Abstract
A disease characterized by tongue and facial muscle atrophy has been recognized sporadically among Pembroke Welsh Corgi (PWC) dogs in Japan. The present study describes the pathologic findings of this canine syndrome. Histopathologic examinations were performed in 2 dogs, including a case of muscular biopsy. Identification and characterization of autoantibodies were attempted by fluorescent antibody test (FAT) and Western blot (WB) by using sera from 7 PWC dogs with typical clinical features, 6 PWC dogs with other clinical signs, and 2 from other breeds with polymyositis. Clinically, the 7 affected PWC dogs exhibited dysphagia with severe tongue atrophy, facial muscular atrophy, and occasional walking difficulty. Histopathologic examinations of the 2 dogs with clinical symptoms revealed moderate to severe inflammatory lesions characterized by lymphohistiocytic infiltration and muscular atrophy in the tongue and/or femoral muscles. The tongue lesions were very severe and accompanied by diffuse fatty infiltration. There were no major lesions in the nervous tissues examined. By FAT, an autoantibody against the cross striation of skeletal muscle was detected in sera from 5 affected PWC dogs. By using WB analysis, the autoantibodies recognized a 42-kDa molecule in striated muscle but not in the nervous tissues. All of the findings indicated that the unique disease of PWC dogs might be generalized inflammatory myopathy, whereas the detailed etiology concerning the dominant involvement of tongue muscles and the role of the autoantibody in the canine disease remain to be clarified.
Keywords
Introduction
Tongue muscular atrophy that results in aphagia, together with facial and temporal muscular atrophy, is a characteristic clinical sign caused by disorders of the cranial nerves, including the glossopharyngeal, trigeminal, facial, and vagus nerves, or of the brain stem, 4,18,22,27 or generalized by diseases such as muscular dystrophy 2 and myasthenia gravis. 3,8 Recently, generalized muscular disease characterized by tongue muscular atrophy has been found sporadically among Pembroke Welsh Corgi (PWC) dogs in Japan. A previous study 17 introduced the clinical and pathologic features in an affected PWC dog. The dog in the previous case had severe aphagia, masticatory, and temporal muscular atrophy, and, occasionally walking difficulty. Based on these clinical signs, some neurologic etiology or muscular dystrophy had been suspected, but the cause has remained unclear.
Canine muscular diseases are classified into the following 3 types by the site of injury. 24 The first classification is myogenic diseases, which result from damage to the muscle. These diseases are divided into inflammatory and noninflammatory myopathies. Inflammatory myopathies are classified as infectious, immune mediated, paraneoplastic, and nutritional. Noninflammatory myopathies are classified into acquired (toxic, metabolic, and so on) and inherited or breed-specific myopathies. Muscular dystrophy is an inherited type of noninflammatory myopathy. 9,25 Two types of canine muscular dystrophy, which are caused by genetic defects of dystrophin 2,33 or laminin alpha 2 (merosin), 29,35 have been reported previously. On histopathologic examination, such types of muscular dystrophy are characterized by generalized diffuse atrophy, degeneration, regeneration, fibrosis, and calcium deposition in the muscle tissues. Neurogenic diseases make up the second group of canine muscle diseases. They are caused by disruption of muscle innervation. Such neurogenic muscular atrophy shows common histopathologic changes, such as group atrophy and fiber type grouping. Myodegeneration, muscular necrosis, cellular infiltrations, muscular regeneration, and vascular lesions are minimal in neurogenic myopathies. 5,9,20,21,30,36 Finally, the third type of canine muscle disease is caused by abnormalities of the myoneural junctions (i.e., myasthenia gravis). 31
Canine immune-mediated inflammatory myopathies include masticatory muscle myositis (MMM), polymyositis (PM), dermatomyositis (DM), and extraocular myositis (EOM). 10,32 Canine MMM is a focal myositis that selectively involves masticatory muscles, including the temporalis, masseter, medial and lateral pterygoid muscles, and rostral portions of the digastricus muscles. 10 Canine PM is a generalized myositis with common clinical signs, including weakness, stiff-stilted gait, and progressive generalized muscle atrophy. 10 Canine DM is generalized myositis that involves the skin and vasculature as well as the striated muscle. 11,15,39 Canine EOM is a focal myositis limited to the extraocular muscles. 1,41
To elucidate the etiology of the muscular disease that demonstrates severe tongue atrophy among PWC dogs in Japan, pathologic examinations by using necropsy and biopsy samples from affected dogs and the detection of an autoantibody against canine muscular and nervous tissues were performed in the present study. The etiology of the disease and the role of autoantibody in the serum from the affected dogs are discussed.
Materials and methods
Cases
The clinical data, including canine breed, sex, age at onset, complete blood cell count, and biochemical profile, were obtained from the medical records of each veterinary hospital where the dogs were presented initially or secondarily. In all cases examined, 7 PWC dogs (dogs 1–7) had typical tongue and facial atrophy. In addition, 6 PWC dogs (dogs 8–13) without tongue atrophy, and 2 dogs (dogs 14 and 15; German Shepherd Dog and Papillon, respectively) with clinical and pathologic evidences of polymyositis were also examined. Magnetic resonance imaging (MRI) examinations were performed in dogs 1, 2, 4–10, and 15 to examine cranial lesions. Serum antibodies against Canine distemper virus and Toxoplasma gondii were examined only in dog 1; antibodies for acetylcholine receptor, nuclear antigens, and type 2M muscle fiber were examined in dogs 1–3 by commercial laboratories. Muscle biopsy was done in dogs 1, 5, 14, and 15, but the specimens of dog 5 were not available to examine. Necropsy was performed on 5 dogs (dogs 1–3, 12, and 13), whereas complete histopathologic examinations were performed only in 2 affected dogs (dogs 1 and 2) and 2 control dogs (dogs 12 and 13). Among 5 PWC dogs with typical tongue atrophy (dogs 3–7), complete clinical profiles, including hematologic and biochemical data and history of treatments, were not available from the medical records.
