Abstract
A 7-year-old, intact male standard Poodle dog with hypothyroidism and atypical hyperadrenocorticism developed acute signs of lethargy, weakness, inappetence, vomiting, and diarrhea. Clinical signs progressed to hind limb proprioceptive deficits, aggressive behavior with obtundation, and an equivocal seizure. Necropsy revealed a mass in the sellar region that histologically consisted of multinucleated giant cells, macrophages, and hemosiderin-laden macrophages with fewer lymphocytes and plasma cells admixed with large regions of cholesterol cleft deposition, fibrin, and prominent Rosenthal fibers. Pituitary tissue was not identified on gross or histologic examination, but the mass was partially bordered by epithelial cells. The histologic characteristics are similar to changes described in the human medical literature as xanthogranuloma of the sellar region and xanthogranulomatous hypophysitis.
Keywords
In domestic animals, diseases of the pituitary and sellar region include pituitary cyst, primary pituitary neoplasia, metastatic neoplasia, 4 craniopharyngioma, suprasellar germ cell tumor, 14 and pituitary abscess. 13 Many of these diseases are also reported in human beings; however, in human beings, a wider array of reported pituitary and sellar disease entities includes inflammation of the pituitary gland (hypophysitis) 7 and xanthogranuloma of the sellar region. 10,12,17
Masses in the sellar region often cause varied clinical signs. Dogs with sellar and suprasellar masses can exhibit behavioral changes, 18 neurologic deficits, and endocrine dysfunction. Behavioral changes might include depression, 22,24 aggression, 8 lethargy, 6 anorexia, 24 polyphagia, 24 and polydipsia. 6 Neurologic deficits might include proprioceptive deficits, 6 circling, 6,24 and cranial nerve deficits, particularly related to compression of the optic chiasm. 16,18,22,24 Last, endocrine disease might manifest as hypothyroidism or diabetes insipidus. 16
A 7-year-old, intact male standard Poodle dog presented to the Oklahoma State University Boren Veterinary Medical Teaching Hospital (Stillwater, Oklahoma) with a 1-year history of polyuria and polydipsia and a 1-week history of inappetence. The week before, the dog had been diagnosed with hypothyroidism on the basis of decreased total thyroxine (T4), and treatment with levothyroxine (0.01 mg/kg every 12 hr) had been instituted.
Other than mild lumbosacral pain, physical examination findings were within normal limits. Because inappetence had coincided with initiation of thyroid supplementation, and euthyroid-sick syndrome was a differential diagnosis, treatment was discontinued to allow for future reassessment of thyroid function. Abdominal ultrasound and radiographs revealed no obvious abnormalities. Adrenocorticotropic (ACTH) stimulation testing demonstrated increased cortisol levels (23.2, reference [ref.] interval: 8.0–17.0 μg/dl) post-ACTH stimulation from a baseline cortisol level of 2 μg/dl, suggestive of possible hyperadrenocorticism. Serum chemistry revealed a mild hypercholesterolemia (330, ref. interval: 92–324 mg/dl). Complete blood count did not reveal significant abnormalities. Urinalysis demonstrated hyposthenuria (specific gravity: 1.004). Lumbosacral pain was treated with deracoxib (1.7 mg/kg orally every 24 hr).
At the 1-month recheck appointment, the dog exhibited hair loss and lethargy consistent with previously diagnosed hypothyroidism. Repeat thyroid testing revealed a low free T4 (4.8, ref. interval: 8–40 pmol/l) with normal thyroid-stimulating hormone and T4. Hypercholesterolemia had increased (435 mg/dl). Thyroid supplementation was reinstituted (levothyroxine, 0.02 mg/kg every 12 hr).
