Abstract
An 11-year-old, male, neutered crossbred Collie dog was presented for a history of polydipsia and polyuria. Diagnostic investigations revealed total and ionized hypercalcemia and an increased concentration of parathyroid hormone–related peptide. Abdominal ultrasonography and contrast-enhanced computed tomography of the abdomen revealed a right-sided, cystic-appearing renal mass. Cytological examination of ultrasound-guided aspirates of the mass revealed high numbers of spindle cells. The mass was removed en bloc via an ureteronephrectomy. Histopathological examination of the mass revealed neoplastic spindle cells in loosely packed and interlacing streams within a myxomatous stroma. Immunohistochemical examination with vimentin, von Willebrand Factor, and α-smooth muscle actin confirmed the mass to be a renal angiomyxoma. A minority of the neoplastic spindle cells showed positive cytoplasmic parathyroid hormone–related peptide immunostaining. The hypercalcemia resolved following surgery, and the parathyroid hormone–related peptide concentration returned to within the reference interval. The dog was no longer polydipsic or polyuric 1 year following surgery. The present report describes a previously unreported renal neoplasm causing paraneoplastic hypercalcemia and highlights the possibility of paraneoplastic hypercalcemia being caused by a benign neoplasm.
An 11-year-old, male, neutered crossbred Collie dog was presented to the Queen Mother Hospital for Animals (Royal Veterinary College, Hatfield, Hertfordshire, United Kingdom) with a 4-week history of polydipsia and polyuria. In the preceding weeks, the dog had also become progressively lethargic and inappetent. Serum biochemical analysis performed prior to referral revealed increased concentrations of total calcium (15.4 mg/dl, reference [ref.] interval: 7.9–12 mg/dl; 3.85 mmol/l, ref. interval: 1.98–3.0 mmol/l) and ionized calcium (6.4 mg/dl, ref. interval: 4.5–5.3 mg/dl; 1.61 mmol/l, ref. interval: 1.25–1.45 mmol/l). The concentration of parathyroid hormone (PTH)-related peptide (PTHrp) was also increased (1.8 pmol/l, ref. interval: <0.5 pmol/l; >1.0 pmol/l suggestive of paraneoplastic production). Serum PTH concentration was within normal limits (42 pg/ml; ref. interval: 18–130 pg/ml), and an adrenocorticotropic hormone (ACTH) stimulation test provided no evidence for hypoadrenocorticism (basal cortisol 3.6 μg/dl, ref. interval: 0.9–4.5 μg/dl; 98.2 nmol/l, ref. interval: 25–125 nmol/l, and post-ACTH cortisol 18.9 mg/dl, ref. interval: 4.5–18.8 μg/dl; 521 nmol/l, ref. interval: 125–520 nmol/l).
On examination, the dog was quiet but alert and responsive, well hydrated, and in good body condition. Marked bilateral temporal and masseter muscle atrophy was noted, but there was no restriction of the range of motion of the temporomandibular joint. Cranial nerve examination was normal. Mild bilateral superficial cervical lymphadenomegally was noted; however, the remainder of the physical examination, including rectal and fundic examinations, was unremarkable. A venous blood gas and electrolyte panel confirmed moderate ionized hypercalcemia (6.36 mg/dl or 1.59 mmol/l). A complete blood cell count revealed mild lymphopenia (0.82 × 10 9 /liter; ref. interval: 1–4.8 × 10 9 /liter), which was attributed to physiologic