Abstract
The objective of the current study was to identify factors associated with avian mycobacteriosis in zoo birds. Inventory data, population health records, and necropsy data from eligible birds in the Zoological Society of San Diego's (ZSSD) collection from 1991–2005 (n = 13,976) were used to describe disease incidence, prevalence, and postmortem findings. A matched case-control study was then conducted to identify factors describing demographic, temporal, and enclosure characteristics, along with move and exposure histories. Cases (disease-positive birds; n = 167) were matched in a 1:7 ratio with controls (disease-negative birds; n = 1169) of similar age and taxonomic grouping. Potential risk factors were evaluated using univariate and multivariable conditional logistic regression. Disease prevalence and incidence were estimated for the study period at 1.2% and 3 cases/(1,000 bird-years at risk), respectively. Lesion characteristics and order prevalence are described. In the multivariable model, case birds were more likely to have been previously housed with a bird with mycobacterial disease involving the intestinal tract (odds ratio [OR] = 5.6, P < 0.01) or involving only nonintestinal sites (OR = 2.0, P < 0.01). Cases were more likely to have been imported into the collection than hatched at the ZSSD (OR = 4.2, P < 0.01). Cases were moved among ZSSD enclosures more than controls (OR = 1.1 for each additional move, P < 0.01). Findings will help guide future management of this disease for zoo bird populations.
Introduction
Avian mycobacteriosis, often referred to as “avian tuberculosis,” is an important disease of wild, pet, and zoo birds. 18,24 Characterized by an insidious onset and slow progression, this chronic wasting disease is caused predominantly by 3 species of mycobacteria: Mycobacterium avium, Mycobacterium intracellulare, and Mycobacterium genavense. 18,24 Many birds never show signs of disease, and the lack of sensitive diagnostic tests makes antemortem diagnosis difficult. 17,24 Often disease is diagnosed during postmortem evaluations, when gross examinations and histopathology reveal characteristic lesions, 24 and tissue cultures can confirm the mycobacterial agent. When antemortem diagnosis does occur, treatment is often not feasible, and antimicrobial therapies are not always effective. Therefore, the humane euthanasia of infected individuals is commonly recommended. 18,24
In addition to the loss of valuable birds directly from disease, avian mycobacteriosis presents difficult management challenges for zoo populations and endangered species recovery programs. Traditionally, most infections are thought to have been acquired from exposure to a disease-positive bird or its contaminated enclosure. 9,24 For this reason all enclosure mates and contacts of affected birds are considered disease suspects until proven otherwise. Disease control has focused on policies to break the bird-to-bird disease transmission cycle through depopulation, quarantine, environmental decontamination, and elaborate surveillance of in-contact birds. 1,5,18,23,24,26 Borrowed from the poultry industry, these control methods were employed in the early 1900s and are still commonly used in zoo bird husbandry today. 7,9,24 These methods have the lasting negative effect of greatly impeding captive population sustainability efforts and endangered species recovery programs.
Currently, there is a need within the zoological community to re-examine the assumption that exposure is the primary risk factor for acquiring infection. This simple relationship does not always explain observed disease patterns. Case series of pet birds 12 and of zoo species 19 have found that birds without apparent exposure sometimes develop disease, while birds kept in close contact with another mycobacteria-positive enclosure mate may never show signs of disease. Such patterns suggest that additional factors and cofactors may contribute to the risk of acquiring infection or developing overt disease. The identification of these risk factors and their interrelationships are needed in order to improve the health and management of zoo birds. To understand avian mycobacteriosis in zoo birds, an epidemiologic study was performed to estimate disease incidence and prevalence, characterize postmortem findings, and identify potential risk factors associated with avian mycobacterial disease in a well-defined, dynamic cohort of zoo birds residing at the Zoological Society of San Diego (ZSSD) between 1991 and 2005.
Materials and methods
Source population
The ZSSD maintains two facilities: the San Diego Zoo (SDZ) on 120 acres of land near downtown San Diego, CA. and the San Diego Zoo's Wild Animal Park (SDZWAP) on 1,500 acres in a rural setting near Escondido, CA. Both facilities have an extensive animal caretaker staff, including clinical veterinarians, and closely monitor the health status of all animals daily.
The source population was defined as all collection birds living at the SDZ or SDZWAP between 1991 and 2005 that were at least 5 months old and that had been part of the collection (and released from quarantine) for at least 30 days. Computerized records from the ZSSD's database of accessioned animals were used to identify the eligible cohort of birds. The dynamic nature of this population, with various birds being moved in and out for reasons related to morbidity, mortality, breeding, and husbandry, precluded the ability to follow all birds for the entire 15-year study. Therefore, population dynamics were reflected through time by considering incidence per year of time that a bird spent in the collection (or bird-years at risk). Birds were included in the target cohort at the time(s) they met all eligibility criteria. Birds remained in the cohort until they no longer met eligibility criteria, they were removed from the population, or the study ended.
Postmortem evaluations and histopathology
All collection birds that died during the study period underwent a postmortem examination including complete histologic evaluation by staff pathologists unless advanced autolysis precluded histopathology. In addition to routine hematoxylin and eosin staining of all tissues, Ziehl-Neelsen acid-fast stain was performed on select tissues of clinical suspects, birds with postmortem lesions suggestive of mycobacteriosis, and enclosure mates of mycobacteriosis-positive birds when exposure history was available. At the minimum, sections of small intestine, liver, and lung were stained with Ziehl-Neelsen acid-fast stain for all suspect and disease-positive birds except in rare cases where 1 or more of these tissues were not available for evaluation. Birds were diagnosed as having avian mycobacteriosis if acid-fast organisms were observed in any tissues during histologic examinations. Select tissues observed to have acid-fast organisms were sent to the National Veterinary Services Laboratory (Ames, IA) or the Microbiology Laboratory (University of California, San Diego Medical Center, San Diego, CA) for mycobacterial culture only for cases in which confirmation or specific identification of mycobacterial species was important for diagnosis or management.
Data on postmortem findings were stored in a searchable, proprietary pathology database that includes text reports and systematized nomenclature of medicine (SNOMED) codes for final diagnoses. Birds diagnosed with mycobacteriosis were identified from the ZSSD pathology database with keyword searches, SNOMED code searches, and previously compiled rosters of disease-positive birds.
