Abstract
A previously beach-stranded, juvenile, male, bottlenose dolphin (Tursiops truncatus) was diagnosed with vertebral osteomyelitis of unknown etiology. Antemortem serological testing suggested past or current Brucella sp. infection; however, this could not be confirmed prior to death despite multiple isolation attempts from aspirates, blood, and biopsies. Systemic antibiotics were administered for over a year to control the suspected infection; however, the animal succumbed peracutely to infection by a highly pathogenic, enterotoxin-secreting Staphylococcus sp. Gross necropsy findings included a fistulous tract leading to locally extensive osteomyelitis of a coccygeal vertebra with sequestra and osteophytes from which a Brucella species was isolated. Histopathological examination of intestine revealed pseudomembranous enteritis with a uniform population of intraluminal Gram-positive cocci. Staphylococcus aureus was isolated in pure culture from the intestine and tested positive for the staphylococcal enterotoxin A gene by polymerase chain reaction analysis. Serum taken shortly before death had endotoxin and elevated antibody titers to staphylococcal enterotoxin A when compared to samples collected during a period of apparent good health 18 months earlier. The isolation of a pyrogenic toxin superantigen-producing staphylococcal isolate, clinical signs, and diagnostic findings in this animal resembled some of those noted in human toxic shock syndrome. The present case highlights the clinical challenges of treating chronic illnesses, complications of long-term antibiotic use, and promotion of pathogenic strains in cases of prolonged rehabilitation of marine mammals.
Keywords
A male bottlenose dolphin (Tursiops truncatus) estimated to be unweaned and 1.5 years of age stranded on the Texas coast in September 1998. Significant clinical signs on presentation included an unhealed shark bite approximately 9 cm × 15 cm on the left dorsolateral abdomen caudal to the dorsal fin, loss of part of the dorsal fin, dehydration, and weight loss. A small area of swelling was noted on the animal’s right peduncle close to where the trunk merges with the flukes. There were no breaks in the skin at the site, and the lesion appeared unrelated to the shark bite. After 6 months of rehabilitation at a Texas facility, the animal was transferred to Mystic Aquarium (Mystic, Connecticut) for continued rehabilitation.
Initial radiographs of the peduncular lesion revealed sclerosis of the affected vertebra with calcification in the adjacent soft tissues but normal intervertebral spacing (Fig. 1a). The lesion was interpreted as inactive, and no etiology was established. Throughout the next 8 months, serial blood sampling revealed that the animal’s white blood cell count levels remained within the high end of the reference interval for the species 33 with occasional, mild elevations of neutrophils and fibrinogen. On 4 occasions during this period, oral administration of enrofloxacin (approximately 6.0 mg/kg) and clindamycin (approximately 5.5 mg/kg) for 7–10 days or just enrofloxacin was associated with a rapid return to normal white cell differential and total cell count. Minor subtle changes in size and palpable temperature were noted in the peduncular lump, but these did not consistently correlate with hematological or behavioral changes. Bacterial isolation was attempted on superficial punch biopsies of the skin and blubber overlying the affected vertebra, as well as a discharge from a fistulous track that developed from the lesion, but no organisms were recovered. However, blood samples taken at this time were seropositive for Brucella antibodies on the card, buffered acid plate agglutination (BAPA), and rivanol tests developed for use with domestic animals (Texas Veterinary Medical Diagnostic Laboratory, College Station, Texas).