stress. A serum biochemical profile revealed marked total hypercalcemia (15.6 mg/dl or 3.9 mmol/l) and mild hypophosphatemia (2.6 mg/dl, ref. interval: 2.7–6.7 mg/dl; 0.77 mmol/l, ref. interval: 0.8–2 mmol/l), both of which were attributed to the raised PTHrp concentration. Urinalysis (within 1 hr of collection) of a cystocentesis sample revealed a refractometric specific gravity of 1.026. Manual dipstick analysis a revealed 3+ proteinuria, 3+ bilirubinuria, and trace hematuria; however, the urine sediment examination was unremarkable. Urine microbial culture (agar disk diffusion method) was negative for bacterial growth. The proteinuria and hemoglobulinuria were attributed to the renal mass. Cytological examination of fine-needle aspirates of the superficial cervical lymph nodes was consistent with reactive lymphoid hyperplasia. Orthogonal thoracic radiographs and lateral lumbar spinal radiographs were unremarkable. Abdominal ultrasonography revealed a large (5.6 cm in diameter), fairly well marginated, heterogeneous mass in the caudal pole of the right kidney (Fig. 1). The mass contained an anechoic (cyst-like) component, with septa. Ultrasound-guided aspiration of the renal mass and the cystic component was performed. Aspirate smears and direct and cytospin preparations of the fluid were stained with a modified Wright–Giemsa stain on an automated stainer. b Cytological examination of the renal aspirates revealed abundant sheet-like arrangements of cells exhibiting mild anisocytosis and anisokaryosis (Fig. 2). Cells were predominantly spindle in appearance with low amounts of wispy basophilic cytoplasm and central oval nuclei with smooth chromatin. Clusters of neutrophils consistent with neutrophilic inflammation, and eosinophilic and blue-gray debris consistent with necrosis, were also observed. The cyst yielded a fluid of low cellularity (0.5 × 10 9 total nucleated cell count), with low numbers of mixed leukocytes admixed with erythrocytes. Macrophages exhibiting erythrophagia and containing heme pigment indicative of prior hemorrhage were also observed. The background was pale, eosinophilic-appearing proteinaceous fluid (fluid protein 58 g/l or 5.8 g/dl). Bacterial culture of the fluid was negative for growth. Computed tomography (CT) of the abdomen revealed that the right-sided renal mass was heterogenous on precontrast and postcontrast images and included 2 small, mineralized foci. Part of the mass was homogeneous and iso-attenuating to water (Hounsfield units = 5) in precontrast images (Fig. 3A) and showed no contrast enhancement in either the early or late phases (Fig. 3B, 3C). The remainder of the mass was heterogeneous and soft-tissue attenuating (Hounsfield units = 40) in precontrast images (Fig. 3A), showing a vascular component in the early phase of postcontrast (Fig. 3B) and heterogeneous contrast enhancement in the late phase (Fig. 3C). The renal vasculature and ureters were preserved bilaterally, but mild medial iliac lymphadenomegaly was observed. Thoracic CT images revealed no evidence of pulmonary metastases. Echocardiography was unremarkable.
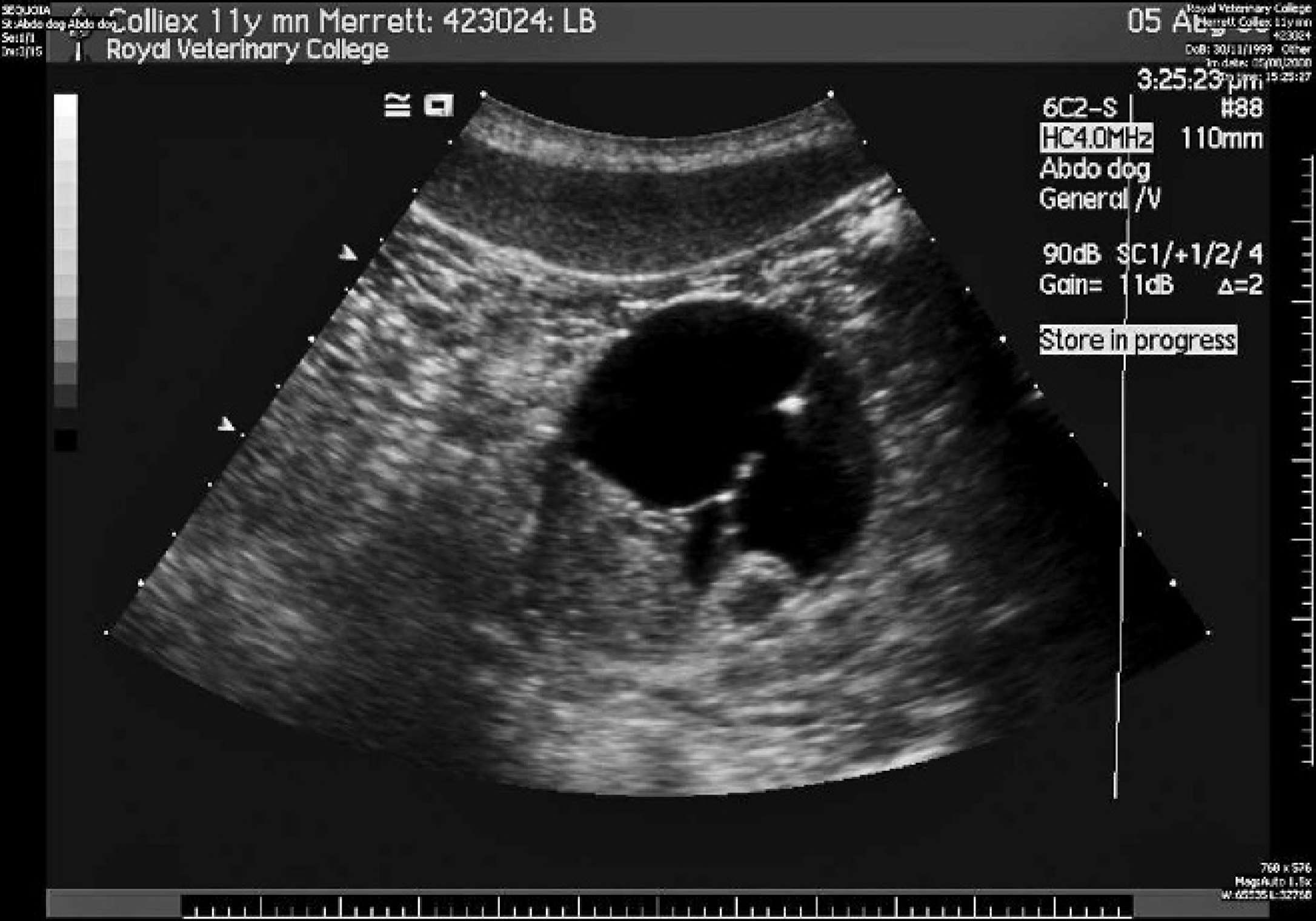
Ultrasound image showing a mass on the caudal pole of the right kidney. The mass has an anechoic (cyst-like) component with septa and a component isoechoic to the renal cortex.
