Abstract
Because interactions between livestock and chamois occur on Alpine pastures, transmission of infectious diseases is considered possible. Thus, the occurrence of Chlamydiaceae, Mycoplasma conjunctivae, and pestiviruses in Alpine chamois (Rupicapra r. rupicapra) of the Surselva region (eastern Swiss Alps) was investigated. In total, 71 sera, 158 eye swabs, 135 tissue samples, and 23 fecal samples from 85 chamois were analyzed. The sera were tested by 2 enzyme-linked immunosorbent assay (ELISA) kits specific for Chlamydophila abortus. Eye swabs, tissue, and fecal samples were examined by a Chlamydiaceae-specific realtime polymerase chain reaction (PCR). Positive cases were further investigated by microarray method. One serum sample (1.4%) was positive in 1 of the ELISAs. Eye swabs of 3 chamois (3.8%) were positive for Chlamydiaceae. The microarray method revealed the presence of Chlamydophila abortus, C. pecorum, and C. pneumoniae. All tissue and fecal samples were negative. With real-time PCR, 3.9% of the chamois tested positive for Mycoplasma conjunctivae. One chamois had a simultaneous infection with M. conjunctivae and 2 chlamydial species (C abortus, C. pecorum). Skin and tongue tissue samples of 35 chamois were negative for pestivirus antigen by immunohistochemistry. It was concluded that in contrast to the findings in Pyrenean chamois (Capra p. pyrenaica) of Spain, the occurrence of Chlamydiaceae in Alpine chamois of the Surselva region is low, and the transmission between domestic and wild Caprinae seems not to be frequent. Comparably, persistent pestiviral infections do not seem to be common in chamois of the Surselva region.
Chlamydophila abortus is the most common infectious abortigenic agent in small domestic ruminants in Switzerland. A previous study revealed that 39% of examined abortions in sheep and 23% in goats were caused by C. abortus. 7 Economic losses due to chlamydial abortion in small domestic ruminants are significantly higher in the canton of Grisons (eastern Swiss Alps) than in other Swiss regions (Grisons: 43% seroprevalence in sheep for C. abortus). 4 The question was raised whether wild ruminants play a role in the transmission of chlamydiosis. In a previous study, a low prevalence of Chlamydiaceae was demonstrated in Alpine ibex (Capra i. ibex) of Switzerland, 16 but the situation in other wild ruminants still remains to be elucidated. Alpine chamois (Rupicapra r. rupicapra) are known to be in contact with domestic sheep and goats on Alpine pastures 21 and could therefore possibly play a role in distribution and transmission of infectious diseases. Infectious keratoconjunctivitis (IKC) is an ocular disease affecting domestic as well as wild Caprinae such as chamois and Alpine ibex. At present, Mycoplasma conjunctivae is regarded as the major cause of IKC, 3 but chlamydiae have also been reported to be involved in the etiology of IKC. For example, in the United States, a Chlamydia sp. was isolated from the conjunctiva of 2 of 7 free-ranging mule deer (Odocoileus hemionus) with IKC in Zion National Park in Utah 24 and was detected in the eyes of bighorn sheep (Ovis canadensis) affected by IKC in Yellowstone National Park in Wyoming. 18
Eradication of bovine viral diarrhea (BVD) is currently performed in Switzerland. Before eradication, the prevalence of persistent infections in cattle of Switzerland was approximately 1%. 6 However, wild ruminants are not tested during this program, as they are not believed to have a significant epidemiologic impact. Bovine viral diarrhea virus (BVDV) and Border disease virus (BDV) are not strictly host-specific, and transmission between species has been described. 23 In wildlife, infection with pestiviruses has been demonstrated worldwide in serologic surveys. 26 A very recent study revealed a seroprevalence of 4.3% for BVDV in Alpine ibex of Switzerland (Marreros et al., submitted), but data on the prevalence of pestiviruses in chamois are lacking at present. The aim of the present study was to determine the occurrence of Chlamydiaceae in Alpine chamois of Grisons (eastern Swiss Alps), using sensitive and specific methods. Furthermore, eye swabs were investigated for M. conjunctivae by real-time polymerase chain reaction (PCR), and cryosections of skin and tongue tissue were tested by immunohistochemistry (IHC) for the presence of pestiviral antigen.
