Abstract
Infectious bovine keratoconjunctivitis (IBK) has been associated with ocular infections by Moraxella bovis, the established etiologic agent of IBK, and more recently, Moraxella bovoculi, a recently described species of Moraxella. To assist in designing rational treatment regimens for M. bovoculi infections associated with IBK, the in vitro susceptibilities of 57 M. bovoculi field isolates cultured from eyes of cattle with IBK in California from 2002 through 2007 were determined. The minimum inhibitory concentration required to inhibit the growth of 90% of organisms (MIC90) of the following 18 antibiotics tested in the present study were: danofloxacin and enrofloxacin: ≤0.12 µg/ml; ampicillin and ceftiofur: ≤0.25 µg/ml; penicillin: 0.25 µg/ml; gentamicin: ≤1 µg/ml; chlortetracycline, oxytetracycline, and tiamulin: 1 µg/ml; florfenicol: 0.5 µg/ml; trimethoprim–sulfamethoxazole: ≤2/38 µg/ml; clindamycin: 2 µg/ml; neomycin and tilmicosin: ≤4 µg/ml; tulathromycin: 4 µg/ml; spectinomycin and tylosin: 16 µg/ml; and sulfadimethoxine: >256 µg/ml. The low MIC90 of these M. bovoculi isolates suggests that commonly used antibiotics for treatment of IBK associated with M. bovis should also be effective against M. bovoculi.
Moraxella bovoculi is a recently identified species of Moraxella first characterized in 2007. 10 The original 18 characterized isolates were cultured from the ulcerated eyes of calves with infectious bovine keratoconjunctivitis (IBK; “pinkeye”) in northern California during summer of 2002. 10 At the present time, it is not known if M. bovoculi plays a primary or secondary role in the pathogenesis of IBK; to date, no published reports have demonstrated Koch’s postulates for M. bovoculi and IBK. Nevertheless, M. bovoculi can be isolated with or without Moraxella bovis from eyes of cattle with IBK. 7 In 2 recent reports, M. bovoculi was isolated from an affected dairy cow and a dairy bull calf during outbreaks of conjunctivitis in adult dairy cows and dairy beef calves, respectively. 4,15
Moraxella bovoculi possesses an RTX (Repeats in the Structural ToXin) operon that encodes an RTX toxin designated MbvA that has 83% deduced amino acid sequence identity to MbxA, the M. bovis cytotoxin. 6 Rabbit antisera against the carboxy terminus of MbvA neutralizes hemolytic activity of M. bovoculi. The presence of this cytotoxin would support a putative role for M. bovoculi in the pathogenesis of IBK especially as previous research showed that corneal lesions similar to IBK could be reproduced with purified M. bovis hemolysin (cytotoxin). 11
To date, the results of vaccine studies with M. bovoculi antigens have not indicated benefit from the use of such antigens in vaccines to prevent IBK. Vaccination of 1 at-risk beef herd with an autogenous M. bovoculi bacterin did not reduce IBK cumulative incidence. 14 In another study, a recombinant M. bovoculi cytotoxin subunit vaccine did not reduce the cumulative proportion of corneal ulcerations associated with IBK. 9 Nevertheless, there are anecdotal reports that vaccination of cattle with M. bovoculi bacterin has helped to reduce IBK in herds from which M. bovoculi was isolated. Such reports suggest that this organism plays an important, however, as yet undefined, role in the pathogenesis of IBK. 4
To further characterize M. bovoculi and to generate data that could assist in developing appropriate treatment recommendations for IBK associated with M. bovoculi, the minimum inhibitory concentrations (MIC) of selected antimicrobial agents for Moraxella bovoculi were determined. The 57 M. bovoculi isolates used in the current study were isolated from naturally occurring cases of bovine keratoconjunctivitis occurring in California cattle during 2002 through 2007; none of the cattle from which the swabs were collected had received antibiotic therapy for IBK at the time of sampling. Eighteen of the isolates had been previously characterized in the original species description of M. bovoculi. 10 Ocular swabs taken from the inferior subconjunctival cul-de-sac of ulcerated eyes were streaked onto trypticase soy agar plates containing 5% sheep or bovine red blood cells and incubated at 35°C for 18 hr. Colonies with morphology characteristic of M. bovoculi were subcultured for purity; such colonies appeared as ≤1 mm diameter colonies that were white to off-white, circular, and convex with an entire margin and a moist-looking surface. 10 Fifty-six of 57 isolates exhibited beta hemolysis when grown on blood agar. Moraxella bovoculi was differentiated from other moraxellae on the basis of characteristic cellular morphology (Gram-negative cocci and diplococci) as well as biochemical and/or molecular testing. 5,10 Isolates identified as M. bovoculi had a positive phenylalanine deaminase test or had an identical 16S–23S interspacer region DNA sequence to that previously published for M. bovoculi. 5,10 Purified colonies were scraped from blood plates and resuspended in a 10% skim milk and 50% glycerol solution and stored at −80°C.