The distribution and severity of lesions for all birds diagnosed with mycobacteriosis were characterized by a board-certified, veterinary anatomic pathologist (RP) based on examination of histologic sections and specially stained sections. Tissues were considered affected if acid-fast bacteria were detected. Severity of disease in each tissue was graded as incidental or significant, depending on the observed degree of organ involvement, inflammation, and tissue reaction. Incidental lesions were those determined to have been clinically insignificant (i.e., organ function was unlikely to have been adversely affected). For birds with lesions classified as significant, some disruption of organ function was determined to be likely and mycobacterial disease was considered to have contributed to or caused their death. Additionally, the abundance of mycobacterial organisms observed in each tissue was qualitatively evaluated. Tissues were classified as having “few” Mycobacteria if acid-fast organisms were only present in some affected areas of the tissue and were present in low numbers within cells. Tissues with more organisms were classified as “moderate to abundant,” and were characterized by acid-fast organisms being relatively easy to detect and observed in at least half of the lesions.
Case and control selection
A matched case-control study was nested within the cohort to allow more intensive evaluation of potential factors associated with avian mycobacteriosis. All birds diagnosed histologically with avian mycobacteriosis were defined as cases. Each case was enrolled in the study at the original date of tissue collection in which acid-fast organisms were observed and diagnosis of avian mycobacteriosis was made. These dates often coincided with the date of death, since the majority of diagnoses were made postmortem.
Each case was matched to 7 controls (birds without a diagnosis of mycobacteriosis) randomly selected from those within the same taxonomic and age group. Taxonomic order was used as the matching factor for all groups of birds except Columbiformes, where pronounced species diversity prompted use of genus as the matching factor. Age matching was included to account for the age-related effects of exposure and disease development. For cases less than 1 year of age, matched controls were between 5 months and 1 year of age. For cases greater than 1 year of age, matched controls were within 1 year of the same age of their matched case, but were no less than 1 year of age. Cases and their matched controls were not required to be the same age at the same time; controls were drawn from among all birds without mycobacteriosis that matched genus and age criteria at any time during the 15-year study period. Data were summarized for each control bird using the date it attained the age of diagnosis of its matched case. If a control had not yet entered the population or had already left the population when it was the exact age of the matched case (n = 325), an alternate date for summarizing control data was randomly selected from within the time period that the control bird was present in the population and within the specified age range. Cases were eligible to be used as controls until 1 year prior to their date of diagnosis, for purposes previously described. 29
Potential risk factors
The variables examined in the present study included demographic characteristics of the bird, time periods in the collection, enclosure characteristics, potential stressors, and exposure to other disease-positive birds. Potential risk factors were assessed from the earliest acquisition date (i.e., the date when the bird first came into ZSSD care) until the date the bird was enrolled as a case or control.
Demographic characteristics were abstracted for each bird from computerized animal management and health records. Independently evaluated characteristics included sex, hatch type (zoo or wild hatched), and whether raised exclusively at the ZSSD, imported from another institution, or brought in from the wild. Because all birds that were hatched in the wild were also imported into the zoo, reference cell coding 21 was used to clarify the relationships between disease status, hatch type, and import status. Mycobacteriosis among wild-hatched, imported birds and zoo-hatched, nonimported birds were each compared with the reference group of zoo-hatched, imported birds.
The relationship between disease status and the amount of time that a bird spent in the ZSSD collection prior to its becoming a case or a control was evaluated by examining the linear effects of the total number of years (total number of days/365). Additionally, disease status was compared between birds living in the collection exclusively prior to the start of a ZSSD-initiated avian mycobacteriosis disease management program in 2001, and those that were either present both before and after 2001 or those living in the collection exclusively post-2001.
Effects of site and enclosure size on disease were assessed for the location where the bird resided immediately before it became a case or control. For birds that had been recently moved (n = 161), the most recent enclosure inhabited for at least 90 days was used. For birds that were enrolled into the cohort prior to spending 90 days in a single enclosure (n = 68), the characteristics of the most recent location were used. Enclosures were first coded as being located at either SDZ or SDZWAP. Then, enclosures were categorized according to size, using actual dimensions in combination with qualitative assessments from bird curators (small, not small) to establish a cutoff for identifying small enclosures. If enclosure measurements were unavailable, approximate dimensions were determined by bird managers and curators. Birds having recently lived in a small enclosure (<46.5 m2 and <142 m3 [<500 ft2 and <5000 ft3]) were compared with birds that had recently inhabited a larger enclosure.
Disease occurrence was compared among birds that were recently housed in outdoor enclosures with unobstructed access to wild birds (such as open-air duck ponds and flamingo pools) and those that had only ever been housed in enclosures with indirect, limited access to wild birds (enclosures that were indoors or those with mesh or wire on all sides and tops). Data describing intensity and species of wild bird contact were not available in the records. Other enclosure characteristics such as environmental conditions of dampness, cleaning methods, and bird density were not analyzed because of changes through time, and subjectivity and incompleteness of these descriptors in historic records.
The number of moves between distinct enclosures and the rate of enclosure moves (the number of moves divided by the number of years that the bird lived at the ZSSD) experienced at the ZSSD up until the time point that a bird was considered a case or control were determined from computerized enclosure history records. Effects of general movements between ZSSD facilities on disease status were also evaluated by comparing birds that had ever moved between the SDZ and SDZWAP and those that had never moved between locations.
Exposure to other mycobacteriosis-positive birds was assessed by identifying study subjects that shared enclosures with future disease-positive birds within 1 year of the subject becoming a case or a control. Sharing enclosures was only considered to be an exposure when it occurred within 1 year of the future disease-positive bird's diagnosis. Computerized records of enclosure histories, and complex functions and macros developed in Excel, a were used to date track all enclosure entries and exits of cases and controls, and their disease-positive contacts. Exposure was assessed for all birds in relation to the disease-positive contacts identified in this study, as well as 31 disease-positive contacts diagnosed prior to 1991. Exposures were further subcategorized as having been exposed to a bird with an intestinal infection or to a bird with only other organ involvement.
Analytical methods
Prevalence of avian mycobacteriosis, with 95% confidence intervals, was calculated for all birds in the ZSSD collection for the time period of 1991–2005. Prevalence was also calculated among only birds with necropsy reports, excluding birds that did not die at the ZSSD and those that died but had a notation in the record that no histopathology was able to be done. Disease incidence was determined by dividing the number of new cases between 1991 and 2005 by the total bird-years at risk for the entire population. Disease-positive birds contributed to the bird-years at risk until 1 year prior to becoming a case. Annual incidence rates and exact 95% confidence intervals were calculated.