The dog's hypercalcemia was managed with crystalloid intravenous fluid therapy (0.9% NaCl with added KCl c as required) at rates of up to 6 ml/kg/hr. Furosemide d (2 mg/kg intravenously every 8–12 hr) was also administered. An exploratory laparotomy revealed an enlarged and irregular right kidney with a 4 cm × 4.5 cm × 4 cm mass raising the capsule at the caudal pole. Grossly, the mass appeared to be contained within the renal capsule, and a right-sided ureteronephrectomy was therefore performed. The kidney and mass were fixed in 10% neutral buffered formalin and submitted for histopathological examination. Following fixation, the kidney was sectioned longitudinally to reveal a soft, well-demarcated, cream-colored, partially cystic mass with mottled red areas occupying the cortex and medulla and extending to the pelvis. The tissue was embedded in paraffin, sectioned at 4 μm, and stained with hematoxylin and eosin. Microscopic examination revealed a well-demarcated, nonencapsulated, and sparsely cellular neoplastic mass replacing the cortex and medulla. Neoplastic spindle cells were arranged in loosely packed and occasionally interlacing streams within an abundant myxomatous stroma (Fig. 4A, B). Neoplastic cells had indistinct borders, a scant-to-moderate amount of weakly eosinophilic fibrillar cytoplasm, and a single spindloid-to-fusiform nucleus with indistinct nucleoli. Anisocytosis, anisokaryosis, and cellular pleomorphism were mild, and no mitotic figures were observed in ten 40× fields. Multifocally within the mass, there were variably sized, thin-walled blood vessels; large areas of necrosis; and occasionally entrapped renal tubules and glomeruli. The overlying renal capsule was mildly thickened.
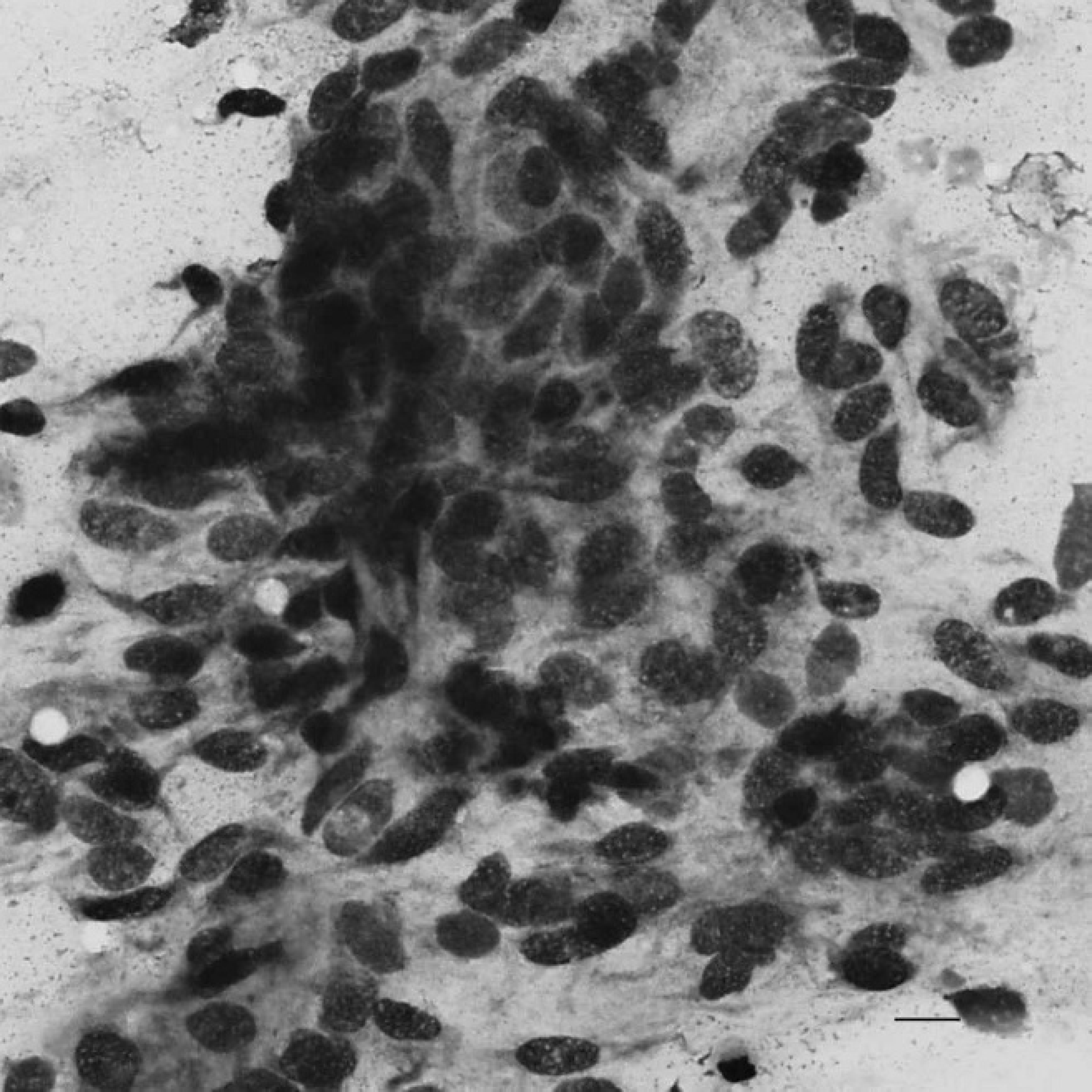
Cytological appearance of ultrasound-guided fine-needle aspirates of the renal mass. Modified Wright–Giemsa. 400×. Bar = 10 μm.