A microdilution system was used for performing sensitivity tests based on methodology from the Clinical Laboratory Standards Institute. 12 Briefly, the frozen isolates were thawed and streaked onto trypticase soy agar plates containing 5% sheep or bovine red blood cells and incubated at 35°C for 18 hr. Two to 3 isolated colonies were used to inoculate 2 ml of brain heart infusion broth a and incubated at 35°C without carbon dioxide for 4–5 hr. The broth culture was then added drop-wise to 0.9% NaCl to a McFarland standard of 0.5 as determined by nephelometer; 10 µl of this suspension was then added to cation-adjusted Mueller–Hinton broth with N-Tris(hydroxymethyl) methyl-2-aminoethane sulfonic acid, b and plates c were inoculated with 50 µl of broth in each well. The plates were incubated at 35°C without carbon dioxide overnight, and a MIC was determined for each antimicrobial drug. The breakpoints for sensitive (S), intermediate (I), and resistant (R) interpretations for the antibiotics tested are shown in Table 1. As no standardized criteria for interpretation of sensitivity exist for Moraxella, breakpoints established for Gram-negative pathogens associated with bovine respiratory disease (BRD) were used when available. 12 Where breakpoints for BRD-associated pathogen were not available, the breakpoints used were from established breakpoints for veterinary isolates. 12 No established breakpoints were available for neomycin or tylosin. The MIC was defined as the lowest concentration of antibiotic that completely inhibited growth; MIC50 and MIC90 were defined as the lowest concentration of antibiotic that was capable of inhibiting growth of 50% and 90% of the M. bovoculi isolates, respectively. Escherichia coli (American Type Culture Collection [ATCC] 25922), Pseudomonas aeruginosa (ATCC 27853), and taphylococcus aureus (ATCC 29213) were used as control organisms in the assays.
Concentrations and breakpoints used for antibiotics tested against 57 isolates of Moraxella bovoculi from cattle with infectious bovine keratoconjunctivitis in California from 2002 through 2007.