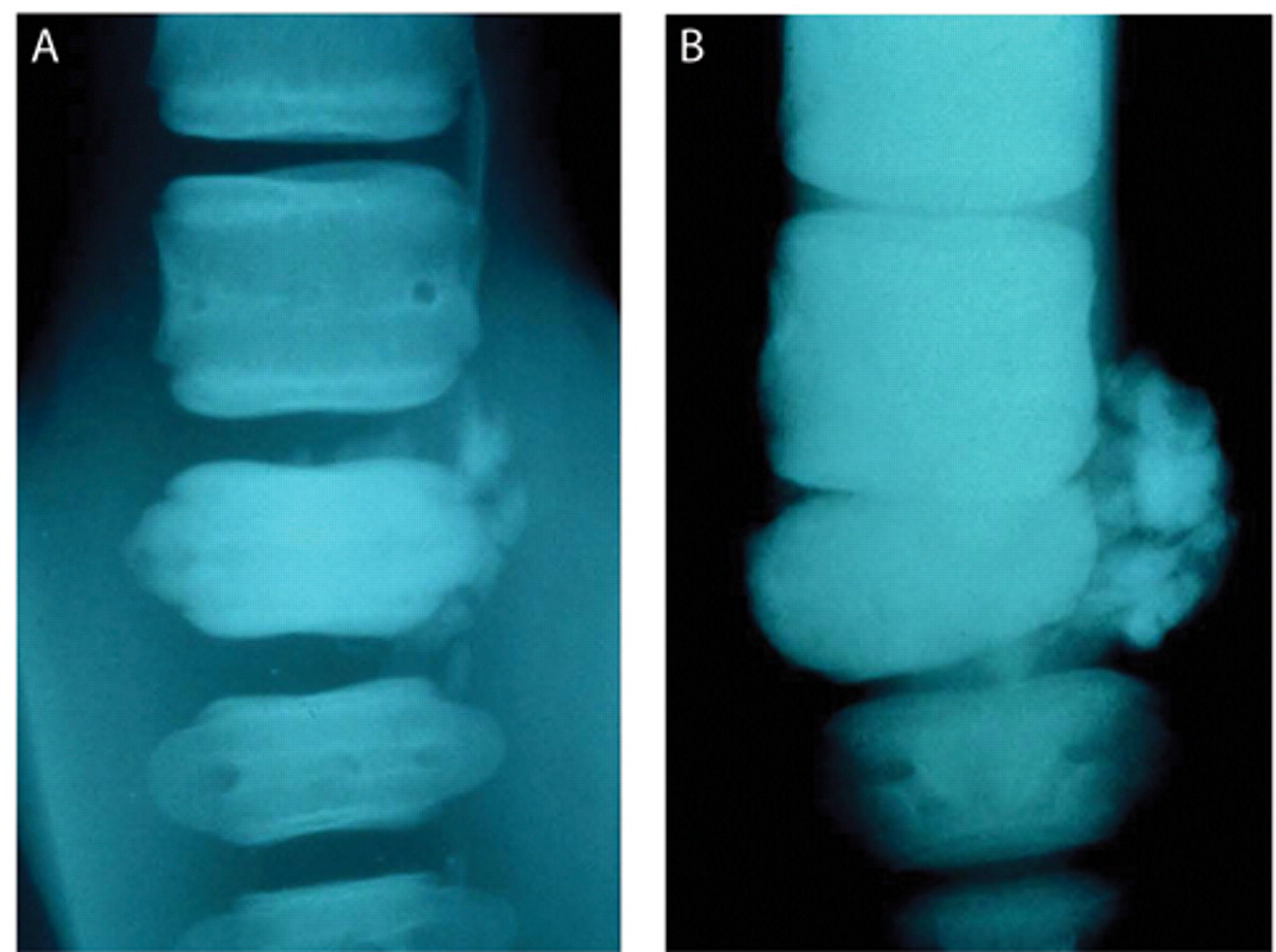
Radiographic imagings of the caudal vertebrae taken at 2 different times; both images are a dorsoventral view with the more anterior vertebrae positioned at the top of the image.
Nine months after transfer to Mystic, the animal became acutely anorexic. Concurrently, the peduncular lesion was noted to be warm to the touch and was thought to have increased in size since the animal’s last examination 1 month previously. A complete blood count showed marked neutrophilia to 8,650 cells/mm 3 from a baseline average for this individual (compiled from multiple samplings over several months during times of normal behavior and apparent good health) of 4,680 cells/mm 3 and an elevated serum fibrinogen level (577 mg/dl from baseline 228 mg/dl). Radiographs of the vertebral lesion revealed further progression of the changes seen in the initial radiographs. These included roughening of vertebral margins, soft-tissue calcification, and intervertebral bone chips, sclerosis of the affected vertebra, and widening of the nutrient foramen. Intervertebral spacing remained normal. A computed tomography (CT) scan of the involved peduncular vertebra revealed a slab fracture involving nearly one-third of the vertebral body with evidence of devascularization and sequestrum formation (Fig. 2a). Additional culture-based attempts to isolate a causative agent, including utilization of Brucella-specific culture methods on blood, aspirates, and deep biopsies, were unsuccessful. Despite an inability to isolate a causative agent, treatment was based on an assumption of Brucella sp. infection of the affected vertebra, which subsequently had sustained a pathological fracture. At various times during the year, systemic antibiotics (e.g., doxycycline a [400 mg orally twice a day for 10 days], ceftiofur b [200 mg intramuscularly every 36 hr for 60 days], clindamycin, c enrofloxacin d as previously employed, and amoxicillin–clavulanate e [2,188 mg orally twice a day for 14 days]) were administered to control the presumed Brucella sp. Intralesional antibiotics included 200 mg of ceftiofur and 1 application of sustained release doxycycline-impregnated biodegradable microspheres (Goertz CEC, Dunn JL, Manning MC, et al.: 2000, Vertebral osteomyelitis in a bottlenose dolphin (Tursiops truncatus): a novel treatment using sustained release antibiotic impregnated, biodegradable microspores. In: Proceedings of the Joint Conference of the American Association of Zoo Veterinarians and International Association of Aquatic Animal Medicine, vol. 31, pp. 380–381). Aspirin f (650 mg orally twice a day) and hydrocodone g (5–10 mg orally twice a day) were administered to control pain.
