Abstract
A 5-year-old female spayed Labrador Retriever dog was referred to the Louisiana State University Veterinary Teaching Hospital for treatment of pneumothorax. Thoracic radiographs and computed tomography showed spontaneous pneumothorax, thoracic lymphadenopathy, pulmonary hypertension, and multifocal pulmonary bulla lesions. At surgery, numerous adult nematodes protruded from the parenchyma of the left caudal and accessory lung lobes and pulmonary arteries. On histopathology, multiple adult filarid nematodes were observed within the pulmonary blood vessels. Broad foci of necrosis of the pulmonary parenchyma were present. The tunica intima of the pulmonary arteries was markedly thickened by intimal fibrosis and medial hypertrophy. The final diagnosis was severe Dirofilaria immitis infection that resulted in pulmonary vascular lesions and focally extensive infarcts of the pulmonary parenchyma with bulla formation and rupture causing spontaneous pneumothorax. The dog received antibiotic and steroid therapy, as well as adulticide treatment, and recovered.
Spontaneous pneumothorax is an accumulation of air in the pleural space, resulting in closed pneumothorax, without iatrogenic or traumatic causes. 1 Traumatic pneumothorax is common in dogs, whereas spontaneous pneumothorax is relatively rare. 9,12 Reported causes include pulmonary blebs or bullous emphysema, lobar emphysema, bacterial pneumonia, pulmonary abscesses, dirofilariasis, paragonimus infection, and neoplasia. 3,5,7,10,12,13 The primary cause of spontaneous pneumothorax in dogs and human beings is rupture of subpleural bullae or blebs. 1,7,12 Bullae are large cystic air spaces within the pulmonary parenchyma radiographically characterized by a barely perceptible rim or no rim around the hyperlucent lesion. 1,6,14,15 Bullae result from destruction, dilation, and confluence of adjacent alveoli, but the pathogenesis is not completely understood. 7,14 A bleb is a smaller accumulation of air between the visceral pleura and the lung parenchyma that forms when air escapes from within the lung parenchyma and becomes trapped between the layers of the visceral pleura. 1,7 Bullae and blebs are associated with congenital bronchial abnormalities, infectious diseases, or pulmonary emphysema, and radiographic differentiation between these 2 lesions may be difficult. 15
Heartworm disease is most common in tropical and subtropical climates but occurs throughout the United States, with an incidence of 45% along the Atlantic and Gulf Coasts and the Mississippi River and its major tributaries. 2,11 To the authors' knowledge, there are only 3 previous reports in dogs of spontaneous pneumothorax secondary to Dirofilaria immitis infection. 3,5,13
A 5-year-old spayed female Labrador Retriever dog was evaluated at a private veterinary practice with a 1-week history of lethargy, vomiting, and diarrhea. A presumptive diagnosis of pneumonia was made, and despite antibiotic therapy, the lethargy persisted and the dog developed a cough 1 week later. At this time a heartworm antigen test a was performed, which was negative. Thoracic radiographs were obtained, and pneumothorax was diagnosed. The dog was not current on vaccinations, and no heartworm or flea and/or tick preventative was being given. There was no history of trauma. The patient was referred to the Louisiana State University Veterinary Teaching Hospital (Baton Rouge, Louisiana) for treatment of the pneumothorax. The radiographs from the referring veterinarian were not available for evaluation. Thoracocentesis was immediately performed, and 1 liter of air was removed from each side of the thorax. On subsequent physical examination, the dog had tachypnea, fever, and a body condition score of 2 out of 5. The dog was placed in an oxygen cage and monitored overnight. A complete blood count and chemistry panel showed thrombocytopenia (165,000/μl, reference [ref.] interval: 200,000–900,000/μl), neutrophilia (26,000/μl, ref. interval: 2,900–12,000/μl), and monocytosis (1,600/μl, ref. interval: 100–1,400/μl). The findings can all be related to the heartworm infection (chronic inflammation/infection); the thrombocytopenia could be due to decreased production related to infection and is usually seen in chronic heartworm disease cases or more commonly after adulticide therapy; the neutrophilia is also a finding in heartworm disease infection, in approximately 20–80% of cases; and the monocytosis could also be secondary to chronic inflammation and/or infection. A Baermann technique fecal examination was negative for lung nematodes. A heartworm antigen testa was strongly positive.