All samples (n = 387) available for the study originated from a total of 85 Alpine chamois. The material consisted of 71 sera, 158 eye swabs (originating from 79 chamois), 135 organ samples (of 42 chamois), and 23 fecal samples. The samples were collected in autumn 2008 in the Surselva region (canton of Grisons, eastern Swiss Alps, 46°36′ to 46°48′ N, 8°42′ to 9°20′ E). This area is known as a risk area for chlamydial abortion in sheep, 4 and interactions between wild and domestic ruminants occur regularly (Brosi and Thoma, personal communication, 2008). The animals used for the study were either shot during the hunting season (September 2008, mainly healthy animals) or were killed by game wardens due to disease symptoms from September to November 2008. Two chamois were found dead. For each chamois, a questionnaire form was completed by the sampling person providing information about the animal and recording disease symptoms. Of 19 chamois, the complete sample spectrum was obtained, which included serum, eye swabs, organs, and fecal samples. The most frequently collected organs included liver, lung, kidney, and genital tract, as well as skin and tongue for the examination of pestiviruses. Of 66 animals, only limited samples were available.

Chamois severely affected by infectious keratoconjunctivitis showing broad corneal ulceration and high amount of mucopurulent exudate.
The DNA extraction was performed using a nucleic acid isolation system, a as previously described, 16 followed by real-time PCR for Chlamydiaceae on a commercial instrument b using the previously described 23S ribosomal RNA gene-based Chlamydiaceae family-specific real-time PCR. 11 A threshold cycle (Ct value) of <38.00 was considered as positive, and all samples were tested in duplicate. The results were interpreted as questionably positive if 1 Ct value was less than 38 and the other sample showed no Ct value. If 1 Ct value was more than 38 and the other sample showed no Ct value, the result was interpreted as questionably negative. The samples with at least 1 positive Ct value were examined using a 23S microarray assay c for chlamydial species identification, as described previously. 5 The serum samples were tested with 2 commercial antibody-detecting ELISAs specific for C. abortus: 1) Pourquier ELISA Chlamydophila abortus serum verification d (hereafter, Pourquier ELISA) validated for sheep, goats, and cattle; and 2) ID Screen C. abortus indirect ELISA e (hereafter, ID Screen ELISA) validated for ruminants, horses, and swine. The 2 ELISAs were performed according to the manufacturer's instructions. In both cases, the final values were determined as a ratio between the corrected optical density of the sample (S) and the mean corrected optical density of the positive control (P), expressed as S/P%. Sera with S/P% values equal to or less than 50% were interpreted as negative, sera with a S/P% of 50–60% were classified as doubtful, and sera with a S/P% higher or equal to 60% were considered positive for antibodies against C. abortus. For chamois sera, reference values for goats were applied with both ELISAs. Presence of M. conjunctivae in the eye swabs was examined by realtime PCR using an lppS-directed hydrolysis probe, as previously described. 21,29 The assay included the presence of an exogenous internal positive control in each reaction to check for the presence of eventual PCR inhibitors and to prevent false-negative responses. Immunohistochemistry for pestiviruses was performed using frozen samples of skin and tongue tissue by 2 antibodies specific for BVDV and pestiviruses, respectively, as described previously. 15
