Abstract
Rickettsial organisms are well-known fish pathogens in both natural and culture environments. This study reports an outbreak of disease in red tilapia larvae caused by piscirickettsia-like organisms (PLOs), which lasted from June until October 2009. Severe mortality was recorded almost exclusively in larvae and postlarvae aged 1–22 days old. Although clinical or gross findings were not evident in diseased fish, histopathology revealed severe necrosis of the epidermis and gill epithelium, with concomitant changes in the underlying skeletal muscle as being the most relevant microscopic lesions. Although PLOs were visible with the routine hematoxylin eosin technique, they were better observed with Giemsa and toluidine blue stains. Transmission electron microscopy revealed that the bacterium was located within the cytoplasm and phagolysosoma-like structures of epithelial cells from the gills and the skin. The bacteria measured 0.9 ± 0.2 μm × 2.1 ± 0.6 mm and had a double cell membrane (the outer one having undulating projections), with variable electron-dense and electron-lucent areas. Ultrastructurally, abundant myelin figures surrounded the microorganisms within host cell cytoplasm. Results indicated that Piscirickettsia-like organisms can cause massive epithelial cell damage associated with concomitant alteration of the electrolyte balance.
The genus Rickettsia are Gram-negative, obligate, intracellular bacteria, composed of multiple pathogenic species having wide host range and geographic distribution. 20 Indeed, rickettsial microorganisms have controversial taxonomy due to the absence of universal consensus regarding the proper criteria needed for designating the species. Rickettsia cause severe and fatal disease in a wide range of animals including fish and humans, although there is no indication that the rickettsial species affecting fish can cause disease in humans or other warm-blooded animals. 12 The Rickettsia life cycle in mammals involves an arthropod as a vector, 20 whereas the disease in fish (named piscirickettsiosis) is most apparent and spread rapidly by fish-to-fish contact throughout high-density population environments. 19 However, some ectoparasites, such as Caligus spp., might be associated with transmitting the bacteria among fish. 21
The first member of the fish rickettsial pathogens was isolated from Pacific salmon in Chile and identified as Piscirickettsia salmonis (Cvitanich JD, Garate NO, Smith CE: 1990, Etiological agent in a Chilean Coho disease isolated and confirmed by Koch's postulates. AFS Fish Health Newsletter 18:1–2), 1,2,11 which shares a close phylogenetic position with Francisella tularensis and Wolbachia persica. 12 Since the initial study was carried out additional reports have documented piscirickettsial organisms preferentially occurring in salmonids. 18 However, other species, such as Epinephelus melanostigma, 4 the ornamental blue-eyed plecostomus fish (Panaque suttoni) from Colombia, 14 the juvenile sea bass (Dicentrarchus labrax), 7 and the white weakfish (Atractoscion nobilis) 5 have also been affected. Piscirickettsia-like organisms (PLOs) have now become well-established pathogens in different fish species worldwide. 4,13,14 Outbreaks of disease caused by PLOs have been described in tilapia in Taiwan, Hawaii, and the continental United States where heavy economic losses have been recorded. 3,6,16,17
Piscirickettsiosis is characterized by systemic distribution of PLO-associated necrotic lesions. Gross lesions in different fish species, including tilapia, have preferentially been described as either necrosis or inflammation of the gills, liver, spleen, intestine, and hematopoietic tissue of the kidney, or as nodular lesions in gills, kidney, spleen, and liver. 6,8,16–18 Less frequently, petechia, frayed fins, and exophthalmia have been present. Microscopically, necrosis is extensive, and the nodules correspond to necrogranulomatous inflammation, also affecting skeletal muscle, subcutis, bone, testes, epicardium, choroid gland, and around the spinal cord. 6,16–18
The current study reports an outbreak of rickettsiosis in red tilapia (Oreochromis spp.), which caused heavy mortality in about 50% of the fish at a freshwater fish farm in southern Colombia from June to October 2009. Although the disease almost exclusively affected red tilapia larvae aged 1–22 days old, other fish at different developmental stages remained clinically healthy. The larvae on this fish farm come from naturally fertilized eggs, which are extracted from the mouths of female fish and taken to an artificial rearing system consisting of plastic trays with a closed water circulation system; this provides the trays with a high water exchange rate with minimum turbulence. The larvae hatch in the trays and stay there for 4–6 days until the yolk sac has become absorbed. Postlarvae are then transferred to net enclosures in earthen ponds where they stay there for 20–30 days and are fed a balanced diet (45% protein), supplemented with hormone 17-α-methyl testosterone for sexual reversion. Water for the rearing system is pumped from a deep well to a tank having layers of granular filtering material at the bottom. The water then goes to a reservoir where it is heated to an optimum temperature (26–27°C) to be distributed to the trays. At the time of the outbreak, the water temperature was at the indicated degrees and contained 5–6 mg/l of oxygen and a pH of 7–7.5. Water for the earthen ponds comes from a small river having similar characteristics.
