Abstract
A 9-year-old, male, captive red panda (Ailurus fulgens fulgens) in an urban zoo in the United Kingdom presented with respiratory distress and weight loss. The animal was euthanatized, and a postmortem examination was performed. The lungs were diffusely consolidated with extensive mineralization. Microscopically, there was extensive obliteration of normal pulmonary architecture by sheets and coalescing nodules of partially mineralized fibrous tissue and granulomatous inflammation centered on large numbers of nematode larvae and eggs. First stage nematode larvae were isolated from lung tissue and were characterized as Angiostrongylus vasorum on the basis of their morphology and sequencing of the 18S ribosomal RNA gene and the entire second internal transcribed spacer. Although A. vasorum has previously been reported in red pandas in a zoological collection in Denmark, this study is the first reported case in the United Kingdom and occurs against a background of geographical spread and increased incidence of disease in domestic and wild canids. Angiostrongylus vasorum should be considered a differential diagnosis for respiratory disease in the red panda and taken into account when planning parasite and pest control programs for zoological collections.
Keywords
A 9-year-old, male, captive-bred red panda was presented to the Bristol Zoo Gardens Veterinary Department (Bristol, UK) in February 2006 with marked paradoxical breathing, harsh lung sounds, and weight loss (1 kg over the past 2 years). Thoracic radiographs revealed a diffuse broncho-interstitial lung pattern with patchy alveolar filling throughout all lobes, in combination with foci of speckled mineralization (Fig. 1). These changes were not evident on radiographs taken 2 years earlier. A provisional diagnosis of chronic bronchopneumonia of unknown etiology was made. A comparative tuberculin skin test in March 2004 had resulted in a reaction at the bovine tuberculosis site. However, a bronchoalveolar lavage specimen obtained in May 2004 was negative for Mycobacterium sp. by culture and polymerase chain reaction (PCR) testing and was also negative for parasites. Because of the poor prognosis for recovery and renewed concern over possible tuberculosis, the animal was euthanatized while still under general anesthesia.
At postmortem examination, which was performed by veterinarians at the Bristol Zoo Gardens, the lungs were noted to be consolidated and mottled red and tan with areas of mineralization affecting all lobes, including at least 50% of the volume of the right and left caudal lobes. Hemorrhage was noted in the enlarged bronchial lymph nodes. The gastrointestinal tract was empty with no fecal material in the colon or rectum. Specimens from the lungs, bronchial lymph nodes, trachea, left eye, testis, skeletal muscle, spleen, heart, kidney, liver, pancreas, brain, thymus, and gastrointestinal tract were submitted in 10% neutral buffered formalin to the Department of Pathology and Infectious Diseases, The Royal Veterinary College, University of London (Hatfield, Hertfordshire, UK). Additional lung tissue was sectioned and frozen at −20°C. Tissue specimens for histological examination were processed routinely, embedded in paraffin wax, sectioned at 5 μm, stained with hematoxylin and eosin, coverslipped, and examined microscopically. Further sections were stained with an acid-fast stain (Ziehl-Neelsen).
Histologically, normal pulmonary architecture was largely obliterated by extensive, often nodular areas of partially mineralized fibrous tissue centered on multiple cross sections and oblique sections through nematode eggs (∼20 μm × 30 μm) and coiled larvae (6–15 μm in diameter; Fig. 2). Small clusters of epithelioid macrophages and multinucleate histiocytic giant cells were scattered within the fibrous tissue. In other fields, sheets of epithelioid macrophages and smaller numbers of histiocytic giant cells, granulocytes, plasma cells, and lymphocytes were also centered on large numbers of nematode eggs and larvae. There were extensive areas of parenchymal necrosis and hemorrhage. Remnant alveolar spaces were dilated, with hyperplasia of the smooth muscle of bronchiolar walls and alveolar ducts. Some of the remaining bronchioles contained larvae, which were free within the lumen. Bronchial lymph nodes were hyperplastic, and the medullary region of one lymph node contained granulomas centered on nematode larvae. Acid-fast stains did not reveal any bacteria. Microscopic lesions in other tissues included testicular and skeletal muscle atrophy, portal hepatitis, and infiltration of the left ventricular myocardium by small numbers of macrophages.