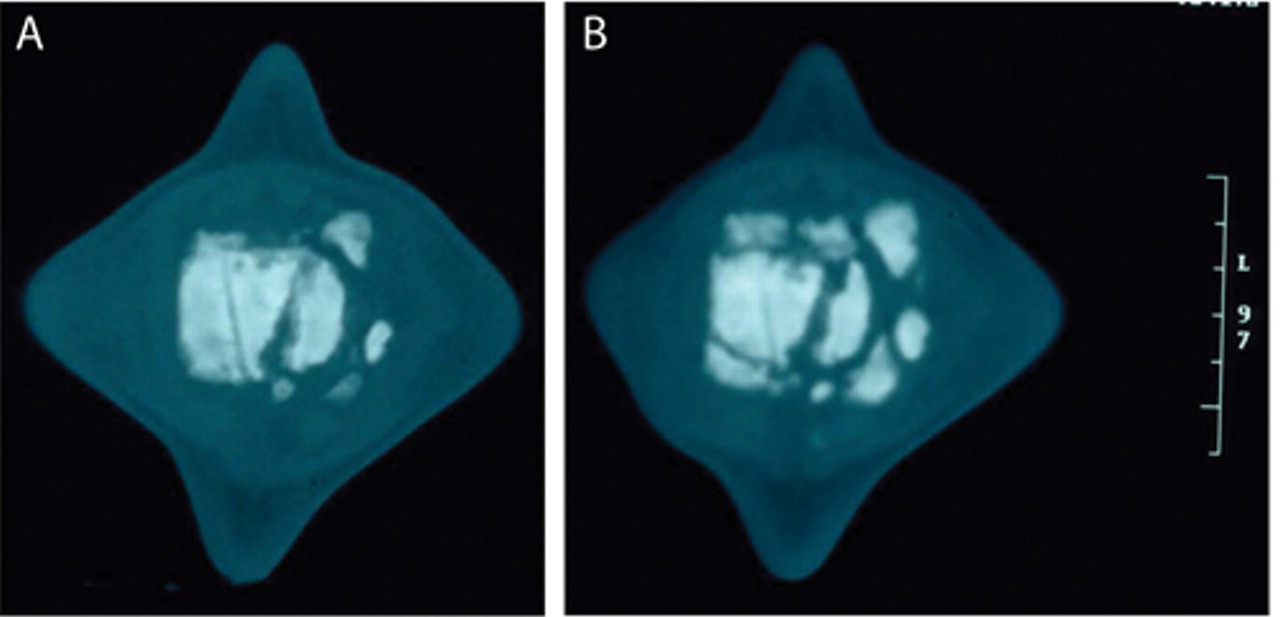
Computed tomography (CT) images of the affected vertebra taken at 2 different times; both images are posterior–anterior views of sagittal sections approximately midway through the vertebral body on right. A centimeter scale is on the right. Note that CT images shown are part of a series; viewing multiple adjacent images is required to visualize the findings.
The animal’s general health continued to deteriorate over the next 6 months with intermittent decreases in food consumption and decreased activity. An acute decrease in the animal’s voluntary use of its flukes and an increase in sensitivity when the flukes were manipulated were noted. A second CT examination 1 year after the first revealed severe progression of osteolysis and osteoproliferation with multiple bony fragments adjacent to the vertebra and in the joint space both anterior and posterior to the affected vertebra (Fig. 2b).