Immunohistochemical stains for vimentin, von Willebrand Factor (vWF), and α-smooth muscle actin (α-SMA) were performed. e , f Briefly, endogenous peroxidases were blocked with peroxidase-blocking solution e for 15 min at room temperature. Antigen retrieval was performed using 0.05% protease XIV g at 37°C for 15 min (vWF) or citrate antigen unmasking solution h in a steamer for 15 min (vimentin and α-SMA). Slides were incubated with primary antibody at the following dilutions: vimentin g at 1:500, α-SMA i at 1:50, or vWF e at 1:50. 3,3-diaminobenzidine tetrahydrochloride e was used as a chromogen, and sections were counterstained with hematoxylin.
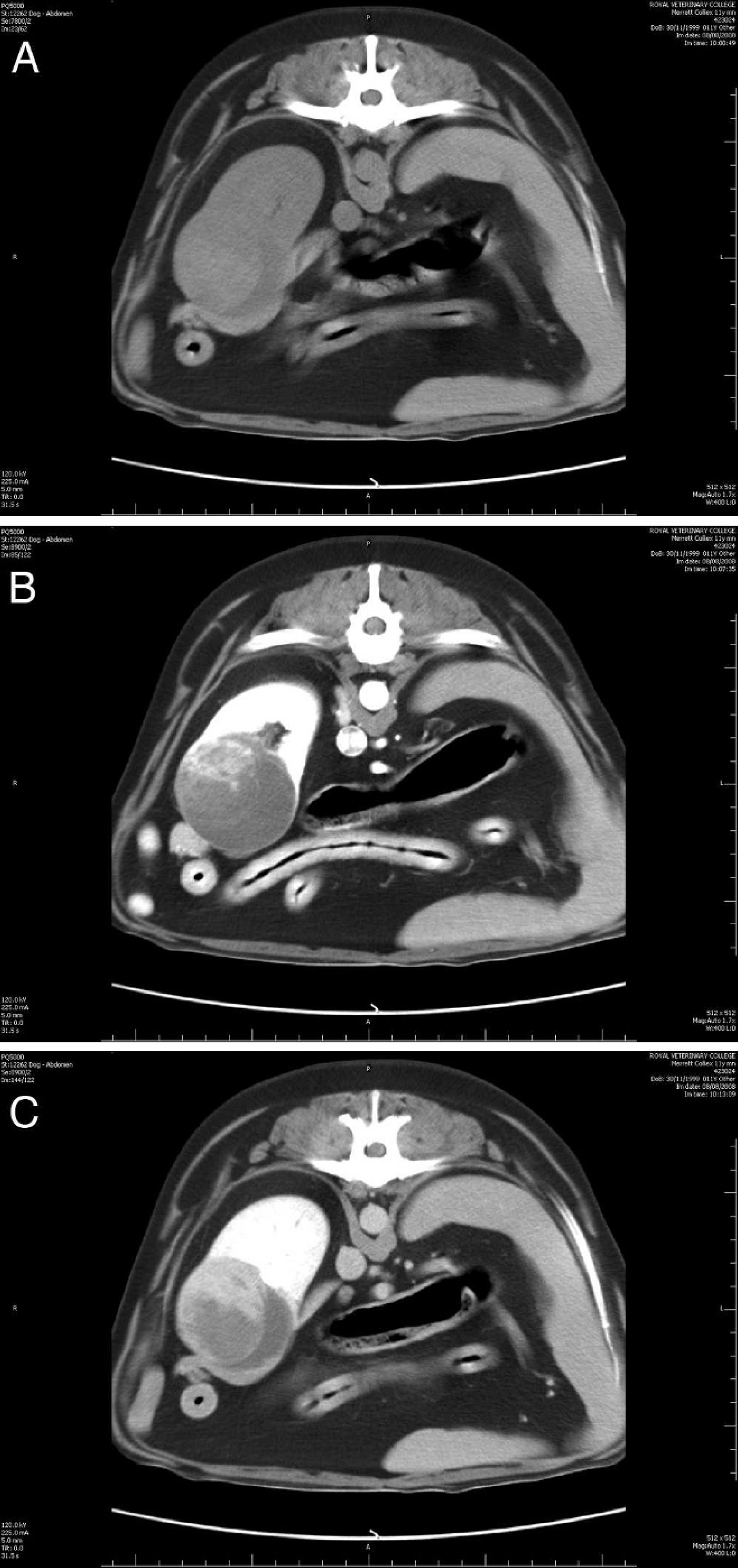
precontrast computed tomographic image of the right kidney. There is a heterogeneous mass deforming the caudal pole of the right kidney ventrally. Two areas are distinguished within the mass, one of similar attenuation to water (Hounsfield units = 5) and one of similar attenuation to blood (Hounsfield units = 40).
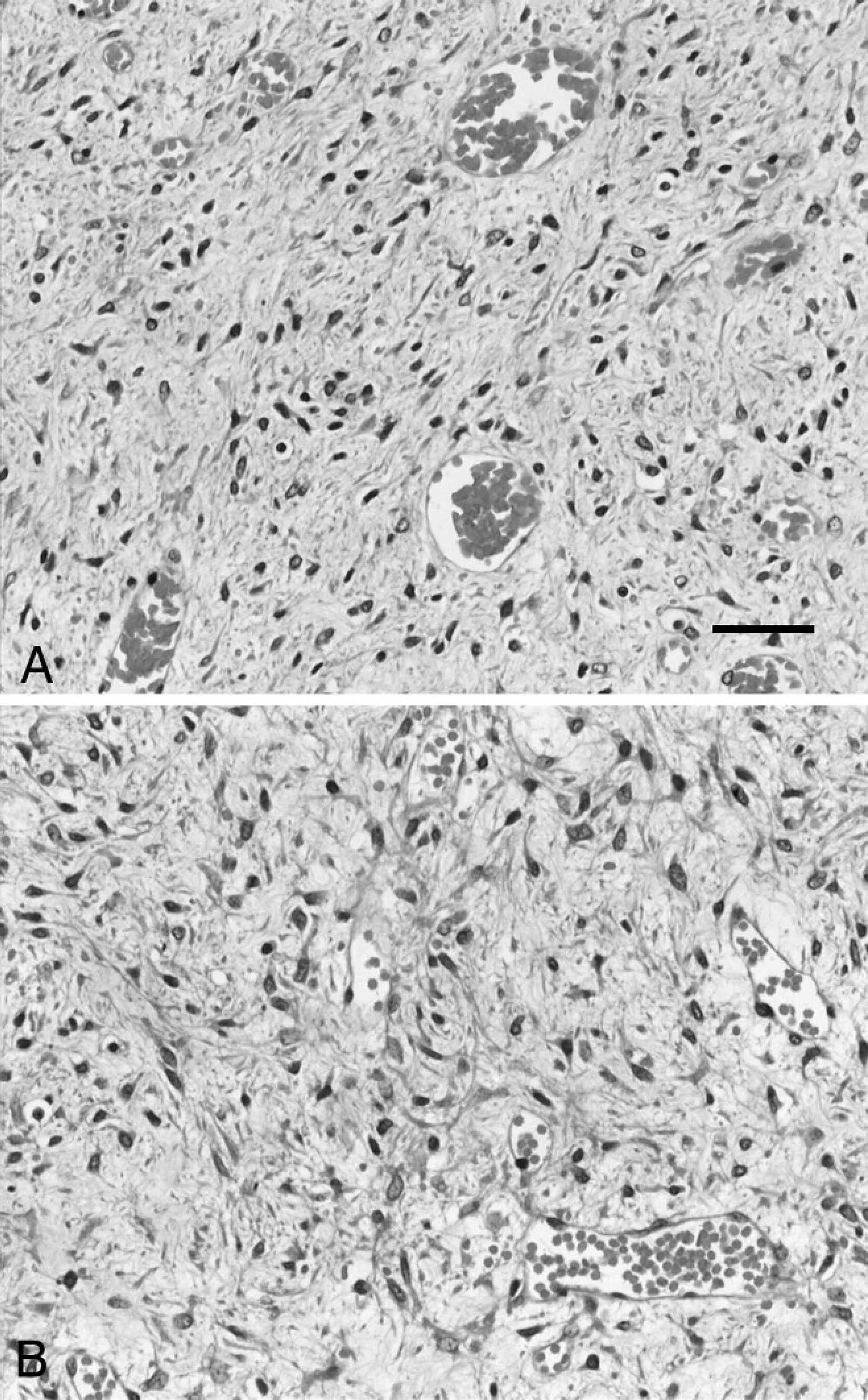
A, the tumor consisted of spindle mesenchymal cells in a loose myxoid stroma. Hematoxylin and eosin. Bar = 50 μm.