Tissue samples and histopathology
Two necropsy cases from PWC dogs (dogs 1 and 2) were examined histopathologically in the present study. Dog 1 was a 4-year-old female, and dog 2 was a 3-year-old female. Both had similar clinical histories of dysphagia and dyspnea because of severe tongue atrophy. At necropsy, tissue samples from the tongue; masticatory muscles, including the temporalis and masseter muscles, brachial, femoral, and oculomotor muscles; and nervous tissues, such as the brain, spinal cord, and trigeminal nerve, and almost all visceral organs were fixed with 10% neutral buffered formalin. In addition, biopsy samples from the temporalis, masseter, latissimus dorsi, and femoral muscles from dog 1 were also examined. Two additional necropsied PWC dogs with other disorders (dogs 12 and 13) and 2 biopsy samples from other breeds with polymyositis (dogs 14 and 15) were also examined histopathologically. For histopathologic examination, formalin-fixed tissues were routinely processed and embedded in paraffin. Paraffin sections of 2-μm thick were stained with hematoxylin and eosin (HE). Fresh muscular biopsy samples were stored at −80°C for the generation of cryostat sections, and cryostat sections of 10-μm thick were also stained with HE.
Serum
Seven serum samples from PWC dogs with severe tongue atrophy (dogs 1–7) were used for fluorescent antibody test (FAT) and Western blot (WB) for the detection and characterization of autoantibodies. As controls, sera from PWC dogs diagnosed with cervical carcinoma (dog 8), muscular atrophy of the temporalis muscle without tongue atrophy (dog 9), masticatory muscle atrophy (dog 10), immune-mediated anemia (dog 11), patent ductus arteriosus (dog 12), and multiple thrombosis (dog 13), and sera from a German Shepherd Dog and a Papillon with PM (dogs 14 and 15) were also examined. These samples were stored at 4°C until used.
Immunohistochemistry
Immunohistochemistry was performed by using a commercial polymer method. a As primary antibodies, anti-sera against human cluster of differentiation 3 a (CD3, 1:50), canine immunoglobulin G b (IgG, 1:200) and canine complement factor 3 b (C3, 1:200), and mouse monoclonal antibodies against human CD20 c (1:400), human B-lymphocyte antigen 36 a (BLA36, 1:100), and HLA-DR alpha-chain a (1:50) were used. Deparaffinized sections were autoclaved for antigen retrieval, then immersed in 0.3% H2O2 methanol to block intrinsic peroxidase activity, and finally in 8% skim milk to block nonspecific binding of the primary antibody. Paraffin sections from the lymph nodes and spleen from dog 12 were used as positive controls.
Cryostat sections were used for immunohistochemistry for laminin alpha 2 and dystrophin. Monoclonal antibodies against laminin alpha 2 d (1:10) and dystrophin e (1:100) were used. After the reaction with primary antibodies, the sections then were incubated with a biotinylated secondary antibody against mouse IgG f (1:200), incubated with peroxidase-labeled streptavidin, g and finally visualized with 3,3′-diaminobenzidine (DAB) and H2O2. Counterstaining was done with Mayer hematoxylin. Cryostat sections of the muscular tissues from dog 13 were used as positive controls.
Fluorescent antibody test
To detect autoantibodies against canine tissues, 10-μm thick cryostat sections were made from the tongue, masticatory muscles, femoral muscle, and esophagus of a necropsied PWC dog with multiple thromboses (dog 13). Cryostat sections were first washed in phosphate buffered saline solution (PBS), and then immersed in 12% skim milk. Sections were incubated at 37°C for 40 min with diluted (1:50) serum samples from all dogs (dogs 1–15). Fluorescent antibody test by using sera from dogs 8–13 and dogs 14 and 15 were performed as presumptive negative or positive controls, respectively. Sections were then incubated at 37°C for 20 min with a fluorescein isothiocyanate (FITC)-conjugated goat anti-canine IgG b (1:200) and subsequently observed by using a fluorescence microscope.
WB analysis
Samples from the tongue, spinal cord, spinal nerve root, and trigeminal nerve of a dog (dog 13) and those from the heart, liver, lung, stomach, kidney, spleen, tongue, masseter muscle, femoral muscle, and brain of a mouse (strain CF 1) were homogenized in 50 mM Tris—HCl buffer (pH 8.0) that contained 150 mM NaCl, 1 mM ethylenediamine tetra-acetic acid, 1% NP-40, 10 mM NaF, 2 mM Na3VO4, 0.1% sodium dodecyl sulfate (SDS), 0.5% diethyloxacarbocyanine and proteinase inhibitor cocktail. h Protein concentrations of the homogenates were determined by Lowry method by using a DC protein assay kit. i Homogenates with equal amounts of protein were mixed in equal amounts with 5% 2-hydroxyethylmercaptan and β-mercap-toethanol in Laemmli sample buffer i and heat treated. Then, homogenates were loaded onto a 10% SDS-polyacrylamide gel electrophoresis gel, electrophoresed, and transferred to a polyvinylidene fluoride membrane. i The membranes were washed in 0.1% Tween 20 in Tris buffered saline solution then probed with serum samples from all 15 dogs (diluted 1:100–1:10,000) at room temperature for 60 min. The analysis by using sera from dogs 8–13 and dogs 14 and 15 was done as presumptive negative or positive controls, respectively. After incubation with horseradish peroxidase—conjugated goat anti-canine IgG b at room temperature for 60 min, positive reactions were visualized with DAB or enhanced chemi-luminescence (ECL) plus WB detection reagent. j
Two-dimensional gel electrophoresis and matrix-assisted laser ionization time-of-flight mass spectrometry analysis
The samples from the tongue muscle of dog 13 were treated and then homogenized in lysis buffer (8 M urea, 4% 3-[(3-cholamidopropyl)dimethylammonio]-1-propanesulfonate [CHAPS], and 40 mM Tris—base). The proteins were separated by the 2-dimensional (2D) gel electrophoresis. The pH 3–10, 7-cm immobiline pH gradient (IPG) dry-strips j were rehydrated in sample buffer (8 M urea, 2% CHAPS, 0.002% bromophenol blue, 20 mM dithiothreitol [DTT], and 2% IPG buffer) that contained sample proteins, and then isoelectric focusing (IEF) was carried out. After equilibration of the IEF strips, the gel strips were placed on 10% polyacrylamide gels, and 2D gel electrophoresis was performed. Then, the gels were used for The WB analysis by using a serum sample from dog 1, and, in parallel, those were also stained by Coomassie brilliant blue. The immunopositive-protein spots by WB analysis were used for matrix-assisted laser ionization time-of-flight mass spectrometry (MALDI-TOF MS) analysis, which was carried out by a commercial company. k
Results
Clinical signs and histopathology of biopsy specimens
Among 7 PWC dogs (5 females and 2 males) with typical tongue atrophy, the mean (SD) age at clinical onset was 3.4 ± 2.14 years (Table 1). No familial relationships among these 7 dogs were recognized.
