Abstract
The aim of the current study was to validate an automated spectrophotometric method for salivary alpha-amylase measurement in pigs and evaluate its possible application as a noninvasive stress biomarker. The analytical validation included intra- and interassay precision, linearity under dilution, and limit of detection. In addition, to study the possible use of salivary alpha-amylase as a possible stress marker, 12 crossbred growing pigs of 3–4 months of age were subjected to restraint stress by a nasal snare for at least 1 min, and saliva samples were obtained at different time points. The results of analytical validation indicated that the method was precise and able to measure alpha-amylase in a linear manner. The results obtained in the stress test showed a significant increase in salivary alpha-amylase activity. Although other factors influencing this enzyme activity should be studied, these preliminary results indicate that salivary alpha-amylase could be a reliable biomarker of stress in pigs.
Keywords
Introduction
In the last decade, there has been a growing concern in producers and consumers about the importance of minimizing the stress in farm animals. In pigs, stress has been considered a cause of production decrease due to an increase in animal susceptibility to infectious diseases, as well as a decrease in the quality of meat products. 2,9,12
To evaluate stress in pigs, the measurement of several analytes, such as catecholamines (epinephrine and norepinephrine) as markers of the sympathetic adrenal-medullary (SAM) system in blood, 13 and cortisol as a marker of activation of hypothalamic-pituitary–adrenocortical (HPA) axis in blood and saliva, 1,20 has been reported. The use of saliva as sample for stress evaluation has several advantages compared with blood since it can be obtained by noninvasive procedures that do not produce additional stress or harm to the animals. In addition, saliva sampling procedures are very simple to obtain and can be taken by personnel with minimum training. 7
In human beings, the use of salivary alpha-amylase as a marker of stress has gained popularity during the last decade. 15 Many studies have shown that alpha-amylase levels rise in response to both physical and psychological stress. 3,6,14 While salivary alpha-amylase levels respond to emotionally arousing pictures as well as to physiological stressors, cortisol reacts only to the physical stressors. 25 Overall, salivary alpha-amylase might be used in parallel with indicators of the 2 stress systems (SAM and HPA), such as cortisol and catecholamines, because it seems to bear additional information. 15 The purposes of the current study were to validate an automated spectrophotometric method for the determination of salivary alpha-amylase in pigs, and to evaluate its possible application as a noninvasive biomarker of stress in such species.
Material and methods
Animals and sample material
Twelve crossbred growing pigs ([Sus scrofa domesticus] Duroc × [Landrace × Large White]) were used for the present study. All animals were 3–4 months of age and were housed in the Experimental Farm of the University of Murcia (Murcia, Spain). Pigs were given ad libitum access to a nutritionally balanced diet and water. The animals were housed in pens with a minimum space of 0.65 m 2 per animal (Council Directive 2001/88/CE of 23 October 2001 amending Directive 91/630/CEE concerning minimum standards for the protection of pigs. In Spanish. Available at http://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=CELEX:32001L0088:FR:HTML. Accessed on October 13, 2010). The temperature in the pens was kept at a minimum of 23°C. Animals set for stress induction were in different pens in order to avoid possible psychological stress of nasal snare on other animals.
Salivary samples were obtained by introducing a small sponge in the pigs' mouth for at least 1 min with the help of a metal rod as previously described. 7,8 The sponges were placed in collection devices a and were centrifuged at 4,000 × g for 8 min. Saliva samples were kept at −20°C until analysis.
Reactives and equipment
Alpha-amylase was determined by a commercial kit b using the International Federation of Clinical Chemistry and Laboratory Medicine (IFCC) method. 10 This is a kinetic spectrophotometric assay that uses 4,6-ethylidene(G7)-p-nitrophenol(G1)-alpha-D-maltoheptaoside (ethylidene-G7PNP) as a substrate of the enzyme. The intermediate product of substrate hydrolysis reacts with alpha-glucosidase, giving p-nitrophenol as the final product of the reaction. The rate of p-nitrophenol formation is directly proportional to the alpha-amylase activity of the sample and can be determined by measuring the absorbance at 405 nm. The volume of the sample was modified to optimize the method to porcine saliva. Reagent volumes were adjusted following the manufacturer's indications. The assay was adapted to an automatic analyzer. c
Cortisol was analyzed with an immunoassay system d that was validated in the authors' laboratory for cortisol measurements in porcine saliva.
