHα. The distribution of INHα immunoreactivity was variable among tumors; the percentage of positively labeled neoplastic cells ranged from less than 10% to 60–100%.
All 18 epithelial tumors were negative for INHα, and 17 out of 18 were positively labeled by HBME-1. The immunolabeling was restricted to the apical portion of the neoplastic cells that lined the tubular, cystic, or papillary projections within the tumor (Fig. 1E). The percentage of labeled cells varied from less than 10% to 60–100%; positive and negative neoplastic cells coexisted in close proximity (Fig. 1F).
The heat-induced antigen retrieval procedure moderately increased the number of cells immunolabeled with HBME-1 (Table 1) compared to untreated samples. The reactivity was improved in 2 out of 6 ovarian epithelial tumors tested (case nos. 12 and 13). The multifocal areas that showed “diffuse/solid” growth patterns were not labeled by HBME-1, and 1 sample (case no. 16) was totally negative.
In human pathology, several studies have identified the immunophenotype of normal ovaries in order to provide a diagnostic tool for differentiating between surface epithelial tumors and sex cord stromal tumors of the ovary. 12 In particular, it was demonstrated that CK7 and HBME-1 were selective markers for normal and neoplastic ovarian surface epithelium,3,12 and granulosa cells demonstrated strong reactions to anti-INHα antibodies. 17 Recent reports found that the expression of CK7 and INHα in canine ovarian tumors was similar to that found in human ovarian tumors. 16 In the present study, the surface epithelium of normal canine ovaries was consistently immunostained by HBME-1. This indicated that, in dogs, as in human beings, HBME-1 can be considered a good marker of ovarian surface epithelium. Among the tumors examined, GCTs were negative for HBME-1. Furthermore, even after heat-induced antigen retrieval, the only cells recognized by HBME-1 were the normal epithelial cells in tracts of surface epithelium that happened to be present in the sections. On the other hand, GCTs were always positive for INHα, in accordance with previous reports in dogs. 16 Seventeen out of 18 ovarian epithelial tumors were positively labeled by the antibody HBME-1, with no differences between malignant and benign lesions. The percentage of immunolabeled cells was variable (Table 1), and immunolabeling was restricted to the apical portions of epithelial cells that lined papillary, tubular, or cystic neoplastic structures. Therefore, in dogs, the observed HBME-1 pattern of immunostaining corresponded to the “thick membrane” pattern described by several authors in human mesotheliomas and in ovarian tumors.5,17
The results indicated that heat-induced retrieval is recommended before applying anti–HBME-1 antibody. This procedure increased the reactivity of ovarian epithelial neoplastic cells in 2 cases (Table 1).
Only 1 adenocarcinoma (case no. 16) was negative for the antibody HBME-1. The case showed a “solid/diffuse” pattern of growth and, in a previous report, showed positive staining for CK7. 15 The lack of HBME-1 staining was also observed in limited areas of other adenocarcinomas considered in the current study. This condition may be due to the loss of microvilli, structures that could be absent in carcinomas characterized by a solid pattern of growth, as suggested previously. 11 Thus, to avoid false-negative immunohistochemical results, it is necessary to collect several samples from ovarian neoplastic masses and to examine different sections.
In conclusion, the present study demonstrated that HBME-1 would be a reliable marker for canine ovarian surface epithelium and derived tumors. The results suggested that this marker merits inclusion in the immunohistochemical panel for the differential diagnosis of canine ovarian tumors.
Footnotes
Acknowledgements
The authors thank Mr. Marco Colombo for photographic support. Dr. Giovanni Tortorella (veterinary service ASL Lecce) is gratefully acknowledged for having provided cases of ovarian tumors, as well as Prof. Luisanna Busincu (Department of Pathology, Medicine Faculty, Sassari) for helping in the diagnosis of some cases of ovarian tumors according to the human WHO classification.
a.
Vector Laboratories Inc., Burlingame, CA.
b.
Serotec Co., Oxford, United Kingdom.
c.
Dako North America Inc., Carpinteria, CA.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