Necropsy data was evaluated by calculating the proportions of diseased birds with characteristic lesions and the associated 95% confidence interval. Taxonomic order predilection for mycobacterial disease was evaluated by calculating order-specific prevalence estimates and their 95% confidence intervals. The proportion of disease-positive birds belonging to a particular taxonomic order was compared with the proportion of disease-positive birds in other orders with multiple pairwise comparisons and Fisher's exact tests. Taxonomic orders were also evaluated to determine whether lesion distribution and disease severity varied significantly by order. Proportions of disease-positive birds with intestinal lesions, respiratory lesions, and severe disease were compared separately with birds in other orders with Fisher's exact tests.
Univariate, conditional logistic regression (accounting for matching) was used to screen for factors to include in the multivariable modeling process and to estimate unadjusted odds ratios (OR), confidence intervals, and Wald statistics. Associations with P ≤ 0.25 were further evaluated in multivariable analysis. 14 A multivariable, conditional logistic regression model (SAS [statistical analysis software] procedure PHREG) was used to calculate the adjusted OR and 95% confidence intervals for case birds relative to controls as a function of the evaluated characteristics and exposure histories. The best subsets method 14,16 was used to identify the best model of each size based on differences in global score chi-square statistics. Variables were then individually included and excluded from the best-identified models to develop the final model. Effects of each factor were assessed based on significance of its Wald statistic and contribution to model fit (comparison of likelihood ratios). The assumption of linearity in the logit for continuous variables was evaluated by refitting the multivariable model, replacing each continuous variable with a 4-level categorical design variable based on quartiles of the continuous distribution. Estimated coefficients of the design variable were plotted against the midpoint of each quartile and visually inspected to verify the scale of the variable. 14 Potential confounding was evaluated by examining change in magnitude of coefficients for other factors in the model (greater than 10% change), with and without the potential confounding variable. 14,21 Variables that were not significant, not confounders, and did not improve the fit of the model (assessed by comparing model likelihood ratios and concordance) were removed. After fitting main effects, potential interaction was evaluated by comparing models with only main effects to those that also included biologically meaningful first-order interaction terms. 14,21 Interaction terms with a statistically significant estimated coefficient were retained. 14 Although matching multiple controls to each case minimizes the potential for effects of influential observations, final candidate models for outliers by calculating the changes in individual parameter estimates when a subject was deleted. 14 Outlier criteria was defined as a greater than 10% change in the estimated coefficients. The final model was selected based on strength and significance of associations, model fit, and biological plausibility. Analyses were conducted with statistical software program SAS. b Statistical significance in this study was defined at the P ≤ 0.05 level.
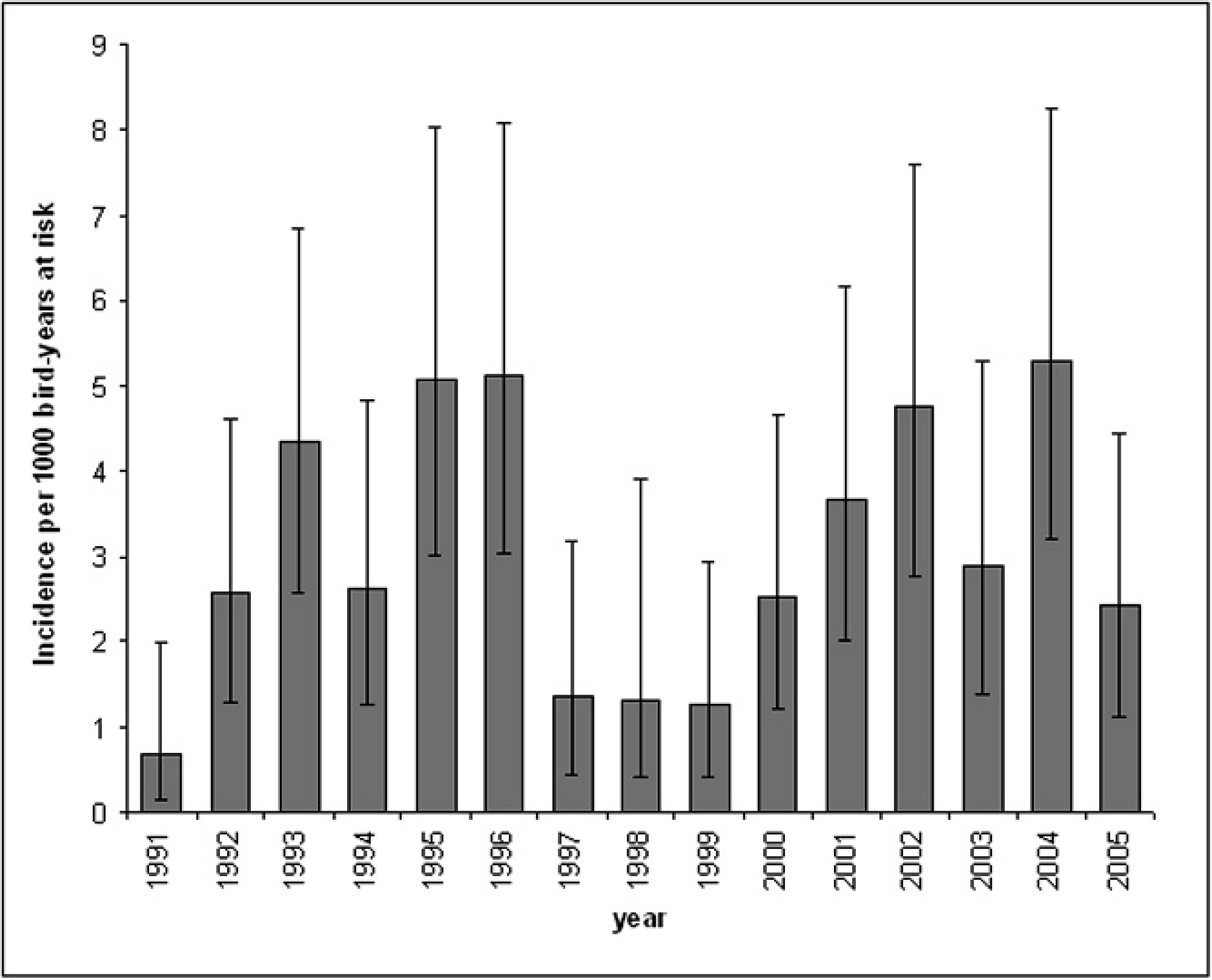
Annual incidence estimates and 95% confidence intervals of avian mycobacteriosis in the Zoological Society of San Diego's bird collection from 1991–2005.