Validation of the analytical method
For the validation of the method, previously described protocols were used, 23,24 and the following parameters were evaluated: 1) precision, 2) linearity under dilution, and 3) detection limit. For precision, 4 pools of saliva, 2 with high and 2 with low alpha-amylase activity were used for the precision studies. Intra-assay precision was calculated by measuring 10 times, in the same analytical series, each of the pools selected. Interassay precision was calculated by measuring each of the pools once a day for 5 different days. The samples were stored in separate vials (aliquots) to avoid the possible effect of repetitive thawing and freezing.
For linearity under dilution, 2 samples with different alpha-amylase activities were analyzed in duplicate to calculate this parameter. The samples were diluted in saline solution containing 75%, 50%, 25%, and 12.5% of the initial samples. The average of the 2 initial measurements was used to calculate the expected value for each dilution.
The detection limit was defined as the lowest concentration of analyte that could be distinguished from a specimen of zero value. It was calculated based on data from 10 replicate determinations of the zero standard (saline solution) as mean value plus 2 standard deviations.
Stress induction
Experimental pigs were stressed for at least 1 min with a nasal snare or loop following the procedure described in the literature. 5,19 As noted by previous studies, 5,21 such a technique of temporarily restraining pigs is commonly used in veterinary practice and represents an acute stressor.
Intra-and interassay coefficients of variation (CVs) obtained for saliva pools with high and low alpha-amylase activity.*
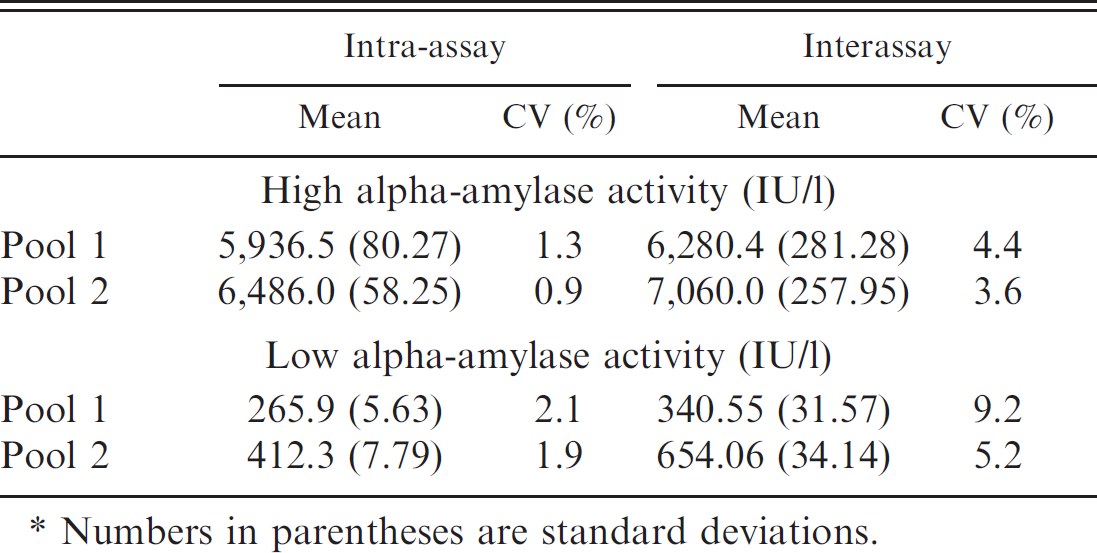
Numbers in parentheses are standard deviations.
Four salivary samples were taken from each animal. The first sample was taken before the immobilization of animals (T − 1), and the remaining samples were taken at 0, 30, and 60 min after restraint stress (T0, T30, and T60, respectively). Samples were brought to the laboratory, and alpha-amylase and cortisol were analyzed as described above. The research protocol used in the current study was approved by the Bioethical Commission of Murcia University according to the European Council Directives regarding the protection of animals used for experimental purposes.