Lateral and ventrodorsal thoracic survey radiographs taken after thoracocentesis showed a predominantly right-sided pneumothorax. The right cranial, middle, and caudal lung lobes were retracted from the thoracic wall and appeared collapsed. The cause of the pneumothorax could not be determined radiographically. On the following day, the condition of the dog was unchanged. Thoracic radiographs were repeated showing unchanged pneumothorax (Fig. 1). The right lung lobes were retracted and decreased in volume. Round, well-circumscribed, radiolucent structure with a thin radiodense (soft-tissue opacity) wall was present in the region of the right caudal lung lobe. Two round to oval, well-circumscribed, radiodense (soft-tissue opacity) structures were present in the cranioventral mediastinum, which were interpreted as sternal and cranial mediastinal lymphadenopathy. A focally increased area of radiodensity (soft-tissue opacity) was present in the hilar region, suggesting tracheobronchial lymphadenopathy. The cardiac silhouette size and shape were unremarkable, but the pulmonary vasculature was difficult to assess due to the severely retracted lung lobes. In the nonretracted, air-filled lobes in the left thorax, the arteries were moderately enlarged.
Radiographic diagnoses of spontaneous pneumothorax, thoracic lymphadenopathy, probable pulmonary bulla in the right caudal lung lobe, and suspected pulmonary hypertension were made. Differential diagnoses included fungal infection, neoplasia (e.g., lymphoma, histiocytic sarcoma), ruptured pulmonary bulla, and abscess. Due to the previous positive heartworm test and the findings of enlarged pulmonary arteries, heartworm infection was also considered. Computed tomography was recommended to better assess the cause and extent of the disease but because the dog showed clinical improvement, the owners refused further diagnostic tests and decided to monitor the dog at home. Two days later, the dog presented with a relapse of the respiratory distress. Four liters of air were removed from the right thorax and 500 ml from the left via thoracocentesis. On physical examination, the dog was bright, alert, and responsive. Respiratory rate after thoracocentesis was normal. Repeated thoracic radiographs showed multiple thin-walled, round, radiolucent structures suspected to be pulmonary bullae in the region of the left caudal lung lobe. Pneumothorax was again present bilaterally and more accentuated on the right side. The cranial and caudal lobar arteries were moderately enlarged.
Computed tomography b of the thorax was performed with the dog under general anesthesia in sternal recumbency on the following day (Fig. 2). Images were acquired with a single-slice helical scanner in the transverse plane at 4-mm-slice thickness without contrast administration. The dog was hyperventilated prior to scanning to prevent motion artifacts. An air-dense region lacking pulmonary vessels peripheral to the retracted lung lobes was noted in the right and left hemithorax. Several round, well-circumscribed, air-dense structures with an isodense to soft-tissue rim were evident in the right cranial, right caudal, left cranial, and accessory lung lobes. Diffuse and severe arterial enlargement was present. Sternal, cranial mediastinal, and tracheobronchial lymphadenopathy were confirmed. Diagnosis of bilateral pneumothorax, multifocal bilateral pulmonary bullae, and thoracic lymphadenopathy was made. Due to the extent of the bulla lesions seen on the computed tomograph, an exploratory thoracotomy was performed. The left and right lungs were reddened. The left caudal lobe was partially resected, and the accessory lung lobe was completely resected. At the cut surface of these lung lobes, approximately 25–50 adult nematodes protruded from the parenchyma and pulmonary arteries. The resected portion of the left lobe was submitted for bacterial culture and histopathologic examination. Thoracostomy tubes were placed on both sides of the thorax. The dog recovered well from anesthesia and was discharged 5 days later with a prescription of antibiotic therapy (amoxicillin trihydrate and clavulanate potassium, c 10 mg/kg twice a day for 7 days; doxycycline, 5 mg/kg twice a day for 7 days) and nonsteroidal anti-inflammatory drug (deracoxib, d 1 mg/kg once a day for 5 days). The owners were instructed to limit the dog's physical activity for 15 days.