Two months after the CT procedure, the animal experienced acute episodes of vomiting and diarrhea and became lethargic and anorexic with severe panleukopenia and mild dehydration. The animal was force-fed fish and freshwater, and ceftiofur b was administered intramuscularly. The following morning, the animal displayed marked depression and disorientation. Blood collection from the vessels of the flukes and caudal peduncle was obtained with uncharacteristic difficulty suggesting decreased peripheral blood flow. Hematology showed persistence of panleukopenia and severe hemoconcentration. Serum chemistry values reflected electrolyte disturbances (hypochloremia, hyperphosphatemia, hyperkalemia, and hypernatremia), liver dysfunction (elevated alanine aminotransferase and aspartate aminotransferase), azotemia, and a profound hypoglycemia (Table 1). By this time, the animal was moribund, and it died following intravenous administration of prednisolone sodium succinate, doxapram, and epinephrine. Major findings of the necropsy conducted on the day of the animal’s death as well as subsequent analyses included gross, histopathologic, serologic, and microbiologic evidence of Brucella sp.–induced vertebral osteomyelitis as well as isolation of an enterotoxin-producing Staphylococcus sp. associated with necrotizing enteritis.
Hematology and serum chemistry: reference intervals and perimortem values.
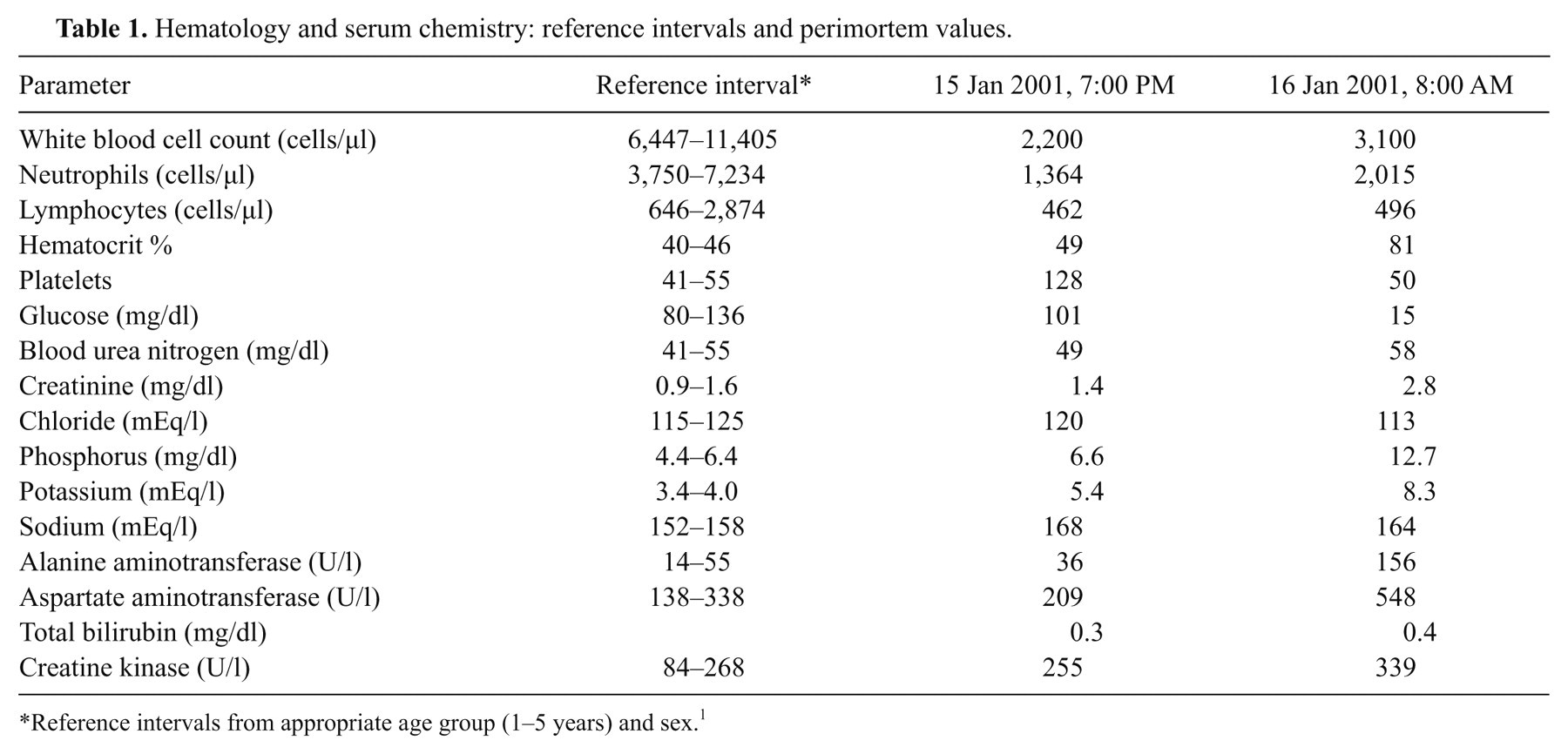
Reference intervals from appropriate age group (1–5 years) and sex. 1
While antemortem tests developed for domestic species indicated the animal had antibodies to Brucella, an active infection was not confirmed until a Brucella sp. was isolated from samples obtained during necropsy, which were taken deeply along the vertebral fascia at the margin of the vertebral body lesion. Samples were inoculated onto trypticase soy agar plates containing 5% bovine serum, 7.5 U/ml of bacitracin, 30 µg/ml of cycloheximide, and 1.8 U/ml of polymyxin B. Cultures were incubated at 37°C in 10% carbon dioxide for a minimum of 3 weeks. The culture isolate was submitted to the National Veterinary Services Laboratory, Brucella Reference Laboratory (Ames, Iowa) to confirm identification. 