Abstract
A 51-chromium-labeled ethylenediamine tetra-acetic acid (51Cr-EDTA) permeability blood test was validated as a method to assess damage to the small intestine in dogs. The test was performed by calculating various percentages from sera after an orally ingested dose solution. The aim of the current study was to determine whether the use of serum or plasma had any influence on the results of the test. A test solution with 3.7 megabecquerel (100 μCi) of 51Cr-EDTA was delivered through an orogastric tube to 13 healthy laboratory Beagle dogs. From each dog, 2 concurrent blood samples were withdrawn from cephalic veins using clotting-factor activator tubes for serum and heparinized tubes for plasma. The samples (n = 26) were taken at 3 and 5 hr after administration of the test solution. Percentages of the orally ingested dose were then calculated in serum and plasma, and their relationship was assessed using correlation analysis. The mean ± standard deviation percentages in serum and plasma after 3 hr were 0.85 ± 0.43% and 0.88 ± 0.49%, respectively, whereas respective percentages in serum and plasma after 5 hr were 0.78 ± 0.52% and 0.81 ± 0.51%. The combined correlation coefficient between the percentages from the sera and plasma samples was excellent (R = 0.98). It was concluded that the 51Cr-EDTA permeability test in blood may be performed using serum or plasma of dogs, and the choice between the 2 samples is one of convenience.
The 51-chromium-labeled ethylenediamine tetra-acetic acid (51Cr-EDTA) intestinal permeability (IP) test provides a means of assessing injury to the small intestine and has been successfully used as a noninvasive diagnostic tool for screening enteropathies in human beings and other species. In dogs, this test has proved its sensitivity in detecting epithelial damage, monitoring improvement in intestinal integrity after successful therapy, identifying a primary permeability defect in Irish Setters with gluten-sensitive enteropathy, and helping diagnose clinically silent bacterial overgrowth in the small intestine. 3,8 11 The 51Cr-EDTA IP test in dogs is performed by orally ingesting a test solution of 51Cr-EDTA dissolved in water, and quantifying the subsequent recovery of the radioisotope in urine or blood. Blood analysis obviates challenges associated with the need for extended and careful urine collection in dogs. It has therefore been shown to be a more practical method than the urine-based means for assessing IP because fecal contamination of urine, catheterization of the urinary bladder, and the housing of dogs in metabolic cages for 6 or 24 hr for complete urine recovery are all obviated. 3,7,11,14,16 The 51Cr-EDTA IP blood test is performed by calculating successive percentage recoveries of 51Cr-EDTA in serum (e.g., at 3 and 5 hr) after oral ingestion of a test solution. To measure radioactivity with the use of a gamma counter, the sample volume must be at least 1 ml. This volume is considerably more difficult to obtain as serum than plasma, particularly in certain patients where size, disposition, or underlying medical conditions could minimize sample availability. 7 It has generally been considered that 51Cr-EDTA has little or no affinity for proteins, but conclusions from such studies may be hampered by the fact that results from protein-binding experiments have been related to the methodology used. 13 Similarly, it has been reported that the binding of 51Cr-EDTA to plasma proteins in in vitro and in vivo studies may not be as negligible as has been believed. 1,2,6,15 Nonetheless, 51Cr-EDTA has been widely used for several years in the clinical assessment of glomerular filtration rates in human and veterinary practice, and the laboratory determination of the radiopharmaceutical has invariably been carried out from either serum or plasma samples. 4,5,17 To the authors' knowledge, the comparability analysis of 51Cr-EDTA in canine serum and plasma is unknown and has not been investigated. The objectives of the current study were to compare the use of serum and plasma samples for measuring 51Cr-EDTA and to determine whether the use of plasma is as well qualified as serum for performing the 51Cr-EDTA IP blood test in dogs.