No clinical signs or gross changes were observed. Tissue samples were submitted in neutral buffered 10% formalin, embedded in paraffin wax, sectioned at 4 μm, and stained with hematoxylin and eosin, Giemsa, Gram, and acid-fast (Ziehl–Nielsen). Slides were observed by light microscopy. Samples for transmission electron microscopy (TEM) were fixed in 2.5% phosphate glutaraldehyde, postfixed in 1% osmium tetroxide, contrasted with uranyl acetate and lead citrate, and embedded in Epon 812 resin. Semithin sections were also prepared and stained with toluidine blue. Samples for microbiology were cultured in BHI -blood agar at 27–28°C.
Microscopically, only the skin and the gills were severely affected. The lesions consisted of severe vacuolation of the cytoplasm of the epidermis and the epithelial cells of the gill; the vacuole merged in the epidermis with resulting formation of vesiclelike structures, necrosis, and detachment of large portions of the epidermal layer, leaving an erosive surface (Fig. 1A, B). Epidermal damage was associated with discoloration of the interstitial substance between the skeletal muscle fibers, suggesting water infiltration within the muscle fibers (Fig. 1B). Giemsa (Fig. 1A, B) and toluidine blue (Fig. 1C, D) staining revealed the presence of different-sized pleomorphic coccobacilli within the epithelial cells of both the gills and skin; the microorganism occupied the cell cytoplasm, giving an appearance of morula. Epidermal cell vacuolation and destruction were more evident in semithin sections stained with toluidine blue and the microorganisms were seen to be freed onto the surface of the fish. Similar lesions were found in gill epithelium (data not shown). Gram and Ziehl–Nielsen stains were negative. Findings indicated that the particular tissue distribution and lesions observed were caused by fatal infection of tilapia larvae with PLOs. The characteristic massive epithelial cell damage, likely associated with concurrent electrolyte imbalance, played a major role in the death of these fish.
Ultrastructurally, the cytoplasm was almost completely occupied by the bacteria in heavily affected epidermal and gill cells to such an extent that no normal cytoplasmic organelles were still visible (Fig. 2A, B). It was extremely difficult to find normal organelles; most of them were destroyed, leaving behind only abundant amounts of their lipid membranes, which adopted different sized myelinlike structure (Fig. 2B). On occasion, these structures surrounded the microorganism or became arranged in a flagellalike manner at 1 or 2 poles of the pathogens (Fig. 2C–E). The nuclei of the host cells were pyknotic or karyorrhectic and became eccentrically displaced to the periphery against the cytoplasmic membrane (Fig. 2A, B). The coccobacilli were pleomorphic, measuring 0.9 ± 0.2 μm × 2.1 ± 0.6 μm. Bacteria showed a variable degree of electron density and most were free within the epithelial cell cytoplasm revealing a high proliferative activity (Fig. 2B, C, E). Occasionally, they were also found within cytoplasmic vacuoles where they were destroyed in a similar manner as by phagolysosomes (data not shown). A double cell wall surrounded the microorganisms and undulating projections were exhibited at some points in the outer one (Fig. 2C, D).