22 The isolate had different growth characteristics and a different dominant antigen from the Brucella sp. isolated from the then only previously documented clinical case of brucellosis in a captive cetacean, 13,25 but further speciation was not attempted. Postmortem radiographs revealed progression of osteolysis and osteoproliferation involving the affected vertebra and further collapse of the intervertebral joint allowing still more contact between the affected vertebra and the adjacent more anterior vertebra (Fig. 1b). The presence of endotoxin in the animal’s serum was confirmed using a Limulus assay. h,9
Tissue samples were collected from adrenal gland, blubber, brain, diaphragm, esophagus, heart, intestine, kidney, liver, lung, lymph nodes, pancreas, skin, spleen, stomach, testis, tongue, thymus, thyroid gland, vertebrae, and paravertebral tissue at the site of the lesion. Tissue samples were fixed by immersion in 10% neutral buffered formalin, trimmed to fit plastic cassettes, processed routinely for paraffin embedment, sectioned at 4-µm width, and stained with hematoxylin and eosin. Additional tissue sections of liver were stained using the Masson trichrome and Verhoeff–Van Gieson techniques, additional sections of intestine were stained using the Brown and Brenn tissue Gram stain, and additional sections of the vertebral lesion were stained using the Grocott methenamine silver, Brown and Brenn tissue Gram, and Ziehl–Neelsen acid-fast techniques. Significant histopathologic findings were observed in the vertebral lesion, intestine, liver, and lymphoid organs.