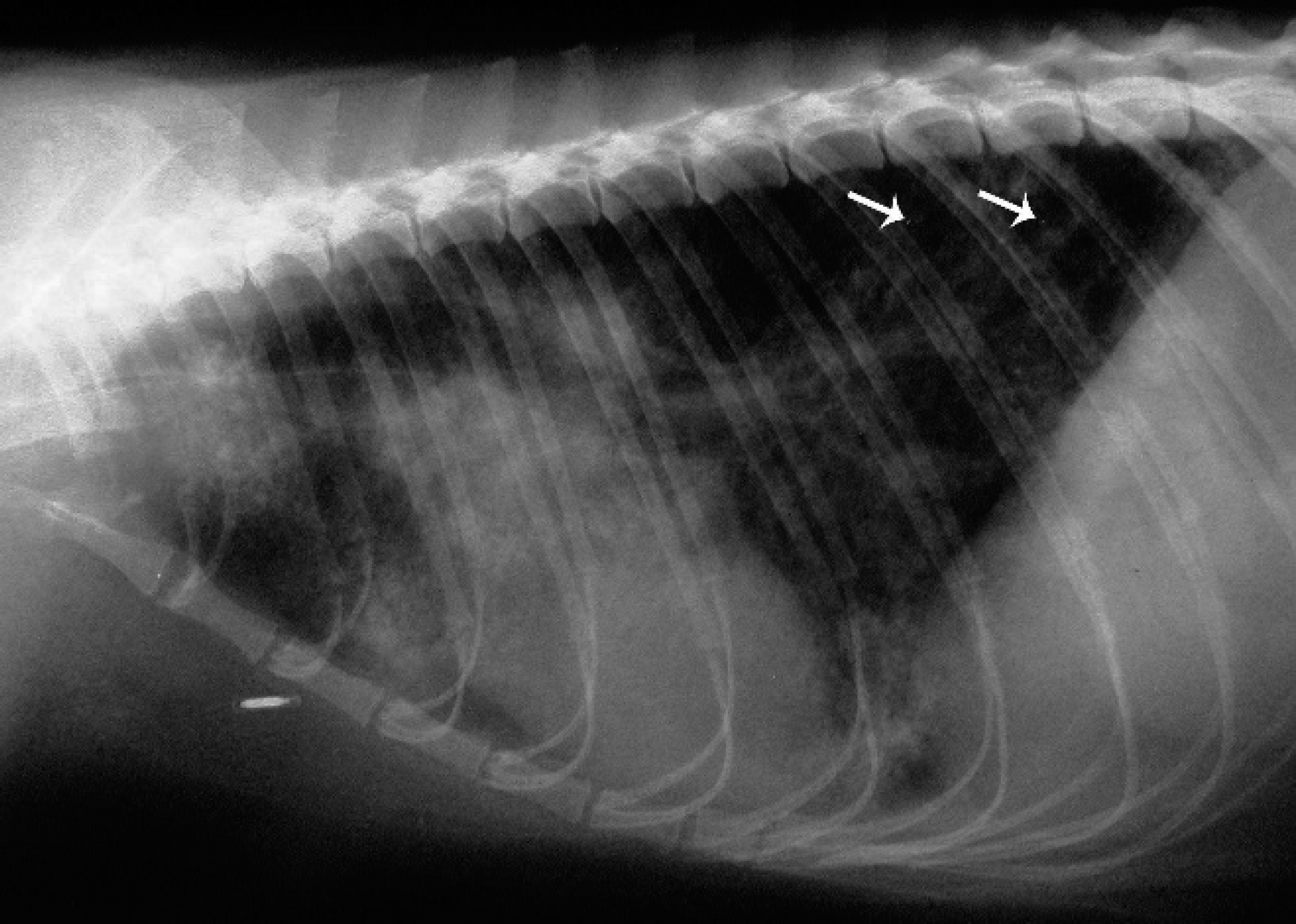
Thorax, red panda. Lateral radiographic view showing a diffuse broncho-interstitial lung pattern, with scattered small foci of mineralization (arrows).