Thirteen male purebred laboratory Beagle dogs supplied by a commercial vendor a were used in the current study. The dogs were acclimatized for at least 1 month before the study commenced. All the dogs were housed at the laboratory dog facilities of the former Faculty of Veterinary Medicine, University of Helsinki (Helsinki, Finland), in indoor pens; the dogs spent about 4 hr daily in outdoor runs. The indoor temperature varied from 18 to 22°C. The dogs were exposed to both natural and artificial lighting, the latter from 7 AM to 4 PM. Feeding throughout the study consisted of 1.5 cans per dog of commercial canned dog food, b which was given twice daily. Water was freely available at all times. At the beginning of the study, the dogs' ages ranged from 17 to 46 months (mean: 23.08 months) and body weight ranged from 13.00 to 17.00 kg (mean: 15.02 kg). To determine the health status of the dogs, physical examinations were carried out, and blood samples were taken from each subject for hematologic and biochemical examinations. Before commencing the study, the dogs were treated with fenbendazole c at a dose of 50 mg/kg orally for 3 consecutive days, and fecal samples were thereafter collected for endoparasite examination. The dogs were cared for in accordance with the prevailing Finnish and European legislation on the use and care of laboratory animals. The experimental protocol was approved by the local Animal Ethics Committee at the University of Helsinki.
Individual test solutions consisted of 3.7 megabecquerel (100 μCi) of 51Cr-EDTA d dissolved in 50 ml of distilled water. The counting standard was retained from the test solution in a 1-ml aliquot, which was further diluted before measuring the radioactivity. The 51Cr-EDTA IP blood test was simultaneously performed on serum and plasma. Food was withheld for 12 to 18 hr before and throughout the study, but water was freely available at all times. After the fasting period, body weights were measured, and a baseline blood sample was taken from each dog. Two concurrent blood samples were withdrawn from the cephalic veins of each dog at 3 and 5 hr after administration of the test solution. One sample was transferred to clotting-factor activator tubes to obtain serum, and the other sample was transferred to heparinized tubes to obtain plasma. After centrifugation of all the blood samples, the standard, serum, and plasma aliquots were measured for 51Cr-EDTA gamma ray emissions by use of a gamma counter. e Gamma ray emissions in the aliquots were concurrently counted for 10 min, and all the analyses were performed within 12 hr of the blood collection period.
Formulas to calculate the amount of 51Cr-EDTA in serum and plasma as a percentage of the orally ingested test dose are as follows:
where cpm represents counts per minute, TSV represents total serum volume, TTSV represents total test solution volume, TPV represents total plasma volume, DF represents dilution factor, BV represents blood volume, BW represents body weight, and Hct represents hematocrit. Estimations of the whole blood and the total serum and plasma volumes were carried out according to formulas described elsewhere. 12,18 Statistical analysis was performed using SPSS 11.0 software. f The data distribution was analyzed by applying the Kolmogorov-Smirnov test and was determined to be Gaussian. Because of the normal distribution, results were expressed as the mean and standard deviation (mean ± SD). The significance of the difference in test results between serum and plasma samples was determined by one-way analysis of variance, and where appropriate, the Student-Newman-Keuls test was used to compare means. The relationship between the results was determined using the Pearson correlation coefficient (R). A P value of less than 0.05 was regarded as statistically significant. The Bland-Altman test was used to analyze the difference and potential bias between the 2 methods. The repeatability coefficient was calculated as 1.96 times the SD of the differences.