Microscopic examination of histologic sections of the affected coccygeal vertebra confirmed a locally extensive osteomyelitis containing sequestra, granulomas, fistulae, and osteophytes, although no etiologic agent was identified in tissue sections stained using hematoxylin and eosin or in tissue sections stained using the Grocott methenamine silver, Brown and Brenn tissue Gram, and Ziehl–Neelsen acid-fast techniques. Histopathologic findings in sections of small intestine revealed necrotizing enteritis with reduced or absent villi, pseudomembranous aggregates of sloughed villous enterocytes along the mucosal surface, and numerous intraluminal Gram-positive, coccoid bacteria, consistent with staphylococci. A coagulase-positive Staphylococcus sp. isolated from the anus, feces, blowhole, intestine, forestomach, and heart was confirmed to be Staphylococcus aureus using the standard coagulase test, colony pigmentation, and trehalose fermentation properties. Staphylococcal genomic DNA was extracted from cultures, purified, and subjected to a multiplex polymerase chain reaction (PCR) technique performed using primers specific for genes encoding 9 staphylococcal superantigens (Fig. 3). 26 Isolates were positive for the gene encoding staphylococcal enterotoxin A. Staphylococcal toxin antibody testing indicated a rising titer. 3,30
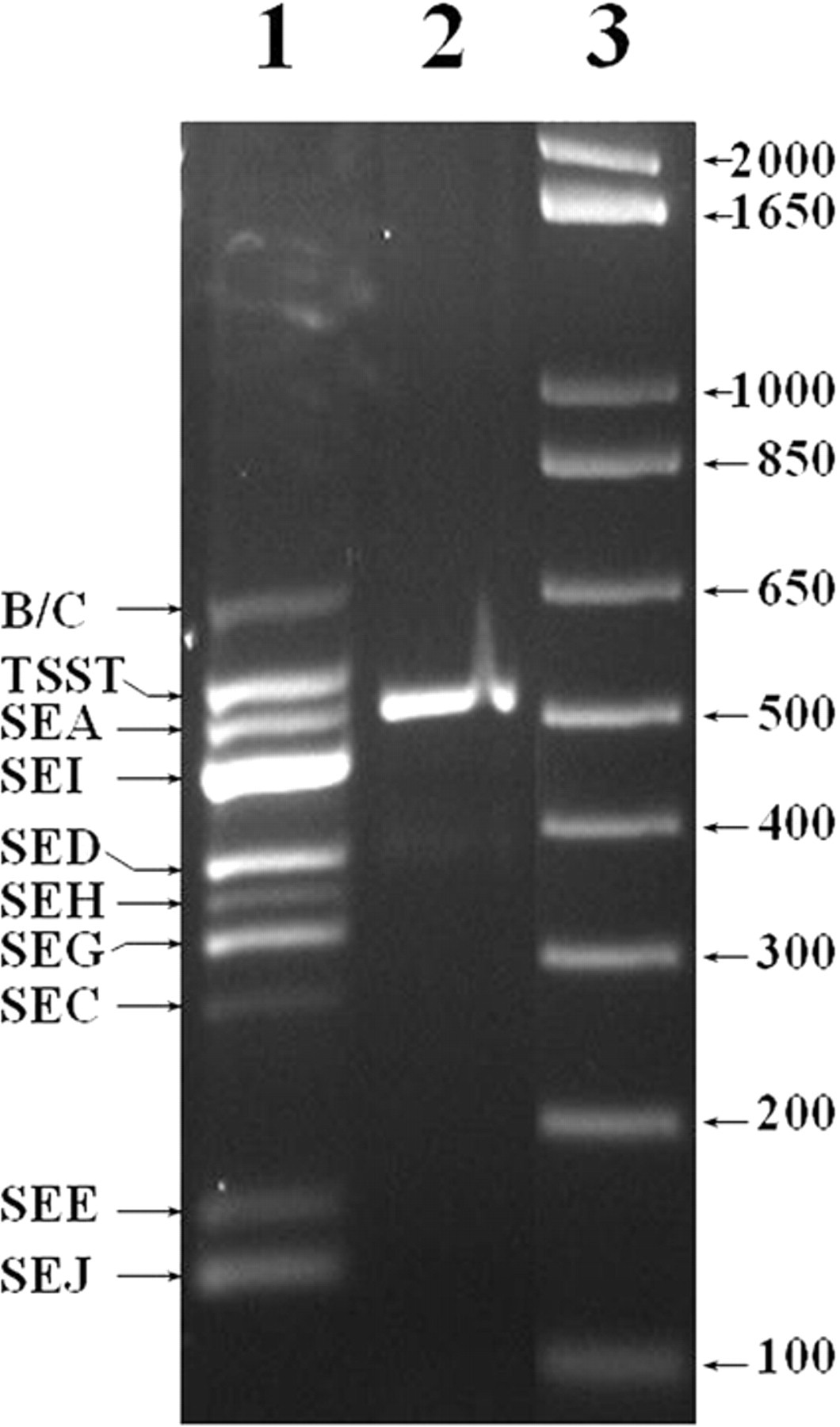
Agarose gel electrophoresis of the 520-bp multiplex polymerase chain reaction (PCR) product from DNA isolated from the Staphylococcus aureus isolate (designated ST-10; lane 2) obtained from the dolphin described in the current study. Lane 1 shows multiplex PCR products for the 9 SE and TSST-1 superantigen controls designated in the left margin (B/C represents SEB and SEC). Lane 3 shows migration of size controls in a commercial product. i
Additional histopathologic findings included diffuse lymphocyte depletion in the spleen and other lymphoreticular organs. Multiple and marked lesions were noted in portal zones of the liver and included arteriolar sclerosis and reduplication, fibrosis, biliary hyperplasia, few portal venular ramifications, and lymphohistiocytic portal hepatitis. These changes could not be attributed as sequelae to the enteritis.