Concurrently, to address the abnormal ACTH stimulation results seen on initial presentation, low-dose dexamethasone suppression testing was performed. Results were within normal limits, making typical hyperadrenocorticism less likely. Because of the persistent polyuria and polydipsia, additional tests were performed to assess other adrenocortical hormone levels. After exogenous ACTH stimulation, there were elevations in cortisol (191.8, ref. interval: 70.6–161.2 ng/ml), progesterone (6.57, ref. interval: 0.22–1.45 ng/ml), 17-OH progesterone (5.31, ref. interval: 0.25–2.63 ng/ml), and aldosterone (458.7, ref. interval: 72.9–398.5 pg/ml). (Cortisol reference intervals vary because tests were performed at 2 separate laboratories.) On the basis of these data, atypical hyperadrenocorticism was diagnosed and subsequently treated with melatonin (6 mg/day), which did not ameliorate the polyuria, polydipsia, and hyposthenuria. The owner declined additional treatments.
Six months after initial presentation, the dog presented with acute signs of lethargy, weakness, inappetence, vomiting, diarrhea, confusion, and wandering. Physical examination revealed dehydration (approximately 7%). Urinalysis revealed continued hyposthenuria (specific gravity: 1.007). Complete blood count and serum chemistry revealed no significant abnormalities. The ACTH stimulation tests performed at this time revealed an elevated cortisol level (18.5, ref. interval: 8.0–17.0 μg/dl) from a baseline level of 1.9 μg/dl. Additional testing to elucidate a potential cause of the polyuria and polydipsia, such as diabetes insipidus, was not performed because of owner financial constraints. After admission to the hospital, the dog acutely developed hind limb proprioceptive deficits and abnormal aggressive behavior. Later, the dog became obtunded and had an equivocal seizure. Because of the deterioration in clinical signs, the dog was euthanized and submitted for postmortem examination.

Brain, dog. Gross image of the fresh brain showing a green–brown, friable mass in the sellar region. Bar = 1 cm.
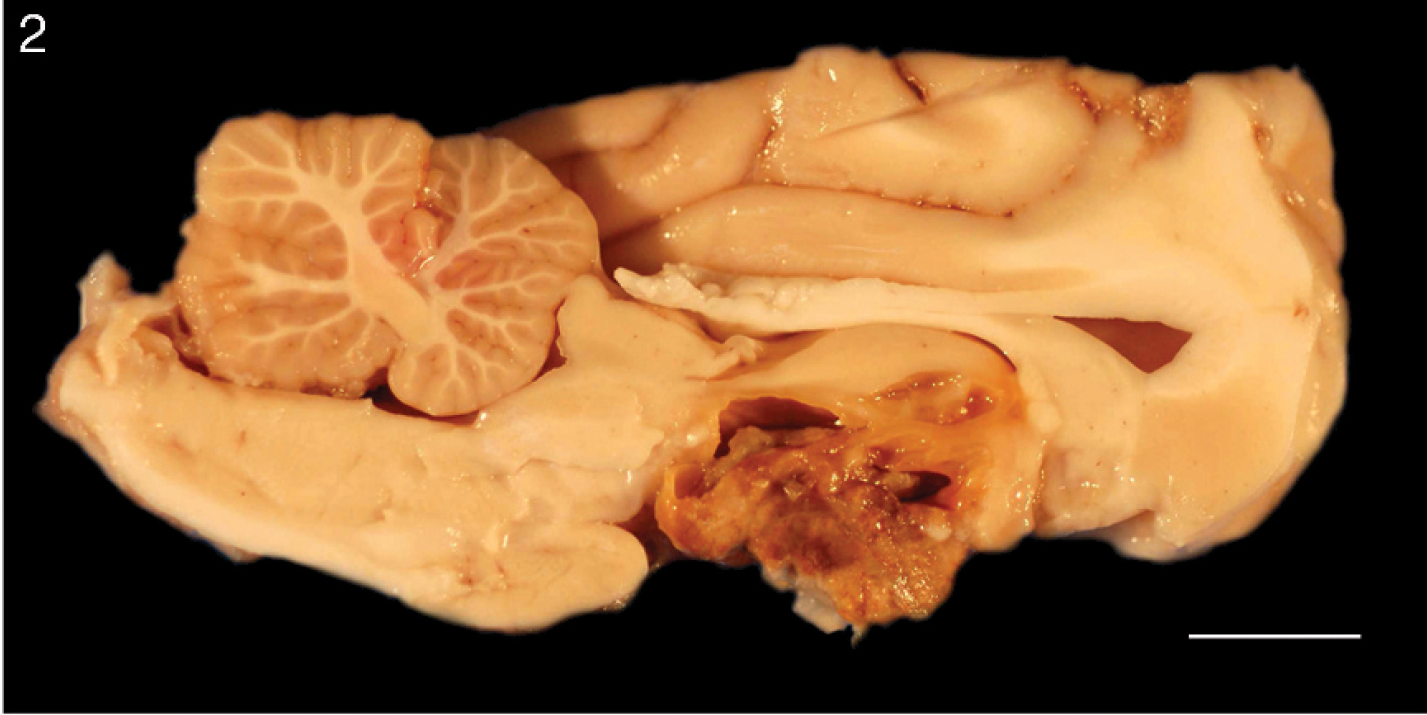
Brain, dog. Gross image showing the cut surface of the mass after formalin fixation. The sellar mass compresses the overlying hypothalamus and contains many small, cystic cavities filled with turbid, brown fluid. The pituitary is not identified grossly. Bar = 1 cm.