Results
A total of 13,976 birds, representing 22 taxonomic orders and 894 different species, were included in the study. Prevalence of avian mycobacteriosis in the total population was estimated at 1.2% (172 of 13,976; 95% CI: 1.1–1.4%). The prevalence of disease when considering only birds with postmortem histopathology evaluations was 3.7% (172 of 4604; 95% CI: 3.2–4.3%). The incidence rate for the entire study period was estimated at 3.0 cases per 1,000 bird-years at risk (172 of 57,535 total bird-years at risk; 95% CI: 2.6–3.5 cases per 1,000 bird-years at risk). The estimated annual incidence rate varied, ranging from 0.7 (95% CI: 0.1–2.0) cases per 1,000 bird-years at risk in 1991 to 5.3 (95% CI: 3.2–8.2) cases per 1,000 bird-years at risk in 2004 (Fig. 1). Incidence rates fluctuated between years, but no overall temporal trends in incidence were observed.
Distribution and characteristics of lesions in affected tissues from the 172 disease-positive birds are summarized in Table 1. Significant mycobacterial lesions were present in at least 1 evaluated tissue in 108 (62.8%; 95% CI: 55.1–70.0%) of the disease-positive birds, whereas only incidental lesions were observed in the remaining 64 (37.2%; 95% CI: 30.0–44.9%). Moderate to abundant numbers of acid-fast organisms were present in at least 1 evaluated tissue in 100 (58.1%; 95% CI: 50.4–65.6%) of the disease-positive birds, whereas only a few organisms were observed in the remaining 72 (41.9%; 95% CI: 34.4–49.6%). Respiratory lesions (125; 74.9%; 95% CI:67.6–81.2%) occurred in more birds than intestinal lesions (95; 58.1%; 95% CI: 50.3–65.8%), but respiratory and intestinal lesions often occurred together (79; 48.8%; 95% CI: 40.8–56.7%).
Distribution and characteristics of mycobacterial lesions among 172 disease-positive birds.
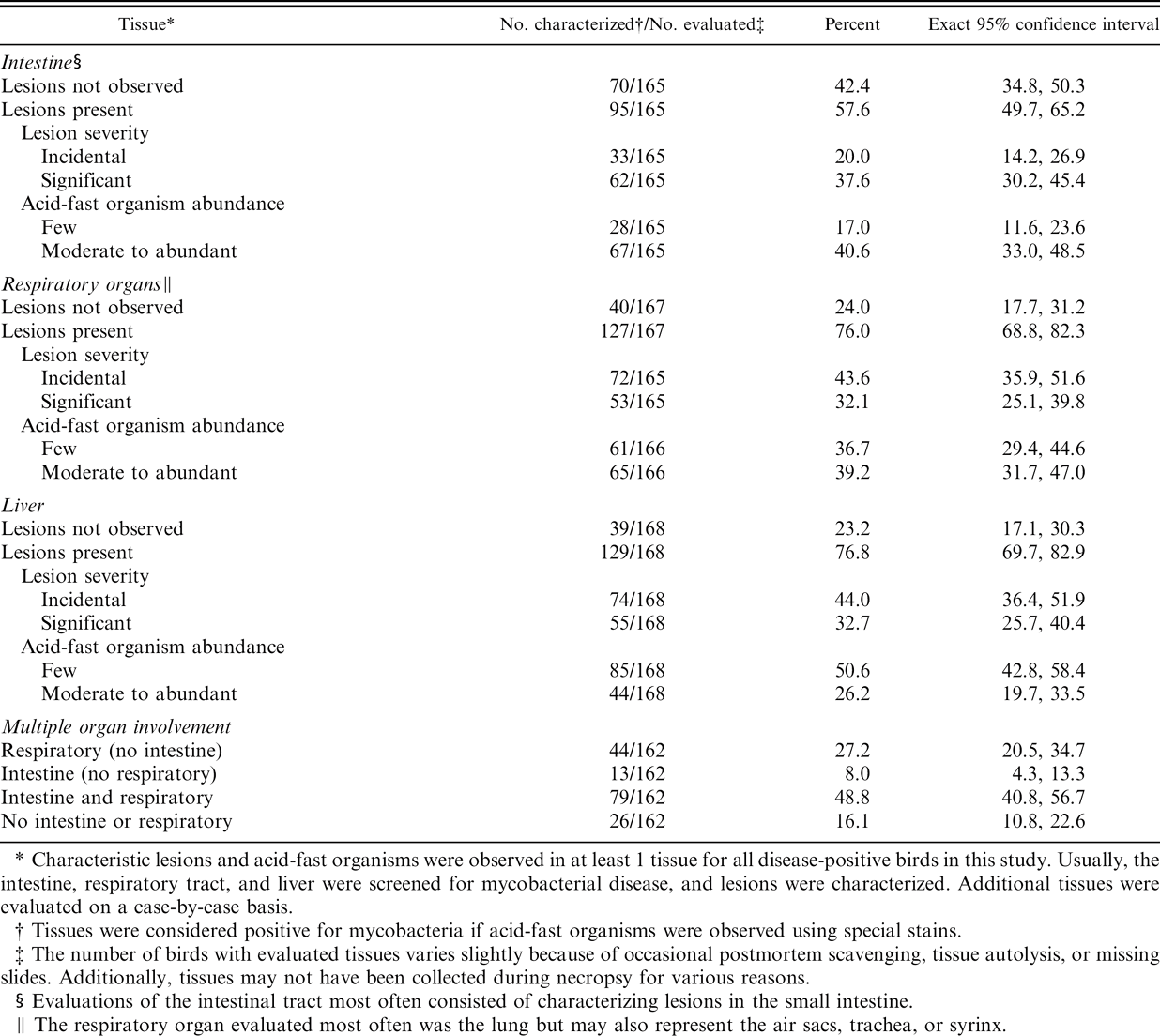
Characteristic lesions and acid-fast organisms were observed in at least 1 tissue for all disease-positive birds in this study. Usually, the intestine, respiratory tract, and liver were screened for mycobacterial disease, and lesions were characterized. Additional tissues were evaluated on a case-by-case basis.