The initial phase of the current case epitomizes the difficulties encountered during treatment of bacterial disease when isolation attempts are not successful. Diagnosis by isolation may be difficult for any Brucella spp., requiring specialized media, unique culture conditions, and sometimes lengthy incubation. The availability of a number of PCR-based diagnostic tests has mitigated many of these problems. Serologic diagnosis in marine mammals has been complicated by the apparent lack of specificity and sensitivity of some techniques developed for detection of exposure in domestic animals. 12,21,27 As such, many institutions perform consensus testing where a number of different serological assays are performed and a seropositive diagnosis is only applied to samples testing positive by multiple means. Retrospectively, serum samples from the subject animal of the present case assayed as having amongst the highest levels of competitive inhibition of samples submitted from thousands of animals when utilizing a new competitive enzyme-linked immunosorbent assay that employed a marine isolate. 24 Notwithstanding these shortcomings, both isolation of Brucella spp. and serologic evidence of infection or exposure have been documented in a variety of wild cetaceans and wild pinnipeds, 1,12,27 and a case of Brucella abortion has been described in a captive bottlenose dolphin. 25 Several Brucella strains have been isolated from marine mammals that are distinct from recognized terrestrial Brucella species 4,16,18 and have a unique insertion sequence in the PCR product of the bp26 gene. 7 Recently, marine Brucella isolates have been divided into 2 strains and named for the preferred host, B. ceti and B. pinnipedialis. 14 While the role of Brucella spp. in osteomyelitis and diskospondylitis has been documented in other species, 32 including a case of presumed marine mammal–associated Brucella spinal osteomyelitis in a human patient, 23 the case reported herein is, to the authors’ knowledge, the first account in a marine mammal. Osteomyelitis is a relatively common diagnosis in cetaceans. Other documented cases of vertebral body osteomyelitis have been associated with Nocardia (Robeck TR, Dalton LM, Young WG: 1995, Nocardia spp. induced chronic suppurative osteomyelitis in a beluga whale. In: Proceedings of the Twenty-sixth Annual International Association of Aquatic Animal Medicine Conference, vol. 26, p. 28), Staphylococcus sp., 2 or have had undetermined etiologies (Stevens R, Hopkins T: 1990, Osteomyelitis in the spine of a bottlenose dolphin (Tursiops truncatus). In: Proceedings of the Twenty-first Annual International Association of Aquatic Animal Medicine Conference, vol. 21, p. 162). The relatively recent discovery of marine origin Brucella and difficulties in isolating Brucella spp. when culture efforts are not specifically aimed at Brucella 12 may have resulted in a failure to determine the etiology in some earlier cases.
Despite a firm suspicion of vertebral brucellosis in the current case, an organism was not isolated antemortem, and consequently, antibiotic sensitivity testing to determine the most appropriate antibiotic was not possible. Antibiotics were used that might have efficacy against Brucella spp. (e.g., doxycycline), but were ultimately not efficacious. The initial administration of doxycycline used an experimental protocol involving the local administration of sustained release antibiotic-impregnated biodegradable microspheres (Goertz CEC, et al.: 2000, Vertebral osteomyelitis in a bottlenose dolphin). While this delivery vehicle has been used successfully for soft-tissue infections in laboratory animals, the successful administration of the microspheres in the present case was hampered by the difficulty encountered in injecting the suspended particles into the rigid, affected tissues. These factors may have contributed to a suboptimal delivery of the drug to the focus of infection. When the animal’s condition continued to deteriorate, this protocol was followed with oral doxycycline, which may not have reached adequate systemic levels due to the large amounts of dietary calcium, in the form of fish bones within the animal’s gastrointestinal tract, that could act to chelate the drug and reduce its absorption. 31 Finally, the sequestra found at the site of osteomyelitis would have had poor exposure to antibiotics, even if appropriate drug levels were achieved systemically, and may have served as persistent niduses of infection.
Most significant in the current case are the effects of chronic illness and of long-term antibiotics on the patient, particularly on commensal and nontargeted bacteria. Chronic infections have been shown to make animals more susceptible to infection with other organisms. Exposure to Brucella antigen has been specifically shown to down-regulate the immune system by reducing lymphocyte blast transformation and Th1-type lymphokine production in mice. 29 Additionally, long-term antibiotic therapy can alter both the resistance patterns and the type of flora present. Increased resistance is observed, and the growth of commensal bacteria is suppressed, allowing the emergence of pathogenic strains and their proliferation. 17,19,28 The continued antibiotic therapy in the present case probably resulted in selecting the toxigenic Staphylococcus that was isolated in pure culture from multiple sites postmortem.