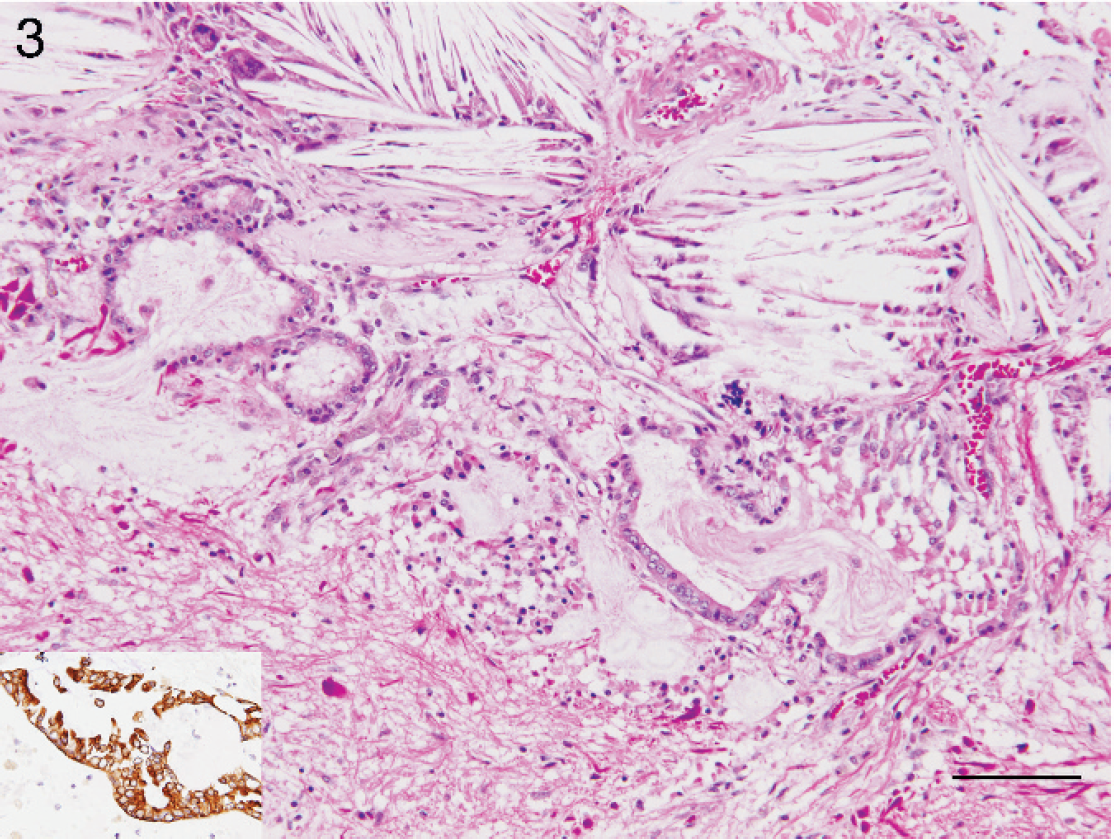
Brain, dog. The sellar mass is composed of cholesterol clefts admixed with multinucleated giant cells, macrophages, hemosiderin-laden macrophages, and variable amounts of hemorrhage, consistent with a xanthogranuloma. Within the mass are several tubules lined by cuboidal epithelium that demonstrates positive immunoreactivity on staining with cytokeratin (see inset). Hematoxylin and eosin. Bar = 100 μm.