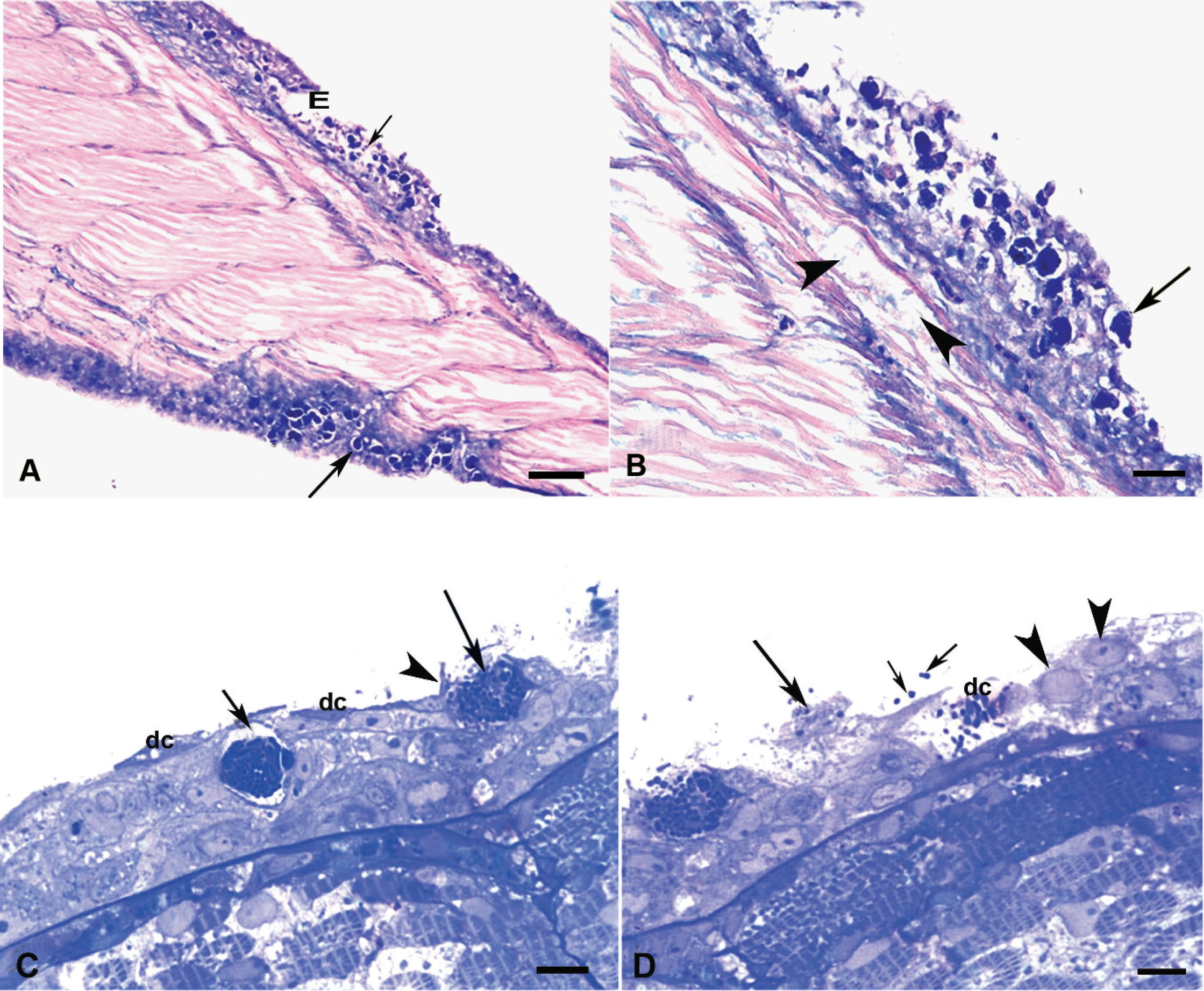
Epidermis of post-larvae tilapia peduncle.