The cause of death in the animal in the current study was likely acute systemic organ failure induced by the toxin-producing enteric staphylococci independently or in combination with concurrent endotoxemia from Brucella sp. osteomyelitis. Staphylococcus aureus exotoxins can immunomodulate hosts, dramatically enhancing host susceptibility to endotoxin and causing shock. 10,15 It could be speculated that the presence of exotoxin and the concurrent release of endotoxin from the Brucella sp. infection resulted in a potentiation of the lethality of both toxins. Interestingly, a 2007 report cites the cases of 2 additional bottlenose dolphins that died with intercurrent Staphylococcal and Brucella spp. infections (Meegan J, Smith CR, Wong SK, et al.: 2007, Brucella sp. infected bottlenose dolphin (Tursiops truncatus) cases in two populations: serologic and clinical evaluations. In: Proceedings of the 38th Annual International Association of Aquatic Animal Medicine Conference, Orlando, Florida). The precise mechanism in the present case could be septic, enterotoxic, or endotoxic shock. The case reported herein manifested both systemic involvement and isolation of an enterotoxin-producing S. aureus, but lacked the demonstration of fever, rash, desquamation, or hypotension included in the clinical definition of toxic shock syndrome. 10 However, it is well documented that some classical symptoms in human beings may not be replicated in other animal species. The missing parameters either were not measured or were difficult to assess and interpret in a marine mammal. In the present case, antemortem indications of systemic involvement included serum chemistry changes consistent with acute deleterious changes in renal and hepatic function (Table 1), vomiting and diarrhea indicating gastrointestinal involvement, and disorientation suggesting central nervous system impairment. Varying degrees of congestion were noted in multiple organs (e.g., lung, liver, spleen, stomach and intestine), together with focal hemorrhage in the superficial gastric lamina propria. The combination of these clinical signs, serum biochemical abnormalities, gross and histopathologic findings in the intestine, and results of serological analyses suggest S. aureus enteritis and toxemia due to enterotoxin A or possibly other superantigen toxins. Isolation of coagulase-positive Staphylococcus spp. from skin and body orifices of clinically normal dolphins is not unusual, and bacteria of this genus have been isolated in cases of respiratory disease (Kinoshita R, Brook F, Vedros N, et al.: 1994, Staphylococcal isolations and clinical cases of Staphylococcus aureus in bottlenose dolphins at Ocean Park, Hong Kong. In: Proceedings of the Thirty-fifth Annual International Association of Aquatic Animal Medicine Conference, vol. 35, p. 159), 5,6,11 renal disease, 20 a cerebral abscess, 8 and skin lesions. 34 The current case highlights the need to avoid complacency with staphylococcal infections and to maintain vigilance to the potential serious complications of isolates capable of toxin production, especially in otherwise compromised animals.
Footnotes
Acknowledgements
This report is a posthumous publication for both David St. Aubin and John Buck. The clinical work up and assessment could not have been accomplished without their earlier contributions. The authors would like to acknowledge the efforts of the Texas Marine Mammal Stranding Network in the rescue and initial rehabilitation of the animal discussed in this case report. The authors thank the administration and staff of the Westerly Hospital, Westerly, RI for providing computerized tomographic scanning services, the Department of Chemical Engineering, University of Colorado and the School of Pharmacy, University of Colorado Health Sciences Center for providing doxycycline-impregnated microspheres for treating the osteomyelitis, and Pfizer Global Research and Development of Groton, CT for serum chemistry analyses. The authors appreciate Gayle Sirpenski’s assistance in record keeping, shipping, editing, photodocumentation, and other support. The suggestions of 2 anonymous reviewers assisted greatly in improving this manuscript. This work is contribution #134 from the Sea Research Foundation.
a.
Vibramycin®, Pfizer Inc., New York, NY.
b.
Naxcel®, Pfizer Inc., New York, NY.
c.
Cleocin HCl®, Pfizer Inc., New York, NY.
d.
Baytril®, Bayer Healthcare LLC, Animal Health Division, Shawnee Mission, KS.
e.
Augmentin®, GlaxoSmithKline, Philadelphia, PA.
f.
Bayer aspirin, Bayer Healthcare LLC, Morristown, NJ.
g.
Hycodan®, Endo Pharmaceuticals Inc., Chadds Ford, PA.
h.
Limulus amebocyte lysate gel-clot assay, Charles River Laboratories International Inc., Wilmington, MA.
i.
1Kb Plus, Life Technologies Corp., Carlsbad, CA.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by USDA grant 99-35201-8581 (GAB), PHS grants AI28401, P20-RR15587, and P20 RR016454 (GAB), and the Idaho Agricultural Experiment Station.