Dog 1 had severe atrophy of the tongue and temporalis muscles, hanging of the lip downward, severe drooling, and dysphagia for 2 years (Fig. 1). Serum creatine kinase (CK) concentration was 570 U/l (normal: <200 U/l) at presentation. The level of thyroid hormone was within the normal range. Serum antibodies against Canine distemper virus, T. gondii, acetylcholine receptor, antinuclear antigens, and type 2M fibers were negative. MRI examination revealed hyperintensive areas on T2-weighted and fluid attenuated inversion recovery images in the right occipital and temporal muscles. These areas were enhanced by contrast agent and were almost intact on T1-weighted images. No abnormalities were found in the brain. Based upon these findings, myositis affecting masticatory muscles area was suspected. Dog 1 had been treated with immunosuppressive therapy for 2 years and died of aspiration pneumonia. Histopathologic examinations of the biopsy specimens from the femoral muscle of dog 1 revealed moderate infiltration of mononuclear cells with mild muscular atrophy (Fig. 2). In the temporalis and masseter muscles, inflammatory changes were minimal, and there also was moderate muscular atrophy, with an increase in satellite cells and interstitial fibrosis. Immunohistochemistry for dystrophin and laminin alpha 2 revealed intact distribution of these molecules in the sarcolemma of the affected dog. Therefore, the case was diagnosed as inflammatory myopathy.
Dog 2 also had severe atrophy of the tongue and mild atrophy of the temporalis muscle. High levels of serum CK (1700 U/l) and aspartate aminotransferase (AST; 221 U/T) were recorded. By MRI examination, no significant lesions were found in the brain, but edema, inflammation, and fat replacement in the ventral part of the temporal muscle and adjacent area were found. Therefore, multiple cranial neuropathy was suspected. Antibodies for type 2M fiber and acetylcholine were negative. The details have been described in a previous report. 17 An immune-modifying dose (2 mg/kg per day) of prednisolone was prescribed for 3 weeks after the first presentation. Then, further treatment with prednisolone (1 mg/kg per day) for 6 weeks was done; meanwhile the tongue atrophy did not improve. The dog died of dysphagia and dyspnea 2 years after the first presentation.
Summary of clinical and pathologic features of dogs examined in the current study. *
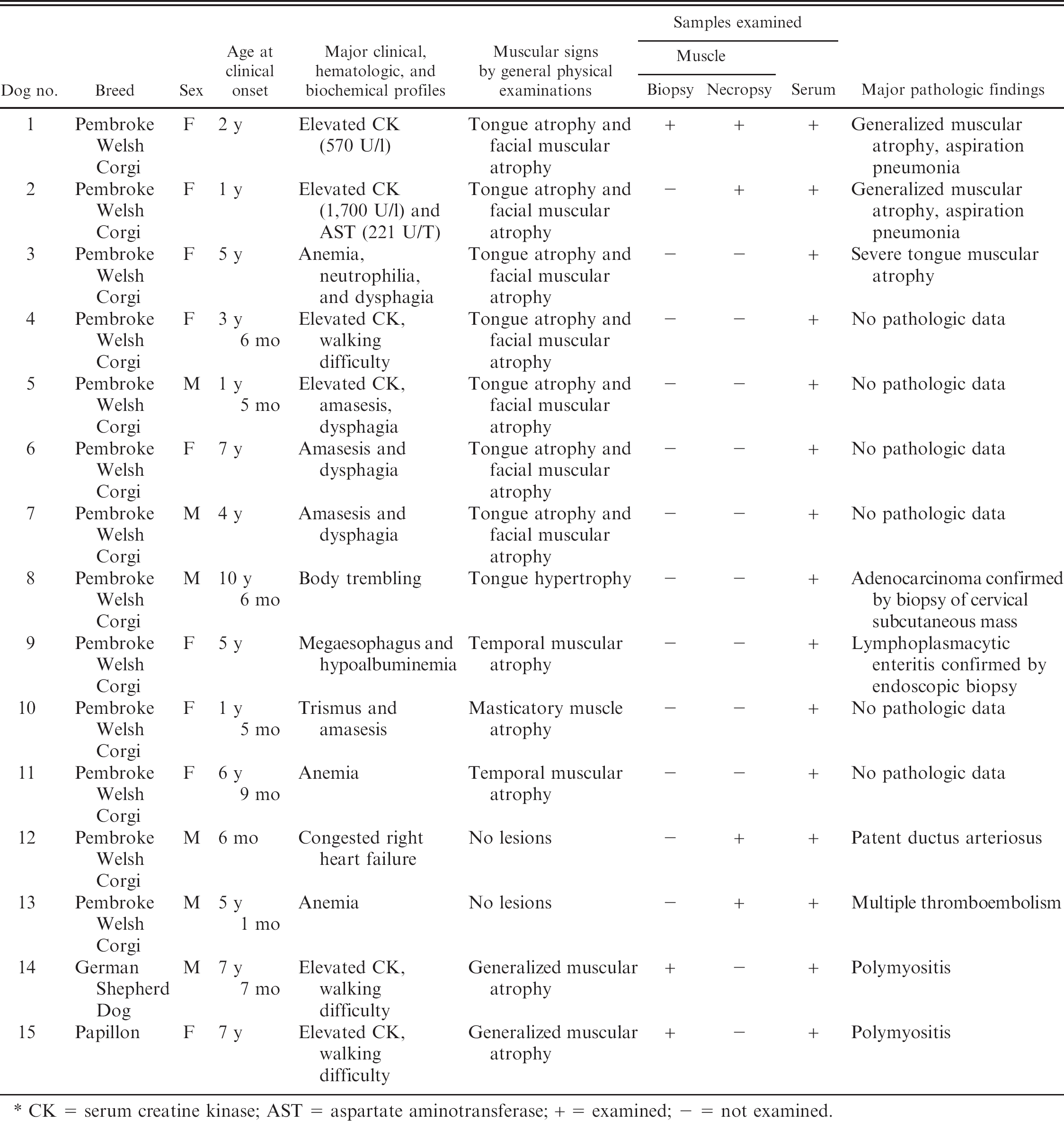
CK = serum creatine kinase; AST = aspartate aminotransferase; + = examined; − = not examined.