Most neoplastic spindle cells stained positively for vimentin, consistent with a mesenchymal origin, and the stain highlighted 2 distinct cellular patterns: a predominant loose myxoid pattern, with low numbers of spindloid-to-stellate cells with fine cytoplasmic processes, and multifocally small, more cell-dense regions. The cytoplasm of endothelial cells lining variably sized vascular channels scattered throughout the tumor stained positively for vWF (Fig. 5). There was positive cytoplasmic staining of abluminal cells for α-SMA in the walls of blood vessels (presumptive pericytes) but also cells within the areas of higher cell density, thus suggesting a myofibroblastic phenotype. Based on routine and immunohistochemical stains, a diagnosis of renal angiomyxoma was made. To investigate whether the neoplastic cells were responsible for the production of PTHrp, appropriate immunohistochemical staining was performed. A minority (<10%) of the neoplastic spindle cells showed mild cytoplasmic PTHrp staining (Fig. 6).
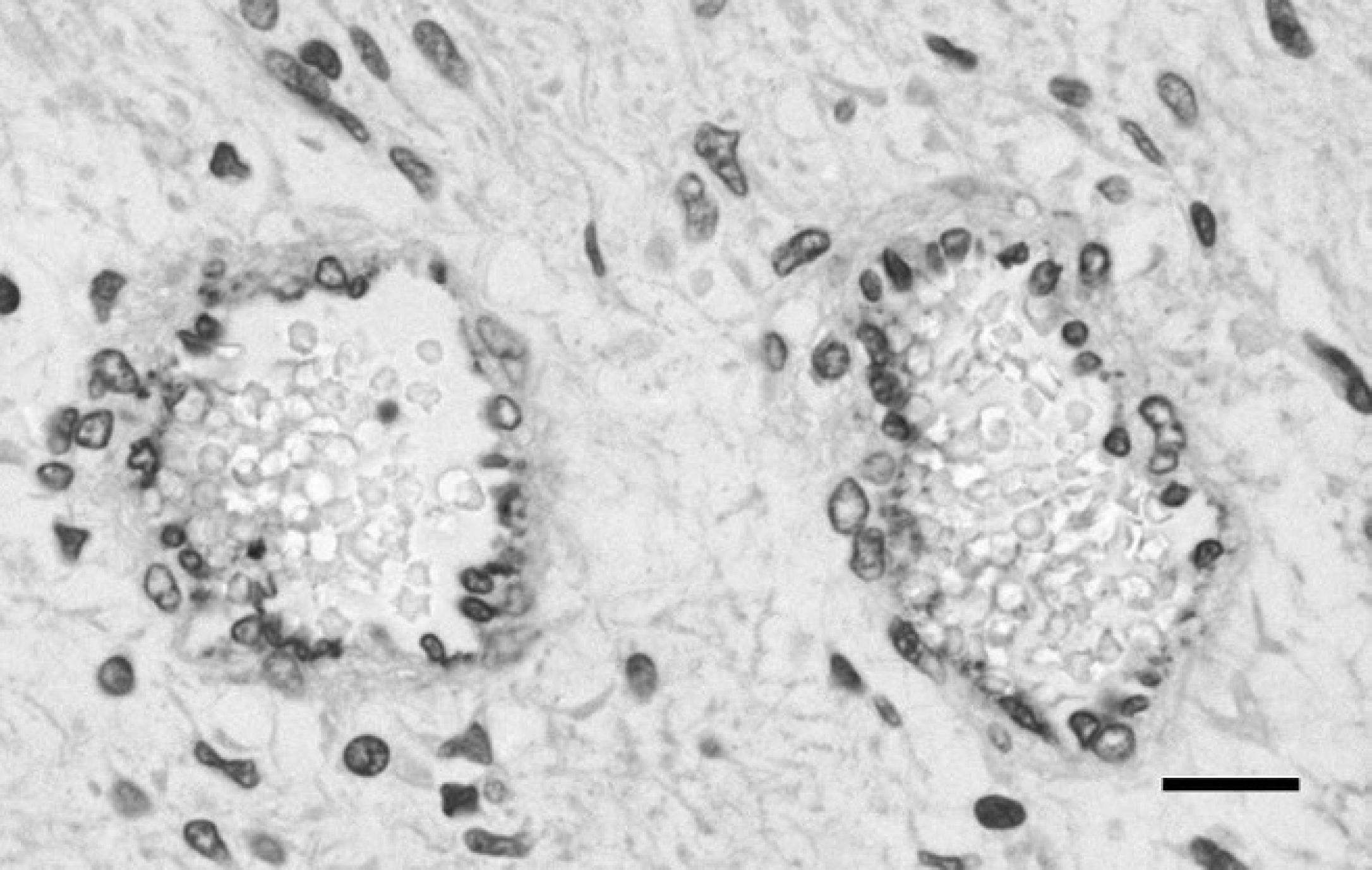
von Willebrand Factor immunohistochemistry identified the vascular component within the loose mesenchyme. 400×. Bar = 50 μm.