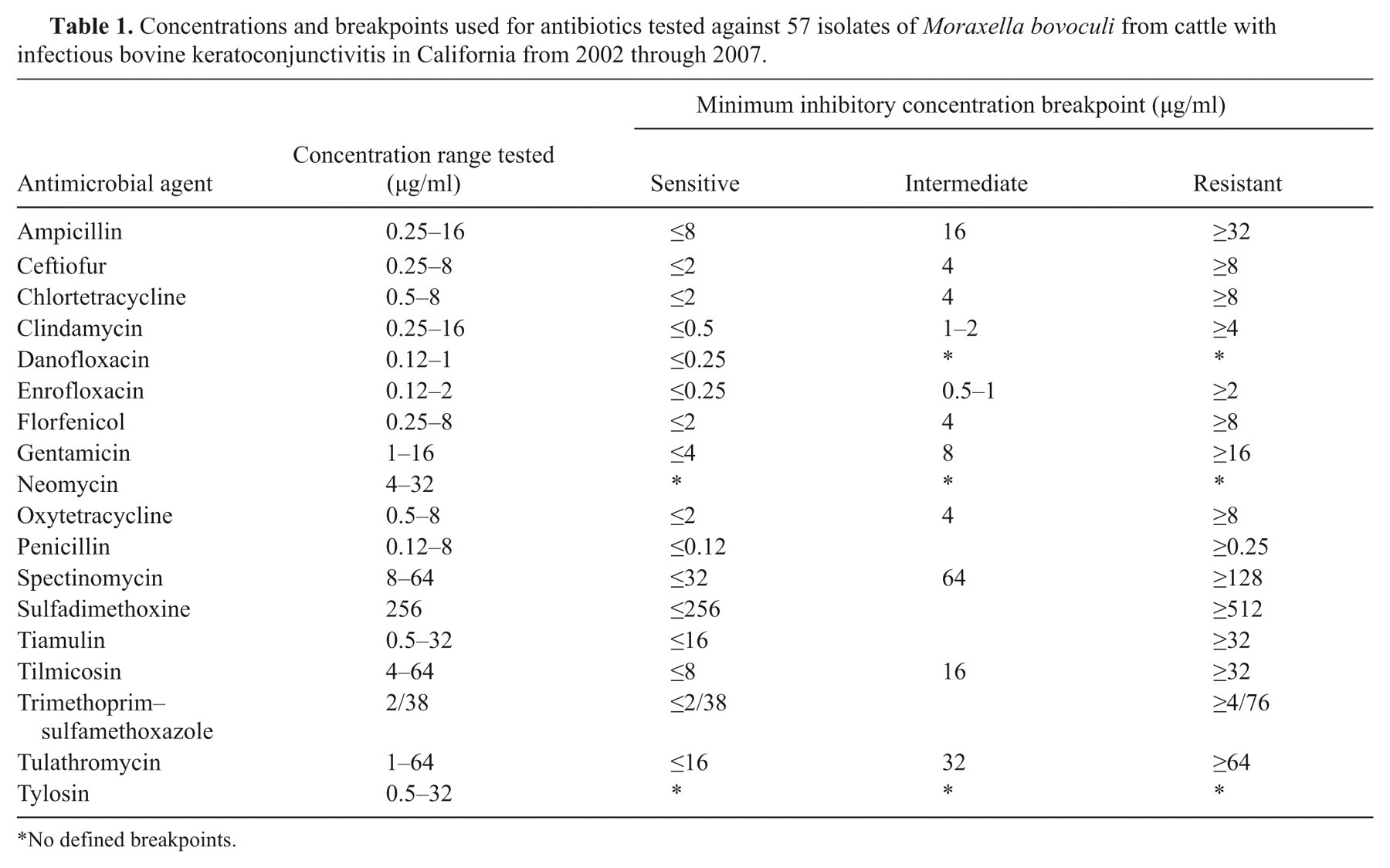
No defined breakpoints.
The MIC90 of 18 antibiotics used in the current study were: danofloxacin and enrofloxacin: ≤0.12 µg/ml; ampicillin and ceftiofur: ≤0.25 µg/ml; penicillin: 0.25 µg/ml; gentamicin: ≤1 µg/ml; chlortetracycline, oxytetracycline, and tiamulin: 1 µg/ml; florfenicol: 0.5 µg/ml; trimethoprim–sulfamethoxazole: ≤2/38 µg/ml; clindamycin: 2 µg/ml; neomycin and tilmicosin: ≤4 µg/ml; tulathromycin: 4 µg/ml; spectinomycin and tylosin: 16 µg/ml; and sulfadimethoxine: >256 µg/ml (Table 2). No resistance was identified among the 57 M. bovoculi isolates to ampicillin, ceftiofur, gentamicin, or tulathromycin. Low resistance (<15%) was observed against chlortetracycline, clindamycin, enrofloxacin, florfenicol, oxytetracycline, penicillin, spectinomycin, tiamulin, tilmicosin, and trimethoprim–sulfamethoxazole. The highest percent resistance was observed for sulfadimethoxine (19.3%; Table 2).