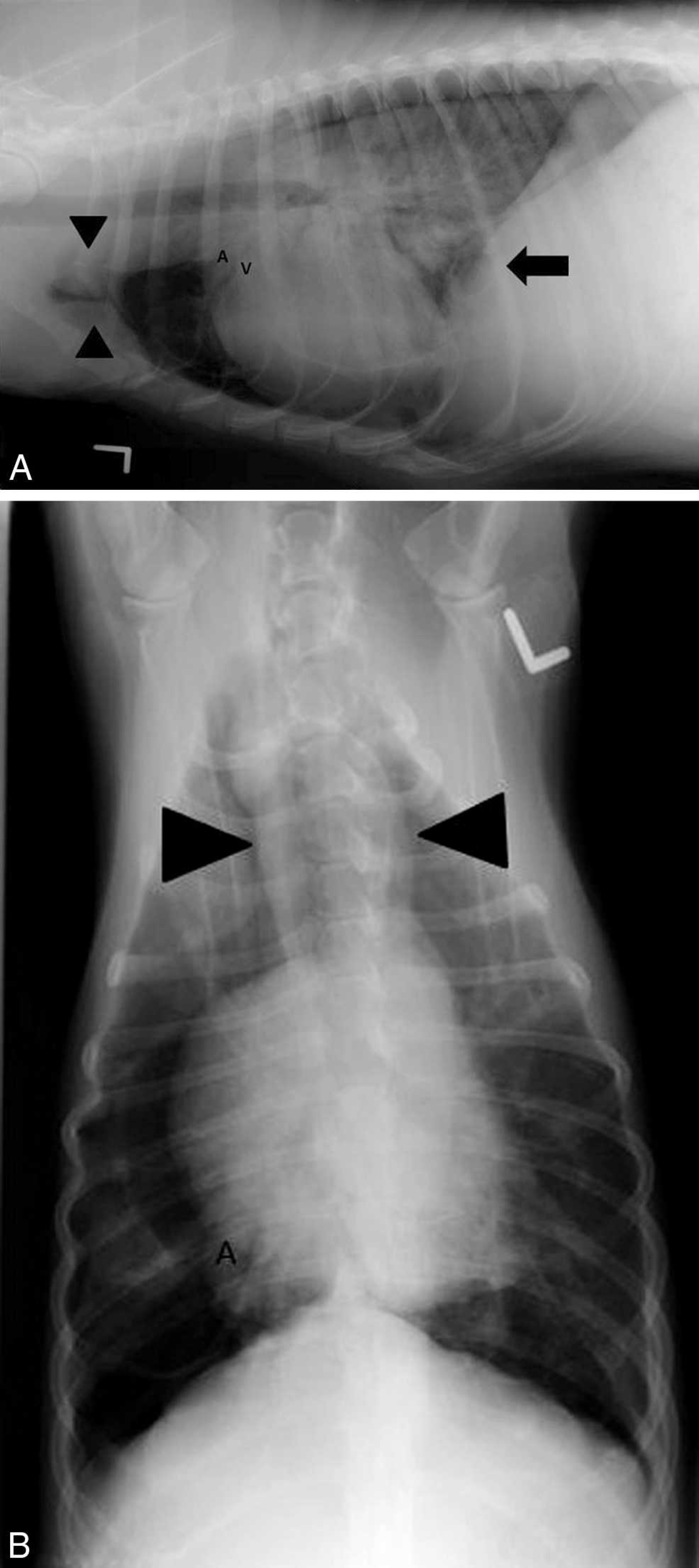
Left lateral (
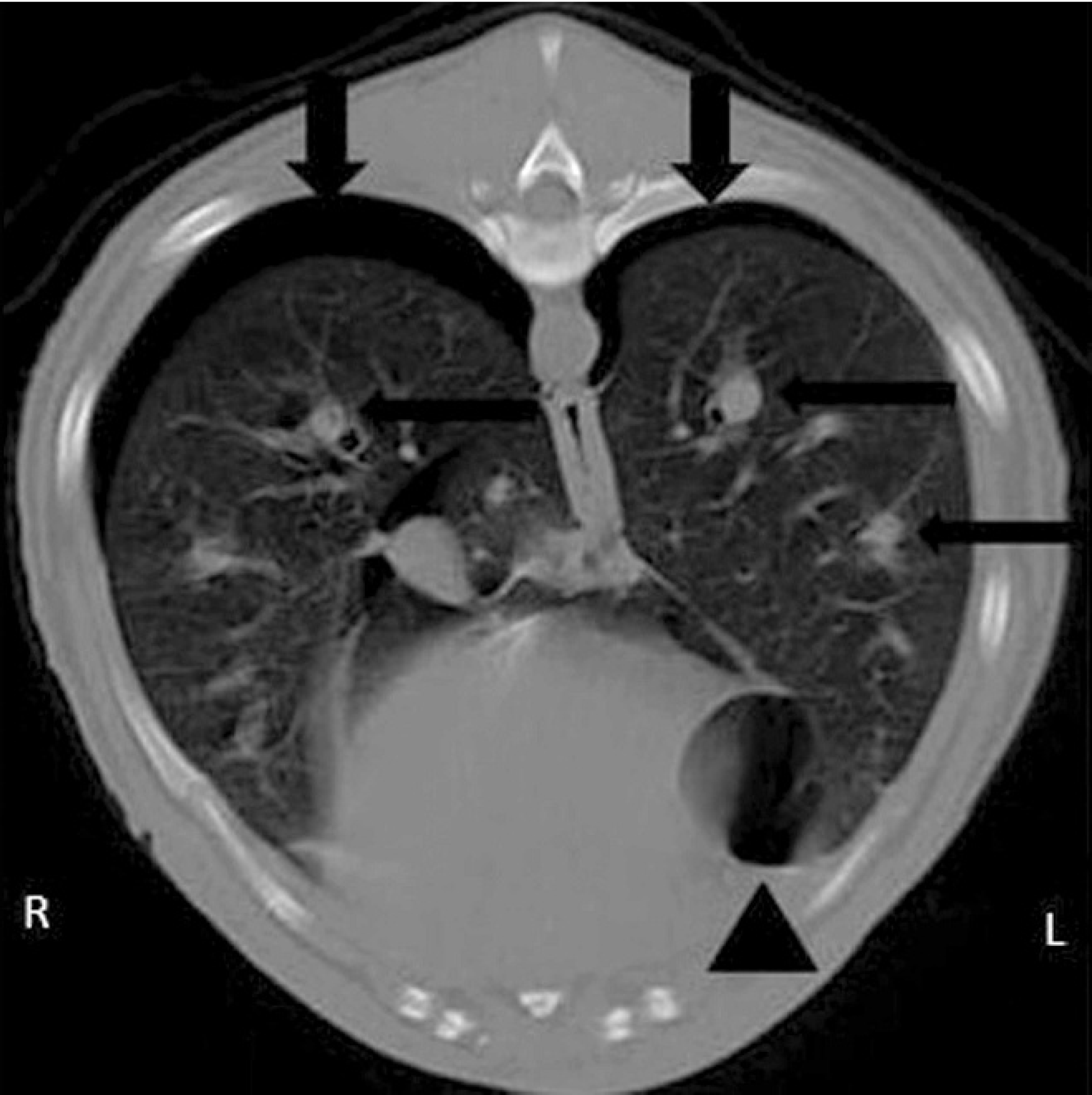
Transverse computed tomographic image in a lung window showing pneumothorax and retraction of the lung lobes in the right and left hemithorax (arrows), bullae in the medial ventral margin of the left caudal lung lobe (arrowhead), and multiple enlarged arteries (thin arrows).
Bacterial culture of the lung was negative. On histopathology, multiple adult filarid nematodes were observed within large and medium-sized arteries of the lung (Fig. 3). Broad areas of necrosis of the lung parenchyma, characterized by accumulations of eosinophilic and basophilic cellular debris mixed with degenerate inflammatory cells (predominantly neutrophils), were present. In the affected areas, the necrosis completely obliterated the pulmonary architecture, and in some areas granulation tissue was present. Throughout most sections of the lung, the alveolar architecture was obscured by marked alveolar histiocytosis, type II pneumocyte hyperplasia, atelectasis, accumulations of hemosiderin-laden macrophages, and rare eosinophils. There was marked smooth muscle hyperplasia scattered throughout the affected lung. The tunica intima of the pulmonary arteries was often irregular with intimal fibrosis and plump endothelial cells, and the walls were markedly thickened by medial muscle hypertrophy and increased fibrous tissue. Some pulmonary arteries contained cross-sections of adult nematodes, characterized by a cuticle, coelomyarian musculature, lateral chords, lateral cuticular ridges, and a digestive and reproductive tract, compatible with D. immitis. The alveolar interstitium and alveolar capillaries in many areas contained markedly increased numbers of cells, predominantly macrophages with lower numbers of lymphocytes and in some areas eosinophils. In lesser-affected areas, alveoli contained lower numbers of these cellular infiltrates and edema and/or extravasated erythrocytes.