Brain, dog. Numerous Rosenthal fibers are scattered throughout the sellar mass. These are dilated astrocytic processes characterized by positive immunoreactivity on staining with glial fibrillary acidic protein (see inset). Hematoxylin and eosin. Bar = 50 μm.
On postmortem examination, the sellar region was filled with a large (1.5-cm-diameter) cavitated mass that was friable and brown to yellow-green (Fig. 1). On cut section, small cystic cavities contained turbid, brown fluid. The mass minimally compressed the overlying brain, and the pituitary gland was not grossly identified (Fig. 2). No other gross lesions were noted.
Tissues were fixed in neutral buffered, 10% formalin solution, routinely processed, embedded in paraffin, sectioned at 4 μm, and stained with hematoxylin and eosin. Histochemical stains were applied to affected sections of brain, including Gomori methenamine silver (GMS), periodic acid–Schiff (PAS), Gram, and Ziehl–Neelsen acid-fast stains. Immunohistochemical (IHC) analysis of the affected tissue was performed, and slides were incubated with glial fibrillary acidic protein a (GFAP; rabbit anti-GFAP; 1:3,000, 30 min) and cytokeratin a (mouse anti-human; clone MNF 116; 1:600, 30 min, 25°C) primary antibodies with the use of a streptavidin–horseradish peroxidase method. Slides were developed with 3,3′-diaminobenzidine chromogen a and counterstained with Mayer hematoxylin. Sections were also incubated with isotype-matched rabbit immunoglobulin (Ig)G (GFAP) and mouse IgG (cytokeratin) antibodies as negative controls. Positive controls consisted of canine brain (GFAP) and canine haired skin (cytokeratin).
Histologic examination of the mass revealed marked granulomatous inflammation consisting of multinucleated giant cells, macrophages, and hemosiderin-laden macrophages with fewer lymphocytes and plasma cells admixed with large regions of cholesterol cleft deposition and fibrin (Fig. 3). Numerous, prominent Rosenthal fibers were scattered throughout the mass and were characterized by markedly swollen, hypereosinophilic astrocytic processes that had positive immunoreactivity on immunostaining with GFAP (Fig. 4). Along the periphery and embedded within the mass were a few small, tubular structures lined by a low cuboidal epithelium that showed positive immunoreactivity on staining for cytokeratin (Fig. 3). Neuroparenchyma overlying the mass was markedly rarefied. Pituitary tissue was not identified histologically. Histochemical stains (GMS, Gram, PAS, acid-fast) for potential infectious agents did not reveal organisms, and granulomatous inflammation was not evident in other tissues.
Histologic examination of the thyroid gland demonstrated degeneration with moderate, multifocal loss of follicles and secondary infiltration by a mixture of macrophages, lymphocytes, and plasma cells. Additional histologic changes included a mild lymphocytic portal hepatitis, moderate pulmonary edema, mild lymph node edema, and mild erosive gastritis. Histologic examination of all other organs revealed no additional abnormalities. The adrenal glands were grossly within normal limits but were not available for histologic examination.
Relating postmortem findings to clinical presentation, expansion of the mass within the cranial vault likely caused the neurologic signs (proprioceptive deficits, aggression, obtundation, and equivocal seizures). Destruction of hypothalamic and pituitary tissue might have led to endocrine disease (atypical hyperadrenocorticism, hypothyroidism). However, deficient pituitary function would be expected to result in hypoadrenocorticism rather than atypical hyperadrenocorticism.