No bacterial growth was obtained in common bacteriological media. This prompted a presumptive diagnosis based on histopathological and TEM analysis, and immediate therapy with oxytetracycline as 99% active ingredient (2.5 g/kg of food per day for 10 days) was thereby established. This was followed by consistent reduction of mortality in postlarvae fish. However, this also precluded any attempts at isolating the microorganism in conventional cell lines. The rearing system was disinfected with quaternary ammonium (0.5 g/l) for 15 min and sodium hypochlorite (1 g/l) for 5 hr.
The current study describes a PLO causing up to 50% mortality in red tilapia larvae. Mortality rates in tilapia eventually exceeding 60% have been reported in other studies. 16 However, in sharp contrast to previous reports, significant differences can be pointed out in the current study. The disease reported in tilapia from the continental United States and Hawaii has predominantly involved juvenile fish and, although fish age and size were not explicitly given, fish size was suitable for performing necropsy. The disease almost exclusively affected the youngest animals (larvae and postlarvae) having less than 1–2-cm length in this study, meaning that necropsy examination was not possible and the fish had to be immediately immersed in fixatives.
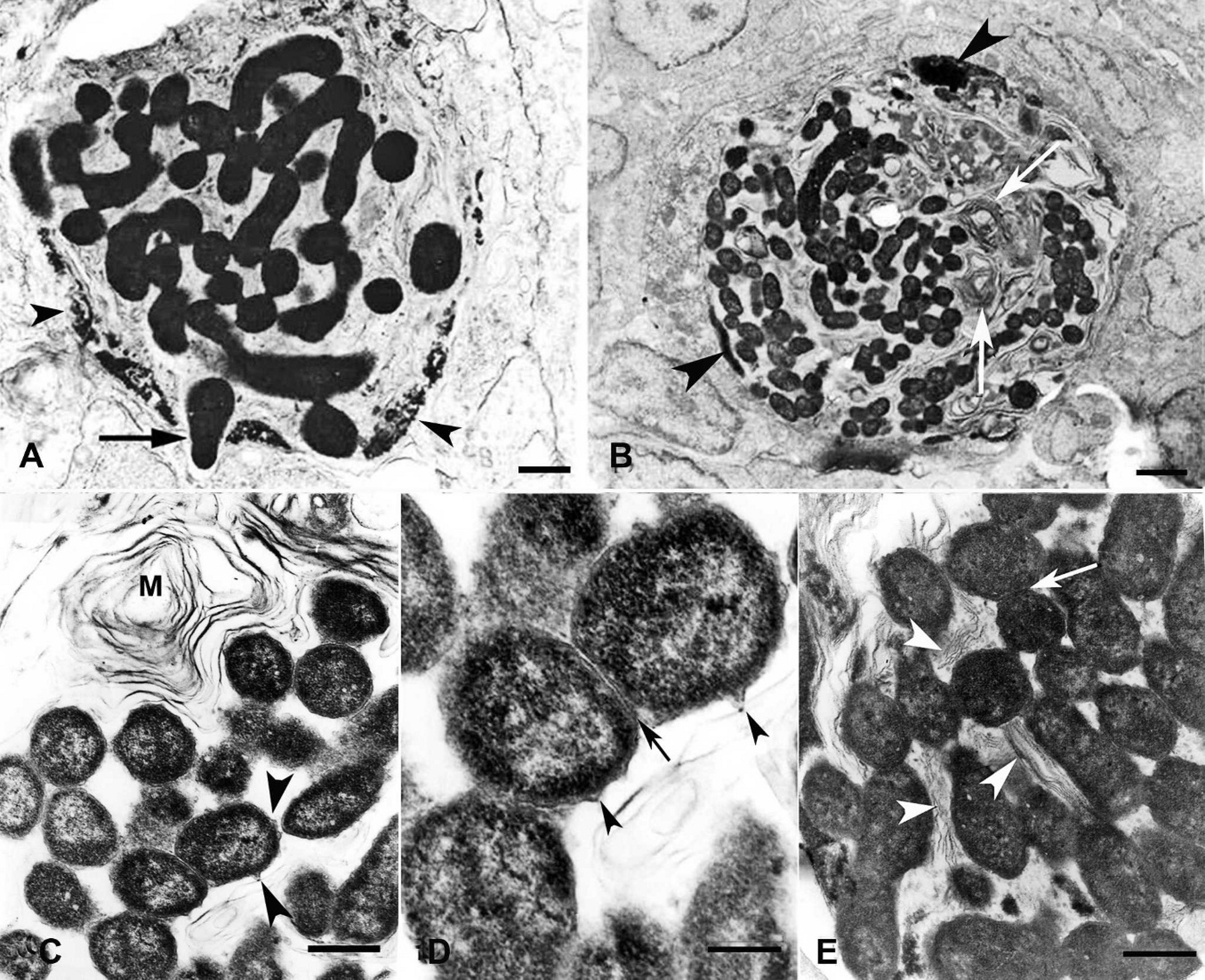
Piscirickettsia-like organisms (PLOs) within host cells in the skin of post-larvae tilapia.