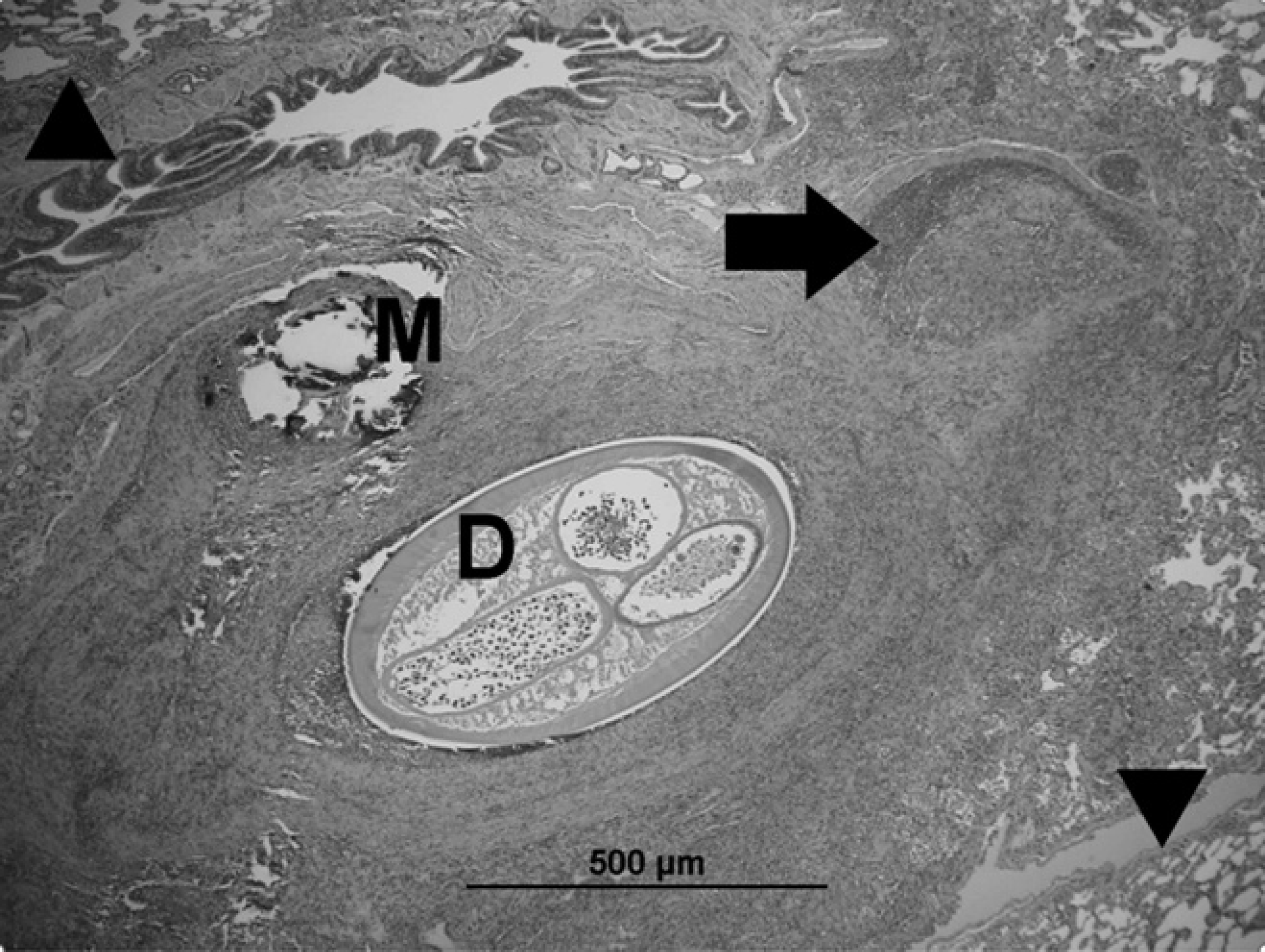
Photomicrograph of lung. Adult dirofilaria occluding a pulmonary artery (D). Abnormal vessel with portions of the tunica intima and media partially obliterated by inflammation and necrosis (arrow). Less affected areas of the lung tissue can be seen (arrowheads). Above the parasite, there is a focal area of fragmented mineralized necrotic material (M) surrounded by inflammation. The bronchiole above is compressed. Hematoxylin and eosin. Bar = 500 μm.