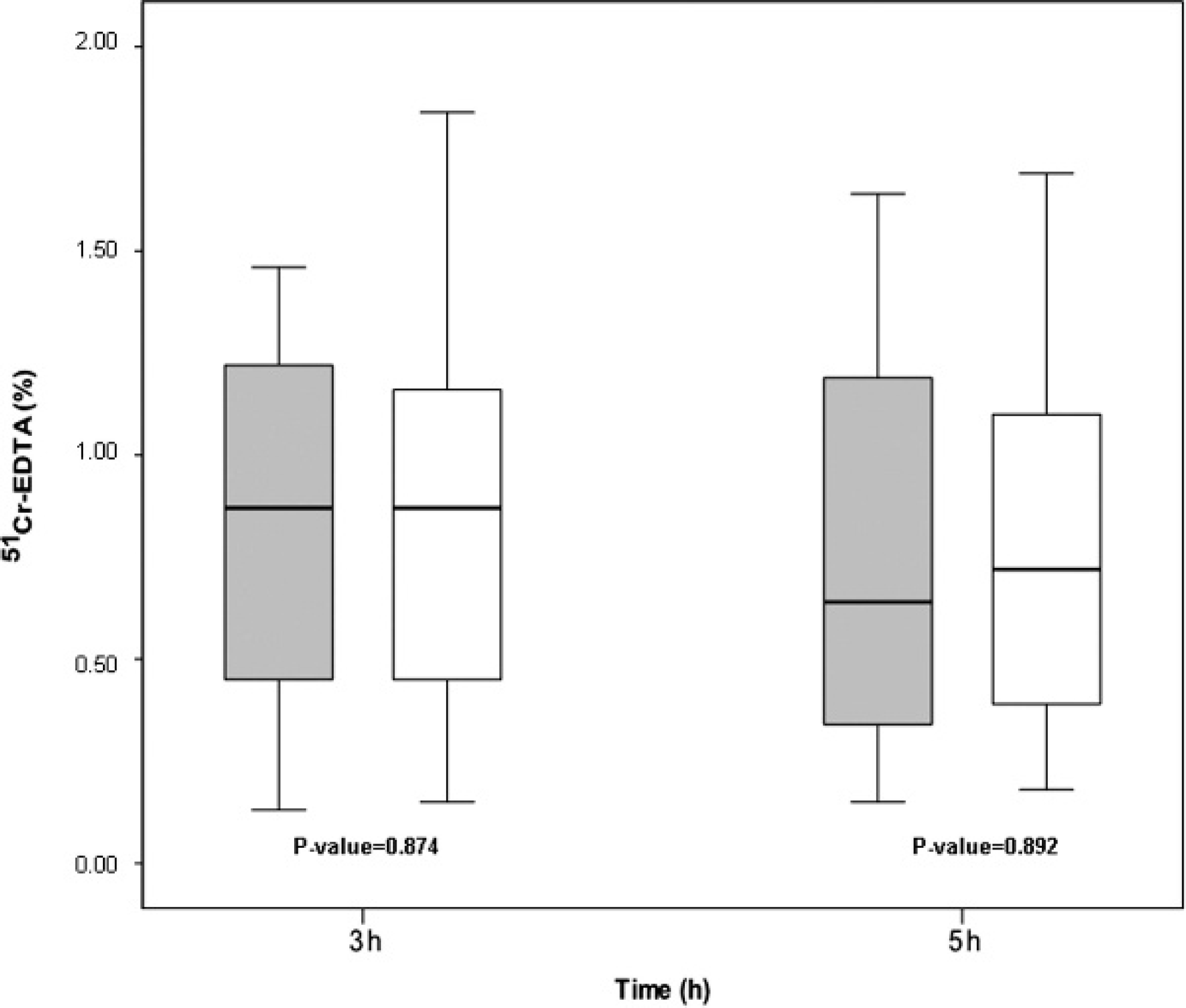
51-chromium-labeled ethylenediamine tetra-acetic acid (51Cr-EDTA) recovery percentages from serum (gray) and plasma (white) samples at 3 and 5 hr after ingestion. The horizontal line in the box is the median (50% percentile), and the upper and lower limits of the box indicate the upper (75%) and lower (25%) quartiles, respectively. The limits of the upper and lower vertical lines are the maximum and minimum data values, respectively. As indicated by the lack of separate dots or asterisks, no outliers were detected in the samples. All data were subjected to statistical analysis.