The dog recovered well from the surgery and at the time of discharge was only mildly hypercalcemic (ionized calcium 5.4 mg/dl or 1.35 mmol/l). Two weeks following discharge, the dog was no longer hypercalcemic (total calcium 11.7 mg/dl or 2.92 mmol/l). Four weeks following discharge, the dog was no longer polydipsic and had normal serum concentrations of PTH (114 pg/ml) and PTHrp (<0.1 pmol/l), while the ionized calcium concentration was mildly decreased (4.7 mg/dl or 1.18 mmol/l). Twelve months following surgery, the dog was reported to be healthy with no clinical signs of polydipsia or polyuria.
Canine primary renal tumors are relatively uncommon, accounting for only 0.6–1.7% of all neoplasms in dogs. 18 Renal tumors of mesenchymal origin account for about 34% of these tumors, while epithelial tumors are the most common form (60%). 1 Within the subset of renal mesenchymal tumors, hemangiosarcomas are the most common type, although other forms, including renal sarcomas and leiomyosarcomas, have also been reported. 1 Angiomyxoma has been reported in the peritoneum of a 2-year-old Flemish cow, 16 but it has not, to the authors' knowledge, been reported in the dog. Angiomyxoma is an extremely uncommon mesenchymal tumor in humans and occurs with the highest frequency in the female pelvis and perineum as a soft (myxoid) vulval, vaginal, or pelvic mass, accompanied by perineal herniation. 2 Histologically, angiomyxomas are characterized by a proliferation of spindle-shaped-to-stellate cells, widely separated by a loose myxoid stroma with a prominent vascular component. 14 In human beings, this tumor has also been reported to occur in the male genital tract as well as the orbit, oral cavity, skin, and umbilical cord. 8–10,25 There are isolated reports of this tumor occurring in the peripelvic renal tissue and the kidney. 5,19 The behavior of angiomyxomas is generally benign, exhibiting slow growth, absence of metastases, and histologic features including a distinct lack of nuclear atypia or mitotic activity. In human beings, the tumor is thought to originate from the myofibroblast progenitor cells within the female genital tract. 6
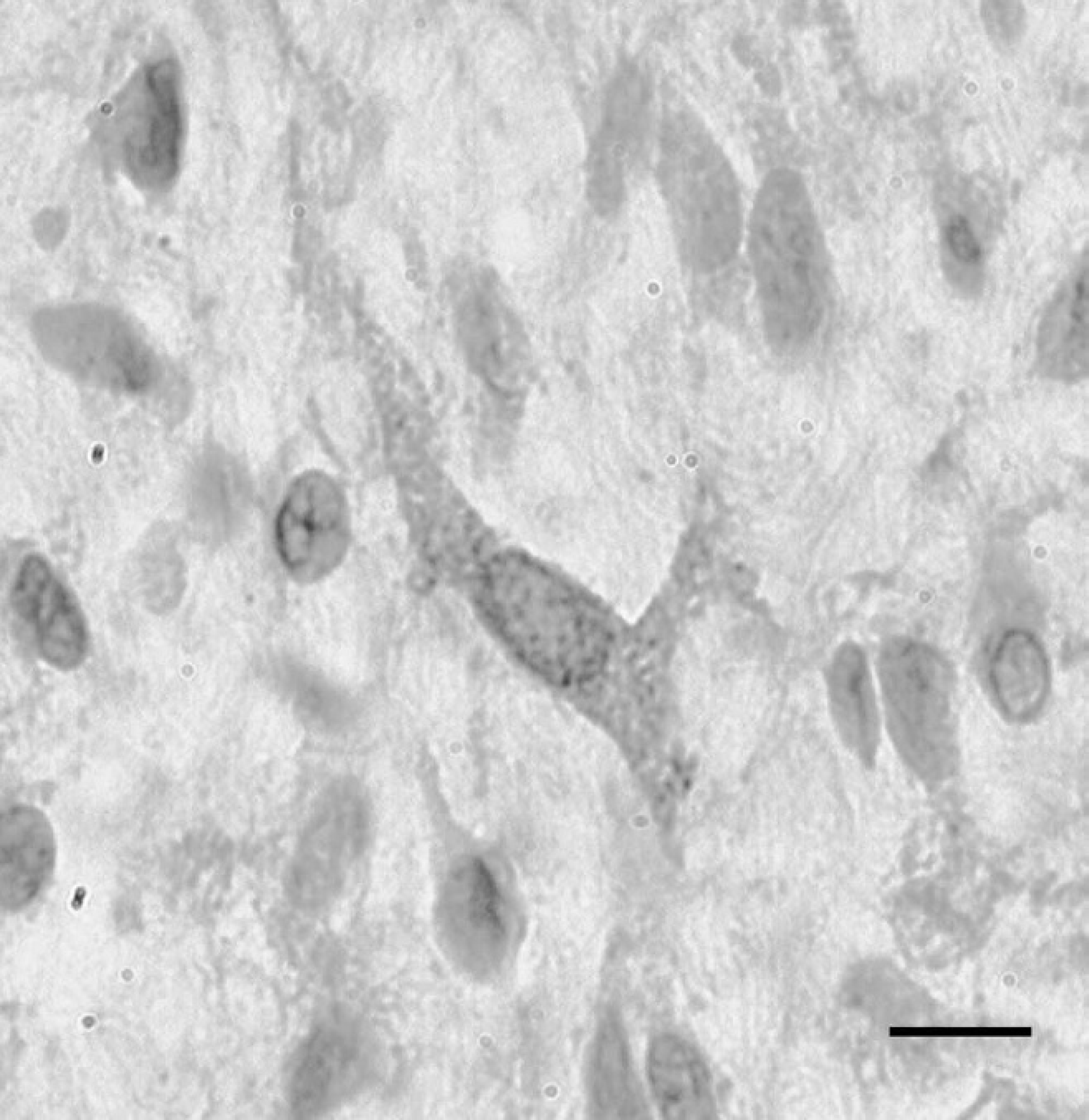
Rare neoplastic stromal cells showed parathyroid hormone–related peptide expression (brown stain). 400×. Bar = 50 μm.