Pathologic findings
At the necropsy of dog 1, almost all muscles exhibited severe-to-moderate atrophy. Histologic examinations revealed severe atrophy of the tongue muscle fibers, with diffuse fat infiltration (Fig. 3). Moderate-to-severe atrophy of muscle fibers with mild interstitial fibrosis also was observed in the other muscles, including the extraocular muscles. Inflammatory changes were mild in all the muscles examined. There were no significant lesions in the nervous tissues, including the brain, spinal cord, trigeminal nerve, and intramuscular nerve branches. No pathologic lesions were observed in other visceral organs except for severe suppurative pneumonia.
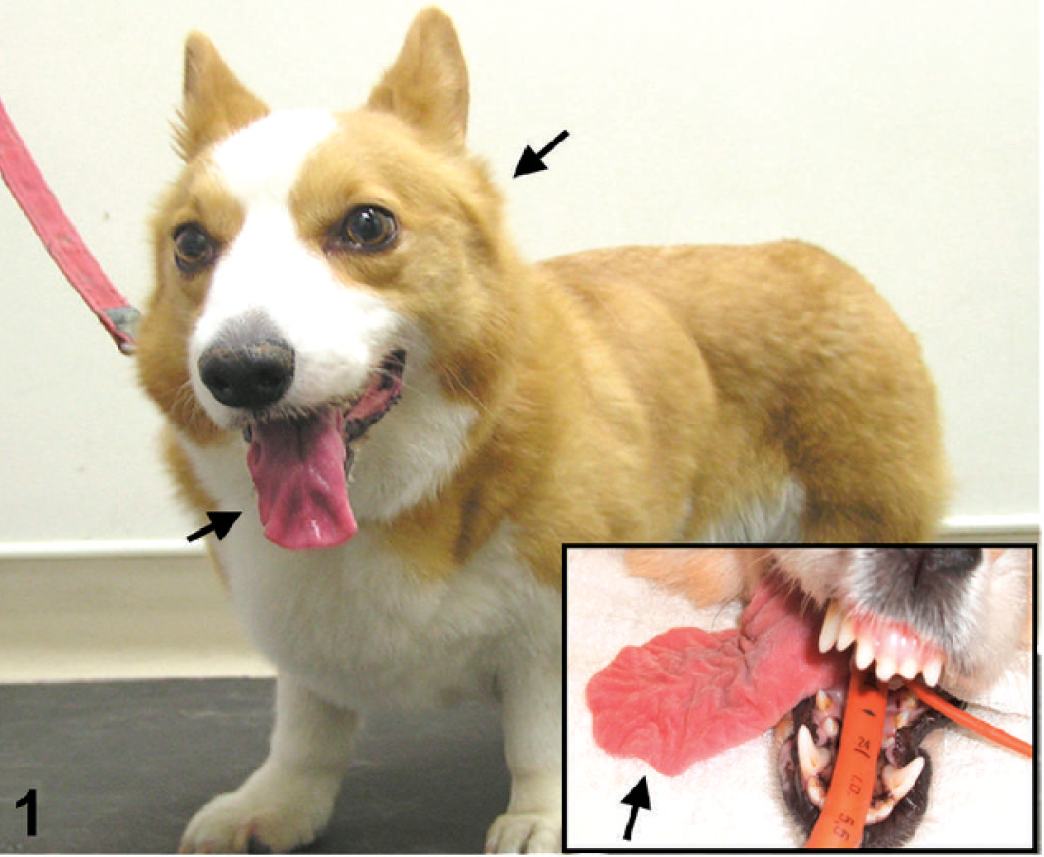
A Pembroke Welsh Corgi (PWC) dog (dog 2) exhibiting tongue and temporal muscular atrophy (arrows). Inset: severe tongue atrophy (arrow) in the other PWC dog (dog 1).

Femoral muscle. Pembroke Welsh Corgi dog (dog 1). Biopsy sample with moderate to severe infiltration of mononuclear cells with mild muscular atrophy. Hematoxylin and eosin. Bar = 200 μm.

Tongue. Pembroke Welsh Corgi dog (dog 1). Severe muscular atrophy with diffuse fatty infiltration. Hematoxylin and eosin. Bar = 500 μm.

Tongue. Pembroke Welsh Corgi dog (dog 2). Severe muscular atrophy with mononuclear cell infiltration, fatty infiltration, and fibrosis. Hematoxylin and eosin. Bar = 500 μm.

Femoral muscle. Pembroke Welsh Corgi dog (dog 2). Moderate mononuclear cell infiltration with moderate muscular atrophy and mononuclear cell infiltration. Hematoxylin and eosin. Bar = 100 μm.

Tongue. Pembroke Welsh Corgi dog (dog 2). Immunohistochemistry for canine immunoglobulin G (IgG;
Histopathologic examination after necropsy of dog 2 revealed severe multifocal to diffuse mononuclear cell infiltration in the tongue with severe muscular atrophy and diffuse infiltration of fatty tissues (Fig. 4). In the lesion, atrophic muscle fibers sometimes had central-chained nuclei, and the number of satellite cells increased. Intramuscular nerve branches in the tongue were well preserved. There also was moderate mononuclear cell infiltration in the femoral muscle, whereas, atrophy of muscle fibers and fat infiltration were minimal (Fig. 5). The masticatory muscles, including the temporalis, masseter, and brachial muscles, exhibited very mild inflammatory lesions, with mild muscular atrophy and interstitial fibrosis. The brain, spinal cord, trigeminal nerve, and their branches, and intramuscular peripheral nerves were intact.
Immunohistochemistry for CD3, CD20, BLA36, and HLA-DR revealed that the mononuclear cells infiltrating the tongue of dog 2 and the femoral muscle of dogs 1 and 2 were predominantly B cells and histiocytes. Few CD3-positive T cells were scattered in the lesions. The cross striation of these muscle fibers in both dogs was positive for canine IgG and C3, which suggests the deposition of these molecules within the structure (Fig. 6).