Subsequent to the histological examination, larvae were removed from frozen lung tissue specimens by flushing with phosphate-buffered saline. The larvae (n = 21) measured 323 ± 18.0 μm in length (mean ± standard deviation) and 20 ± 2.9 μm in diameter and terminated in a sinusoidal projection with a pronounced dorsal notch, characteristic of both Angiostrongylus vasorum and Aelurostrongylus abstrusus (Fig. 3). The larvae were shorter and thicker than first-stage larvae (L1) of A. vasorum described previously 10 but fell within the range of lengths in a 1994 case 1 and were shorter than A. abstrusus L1 described in a 1949 study. 9 The dorsal caudal spine characteristic of A. vasorum was not visible in all specimens.
To identify the L1 larvae by molecular characterization, DNA was extracted from a 5-mg section of frozen lung tissue with a commercial extraction kit a according to the manufacturer's instructions. A 1,708-bp region of the 18S ribosomal RNA (rRNA) gene was amplified with the primers NC18SF1 (5′-AAAGATTAAGCCATGCA-3′) and NC5BR (5′-GCAGGTTCACCTACAGAT-3′). 6 The entire second internal transcribed spacer (ITS-2) was amplified with the primers NC1 (5′-ACGTCTGGTTCAGGGTTGTT-3′)andNC2(5′-TTAGTTTCTTTTCCTCCGCT-3′). 8 Polymerase chain reaction assays were performed in a final reaction volume of 25 μl that consisted of 2.5 μl of 10× polymerase buffer a (TrisCl, KCl, [NH4]2 SO4, 15 mmol MgCl2, pH 8.7), 0.5 μl each of deoxyribonucleotide triphosphate (dNTP; 10 mmol each), 0.625 μl of each primer (12.5 ng/ml), 0.125 μl of HotStarTaq DNA polymerase a (5 U/ml), 19.625 μl of dH2O, and 2 μl of DNA. Targets were amplified following PCR conditions of 95°C for 15 min; followed by 40 cycles of 95°C for 30 sec, 55°C for 30 sec, and 72°C for 1 min; followed by a final extension step of 72°C for 5 min. The PCR products were electrophoresed on a 1% (w/v) agarose gel and visualized with the use of ethidium bromide and ultraviolet illumination. The PCR products were purified with a commercial purification kit b and ligated into the pCR2.1-topoisomerase I (TOPO) plasmid vector, then transformed into TOP10 One Shot chemically competent Escherichia coli with a TOPO Taq-amplified cloning kit. c Cloned plasmids were purified and then sequenced with the use of a kit d with M13 forward and reverse primers. Sequences for the red panda sample were deposited in the GenBank database under the accession numbers EU915247 and EU915248 for the 18S rRNA gene and ITS-2, respectively. The 18S rRNA gene sequence was 100% homologous with A. vasorum (GenBank EF514916 and AJ920365), and the ITS-2 sequence was 99% identical to A. vasorum (GenBank EU627592).