Tissues were considered positive for mycobacteria if acid-fast organisms were observed using special stains.
The number of birds with evaluated tissues varies slightly because of occasional postmortem scavenging, tissue autolysis, or missing slides. Additionally, tissues may not have been collected during necropsy for various reasons.
Evaluations of the intestinal tract most often consisted of characterizing lesions in the small intestine.
The respiratory organ evaluated most often was the lung but may also represent the air sacs, trachea, or syrinx.
Select tissues from 77 of the 172 disease-positive birds were submitted for mycobacterial culture. Of the 50 samples in which the mycobacteria were isolated, M. avium subsp. avium was cultured from 30 (60%) birds; M. genavense was identified in 11 (22%) birds; and M. intracellulare was isolated from a single bird. Additionally, 8 (16%) birds had culture isolates identified as M. avium complex, which is comprised of a group of closely related species, including M. avium and M. intracellulare.
The 172 birds diagnosed with mycobacteriosis represented 12 of the 22 taxonomic orders included in the cohort (Table 2) and 96 different species and subspecies. Prevalence varied among taxonomic groups and was lowest in Ciconiiformes and Falconiformes; moderate in Phoenicopteriformes, Anseriformes, Psittaciformes, Passeriformes, Coraciiformes, and Galliformes; and highest in Columbiformes. Most birds were diagnosed at greater than 1 year of age, but 6 birds were diagnosed when less than 1 year old.
Prevalence of intestinal lesions in diseased birds varied between taxonomic groups (Table 3). Intestinal lesions were found less often in Anseriformes than in Columbiformes, Coraciiformes, Passeriformes, and Psittaciformes (P < 0.01). Intestinal lesions were also less prevalent in Galliformes than Columbiformes or Psittaciformes (P < 0.05). Respiratory lesion prevalence was consistently high in all groups with at least 2 cases, with no significant differences. Most cases had significant levels of disease and this also did not differ statistically between orders.
Numbers of birds affected with avian mycobacteriosis by taxonomic order.
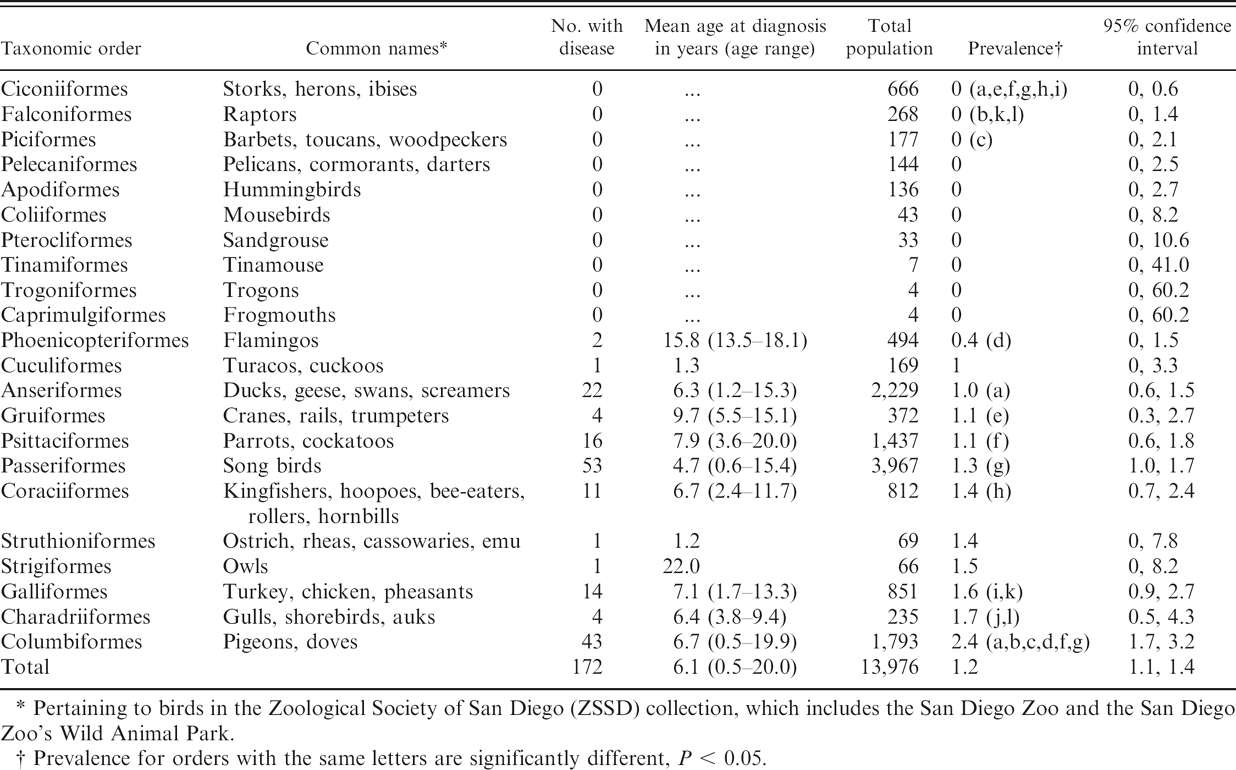
Pertaining to birds in the Zoological Society of San Diego (ZSSD) collection, which includes the San Diego Zoo and the San Diego Zoo's Wild Animal Park.
Prevalence for orders with the same letters are significantly different, P < 0.05.
For the matched case-control analysis, 167 mycobacteriosis-positive birds and 1,169 nondiseased birds were selected. Five cases were not selected for further evaluation because of lack of available controls. In the univariate matched analysis, 6 of the 16 variables evaluated were significantly associated (P ≤ 0.05) with increased or decreased odds for avian mycobacteriosis (Table 4). Three additional variables were selected for further consideration in the multivariable modeling based on the screening criterion of the P ≤ 0.25 level. The final model included 4 of the 9 factors evaluated in the multivariable analysis: exposure to a disease-positive bird with an intestinal infection (OR = 5.6); exposure to a disease-positive bird with any type of infection other than intestinal (OR = 2.0); importation from another institution or from the wild (OR = 4.2); and having more moves within the ZSSD (OR = 1.1 per additional move; Table 5). No interactions, additional confounding variables, or influential observations were identified.