Detection of serum autoantibody by FAT
The presence of serum autoantibodies against muscular tissues was examined by using sera from 7 cases of affected PWC dogs and 8 control dogs. The FAT, by using normal canine tongue tissue as the antigen and FITC-conjugated antibody against canine IgG without test sera, revealed nonspecific green fluorescence signals on the sarcolemma. The FAT revealed significant specific fluorescence on cross striations in addition to signals on the sarcolemma when 5 serum samples from PWC dogs with typical tongue atrophy (dogs 1, 2, 4–6) and 1 sample from a control PWC dog (dog 8) were applied (Fig. 7). However, no or only nonspecific sarcolemmal signals were observed in the other 7 and 2 samples from control and affected dogs (dogs 3 and 7), respectively (Table 2).
Identification and characterization of autoantibody by WB analysis
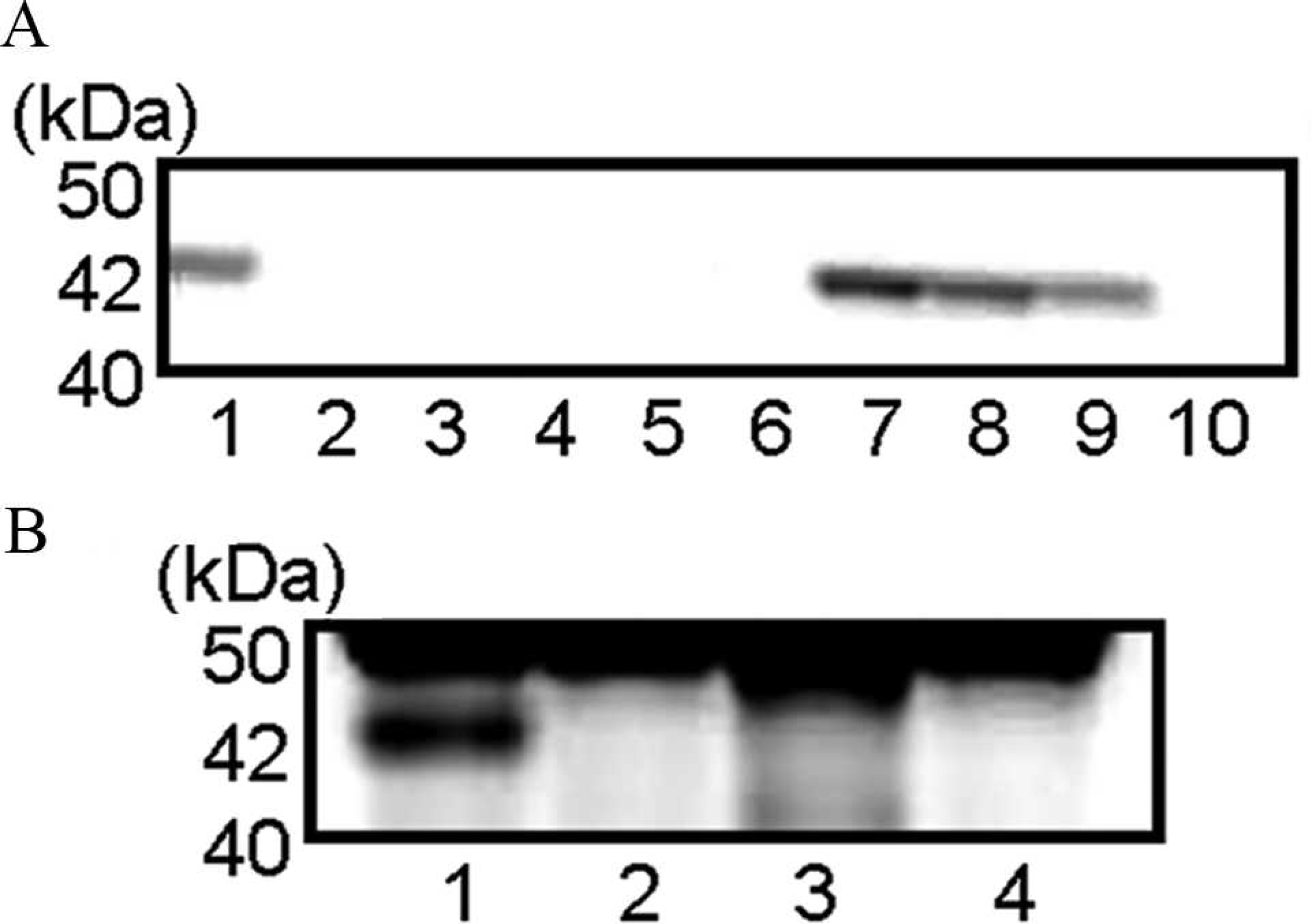
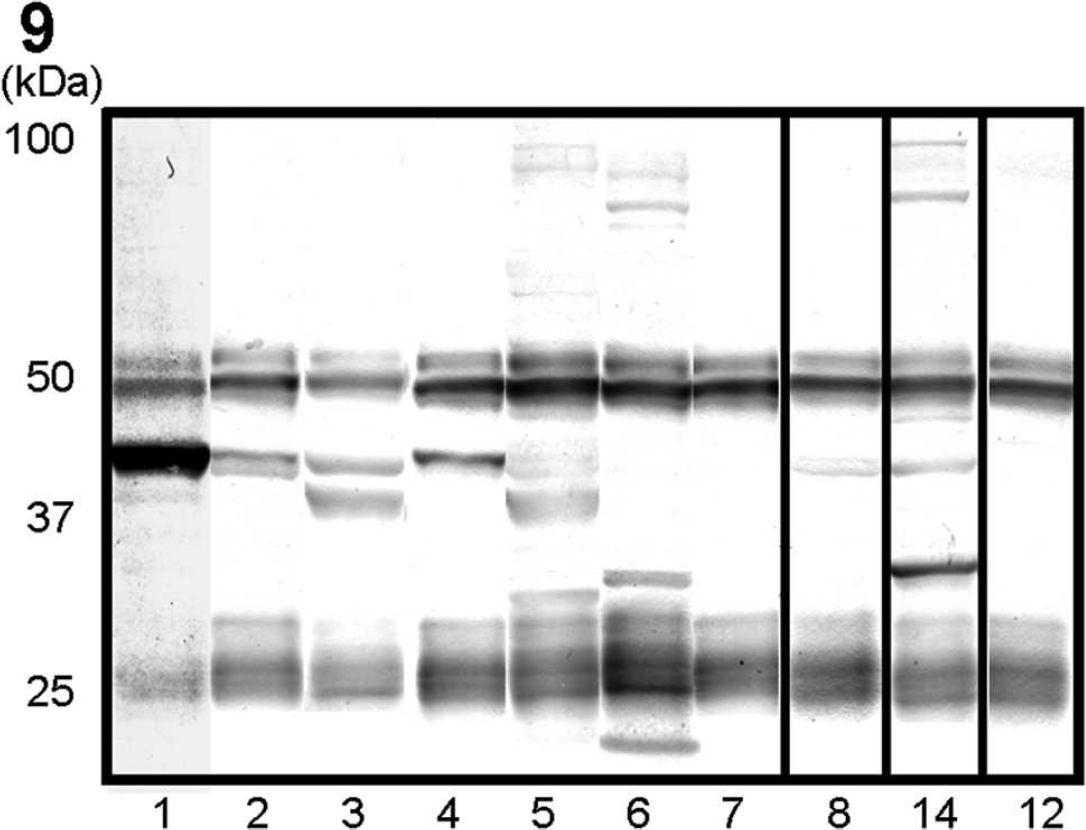
The WB analysis by using mouse tissue antigens and the serum from dog 1 revealed that the serum recognized an antigen of 42 kDa in the heart, tongue, and masseter and femoral muscles, which suggests that an autoantibody in the serum reacts with mouse striated muscles (Fig. 8A). Furthermore, The WB analysis by using canine antigen prepared from the tongue of a control dog and the serum from dog 1 revealed intensely immunopositive bands at 42 kDa in the samples from the tongue, masticatory, and femoral muscles, and the esophagus. Several weak immunopositive bands at 25 kDa and 50 kDa were also found but were considered to be nonspecific reactions. The significant reaction was confirmed only in the striated muscles and not in nervous tissues, including the medulla, spinal cords, spinal nerve roots, and trigeminal nerves (Fig. 8B). The results of WB to examine autoantibodies