No clinical signs or gross changes were noted in the current study, partly because of fish size, but also because of the acute course of the disease. Although gross changes in the skin have been appreciated in previous work on tilapia, no description of such changes has been documented microscopically. In the present work, the most seriously affected tissues were the gills and skin, acute necrotic lesions being observed, which likely compromised the physiological electrolytic balance and supported the acute course of the infection, thereby leading to high fish mortality. The lesions described were in sharp contrast to the multifocal granulomas in internal organs, at the base of the gills and the gill filaments, previously documented in tilapia. 16,17 The skeletal muscle became affected after the development of lesions in the epidermis, manifesting characteristics of water infiltration. 10 Epidermal lesions have been described as being hyperplasic in the white weakfish (A. nobilis) 5 and hemorrhagic or ulcerative type in Coho salmon (Oncorhynchus kisutch). 2 However, such lesions were not appreciated in tilapia larvae in the present study. The acute course of the outbreak may have hampered the development of more chronic lesions, which have been documented by other investigators. 12,15
The coccobacilli complied with most characteristics described for PLOs in tilapia and other fish species in TEM (i.e., their free location in host cell cytoplasm and within phagolysosome-like structures [not shown], showing variable electron-dense and electron-lucent areas, and exhibiting a double cell wall and an undulating outer membrane). However, there were three main differences from previous reports. 16,17 The microorganisms in this study were not as pleomorphic. They were substantially larger (0.9 ± 0.2 μm × 2.1 ± 0.6 μm) than PLOs previously reported from tilapia (0.56 ± 0.14 μm × 0.7 ± 0.20 μm in 2003 16 and 0.57 ± 0.1 μm × 0.8 ± 0.2 μm in 2005 17 ), and they were associated with intense myelin figures in host cell cytoplasm (which has not been documented previously). Whether such morphological differences are compatible with a novel species of rickettsia is something that cannot be confirmed at present, and requires future research.
Although it is not clear whether the PLOs causing the documented outbreak of disease in red tilapia were the same as those described in other outbreaks in the same or different fish species, 2 major differences from previous reports were documented in this study: clinical and gross changes were not observed in the affected animals and the nongranulomatous nature of the lesions has not been previously documented. Three main differences were described by TEM, namely microorganism morphology and size and the associated structures surrounding the pathogen.
A severe disease affecting red tilapia larvae in southern Colombia has thus been documented. The disease was caused by a microorganism having an intracellular location on gill and skin epithelial cells leading to the development of severe epithelial cell damage and subsequent electrolyte imbalance. 10 The microorganism's morphologic characteristics and the lesions induced in these tissues indicated a PLO having some physical and morphological differences from those reported previously. These findings should be taken with care, as isolating the microorganism in tissue culture was not attempted because of the immediate antibiotic treatment. Further characterization of these bacteria was thus not possible, and it is not clear whether this pathogen was the same organism associated with mortalities in farmed tilapia or even in other fish species.
Acknowledgements. This report was supported by a grant from the Colombian Ministry of Agriculture and Rural Development (MADR). The authors are grateful to Mr. Gilberto Cordoba (histotechnician) from the Universidad Nacional de Colombia's Pathology Laboratory and Dr. Paula Esquinas for TEM work.