Statistical analysis
For the validation study, arithmetic means, medians, and coefficients of variation (CVs) were calculated by routine descriptive statistical procedures. Linearity under dilution was accomplished by ordinary linear regression analysis comparing the measured levels of alpha-amylase activity with the expected levels. For the stress-induction study, the Kolmogorov–Smirnov test was performed to assess the normality of data, giving a nonparametric distribution. Data were then log transformed, and 1-way analysis of variance of repeated measures and Tukey posttest were used for statistical processing. Pearson correlation coefficient was calculated between salivary alpha-amylase activity and cortisol concentration. All statistical analyses were performed using a statistical package e and a spreadsheet. f The significance level was set at P < 0.05.
Results
Validation of the analytical method
Results of the precision study are shown in Table 1. The intra-assay CVs were <1.4% for the pools containing high alpha-amylase activity and <2.2% for pools with low enzyme activity. The interassay CVs were <4.5% for the pools containing high alpha-amylase activity and <9.3% for pools with low enzyme activity. The linear regression analysis performed provided a correlation coefficient >0.998 in pools with high and low levels of alpha-amylase activity (Figs. 1, 2). The analytical limit of detection was 11.65 IU/l.
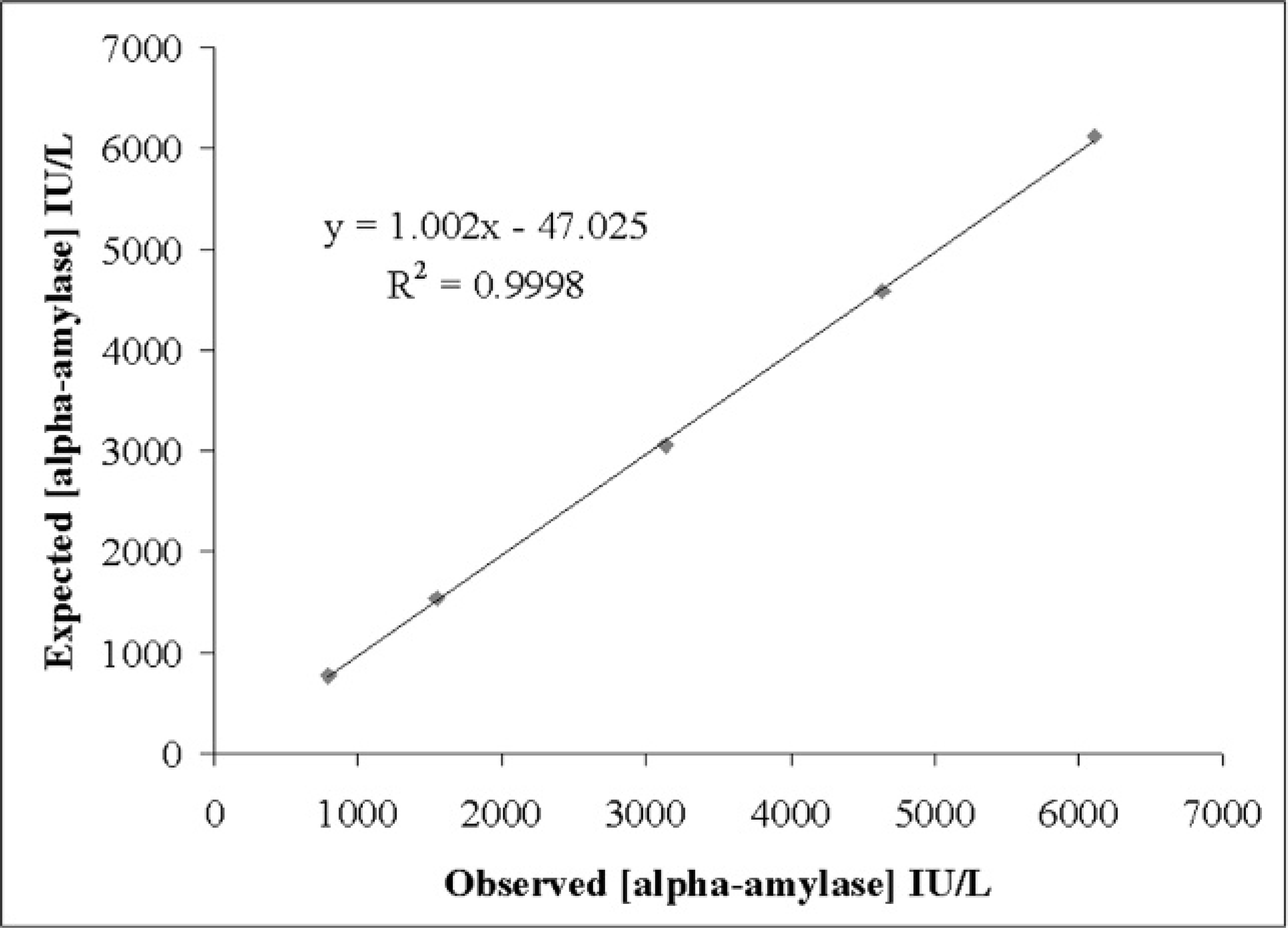
Linearity under dilution of a porcine saliva pool with high alpha-amylase activity.