Numbers of birds affected with avian mycobacteriosis by taxonomic order and type of lesion.*
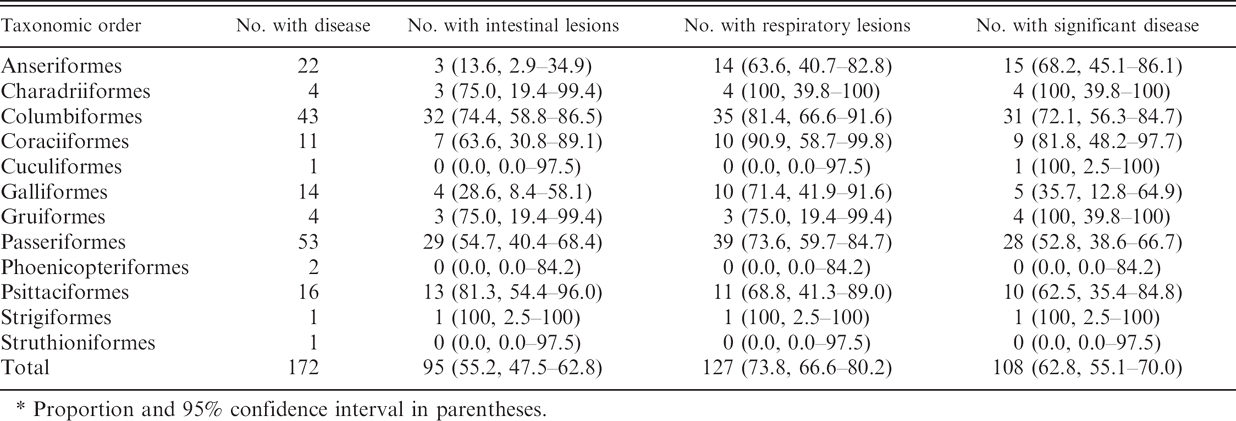
Proportion and 95% confidence interval in parentheses.
Risk factors for avian mycobacteriosis infection with univariate conditional logistic regression analysis.
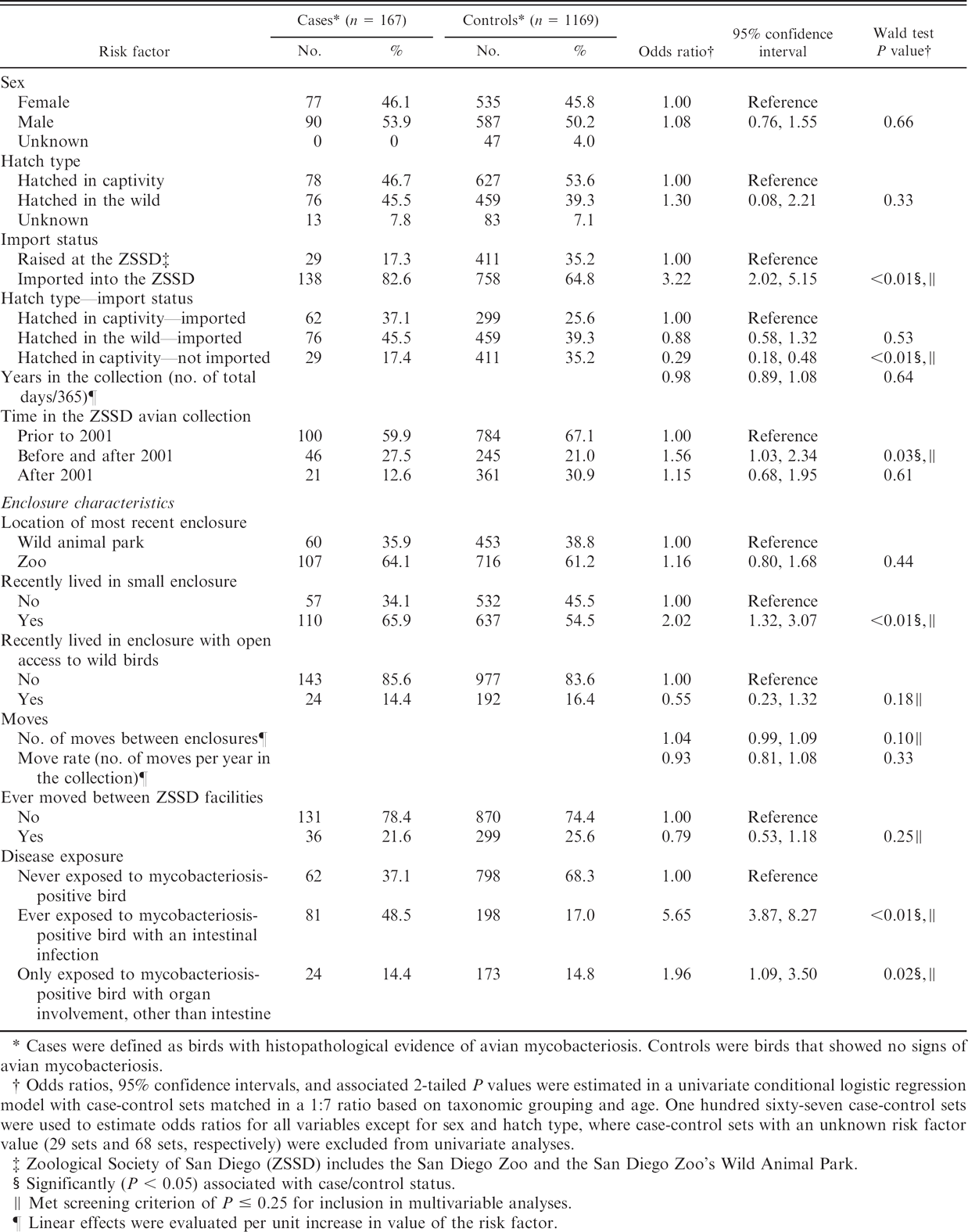
Cases were defined as birds with histopathological evidence of avian mycobacteriosis. Controls were birds that showed no signs of avian mycobacteriosis.
Odds ratios, 95% confidence intervals, and associated 2-tailed P values were estimated in a univariate conditional logistic regression model with case-control sets matched in a 1:7 ratio based on taxonomic grouping and age. One hundred sixty-seven case-control sets were used to estimate odds ratios for all variables except for sex and hatch type, where case-control sets with an unknown risk factor value (29 sets and 68 sets, respectively) were excluded from univariate analyses.