To the authors' knowledge, sellar lesions, suprasellar lesions, or both, like the one in the present study, have not been described in animals. In human beings, several entities can produce a change similar to the one seen here, including xanthogranuloma of the sellar region, Rathke cleft cysts, xanthogranulomatous hypophysitis, and craniopharyngioma. Though these lesions are considered clinically separate entities, they can share similar and sometimes overlapping pathogeneses. 1,9,15,17,19,20,26
Xanthogranuloma of the sellar region 10,12,17 or suprasellar region 21 is a rare disease entity recently described in the human medical literature. Grossly, the lesion is a cystic, red to brown mass 10 that contains yellow–brown fluid. 12 Histologically, sellar xanthogranulomas are composed of macrophages, multinucleated giant cells, cholesterol clefts, hemosiderin, and variable epithelial cell clusters 10,17,21 that can be squamous or ciliated. 10,17 Reports in the human literature do not describe Rosenthal fiber formation in conjunction with this entity. Most probably, the Rosenthal fibers arose from astrocytes in the adjacent brain parenchyma as a reactive process and are not a specific feature of sellar xanthogranulomas. The cause of sellar xanthogranuloma is unknown, and it has been postulated to be secondary to rupture of a cyst. 17
Cysts in the sellar region of human beings include Rathke cleft cysts, arachnoid cysts, dermoid and epidermoid cysts, 7 or oral radicular cysts. Cysts with a sellar location reported in dogs include Rathke cleft cysts, 8 cysts derived from the craniopharyngeal duct, 4 and cysts that result from the failed formation of the adenohypophysis, resulting in pituitary dwarfism. 4,11 Intracranial arachnoid cysts have also been reported in dogs, but these cysts were located in the quadrigeminal cistern and the cerebromedullary angle rather than the suprasellar space. 25 Cyst rupture might have incited inflammation in the surrounding tissue, leading to the lesion seen in the current case. 4
Alternatively, this lesion could have resulted from inflammation of the pituitary gland. In human beings, hypophysitis is subdivided into primary and secondary disease processes. Primary hypophysitis is classified as lymphocytic, granulomatous, and xanthomatous, 5,7 with some authors further specifying necrotizing and xanthogranulomatous forms. 23 Xanthogranulomatous and xanthomatous hypophysitis have been postulated as forming a spectrum of inflammatory disease that includes the suprasellar xanthogranuloma 2 ; however, the etiology of primary xanthogranulomatous hypophysitis is unknown. 23 Primary lymphocytic hypophysitis might be an autoimmune phenomenon. 2 Granulomatous hypophysitis in human beings can be primary idiopathic or secondary to systemic granulomatous diseases, including fungal infection, mycobacterial infection, brucellosis, syphilis, sarcoidosis, Wegener granulomatosis, Crohn disease, or Whipple disease. 7 Both granulomatous and lymphocytic hypophysitis have occurred secondary to ruptured Rathke cleft cyst. 1,9,15,19,20 To the authors' knowledge, primary hypophysitis has not been reported in the veterinary literature.
Finally, a craniopharyngioma could have led to the lesion in the present study. Craniopharyngioma is a neoplasm arising from remnants of the Rathke pouch and is seen most commonly in young dogs. 3 In human beings, the adamantinomatous form of craniopharyngioma often contains areas of xanthogranulomatous inflammation, as seen in the current case. 17 Neoplastic cells were not identified in examined sections, making craniopharyngioma a less likely diagnosis, although some craniopharyngiomas could be hypocellular. In these cases, it is difficult to distinguish between craniopharyngioma and Rathke cleft cysts. 7,26
Thus, the differential diagnoses in the present case include xanthogranuloma of the sellar region, ruptured pituitary cyst (e.g., Rathke cleft cyst), xanthogranulomatous hypophysitis, and hypocellular craniopharyngioma. The histologic changes of granulomatous inflammation with multiple cholesterol clefts strongly suggest that this is a sellar xanthogranuloma. Sellar xanthogranuloma has not previously been described in domestic animals, and it should be included in a list of differential diagnoses for canine sellar and suprasellar masses.
Acknowledgements. Many thanks to Dr. Brian Summers and the neuropathologists at the University College London for help in interpreting this case. The excellent technical support of the Oklahoma Animal Disease Diagnostic Laboratory histology department, including Mr. Curtis Andrews and Ms. Darlene Girocello, is greatly appreciated.
Footnotes
a.
Dako Denmark A/S, Glostrup, Denmark.