against tongue muscle tissues are shown in Figure 9 and Table 2. Sera from 6 of 7 PWC dogs with tongue atrophy showed positive results. Single immunopositive bands at 42 kDa (dogs 1, 2, and 4), double positive bands at 37 and 42 kDa (dogs 3 and 5), and triple positive bands at 15, 30, and 83 kDa (dog 6) were detected. However, there were no specific bands in the sample from dog 7. No specific immunopositive bands were detected in sera from 6 (dogs 9–14) of 8 control dogs (dogs 8–15). A single positive band at 42 kDa was detected in the serum from dog 8. Moreover, in serum from a German Shepherd Dog with PM (dog 14), multiple positive bands at 30, 42, 85, and 100 kDa were recognized.
Identification of autoantigens by MALD-TOF MS analysis
By 2D gel electrophoresis and WB analysis by using 42-kDa proteins from the tongue muscle of a control dog and the serum from dog 1, the intense immunopositive spot was detected. The MALDI-TOF MS analysis revealed that the proteins contained CK, mitochondrial 2 (sMtCK) isoform 4, and ubiquinolcytochrome-C reductase complex core protein 2 (UQCRC2) and mitochondrial precursor (Complex III subunit II) isoform 1.
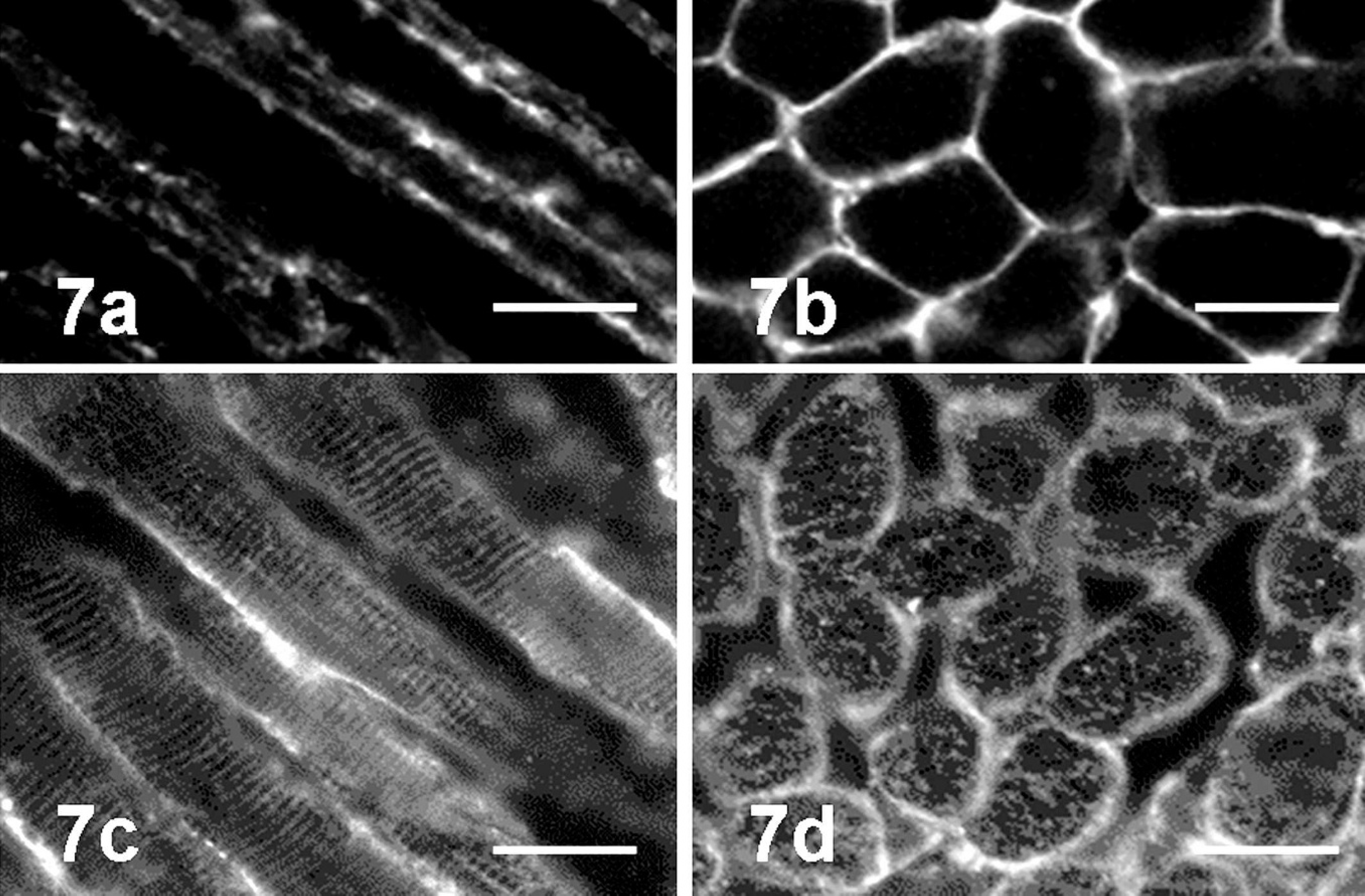
Fluorescent antibody test by using normal canine tongue tissue as antigen. In control serum (dog 12,
Results of fluorescent antibody test (FAT) and Western blot (WB) analysis. *
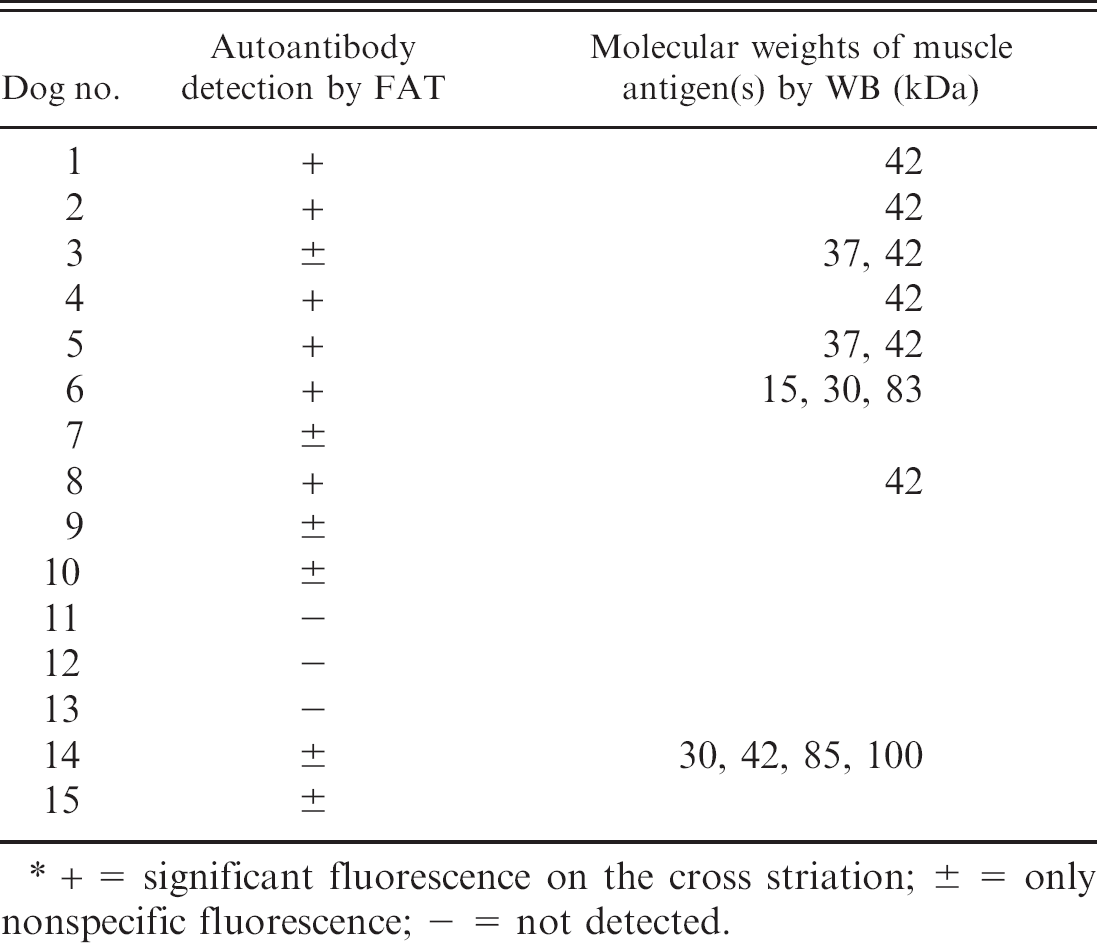
+ = significant fluorescence on the cross striation; ± = only nonspecific fluorescence; − = not detected.
Discussion
The present study revealed the pathologic features of a unique disease in PWC dogs with severe tongue muscular atrophy. The disease was initially suspected to be neurogenic because the clinical signs were suggestive of cranial nerve disturbances. However, the results of the present study