Results of restraint stress
The levels of alpha-amylase activity obtained during the stress induction assay are shown in Figure 3. Statistical analysis showed that the salivary alpha-amylase activity increased significantly between the values of time T − 1 and T30 (P < 0.05) and between the values of time T − 1 and T60 (P < 0.05). The concentrations of cortisol in the pigs' saliva obtained during the stress induction assay are shown in Figure 4. A statistically significant increase of salivary cortisol between the values of time T − 1 and T0 (P < 0.001), between T − 1 and T60 (P < 0.05), and between T0 and T30 (P < 0.01) were observed. No significant correlation was observed between salivary alpha-amylase activity and cortisol concentration (Pearson correlation coefficient = –0.097, P = 0.597).
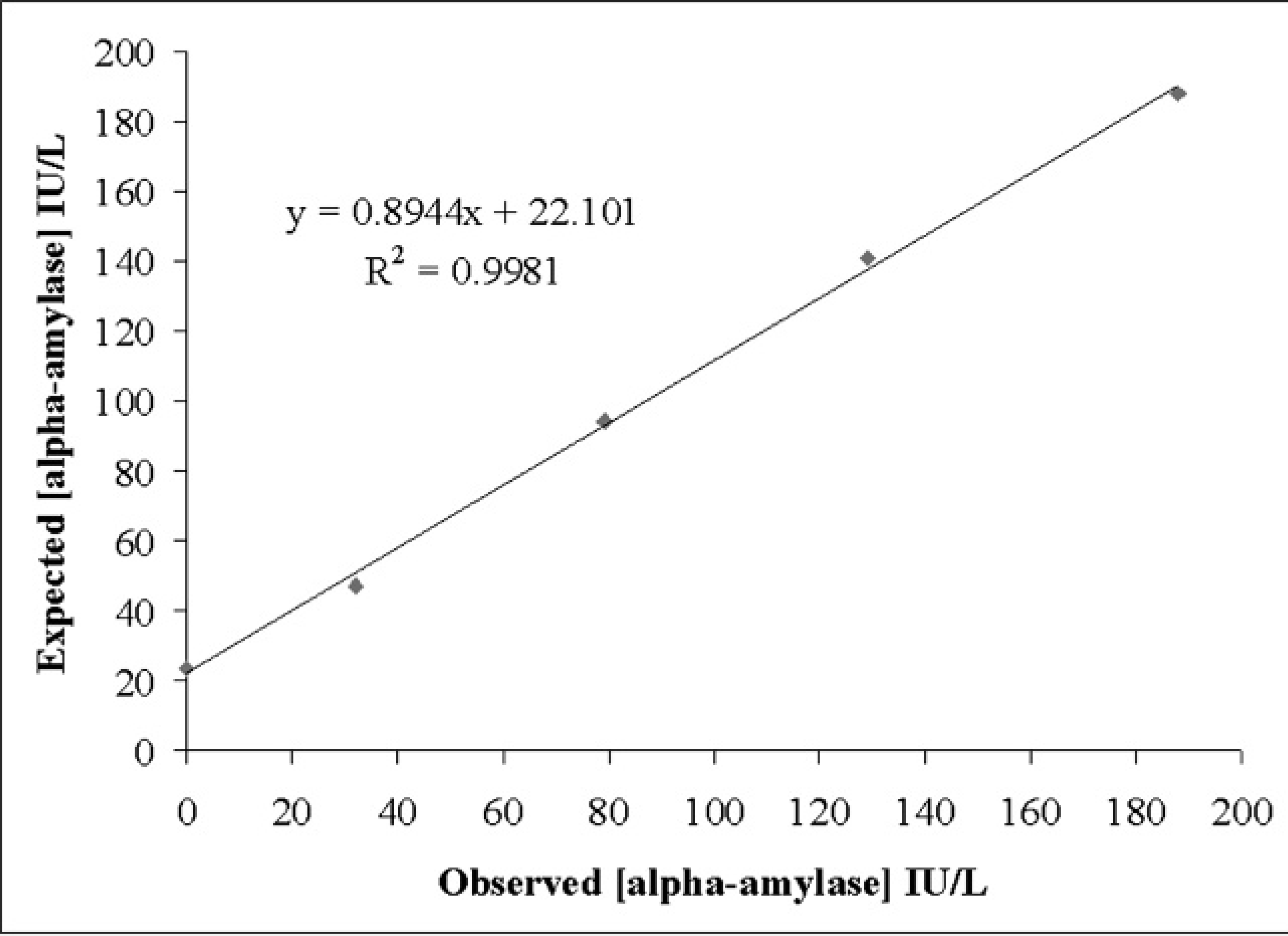
Linearity under dilution of a porcine saliva pool with low alpha-amylase activity.
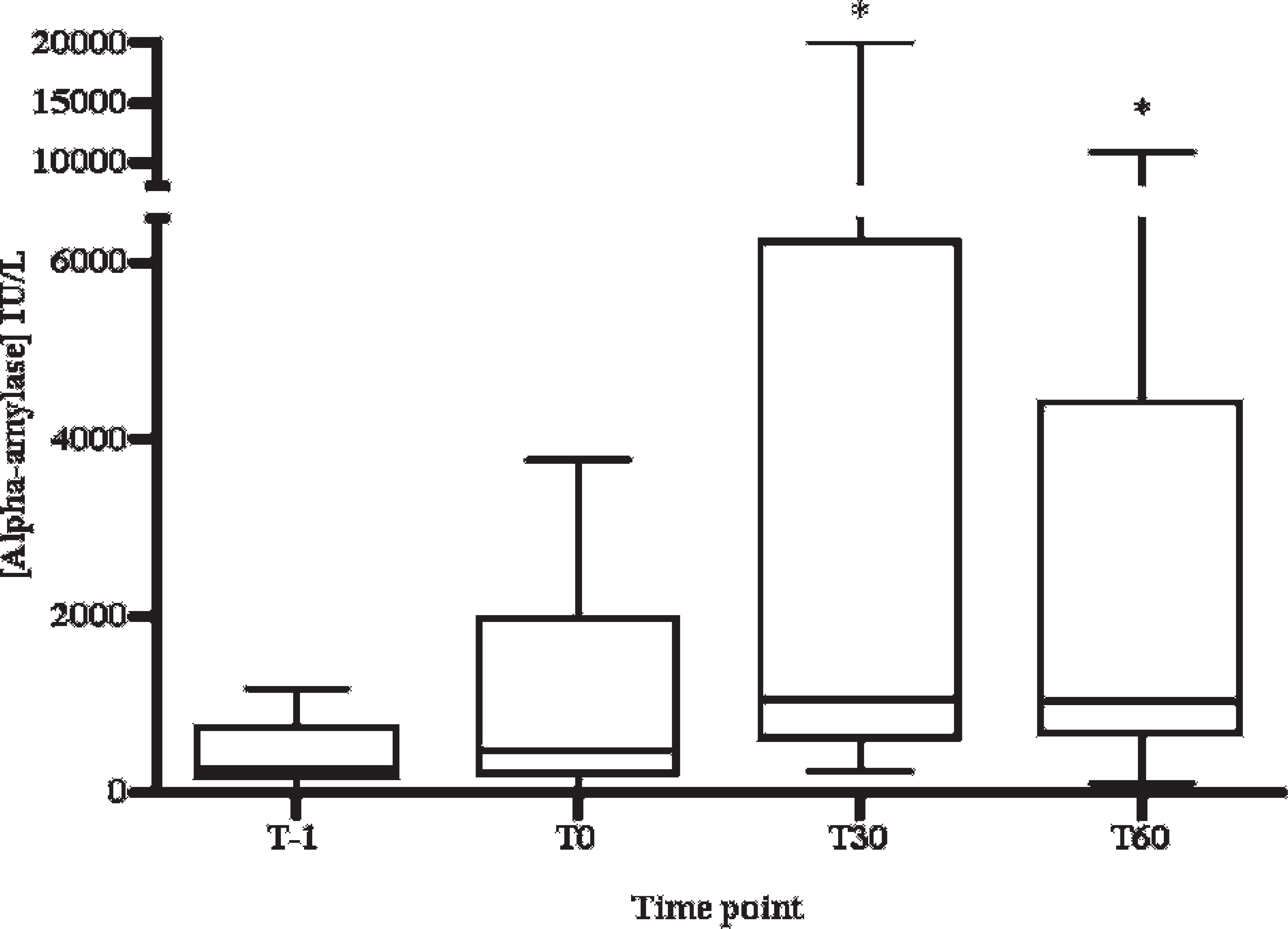