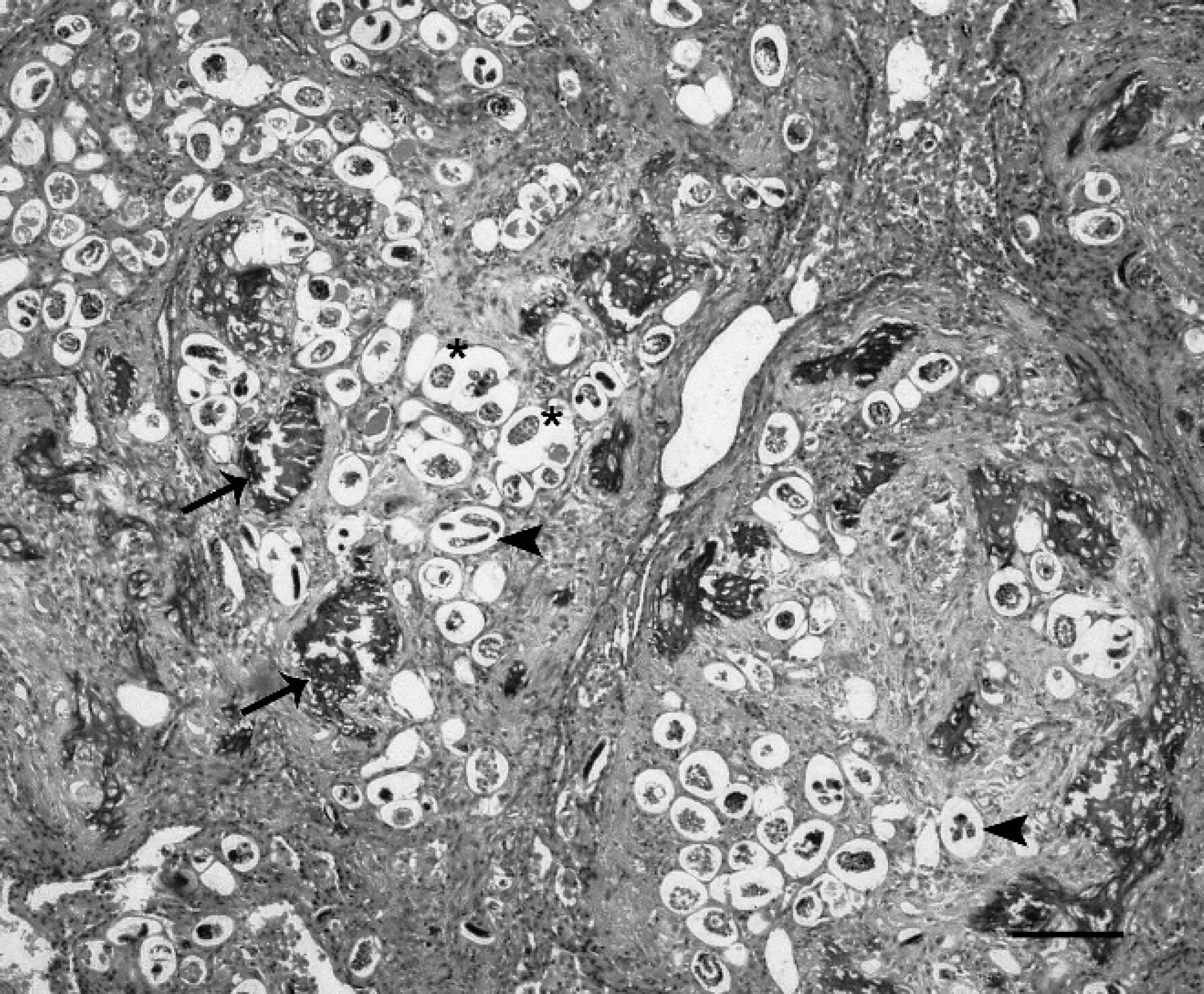
Lung, red panda. Most of the normal parenchymal architecture is obliterated by coalescing nodular areas of fibrous tissue that are partially mineralized (arrows) and surround numerous sections through nematode larvae (arrowheads) and eggs (asterisks). Hematoxylin and eosin. Bar = 100 μm.