indicated that the disease was an immune-mediated inflammatory myopathy. Because the canine syndrome has been recognized sporadically only in PWC dogs in Japan, some inherited basis has been suspected on the etiology of the canine disease. However, any evidence of familial relationship among affected PWC dogs was not confirmed. The mean (SD) age was 3.4 ± 2.14 years old, which suggests a predominant occurrence in young adult PWC dogs. Most apparent biochemical profile was elevated titer of serum CK in both dog 1 and 2, which indicates the injury of skeletal muscles. On pathologic examination, the lesions in the tongue muscle were most severe in 2 affected PWC dogs examined, whereas other muscles, including the femoral muscles, were also involved. The results might explain the clinical signs that affected PWC dogs exhibit (mild-to-moderate walking difficulty together with severe tongue and facial atrophy). The presence of significant inflammatory changes in the tongue and femoral muscles in the early onset of the disease and intact distribution of dystrophin and laminin alpha 2 indicated that major muscular dystrophies were not likely for the present muscular disease in PWC dogs. Serum antibody against acetylcholine receptor—detected dogs with myasthenia gravis was negative in affected dogs (dogs 1–3). Complete pathologic examinations of 2 necropsied dogs revealed no significant lesions in either the central or peripheral nerve tissues, including the cranial nerves. These findings support the interpretation that the present muscular change in PWC dogs is not neurogenic. A degenerative myelopathy has recently been reported in PWC dogs 19 ; however, the clinical symptoms, age of onset, and pathologic lesions are quite different from those observed in the affected dogs described in the current report.