Salivary alpha-amylase activity in restraint stress–induced pigs by nasal snare. The first sample was taken before the immobilization of animals (T − 1), and the remaining samples were taken at 0, 30, and 60 min after restraint stress (T0, T30, and T60, respectively). Horizontal lines indicate median values for each time point. Error bars indicate 2.5 and 97.5 percentiles. * = P < 0.05 with T − 1.
Discussion
Saliva could be considered as an ideal sample for analysis of stress markers in animals because it is welfare friendly (it is noninvasive and produces minimal stress in the animal) and sampling is simple, 7 inexpensive, 27 and repeatable. 16 Stress is a multifactorial phenomenon that requires a multidimensional measurement approach. 15 The study and applications in farm animals of new stress markers, such as salivary alpha-amylase, could be of great interest as a better evaluation of stress and assessment of animal welfare. In the current study, an automated method originally used for human beings for measurements of alpha-amylase activity was adapted and validated in pig saliva samples, and a preliminary evaluation for the potential use of alpha-amylase activity as a marker of stress in pigs was made.
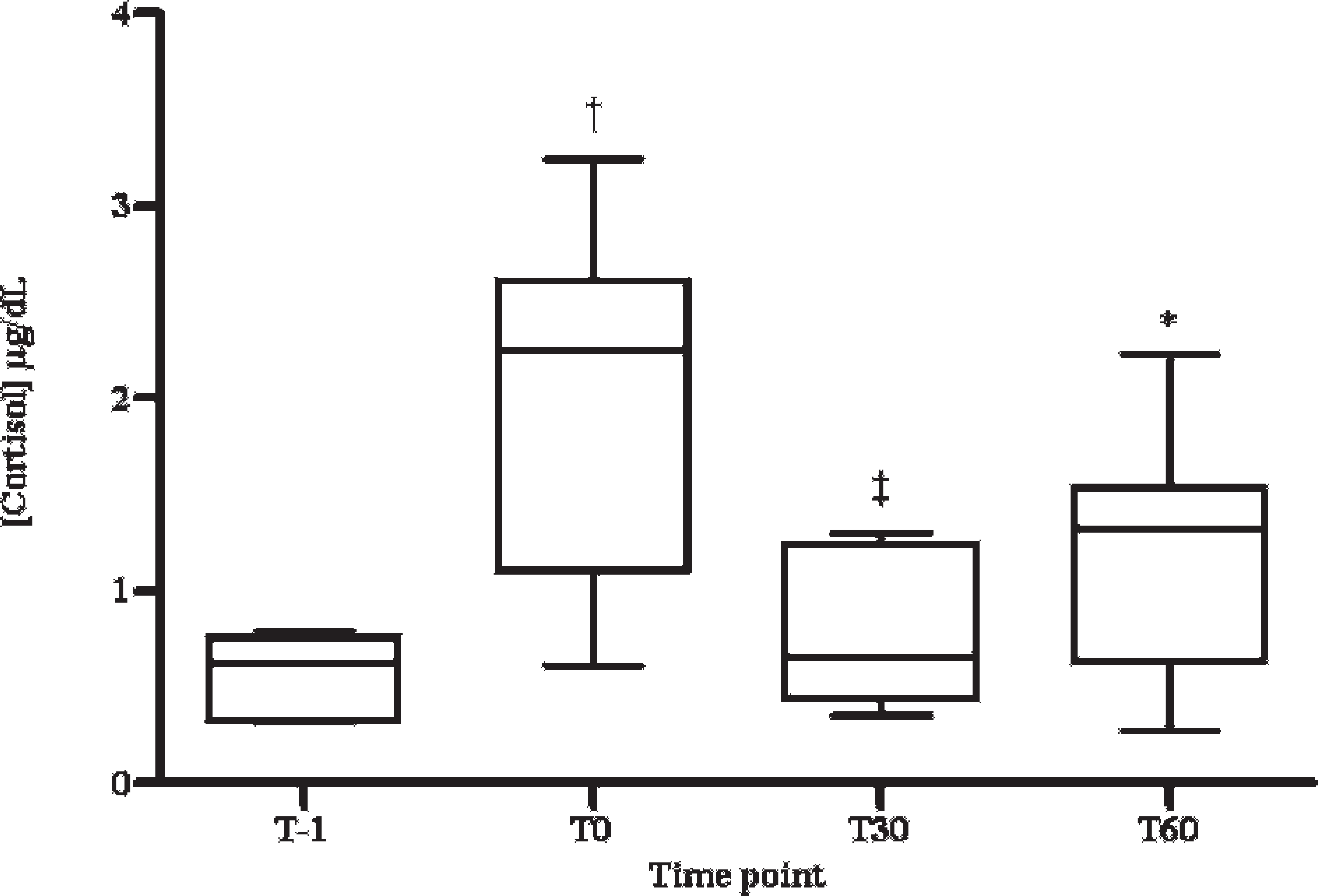
Salivary cortisol concentration in restraint stress–induced pigs by nasal snare. The first sample was taken before the immobilization of animals (T − 1), and the remaining samples were taken at 0, 30, and 60 min after restraint stress (T0, T30, and T60, respectively). Horizontal lines indicate median values for each time point. Error bars indicate 2.5 and 97.5 percentiles. * = P < 0.05 with T − 1; † = P < 0.001 with T − 1; ‡ = P < 0.05 with T0.
The precision study of this method showed intra-assay CVs for pools with high and low alpha-amylase activity of <1.4% and <2.2%, respectively; and interassay CVs for pools with high and low enzyme activity of <4.5% and <9.2%, respectively. The validation results indicated that the method was acceptable for alpha-amylase measurements given that it is generally accepted that imprecision should be lower than 10% for analytical determinations. 23 Although no studies of validation of methods for alpha-amylase in porcine species have been reported in the veterinary literature, the results obtained in the present study were similar to a validated method for human saliva in which intra-assay CVs for pools with high or low alpha-amylase activity were <2.5% and <7.2%, respectively; and the interassay CVs for pools with high or low enzyme activity were <3.6% and <5.8%, respectively. g
Since to the authors' knowledge no reference material or standards for porcine alpha-amylase are commercially available, accuracy was indirectly estimated in the study by linearity assays. The high linear regression coefficients obtained for the results of serially diluted samples showed that this method measures alpha-amylase activity in a linear manner. The detection limit obtained for the method was low enough to detect alpha-amylase activity in saliva from all the animals used in the current study. However, despite its low limit of detection, the method appears to underestimate the low activity samples (i.e., linearity results showed that with values lower than 150 UI/L, an underestimation of 10–20 IU/L occurred).