The final diagnosis was severe D. immitis infection that resulted in vascular lesions and broad areas of necrotic lung parenchyma with bulla formation and rupture causing spontaneous pneumothorax. Physical examination 2 weeks after discharge was within normal limits. Thoracic radiographs revealed resolution of the pneumothorax but the continued presence of a diffuse interstitial pattern and several enlarged, tortuous vessels, including the main pulmonary artery. The dog was treated with melarsomine dihydrochloride e (single dose of 65.5 mg intramuscularly) and prednisone (0.5 mg/kg twice a day for 7 days). The dog received a second dose of melarsomine 50 days later (owners did not return on the regular time of 30 days that is recommended for treatment with melarsomine). At that time, no abnormalities were found on physical examination, and the dog had reportedly been clinically healthy at home. Four months after treatment, the dog was reported by the owners to be healthy at home.
Only 3 previous reports of spontaneous pneumothorax secondary to D. immitis infection exist in dogs. 3,5,13 In one report, the emphysema was believed to be a consequence of pulmonary hypertension resulting from the severe heart-worm infection. Ischemia due to thromboembolism of the terminal branches of the pulmonary artery was thought to have predisposed to infarction and rupture of alveoli with subsequent pneumothorax. 5 In another case, secondary bacterial infection and rupture of an abscess led to pneumothorax. 3 In the case described herein, the vascular lesions appear to have resulted in broad areas of infarcted lung parenchyma with subsequent rupture causing spontaneous pneumothorax.
The current generation of heartworm antigen tests identify most “occult” (microfilaria-negative) infections consisting of at least 1 mature female worm and are nearly 100% specific. False-negative test results occur most commonly when infections are light, female worms are still immature, only male worms are present, and/or the test kit (for test kits requiring refrigeration) or sample has not been warmed to room temperature. False-negative results also can occur erratically with any test, and unexpected negative results can be reconciled by retesting or using a different test. 11
Radiography is an excellent technique for diagnosis of spontaneous pneumothorax but not for detection of bullous lesions. 1,7 In dogs, the accuracy of detection of bullae or blebs on radiographs has ranged from 0% to 50%. 1 In one study of dogs with spontaneous pneumothorax, thoracic radiographs failed to reveal the pulmonary lesions in 11 of 12 dogs. Although these dogs had no signs of pulmonary disease based on radiographs, pulmonary bullae were detected surgically in all dogs. 7 In another study, radiography failed to identify 13 of 17 affected lung lobes, and computed tomography was more sensitive for detection of these lesions. 1
Computed tomography was important in the case reported herein, because bullae were not reliably identified and the full extent of these lesions could not be assessed on radiographs. Computed tomography also provided better assessment of the extent of the pneumothorax and lung lobe collapse. Furthermore, it allowed the surgeons to decide on a surgical approach.
Thoracic radiographs are important in animals with spontaneous pneumothorax as a screening test to identify other potential causes, such as neoplasia, abscess, and dirofilariasis. 7 The radiographic appearance of dirofilariasis is well documented and dependent on severity and duration of parasite infestation. 4,8 The severity of pulmonary arterial changes seems to be more related to the number of adult nematodes present than to duration of infection. 16 In the present case, enlargement of some arteries was the only sign of possible heartworm infection, but assessment of the vasculature was compromised because of the severity of the lung lobe collapse. Furthermore, the presence of pneumothorax and possible bullae were not expected findings for heartworm infection.
Exploratory thoracotomy is the treatment of choice for dogs with spontaneous pneumothorax. 12 Conservative therapy usually results in persistence or recurrence of pneumothorax. 7 In endemic areas, D. immitis should be considered in the differential diagnosis of dogs who present with spontaneous pneumothorax, even when minimal or no signs of heartworm infection or bullous lesions are identified radiographically. In these cases, computed tomography may be helpful for complete assessment of the lesions, final diagnosis, and surgical planning.
Acknowledgements. The authors acknowledge Dr. Leslie McLaughlin from the Department of Pathobiological Sciences of Louisiana State University for her help with the histopathology images.
Footnotes
a.
SNAP®, IDEXX Laboratories Inc., Westbrook, ME.
b.
Picker 5000 Falcon, Universal Systems, Salt Lake City, UT.
c.
Clavamox®, Pfizer Inc., New York, NY.
d.
Deramaxx®, Novartis International AG, Basel, Switzerland.
e.
Immiticide®, Merial Ltd., Duluth, GA.
