Abstract
The tube coagulase test, an invaluable laboratory tool for identifying Staphylococcus aureus, is most often done using rabbit plasma. However, there is evidence that depending on the origin of the isolates, other plasmas may be superior. The current study sought to compare the utility of dog and rabbit plasma in the coagulase test for S. aureus isolated from canine (n = 28), bovine (n = 29), and human (n = 30) hosts. Overall, coagulation times were significantly faster for dog (2.38 hr) than rabbit (3.19 hr) plasma. When coagulation times were compared by isolate origin, no significant differences were found for rabbit plasma, whereas bovine isolates clotted dog plasma significantly faster (1.86 hr) than canine (2.79 hr) or human (2.38 hr) isolates. Investigators should be aware that rabbit plasma may not be the ideal coagulase-testing medium for S. aureus from all sources.
Staphylococcus aureus is one of the most common causes of infections in people worldwide, and it is increasingly recognized in companion animals, including dogs. 4,9 Recent reports of the increasing prevalence of methicillin-resistant S. aureus (MRSA) in people and animals have raised awareness in the veterinary community about this potential pathogen. 6,7 Staphylococcus aureus is identified using a number of standard biochemical tests, including the tube coagulase test, typically using rabbit plasma. 10
Previous studies have documented variability in the coagulability of different plasmas for S. aureus isolated from different hosts. 1–3,5 This variability may be intrinsic to the plasma or due to the presence of S. aureus biotypes, which preferentially clot certain plasmas. 1,5 The importance of identifying S. aureus necessitates sensitive and timely coagulase test procedures, particularly in the case of MRSA, where therapeutic and infection control measures are markedly different than for susceptible organisms. The purpose of the current study was to evaluate the relative utility of dog and rabbit plasmas in the tube coagulase test for S. aureus isolated from canine, bovine, and human hosts.
Eighty-seven archived S. aureus isolates were tested. A total of 30 were of human origin, and 29 and 28 were from bovine and canine hosts, respectively. Commercially prepared rabbit plasma a and dog blood bank plasma b were used. A single well-isolated colony was inoculated into 0.5 ml of plasma and incubated at 35°C for 4 hr, followed by room temperature incubation. 10 Samples were evaluated hourly for clot formation by gently tilting the test tube.
Statistical analyses were done using SPSS version 17.0. c Dog and rabbit plasma coagulase times for all samples were compared using a univariate analysis of variance (AN-OVA) with Bonferroni correction. Differences in the time to clot formation for dog or rabbit plasma between isolates from each host were compared using one-way ANOVAs. Differences in the time to clot formation for dog and rabbit plasma between isolates from the same host species were compared using t-tests. P ≤ 0.05 was considered significant for all analyses.
Overall, the time to clot formation was significantly faster for dog (mean: 2.38 hr) than rabbit (mean: 3.190 hr) plasma (P < 0.001; Table 1). No significant differences were found in time to coagulate rabbit plasma between isolates from different hosts; however, bovine isolates coagulated dog plasma faster (mean: 1.86 hr) than either canine (mean: 2.79 hr) or human (2.50 hr) isolates (Table 1). Bovine and human isolates coagulated dog plasma significantly faster than rabbit plasma, whereas no significant differences were found among canine isolates (Table 1).
Previous studies have shown that S. aureus isolates coagulate plasmas of different origins to varying degrees, including false-negatives, suggesting that multiple plasmas may need to be used. 1,3 However, because these data were collected in the 1970s and 1980s before some staphylococcal species (e.g., Staphylococcus pseudintermedius) were recognized, the identity of those collections and the specificity of the data to S. aureus are uncertain. Additionally, because direct comparisons between dog and rabbit plasma have not been reported, the present study fills an important gap in the literature.
The current study demonstrates that there are significant differences in the coagulase test using dog and rabbit plasmas. Although the time to clot dog plasma was significantly faster overall, suggesting an intrinsic superiority to rabbit plasma, there was also variability between isolates from different host species, giving credence to the biotype hypothesis. Although no false-negatives are described in the present report, the archived samples tested were initially identified using rabbit plasma and may not be representative of all clinical isolates, thus potentially overestimating the utility of rabbit plasma.
Coagulation times of canine and rabbit plasma for Staphylococcus aureus isolated from different species.
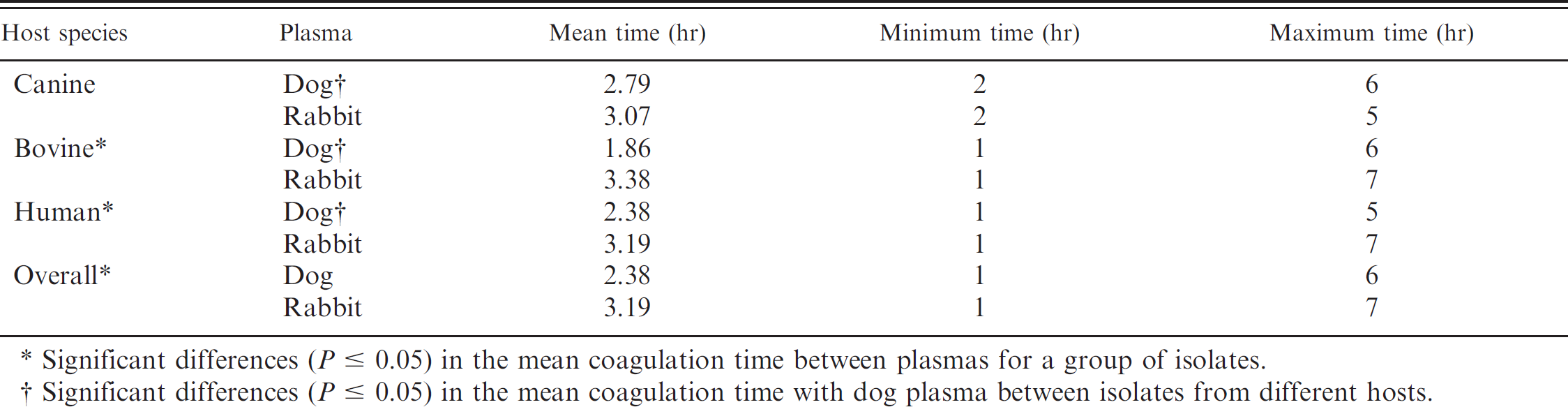
Significant differences (P ≤ 0.05) in the mean coagulation time between plasmas for a group of isolates.
Significant differences (P ≤ 0.05) in the mean coagulation time with dog plasma between isolates from different hosts.
Significant differences were not found between canine and human isolates, possibly reflecting the previously described genetic similarity between canine and human S. aureus collected from a single geographic location. 9 Additionally, the significant differences found in the time to clot dog plasma between bovine and human isolates are consistent with previously reported genetic dissimilarities between bovine and human strains. 8 In the future, it would be useful to compare these data to established techniques for evaluating the relatedness of S. aureus, such as spa typing and multilocus sequence typing.
Investigators should be aware of the potential limitations of only using rabbit plasma, particularly when testing samples from different hosts. Future studies including plasmas and isolates from a greater number of hosts should be done to define the utility of various plasmas for different diagnostic applications.
Acknowledgements
The authors would like to acknowledge the Companion Animal Health Fund for funding this study.
Footnotes
a.
BD Diagnostic Systems, Sparks, MD.
b.
Canadian Animal Blood Bank, Winnipeg, Canada.
c.
SPSS Inc., Chicago, IL.