All the dogs were determined to be healthy based on physical examinations and after interpretations of hematologic and serum biochemical analysis. Blood urea nitrogen and serum creatinine concentrations suggested normal renal function. Exocrine pancreatic function was considered normal based on measurements of serum trypsin-like immunoreactivity concentrations. Examinations for fecal endoparasitic ova were negative. Mean ± SD percentages in serum and plasma after 3 hr were 0.85 ± 0.43% and 0.88 ± 0.49%, respectively, whereas the respective percentages in serum and plasma after 5 hr were 0.78 ± 0.52% and 0.81 ± 0.51%. No significant differences were detected between the percentages of 51Cr-EDTA in serum and plasma samples at 3 and 5 hr (Fig. 1). However, statistically significant correlations were found between serum and plasma levels of 51Cr-EDTA at 3 hr (R = 0.96, P < 0.0001) and 5 hr (R = 0.99, P < 0.0001; Fig. 2). The combined correlation coefficient between the percentages from the serum and plasma samples was excellent (R = 0.98). The findings of the present study indicate that the levels of 51Cr-EDTA in both serum and plasma after oral ingestion of the probe molecule in dogs are strongly correlated. This demonstrates that the 51Cr-EDTA IP blood test in dogs can be equally performed from serum or plasma samples, and it supports