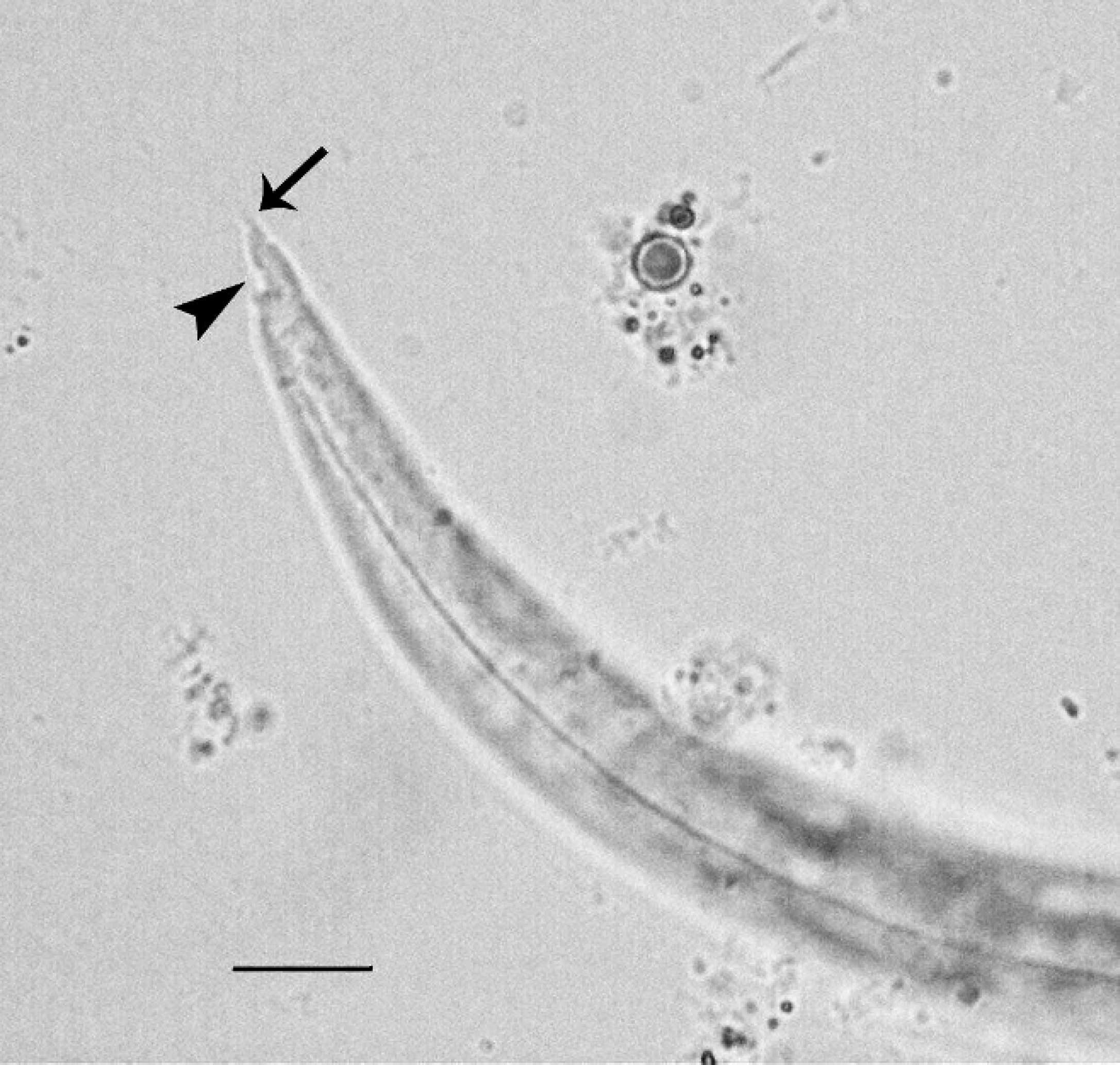
Larva extracted from lung, caudal portion. Note the deep dorsal notch (arrowhead) and the sinusoidal kink in the tail tip (arrow). Bar = 10 μm.