Number of Moraxella bovoculi isolates (n = 57) with the minimum inhibitory concentration (MIC) of antibiotic tested and lowest concentration of antibiotic that was capable of inhibiting growth of 50% and 90% of isolates (MIC50 and MIC90, respectively).
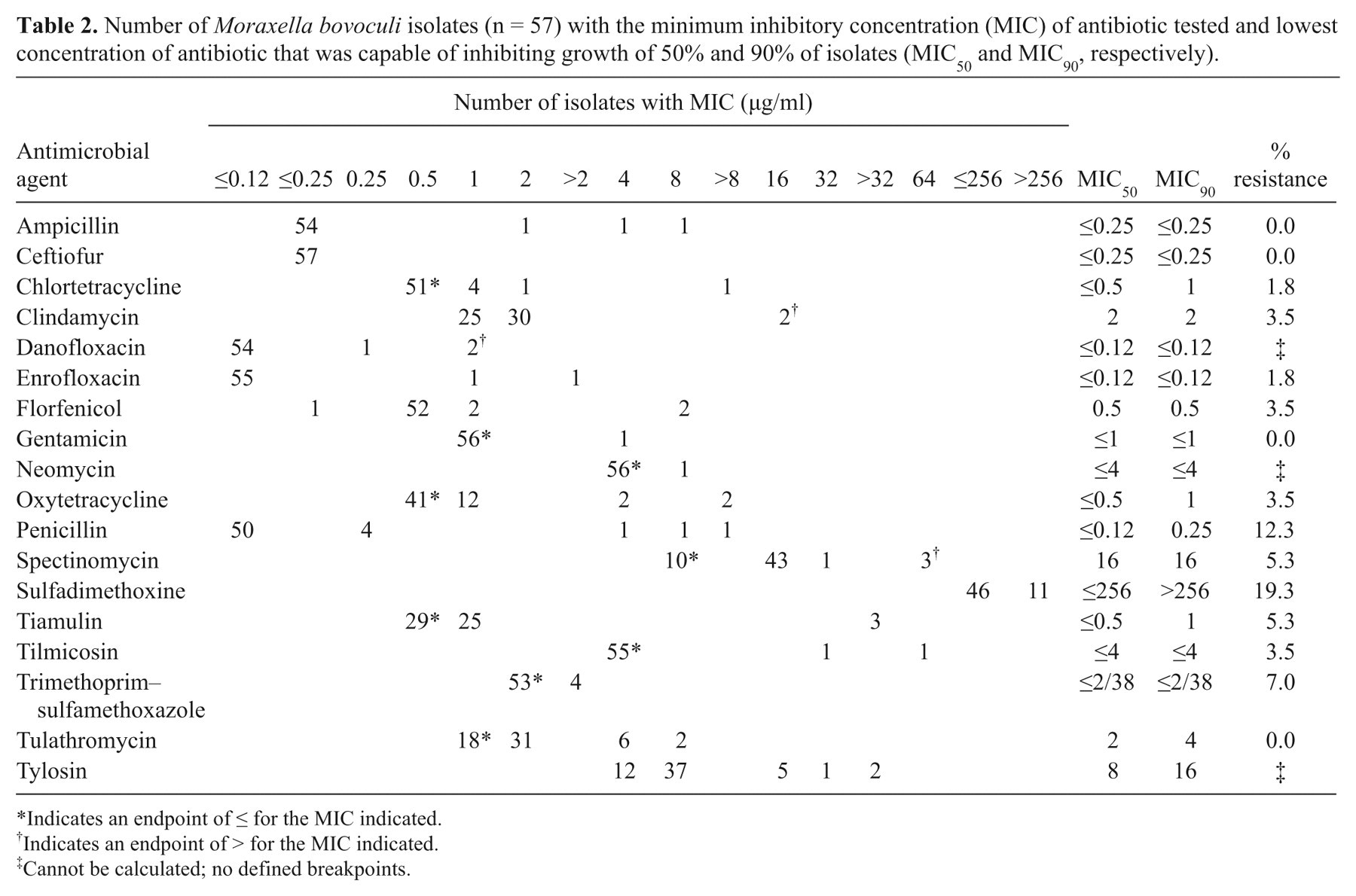
Indicates an endpoint of ≤ for the MIC indicated.