In field observations performed by hunters and game wardens, 64 of 85 investigated chamois were declared healthy, whereas pathological changes were reported in 21 animals. Eye lesions, such as corneal ulceration, corneal opacity, mucopurulent discharge, or blindness, were found in 11 animals (12.9%), whereas poor body condition of unknown cause was reported in 7 chamois. Figure 1 shows an example of a chamois with severe eye lesions. Furthermore, pulmonary lesions consistent with pneumonia were seen in 2 chamois. One of these 2 chamois was found dead and the other was shot due to poor body condition. In addition, 1 chamois found dead showed abscesses in lung and liver. Details of results from 6 chamois (nos. 1–4, 35, and 82) that were positive for at least 1 test are given in Table 1. All 71 serum samples investigated were negative for antibodies against C. abortus by Pourquier ELISA, whereas in the ID Screen ELISA, 1 serum (1.4%) reacted positive (animal no. 4, clinically healthy). Eye swabs (left and right) from 79 chamois were available for the Chlamydiaceae real-time PCR. Of these, 3 animals (3.8%) showed positive results. By microarray investigation, the following chlamydial species could be identified: mixed infection with C abortus and C. pecorum in one eye and C. pecorum monoinfection in the other eye of chamois no. 1; C. pneumoniae unilateral in animal no. 2; one eye of chamois no. 3 was positive by real-time PCR for Chlamydiaceae but the chlamydial species could not be determined by microarray (Table 1). Eye swabs from 76 chamois were analyzed by real-time PCR for M. conjunctivae: 3 chamois had to be completely excluded from the study due to insufficient quality of the samples for this assay. Furthermore, for 8 of 76 animals, only 1 eye swab per chamois could be included in the current study. Three of 76 animals (3.9%) were positive for M. conjunctivae. Animal nos. 1 and 82 were positive in both eyes, whereas chamois no. 35 was only positive in 1 eye (Table 1). Remarkably, in chamois no. 1, a mixed infection with C. abortus, C. pecorum, and M. conjunctivae was found (Table 1). This animal was blind, showed corneal lesions in both eyes, and was emaciated. Chamois nos. 35 and 82 showed eye symptoms as well, whereas chamois nos. 2 and 3 were healthy. All organs (n = 135, originating from 42 chamois) and fecal samples (n = 23) were negative by realtime PCR analysis for Chlamydiaceae. Among the group of 64 healthy animals, 3 (4.7%) were positive for Chlamydiaceae by either PCR or ELISA, whereas in the group of animals showing eye lesions, 3 of 11 chamois (27.3%) were positive for Chlamydiaceae and/or M. conjunctivae. All 68 skin and tongue tissue samples of 35 chamois tested negative for pestivirus antigen by IHC.
Details of chamois (n = 6) positive by either serology for Chlamydophila abortus or real-time polymerase chain reaction (PCR) for Chlamydiaceae and/or real-time PCR for Mycoplasma conjunctivae *
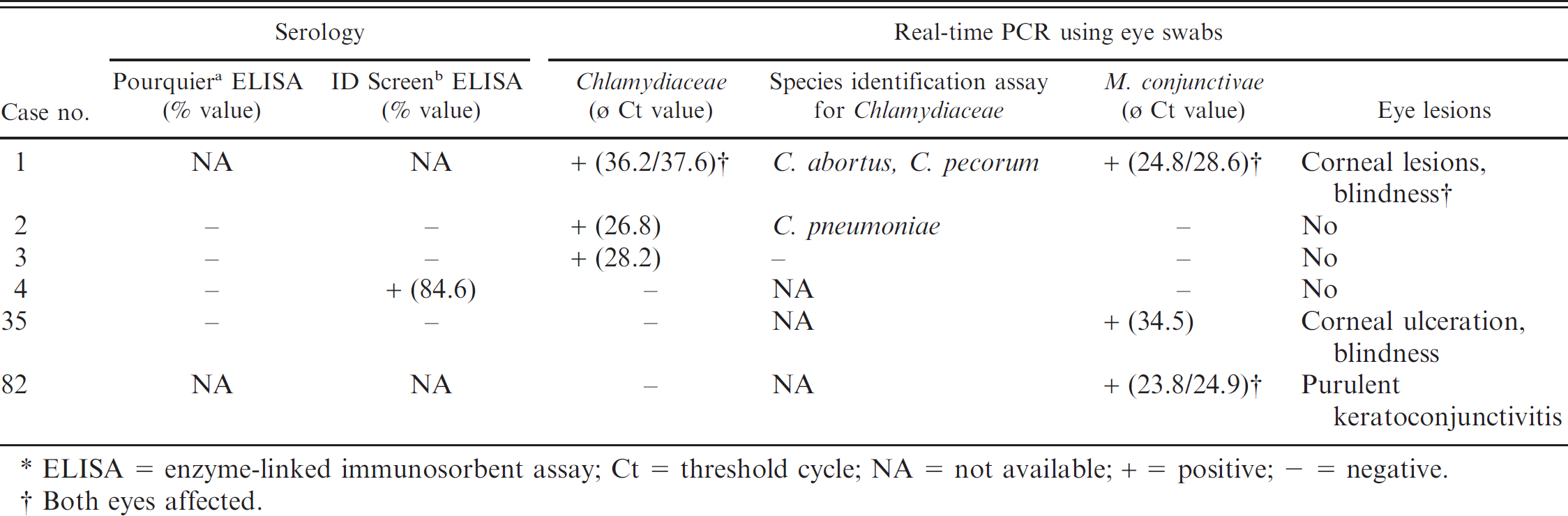
ELISA = enzyme-linked immunosorbent assay; Ct = threshold cycle; NA = not available; + = positive; – = negative.