The gross, microscopic, and parasitological findings indicated that A. vasorum infection was the cause of the respiratory signs and pulmonary inflammation in the red panda in this study. Angiostrongylus vasorum is a metastrongyloid nematode that is known to cause cardiopulmonary disease in dogs, foxes, and other canids, with reports in a few other species, including Eurasian bad-gers. 1,2,5,11,14,16 Its geographical distribution is patchy but appears to be expanding in several parts of the world, 13 and surveys of foxes reveal the parasite to be currently endemic in much of the southern half of the United Kingdom, 14 with cases in dogs recently appearing in Bristol (unpublished data). In Copenhagen, also in an endemic area, fatal disease has been reported in several red pandas in the city's zoo in recent years (Grøndahl C, Monrad J, Dietz HH, et al.: 2005, Angiostrongylosis in red panda [Ailurus fulgens fulgens]. Proceedings of the Institute for Zoo and Wildlife Research, Berlin, 42:117–118; Jensen HE, Dietz HH, Grøndahl C, et al.: 2005, The pathology associated with Angiostrongylus vasorum infection in the red panda [Ailurus fulgens fulgens]. Proceedings of the 23rd meeting of the European Society of Veterinary Pathology, pp. 70–71).
Ovoviviparous female worms are located in the cardiac right ventricle and pulmonary vasculature, depositing eggs that pass via the blood to the lungs. First-stage larvae hatch rapidly, penetrate into the alveoli, and are coughed up and swallowed, eventually being passed in the feces. Further development to infective third-stage larvae (L3) occurs within a few weeks in snail or slug intermediate hosts. Definitive hosts are infected by ingestion of the intermediate hosts or of paratenic hosts, including frogs. 1 Disease is due to proliferative endarteritis and thrombosis in response to the adult worms in the pulmonary arteries, and pneumonia is caused by the larvae and eggs. 4 Infected canids can develop chronic right-sided heart failure, progressive respiratory signs and failure, or both if untreated, 1 and coagulation disorders have also been reported. 11 Histological lesions reported in the lungs of canids with severe disease include granulomatous inflammation, variably severe interstitial fibrosis, thrombosis of pulmonary arteries with fibrin and parasites, arterial fibrosis and recanalization, arterial smooth muscle hyperplasia, and necrosis. 3,4 Granulomas associated with nematode eggs and larvae can also occur in other tissues, including the tracheobronchial lymph nodes (as in this case), brain, kidney, spleen, and adrenal gland. 3,4 The pathological lesions in the red panda in this study were therefore similar to those reported in dogs and foxes with severe disease, although pulmonary mineralization was more extensive. Arterial lesions were not noted histologically, but unfortunately, only a few small specimens of lung tissue were submitted. Other cases of A. vasorum infection reported in red pandas also featured severe granulomatous inflammation in the lung parenchyma, including mineralization and some foci of osseous metaplasia, associated with severe and sometimes lethal clinical disease (Grøndahl C, et al.: 2005, Angiostrongylosis in red panda).
How the red panda in this study became infected with A. vasorum is not known, but it is presumed that the animal ingested an infected intermediate or paratenic host. Exposure could have occurred through entry of infected slugs or snails into the enclosure, either by their own movement or with food or other material; slugs or snails caught through the enclosure wire; or infection of slug and snail populations within the enclosure from inadvertently introduced dog or fox feces. The red panda was often observed hunting in the enclosure, and ingestion of slugs or snails is therefore plausible. However, studies on human outbreaks of abdominal angiostrongyliasis caused by A. costaricensis suggest the source of infection was food contaminated with mucus from infected mollusks rather than the mollusk itself, which indicates another possible route. 12,19 The prepatent period for A. vasorum in the normal definitive host is 38–57 days. 1 The pulmonary lesions in the red panda in this case, especially the extensive mineralization and fibrosis, indicated long-standing infection with large numbers of parasites. Routine parasitologic fecal examinations had been conducted every 6 months. However, at the time, zoo policy was only to treat if results were positive, which they were not in this case. Detection of A. vasorum L1 would require repeated fecal examination by smear, zinc sulfate centrifugation–flotation, or, ideally, the Baermann technique. 15 No anthelmintics had been administered after a single dose of ivermectin e in October 2004, the efficacy of which against A. vasorum infection is uncertain. Up to June 2003, treatment with fenbendazole had been periodic/Fenbendazole, although not licensed for this purpose, is known to reduce A. vasorum burdens in dogs at a dose rate of 50 mg/kg daily, although up to 21 days of treatment might be needed to eliminate infection. Milbemycin oxime and moxidectin are licensed for reduction of A. vasorum infection in dogs, although treatment with moxidectin has previously failed to prevent death of infected red pandas (Grøndahl C, et al.: 2005, Angiostrongylosis in red panda).