Indicates an endpoint of > for the MIC indicated.
Cannot be calculated; no defined breakpoints.
In the United States, only oxytetracycline and tulathromycin currently have drug label claims for the treatment of IBK associated with M. bovis. The MIC50 of oxytetracycline in the present study (≤0.5 µg/ml) was identical to the MIC50 previously reported for M. bovis (0.5 µg/ml). 17 The MIC90 of oxytetracycline in the current study was, however, considerably lower than the MIC90 previously reported for M. bovis (32 µg/ml17). Based on the low MIC90 of oxytetracycline and tulathromycin, it is expected that both of these antibiotics would also be effective against M. bovoculi.
The MIC50 and MIC90 values for florfenicol in the present study (0.5 µg/ml) were identical to values previously reported for florfenicol against M. bovis. 8 The MIC50 and MIC90 values for ceftiofur reported in the current study (≤0.25 µg/ml) were similar to previously reported MIC50 (0.12–0.13 µg/ml13,17) and MIC90 (0.12 µg/ml13 and 0.25 µg/ml17) values for M. bovis isolated from cattle with naturally occurring IBK. As both florfenicol and ceftiofur have been reported to be efficacious against M. bovis associated with IBK, 8,13 it would be expected, based on the MIC values, that these drugs would also be efficacious against M. bovoculi associated with IBK.
Although relatively few antibiotics are currently approved to treat IBK associated with M. bovis in cattle in the United States, a variety of antibiotics from among the antibiotics tested in the current study have been reported to be effective against M. bovis. Along with oxytetracycline, tulathromycin, florfenicol, and ceftiofur, clindamycin, 16 tilmicosin, 19 ormetoprim–sulfadimethoxine (related to trimethoprim–sulfamethoxazole), 18 and penicillin 1-3 have been shown to be effective in treating IBK associated with M. bovis. Of these antibiotics, M. bovoculi had the highest percent resistance to penicillin (12.3%); whether or not the higher percent resistance is correlated with any reduced penicillin treatment response in the field against M. bovoculi–associated IBK is currently not known. At the present time, no controlled studies evaluating specific antibiotic treatment responses in cattle with IBK associated with M. bovoculi have been reported. Because M. bovis and M. bovoculi are routinely isolated together from the same IBK-affected eye, 7,9 separating actual clinical responses attributable to M. bovoculi versus M. bovis may prove difficult.
The relatively low MIC values reported for M. bovoculi in the present study support the conclusion that commonly used antibiotics against M. bovis should also be effective against M. bovoculi. Further investigations are needed to make comparative recommendations as to which antibiotics will be most effective in treating IBK associated with M. bovoculi.
Footnotes
Acknowledgements
The authors thank Spencer Jang, Anita Wong, and Eileen Samitz for technical assistance and use of the Trek Diagnostic System in the Clinical Microbiology Laboratory at the William R. Pritchard Veterinary Medical Teaching Hospital, University of California, Davis, CA.
a.
Difco Laboratories, Detroit, MI.
b.
Trek Diagnostic Systems Inc., Westlake, OH.
c.
Sensititre®, Trek Diagnostic Systems Inc., Westlake, OH.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
Funding for completion of this work was provided by the University of California School of Veterinary Medicine Dean’s Office Start Up Funds for New Faculty Research.