Zoological Society of San Diego (ZSSD) includes the San Diego Zoo and the San Diego Zoo's Wild Animal Park.
Significantly (P < 0.05) associated with case/control status.
Met screening criterion of P ≤ 0.25 for inclusion in multivariable analyses.
Linear effects were evaluated per unit increase in value of the risk factor.
Multivariable model of factors associated with avian mycobacteriosis.*
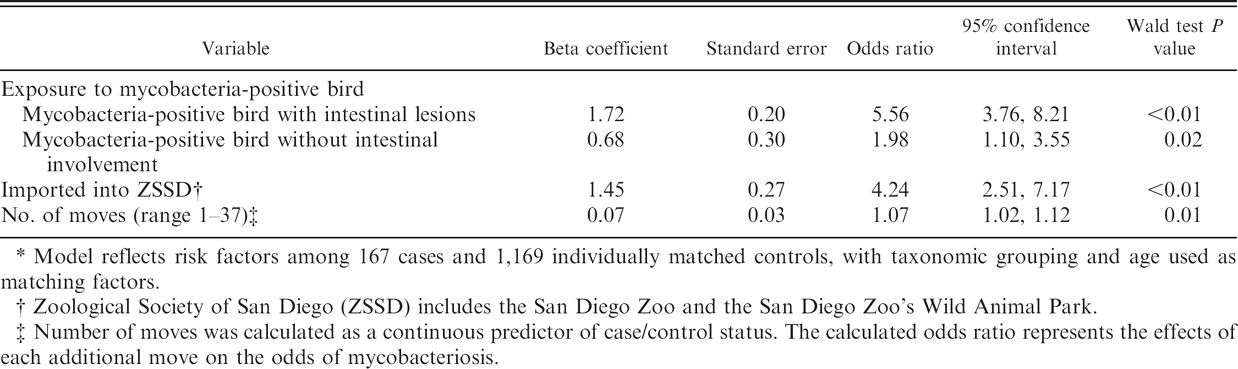
Model reflects risk factors among 167 cases and 1,169 individually matched controls, with taxonomic grouping and age used as matching factors.
Zoological Society of San Diego (ZSSD) includes the San Diego Zoo and the San Diego Zoo's Wild Animal Park.
Number of moves was calculated as a continuous predictor of case/control status. The calculated odds ratio represents the effects of each additional move on the odds of mycobacteriosis.
Discussion
Mycobacteriosis has been recognized in bird collections for nearly a century, 8 but successful disease control or elimination has remained elusive. Characterizing the incidence, prevalence, and risk factors for mycobacteriosis is an important step in planning interventions. The extensive databases at ZSSD, encompassing historical information on acquisition, management, and health for thousands of birds, present a valuable foundation for identifying populationwide patterns of disease. Epidemiologic study methods, such as cohort and matched case-control designs, facilitate control of biases while estimating contributions of interrelated factors to naturally occurring disease. Combining these resources provides a model system for identifying new avenues for possible infection control in bird collections.
The persistent low disease incidence and prevalence estimates for mycobacteriosis during the 15 years of the present study are comparable or slightly lower than those reported in other zoos, 1,5,8,17,23 albeit recent reports from peer-reviewed sources are lacking. Although mycobacteriosis is difficult to diagnosis in living animals, all dead birds were necropsied and screened for Mycobacteria using histologic techniques, making it unlikely that clinically significant cases of avian mycobacteriosis were missed in making these estimates. Cases may not have been identified if lesions were not recognized grossly or selected for histologic evaluation; if tissues were not of sufficient quality to evaluate postmortem changes; if birds moved out of the defined cohort during the study period before diagnosis; or if diseased birds were still alive and had not yet been diagnosed at the end of the study. Therefore, the true prevalence remains unknown but would likely fall between the values calculated from the entire cohort and from the subgroup of birds receiving postmortem examinations.
Although no trends were observed over time despite implementation of a focused screening and enclosure quarantine program, the long incubation and preclinical periods following exposure, which can vary widely, may have masked any short-term trends in incidence. Additionally, quarantine prevents group-to-group spread but does not curb spread within a quarantined enclosure. This transmission, coupled with increased case finding through screening, may overshadow program effects on population-level incidence. More detailed assessment of disease incidence among subgroups of quarantined and nonquarantined birds may be needed with long-term follow-up to account for the very slow progression of disease.
Avian mycobacteriosis has traditionally been described as primarily an intestinal disease, passing between susceptible hosts by the fecal-oral route of transmission. 18,24 In this large cohort, respiratory lesions were detected in 76% of mycobacteriosis-positive birds that were evaluated for respiratory disease. Respiratory-only infections that did not include intestinal involvement were observed in 27% of cases. In contrast, intestinal lesions were observed in 58% of evaluated cases, with only 8% of cases having intestinal involvement without concurrent respiratory lesions. These findings suggest that inhalation of Mycobacteria may be an important route of infection and transmission and should be addressed in disease control plans. Most recommended control programs, such as the one implemented in 2001 by the ZSSD, are based on increased screening for intestinal infection. Based on these results from the current study, respiratory organs are often affected, and screening for respiratory disease may also be warranted.
There has been discrepancy in the literature on which bird species are most susceptible to avian mycobacteriosis. Nearly all bird species can be infected, but susceptibility has been reported to vary across species. 24 Anseriformes, Gruiformes, and Galliformes have often been considered to be the most susceptible; however, this may reflect the larger numbers of these species in zoo collections rather than a measure of susceptibility. 24 For example, an estimated 298 birds would need to be necropsied to be 95% certain of finding at least 1 case if the actual prevalence of mycobacteriosis was 1% among birds in a given order. 25 All 3 of the orders that have been considered to be the most susceptible had reasonably large numbers of representative birds, and cases were found in the current study. However, among Columbiformes, which were also represented in large numbers, significantly higher proportions of diseased birds were identified than were observed in most other orders. Similarly, Ciconiiformes were found to have significantly fewer cases than other orders. Although sample sizes were too limited to compare prevalence estimates for many bird groups, the observed variability may represent genuine genetic diversity in susceptibility to mycobacteriosis or serve as a proxy for differences in other risk factors that cluster in these bird groups. In general, there were no obvious trends in prevalence that might indicate relationships between exposure and broad feeding behaviors or habitat preferences (i.e., ground, arboreal, or aquatic). However, the identification of taxonomic groups with both high and low prevalence levels provides an avenue for further investigations of disease susceptibility, resistance, and transmission dynamics.