An increase in alpha-amylase levels was found when our model of stress was applied. However, a high variability in responses of alpha-amylase to stress among animals was observed. For instance, between T − 1 and T30 there was no or little increase (of less than 2-fold) of alpha-amylase in 4 animals; the number of animals with increases between 2- and 20-fold was 6, and the number of animals in which it was more than 20-fold was 2. It happened similarly for the interval between T − 1 and T60 (Table 2). In human beings, a very wide range in amylase concentrations in resting saliva as well as amylase response after a stress has been reported among different individuals, and even in some cases salivary amylase did not increase after a stress application. 22 This variability of alpha-amylase values and responses to stress in different animals could be explained on the basis of the complexity of the stress reaction 4 as well as the idiosyncrasies of the individual (i.e., genetic factors or prior experience) that could influence the manner that each animal could respond in a different form to the same stressor stimulus. 20 This variability also occurs in the case of other analytes, like cortisol, used to assess stress in the saliva of pigs. 9,11
Variability in responses of salivary alpha-amylase activity in pigs after stressor stimulus.*

T = time. The first sample was taken before the immobilization of animals (T − 1), and the remaining samples were taken at 0, 30, and 60 min after restraint stress (T0, T30, and T60, respectively).
In the animals that had more than a 2-fold increase of alpha-amylase after the stress induction, the underestimation of the method would not have a major influence on the results. However, in the future, more sensitive methods should be developed in order to detect low alpha-amylase concentration in porcine saliva samples, especially in cases of minor increases in alpha-amylase that could occur in some stressful conditions in particular individuals.
Similar to the results of other studies, a correlation between salivary alpha-amylase and salivary cortisol was not found in the current study. 15,26 This result could be explained by the different mechanisms of release of both biomarkers. 11,15,26 The different kinetics of response, the diverse information provided about the mechanism of stress, as well as the variability of the response of the different analytes support the idea that evaluating a panel of stress-responsive analytes (such as cortisol and alpha-amylase) would be more valuable than evaluating a single analyte in saliva.
It would have been desirable to measure the levels of epinephrine and norepinephrine in order to evaluate the SAM system and to compare the behavior of catecholamines and alpha-amylase in the model of stress. However, salivary cathecholamine concentrations are several folds lower than those of venous blood and do not reflect acute changes in blood catecholamine concentrations, so it could be hypothesized that salivary cathecholamine levels are a poor index of the changes in sympathetic activity. 22 In addition, it would be necessary to concurrently conduct blood sampling for measurements of these analytes. Such a procedure could elicit spurious increases in stress markers due to the distress and concomitant pain caused to the animal and could be a confounding effect in the stress model proposed. 17,22
The current study described an automated method for the measurement of alpha-amylase in saliva of pigs that is simple, robust, and allows a high throughput of samples, which is very convenient when a high number of animals has to be sampled, such as in population studies at the farm level. However, the underestimated low levels of such enzyme could be considered a limitation of the method and, therefore, other assays able to detect low values should be developed in the future. In addition, it was observed that alpha-amylase increases after a model of stress induction in pigs. Although the increase of alpha-amylase in stress conditions is well documented in human beings, 14,18,25,28 many factors could influence amylase production such as the intake of food, exercise, or circadian rhythm 17 ; therefore, the current study should be considered as preliminary. Further evaluation of the factors that could have influenced alpha-amylase in pigs, such as feeding, circadian rhythm, or hydration status, should be performed as well as additional studies to further elucidate the mechanisms that can influence the activity of salivary alpha-amylase and to gain knowledge about the behavior of this enzyme before its use is recommended as a marker of stress.
Acknowledgements
This study was supported by the Science and Innovation Ministry of Spain (project no. AGL2009–08509) and Seneca Foundation, Science and Technology Agency of the Region of Murcia (Spain).
Footnotes
a.
Salivette®, Sarstedt AG & Co., Nümbrecht, Germany.
b.
α-Amylase, Beckman Coulter Inc., Fullerton, CA.
c.
Cobas Mira Plus, ABX Diagnostica, Montpellier, France.
d.
Siemens Health Diagnostics, Deerfield, IL.
e.
Version 5.0, GraphPad Software Inc., La Jolla, CA.
f.
Excel®, Microsoft Corp., Redmond, WA.
g.
Salivary α-Amylase Assay Kit, Salimetrics LLC, State College, PA.