Although MMM, EOM, PM, and DM are known as immune-mediated myopathies in dogs, the clinicopathologic features of the disorders are quite different from those of the present disease in PWC dogs. Canine MMM selectively affects the muscles of mastication, and the autoantibody generated in MMM dogs recognizes a unique type 2M muscle fibers. 23 The autoantibody in MMM recognizes a 150-kDa protein that is a member of myosin-binding protein-C family, masticatory myosin-binding protein-C. 40 This protein is specifically included in masticatory muscle type 2M fibers. In canine MMM, an enzyme-linked immunosorbent assay to confirm the presence of an autoantibody by using mastication muscle myosin as the antigen is currently used as a routine diagnostic test. In the present study, serum antibody against type 2M fibers was negative in 2 affected dogs (dogs 1 and 2) examined. This finding suggests that the autoantibody generated in MMM was different from the muscular atrophy of PWC dogs.
Western blot analysis by using canine antigen prepared from a normal tongue. Sera examined were from dogs 1–7 (Pembroke Welsh Corgi [PWC] dogs with tongue atrophy), dog 8 (PWC dog diagnosed with cervical carcinoma), dog 12 (PWC dog with patent ductus arteriosus), and dog 14 (German Shepherd Dog with polymyositis). The lane number corresponds to each dog number. A single specific band at 42 kDa was recognized in lanes 1, 2, 4, and 8; double bands at 37 and 42 kDa in lanes 3 and 5; triple bands at 15, 30, and 83 kDa in lane 6; and quadruple bands at 30, 42, 85, and 100 kDa only in lane 14. There were no specific bands in lanes 7 and 12.
In human PM, some autoantibodies were detected, but these antibodies were also detected in other myositis, such as DM, and in other immune-mediated disease, such as rheumatoid arthritis. 7,12,16 These autoantibodies recognize components of the translational system, such as transfer RNA synthetase or signal recognition particles, or components of the nucleosome remodeling complex, such as MI-2, and these proteins are not muscle specific. In canine PM, a sarcolemma-specific autoantibody has been identified, particularly in Boxer and Newfoundland dogs. 14 The autoantibody found in PWC dogs with myopathy in the present study recognized cross striations of the skeletal muscle, which indicates that the autoantibody in PWC dogs recognizes a different antigen from those in canine PM. Therefore, it was concluded that the myopathy in PWC dogs is a novel immune-mediated disease that belong to a different category than canine MMM, PM, DM, and EOM.
However, the pathologic and immunohistochemical features of the muscle lesions in affected PWC dogs had some similarity to those of canine PM and MMM. The generalized muscle involvement and immunoreactivity of the autoantibody against all striated muscles were consistent with those in canine PM, whereas severe tongue muscle involvement is unusual. On immunohistochemical examination, HLA-DR—positive cells (macrophages or dendritic cells), CD20, and BLA36-positive cells (B cells) were dominant in the muscular lesions of affected PWC dogs. Previous reports demonstrated that the dominant inflammatory cells in canine MMM were also B cells, dendritic cells, and macrophages, whereas those in canine PM were T cells and macrophages. 6,26,28,34 Thus, the inflammatory pattern of the muscular lesions in PWC dogs was different from that in canine PM but was similar to that in MMM.
In the present study, an autoantibody against cross striations of the skeletal muscle was found in the sera from PWC dogs, with typical clinical signs. Because the myopathy in PWC dogs, especially tongue lesions, might be irreversible, early diagnosis is very important to prevent the progression of the disease. In dog 1, muscular biopsy revealed inflammatory changes characterized by lymphohistiocytic infiltration in the muscles examined, whereas, such inflammatory lesions were not prominent at necropsy after 2 years with aggressive immunosuppressive therapy. The serum of dog 1 obtained at muscular biopsy had the significantly highest titer of autoantibody by both FAT and WB among all tested sera (data not shown). In contrast, dog 2, with significant inflammatory muscular changes at necropsy, had no aggressive immunosuppressive therapy for approximately 1 year before necropsy, although the case had a history of prednisolone treatment for a total of 9 weeks after initial presentation at the veterinary hospital. All these findings suggest that the initial lesions of the disease in PWC dogs are inflammatory muscular changes mainly located in the tongue and facial muscles and that those inflammatory changes might be relapsing without effective immunosuppressive therapy. Fatty infiltration and interstitial fibrosis in the affected muscles observed at necropsy of dog 1 might represent end stage of the muscular lesions. In addition, some cases with typical lesions did not have a significant amount of detectable autoantibody by FAT (dogs 3 and 7) and/or WB (dog 7). The use of immunosuppressive treatments may explain the negative results in these cases. It suggests that immunosuppressant therapy was partly effective to prevent the prognosis of the muscular lesions and that detection of an autoantibody in the serum should be done before treatment to diagnose myopathy in PWC dogs.
The present autoantibody recognized cross-striation antigens in muscle fibers. By MALD-TOF MS analysis, the target proteins were identified as sMtCK and UQCRC2. A sMtCK is located in mitochondria and plays a central role on energy transduction. A UQCRC2 is part of the mitochondrial respiratory chain and plays a major part in the mitochondrial respiration. Because sMtCK is known as a sarcomere-specific protein in human, 13,38 it is highly possible that the molecule is the major antigen of the autoantibody. Therefore, the muscular lesions in PWC dogs were widespread in various locations in striated muscles. However, the tongue was most severely affected clinically and pathologically. Different distributions of the molecule in canine muscles may reflect the different severities of the lesions. Because there have been no reports concerning the distribution pattern of sMtCK and UQCRC2 in canine muscular tissues, further studies that use the antibodies against sMtCK and UQCRC2 will be needed.
Concerning the mechanism of autoantibody production, it is supported that the combination of inherited background and environmental factors, such as infection and injuries, might be the triggers of autoantibody production. 37 In these courses, the target molecules in tongue muscles are exposed, which induces the immune response for autoantibody production, and leads to the spread of the lesions into other systemic muscles.
Footnotes
a.
Envision®, Dako Japan, Kyoto, Japan.
b.
Bethyl Laboratories Inc., Montgomery, TX.
c.
Thermo Fisher Scientific Inc., Fremont, CA.
d.
Kindly provided by Dr. Eva Engvall, Department of Pathology, University of California San Diego, La Jolla, CA.
e.
Chemicon International Inc., Temecula, CA.
f.
Kirkegaard & Perry Laboratories, Gaithersburg, MD.
g.
Dako Denmark A/S, Glostrup, Denmark.
h.
Roche Applied Science, Penzberg, Germany.
i.
Bio-Rad Laboratories, Hercules, CA.
j.
Amersham, Buckinghamshire, United Kingdom.
k.
APRO life Science Institute Inc., Tokushima, Japan.