the use of plasma when a small amount of blood is collected from the patient. These correlations were additionally analyzed using the Bland-Altman test (Fig. 3), which revealed no differences in 51Cr-EDTA levels between serum and plasma samples. The regression line of differences versus means and 95% confidence intervals was also included, illustrating a linear regression between the 2 sample tests. Therefore, the IP test measured in blood showed no differences in 51Cr-EDTA levels of serum or plasma, evidencing that there are no differences in the 2 methods used for assessing IP in dogs.
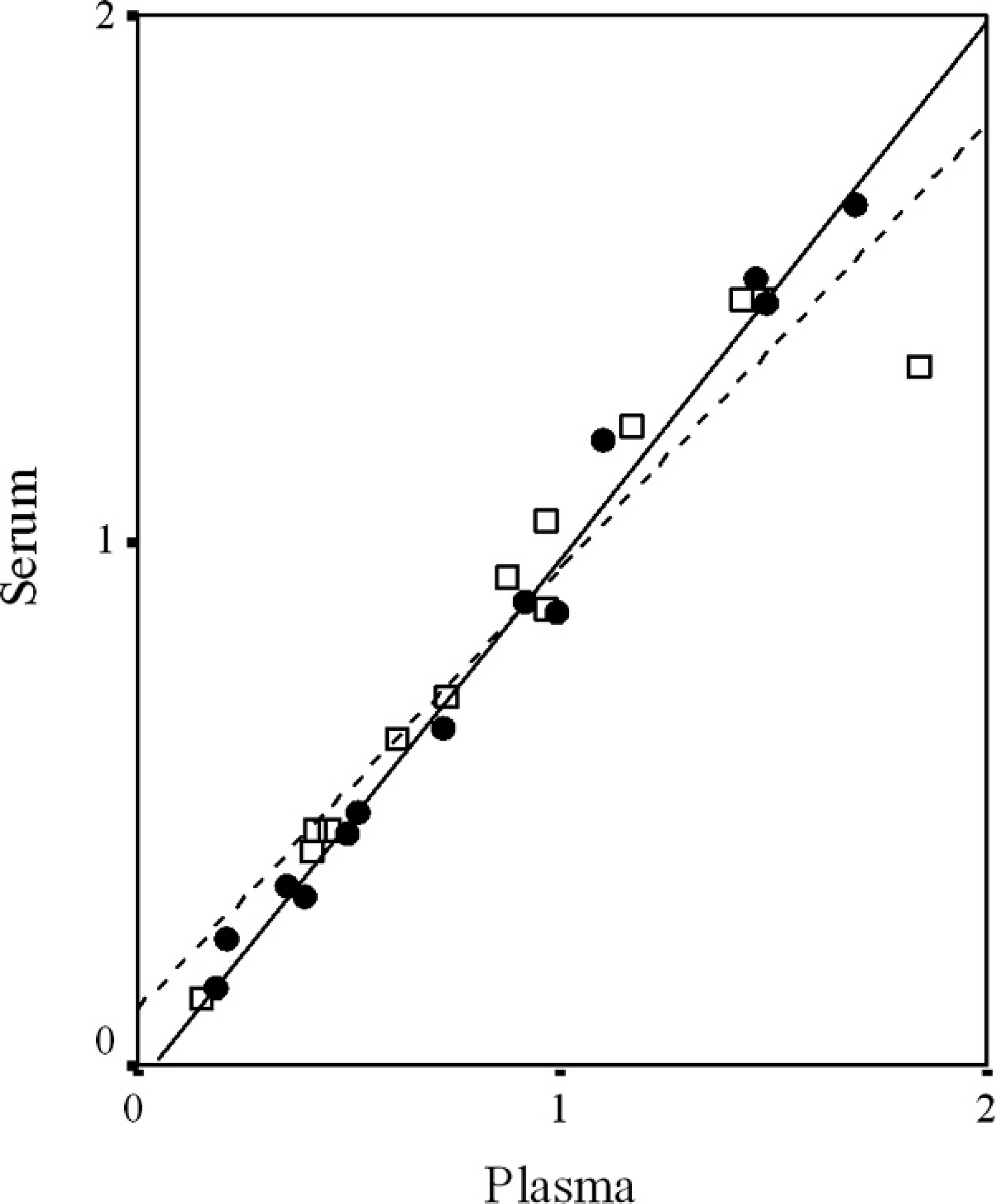
Relationships between serum and plasma levels at 3hr (□ and discontinuous line) and 5 hr (• and continuous line) after ingestion of 51-chromium-labeled ethylenediamine tetraacetic acid (51Cr-EDTA) dissolved in water. Lines show the Pearson correlation (linear adjustment) at 3 hr (R = 0.96, P < 0.0001) and at 5 hr (R = 0.99, P < 0.0001).
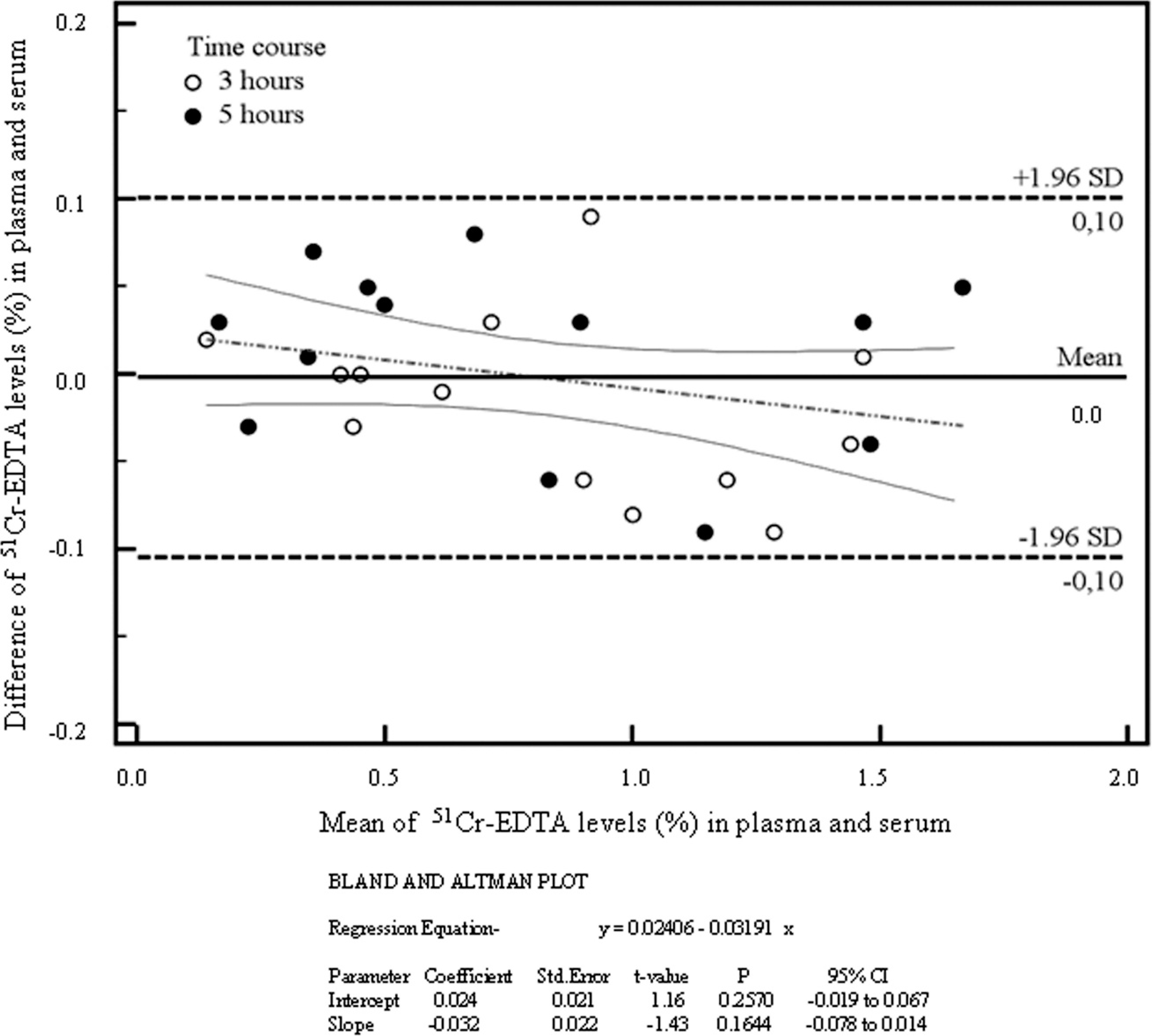
A Bland-Altman plot (difference plot), which is a method of data plotting used in analyzing the agreement between the 2 different methods (serum and plasma). Limits of agreement during Bland-Altman analysis are usually specified as bias ± 1.96 SD (average difference ± 1.96 standard deviation of the difference). The regression line of differences versus averages and the 95% confidence intervals (CIs) have been included (continuous and discontinuous lines, respectively). 