While the cohort study provided incidence measures in the population and species groups, the matched case-control analysis allowed more intensive characterization of potential risk factors. Exposure to a disease-positive bird was an important predictor of case and control status. The even higher odds ratio estimated from exposure to a bird with an intestinal infection is consistent with the prevailing hypothesis that shedding of mycobacteria into the environment is a source of infection for other birds. 24 However, this data could not directly test bird-to-bird transmission hypotheses because exposure to a diseased bird, by definition, may also implicate exposure to a common environmental source.
Associations with exposure could be overestimated if enclosure mates of previous cases received a more thorough diagnostic screening for mycobacteriosis than other birds. There could have been a countering detection bias if birds from enclosures without any diagnosed cases were screened more carefully to be sure that these locations remained free of contamination. Knowledge about prior exposure was available at the time of postmortem examination for some birds but was often very limited. Classification of exposure for this study was a later, complex task involving reconstruction of bird movements over the 15 years through joining multiple data sources. Therefore, a complete inventory of exposed birds, the amount of time they spent in the same enclosure together, and the characteristics of the exposure were unknown at the time of postmortem examination. The extent to which screening varied among all birds in this study is unknown. However, a requirement for thorough postmortem examinations including histopathology for all birds as part of normal operating procedures, such as that practiced by the ZSSD, is the best way to counter these potential biases.
A substantial proportion of the cases (37%; 62 of 167) were never known to have been exposed to another disease-positive bird, which presents a major gap in current hypotheses related to transmission dynamics and could compromise control efforts. Exposures to mycobacteriosis-positive birds that occurred prior to the defined, 1-year period could have played a role in infection acquisition and development. Birds moving into an enclosure after removal of a disease-positive bird may also have been exposed to Mycobacteria but were not considered “exposed” in this study because extensive environmental cleaning was often done before the move. However, some sources of environmental exposure may have persisted. Soil and water, which are considered reservoirs for nontuberculous mycobacterial infection in humans, commonly harbor species of Mycobacteria that are pathogenic to birds. 6,20,27 A recent investigation 22 found a lack of evidence for bird-to-bird transmission in a molecular strain-typing study of mycobacteriosis in zoo birds. Another study 15 found a high level of diversity among isolates from a single epidemic of mycobacteriosis in lesser white-fronted geese (Anser erythropus). These investigations suggest that environmental sources of pathogenic mycobacteria may be more important than previously recognized.
The higher odds of being a case among birds that were imported into ZSSD may indicate previous exposure to other bird or environmental sources outside of ZSSD. Imported birds may also have experienced stress related to shipping that could increase their susceptibility to infection and disease. Experimental studies have highlighted the importance of immunosuppression on mycobacterial infection and have shown that the character and quantity of tuberculous lesions in chickens were related to both the genetic makeup and stress. 10,28 Findings from epidemiologic studies 3,4 on wildfowl populations demonstrate associations between onset of disease and the time of year, suggesting that seasonal stress (from breeding) and/or climate-induced stress may play a role in onset of disease. Stress is also speculated to result from malnutrition, overcrowding, pinioning, concurrent disease, social aggression, and handling. 24
Cases experienced more moves between enclosures than did controls in this population. Birds at the ZSSD, as in most zoo collections, were often moved for breeding, behavior, husbandry, and management purposes. Moves between enclosures may promote stress from social integration/separation and excess handling, or may increase the opportunity for exposure to other infected birds, contaminated environments, or other unknown cofactors for disease. Quarantine and hospital moves were not included in move calculations to decrease the effects of moves that may have resulted from the clinical presentation of symptoms related to mycobacterial disease. Reducing the overall number of moves between enclosures and minimizing stressors may help to decrease the overall incidence of disease.
Risk factors may differ for disease caused by various species of Mycobacteria. Some have suggested that infections caused by recognized avian pathogens may differ between birds. For example, 1 study 19 concluded that M. genavense may not be easily transmitted between birds and may have limited pathogenicity. Similarly, M. intracellulare may be minimally pathogenic for some species, whereas infection with M. avium may more readily produce disease. 2 Unfortunately, the species of Mycobacteria infecting a particular bird was unknown for the majority of cases in the current study because cultures were not always performed and often did not yield further speciation. Therefore, risk factors specific to different mycobacterial species could not be assessed. Of the 50 birds with culture isolates, M. avium was cultured most often. This finding contrasts that of others 11,13,19 where M. genavense was the most commonly isolated organism from birds in Europe. Differences in findings may reflect regional differences in predominant species of Mycobacteria or differences in culturing methods.
The management and disease control strategies used by the ZSSD over the study period were generally those recommended to decrease mycobacterial transmission in zoos and are characteristic of many zoological institutions. Therefore, results from this study may provide a useful model for identifying birds at risk for contracting avian mycobacteriosis in other institutions. Some factors identified in this study, such as frequent intra-institutional moves, may be more common to ZSSD management than other zoos. Conducting similar analyses at institutions where different management strategies are implemented may provide additional insight into factors associated with mycobacterial infection. Analyzing data from diverse avian populations that have traditionally had a higher prevalence may also be valuable for identifying predictors of disease, as datasets yielding a larger number of cases would contribute to a more powerful analysis.
Results from this study indicate that the factors explaining the variation between birds with and without avian mycobacterial disease can be quantified and should be considered when managing disease in a collection. Recognition of the characteristics of birds with avian mycobacteriosis described herein lays a foundation to help guide future management of this disease for zoo bird populations.
Acknowledgements
This study was supported by the Bud Heller Foundation and the Zoological Society of San Diego. The authors thank J. P. Montagne, Yvonne Cates, April Gorow, and Julie Concha for technical support. The authors thank the veterinary staff, the bird curators, managers, and keepers that helped with interpretation aspects of this study. Among these contributors, special thanks go to David Heckard, Dr. Nadine Lamberski, Michael Mace, David Orndorff, David Rimlinger, Dr. Meg Sutherland-Smith, and Pat Witman.
Footnotes
a.
Excel, Microsoft Corporation, Redmond, WA.
b.
SAS v. 9.2, SAS Institute Inc., Cary, NC.