Several other metastrongyloid nematodes can infect red pandas, including Aelurostrongylus sp., which was diagnosed in 42% of 31 animals in a fecal parasitological survey conducted at Chongqing Zoo, China, 18 as well as Crenosoma (Grøndahl C, et al.: 2005, Angiostrongylosis in red panda) and Filaroides sp. (J. McGarry, personal communication, 2008). Larvae of these species would also be detected by Baermann extraction. However, identification on the basis of morphology alone can be difficult, especially in distinguishing between A. vasorum and A. abstrusus. The morphology of the larvae in this case differed subtly from those of A. vasorum typically found in dogs, being slightly larger in diameter and with a less obvious dorsal spine. Host-induced phenotypic variation is possible in this species, or freezing of lung tissue possibly altered larval morphology. Molecular analysis was required to identify the parasites definitively and suggests that incorrect conclusions can be reached if parasites are identified in unusual hosts on the basis of larval morphology alone (i.e., in the absence of identification of adult worms in the pulmonary vasculature; Jensen HE, et al.: 2005, The pathology associated with Angiostrongylus vasorum infection in the red panda). Xenodiagnosis (i.e., infecting snails and then feeding them to captive foxes followed by examination for evidence of parasitic infection; Grøndahl C, et al.: 2005, Angiostrongylosis in red panda) is impractical as a routine diagnostic test.
The case in this report highlights the need for preventative health plans for captive red pandas in A. vasorum–endemic areas, of which zoo veterinarians should be aware. Opportunities for exposure to slugs and snails should be limited as far as is practical, and feces should be routinely examined for L1, noting that routine salt flotation is not sufficient. No proven anthelmintic prevention protocol for red pandas exists; however, anecdotal reports suggest some success with milbemycin oxime (11.5 mg per 5 kg body weight PO [by mouth], monthly; Grøndahl C, et al.: 2005, Angiostrongylosis in red panda). This drug has been used successfully to treat infections in dogs. 7 Fenbendazole or moxidectin are also effective in eliminating infection in dogs 17 and might have some prophylactic effects, but this is not yet proven. Angiostrongylus vasorum should be considered a differential diagnosis by clinicians and pathologists in cases of respiratory disease in red pandas captive in endemic areas, and prompt treatment should be instigated where it is suspected.
Acknowledgements. The authors thank Dr. Mark Fox of the Department of Pathology and Infectious Diseases, The Royal Veterinary College, for his review of the manuscript, and the Morris Animal Foundation for support through First Award D06CA-308 “Molecular epidemiology of French heartworm infection in dogs and foxes,” which enabled development of the molecular diagnostics.
Footnotes
a.
DNeasy®, Qiagen GmBH, Hilden, Germany.
b.
QIAquick®, Qiagen GmBH, Hilden, Germany.
c.
TOPO® TA Cloning Kit, Invitrogen, Carlsbad, CA.
d.
ABI PRISM™ Dye Terminator Cycle Sequencing Core Kit, Applied Biosystems, Foster City, CA.
e.
Ivomec®, Merial Animal Health Ltd., Harlow, Essex, United Kingdom.
f.
Panacur®, Intervet Inc., Walton, Milton Keynes, United Kingdom.