51Cr-EDTA = 51-chromium-labeled ethylenediamine tetra-acetic acid.
In conclusion, the choice between serum or plasma analysis of the probe molecule when performing the 51Cr-EDTA IP blood test in healthy dogs is not relevant and is solely one of convenience. Furthermore, the use of plasma during the 51Cr-EDTA IP blood test may be preferable to the use of serum in certain patients in which a lower volume of blood sample is anticipated, such as in juvenile or miniature dog breed patients.
Acknowledgements. The authors are grateful to the Finnish Veterinary Foundation and the Helvi Knuuttila's Foundation for financial support. This study was conducted at the former facilities of the Experimental Unit, Faculty of Veterinary Medicine, University of Helsinki, Finland. The authors thank Dr. Roy Siddall for reviewing this manuscript.
Footnotes
a.
Harlan-Winkelmann GmbH, Borchen, Germany.
b.
Pedigree®, Fortivil 400 g, Waltham®, Masterfoods Ltd., Helsinki, Finland.
c.
Axilur®, Intervet International, Boxmeer, The Netherlands.
d.
Nycomed Amersham plc, Little Chalfont, Buckinghamshire, United Kingdom.
e.
LKB-Wallac 1270 Rackgamma II gamma counter, LKB-Wallac, Turku, Finland.
f.
SPSS Inc., Chicago, IL.