Both eyes affected.
The occurrence of C. abortus antibodies assessed in the present work by 2 different specific ELISAs was low (0 and 1.4%, respectively). These results are comparable to those recently obtained in Swiss Alpine ibex using the same tests (seroprevalence of 1.5%). 16 The 2 ELISAs gave slightly different results: when using the Pourquier ELISA kit, all 72 sera tested negative, whereas a single serum reacted positive in the ID Screen ELISA. The noncongruent results in 1 serum could possibly be explained by the fact that different specific antigens (polymorphic outer membrane protein [POMP] vs. major outer membrane protein) were used. In contrast to the results of the present study, a recent study 22 reported a very high seroprevalence (40%) in Pyrenean chamois (Rupicapra p. pyrenaica) from Spain. Those investigators used a C. abortus-specific in-house ELISA directed against POMP, which is somewhat comparable to the Pourquier ELISA used in the current study. Hence, it was concluded that the differing seroprevalences are more likely due to the different geographic localization of the 2 chamois subspecies than to differing performance of laboratory tests. Using a real-time PCR for the detection of Chlamydiaceae in eye swabs, 3 chamois showed positive results, 2 in a single eye and 1 in both eyes. Microarray revealed 3 different chlamydial species: C. pecorum, C. abortus, and C. pneumoniae. In 1 chamois, the chlamydial species could not be determined, whereas in another animal, a mixed infection with C. abortus and C. pecorum was detected, which is consistent with the previous finding of 4 ibexes showing mixed infections by the same chlamydial species in the Surselva region. 16 Interestingly, 1 chamois was positive for C. pneumoniae, which has been detected in an ibex from the same region as well. 16 Neither the ibex nor the chamois infected with C pneumoniae showed any clinical eye lesions, which is consistent with the findings of a previous study where no association between the presence of chlamydial DNA (C abortus, C. pecorum, C. suis) in the eyes of sheep and the onset of clinical disease was found. 20
To the authors' knowledge, this is the first description of C pneumoniae in chamois. Pathogenic role and zoonotic potential of C. pneumoniae in wild ruminants remains to be elucidated. In the present study, no correlation between positive PCR results for Chlamydiaceae and positive C. abortus serology was found. The findings confirm the results of a previous study, where the investigators found no agreement between positive PCR results for Chlamydia in the male genital tract and semen of small ruminants and positive serology results for C. abortus. 25 However, good correlation between the presence of C. abortus DNA in conjunctival swabs of sheep and seropositivity was reported previously. 20 All organs (n = 135) and fecal samples (n = 23) analyzed were negative by real-time PCR, indicating that there was neither systemic chlamydial infection nor intestinal infection with fecal shedding. In summary, considering the results obtained by ELISA and real-time PCR, it was concluded that, similarly to the situation in Alpine ibex, C abortus is a very rare infectious agent in the Swiss Alpine chamois population. Thus, Alpine chamois are not responsible for the high seroprevalence observed in sheep in Grisons. Further studies are planned to elucidate the situation in red deer (Cervus elaphus) and roe deer (Capreolus c. capreolus) of Switzerland.
In the Swiss Alps, IKC is frequent in free-ranging Alpine chamois and ibex. 12 Infectious keratoconjunctivitis describes a contagious medical condition, and several infectious agents can be involved. Other microorganisms isolated from ruminants with symptoms of keratoconjunctivitis were, for example, Moraxella (Branhamella) ovis, Listeria monocytogenes, or Chlamydia psittaci, 1 although the general consensus is that M. conjunctivae is the main causing agent of IKC. 3,21,29 Simultaneous infections with M. conjunctivae and Chlamydiaceae have not yet been reported in the literature. In the current study, 1 chamois (animal no. 1) showed a mixed infection with M. conjunctivae and 2 Chlamydiaceae spp. (C. abortus and C. pecorum). This animal was emaciated and had severe eye symptoms. Although it has been reported that M. conjunctivae load in the eyes seems to be responsible for development of IKC, 21,29 one cannot exclude that clinical symptoms may also be enhanced by simultaneous infection with M. conjunctivae and Chlamydiaceae. For single infections with M. conjunctivae, frequency of occurrence is difficult to estimate as outbreaks are associated with the presence of infected sheep on Alpine pastures during summer months. 3 In the present study, 3 of 11 chamois affected by ocular disease (27.3%) were positive for M. conjunctivae, which is comparable to a previous study, 13 where among a sample of 28 chamois affected by IKC, M. conjunctivae was identified in 14 animals (50%) by conventional PCR, but which is inconsistent with more recent studies using real-time PCR whereby the frequency of M. conjunctivae in the eyes of IKC-affected animals was reported to be much higher. 21,29 The presence of pestiviruses in wildlife populations should be monitored to create long-term successful eradication programs. 19,27
In the United States, where persistently infected (PI) white-tailed deer (Odocoileus virginianus) were detected, there is major concern that those animals could endanger a complete eradication of BVDV. 8 In addition, persistent infections have been described in other wild ruminant species such as mouse deer (Tragulus javanicus) and eland (Taurotragus oryx). 10,14,28 During investigations for the reason for population decreases in Pyrenean chamois in the French and Spanish Central Pyrenees, 21 animals found dead or dying were necropsied. Organs of all chamois were positive for pestivirus by IHC. 17 Because PI animals are important regarding BVD eradication programs, IHC was used to detect PI animals. In the current study, all skin and tongue tissue samples (n = 68) originating from 35 chamois were negative for BVDV and pestivirus antigen when investigated by IHC. The low number of positive samples found in the present study contrasts with previous reports, most of them based on serology. For example, 16% of seropositive chamois were found in the Central Pyrenees, and 25.5% of the investigated chamois were positive in an Italian study. 2,19 There, the investigators stated that presence of BDV among extensively kept domestic sheep may be the cause for the high pestivirus antibody prevalence in that chamois population. A recent Swiss study in sheep in Grisons showed a seroprevalence of 32.4% and 33.7% for BVDV-1 and BDV-1, respectively. 9 Although no evidence for PI chamois could be found in the present study, the existence of PI chamois in Grisons cannot be excluded, as the number of sampled animals was small (35 chamois). In summary, despite the limited number of samples investigated, it is concluded that the occurrence of Chlamydiaceae and pestiviral infections in chamois of the Surselva region (Grisons, Switzerland) is low, and that transmission between domestic and wild Caprinae seems unlikely.
Acknowledgements. The present study was supported by the Federal Veterinary Office, Bern, Switzerland (project 1.08.18). The authors are very grateful to the hunting authorities, game wardens, and hunters of Grisons for the excellent collaboration and for providing numerous chamois samples. Thanks to Dr. med. vet. G. Brosi for the professional coordination of the sampling and Dr. med. vet. G. Bearth and Dr. med. vet. U. Probst for providing the laboratory equipment during fieldwork. Many thanks to M. Schmid who provided the photograph for Figure 1. The authors are grateful to ID Vet diagnostics for providing ELISA kits and to the laboratory technical staff of the Institute of Veterinary Pathology and of the Clinical Laboratory, Vetsuisse Faculty, University of Zurich for technical help.
Footnotes
a.
MagNA Pure® LC System, Roche Diagnostics GmbH, Mannheim, Germany.
b.
ABI 7500, Applied Biosystems, Foster City, CA.
c.
ArrayTube, Clondiag Chip Technologies GmbH, Jena, Germany.
d.
Institut Pourquier, Montpellier, France.
e.
ID Vet Innovative Diagnostics, Montpellier, France.