The diagnosis of angiomyxoma in the dog in the current study is unique for several reasons. This tumor has not been previously diagnosed in this species, and its anatomical site was unusual. Interestingly, paraneoplastic hypercalcemia has not been reported in any species in association with this tumor. Human angiomyxomas are reportedly very large (measuring up to 60 cm) and are gelatinous when sectioned, which was also noted grossly in the present case. 2
The most interesting feature of the current case was the paraneoplastic hypercalcemia. The ionized hypercalcemia and concurrent raised serum PTHrp concentration, both of which normalized following surgical excision of the renal tumor, implied that the hypercalcemia was referable to the tumor. Paraneoplastic syndromes are relatively uncommon in canine renal neoplasms, but several distinct forms including polycythemia, leukocytosis, hypertrophic osteopathy, and hepatopathy have been reported. 18 The pronounced masticatory muscle atrophy noted in the present case is unlikely to have been related to the renal neoplasm. Paraneoplastic hypercalcemia is typically associated with the production of PTHrp, but other mediators such as calcitriol, interleukin (IL)-1β, IL-6, prostaglandins, transforming growth factor (TGF)-β, and TNF-α have also been implicated in this process. 23 Parathyroid hormone–related peptide is produced in many normal tissues, including mesenchymal, epithelial, and endocrine tissue, and in the central nervous system and has functions at sites such as cartilage, breast, skin, placenta, and the brain. 4 A raised serum PTHrp concentration is considered to be a specific marker of neoplasia, but renal insufficiency (in the absence of neoplasia) is reportedly also a cause of increased concentrations of PTHrp. 4 There were no biochemical indicators of reduced renal function prior to surgery in the dog in the current study. Serum PTH concentrations in animals with humoral hypercalcemia would be expected to be low or undetectable due to negative feedback from hypercalcemia. However, in an extremely small percentage of dogs with humoral hypercalcemia, the PTH concentration can be normal or elevated. 4 This may be due to the presence of cross-reacting heterophilic antibodies interfering with the assay. 21 In addition to this, assays for PTH messenger RNA have failed to demonstrate transcription of PTH in multiple human and animal (nonparathyroid) tumors, indicating that PTH is a rare cause of paraneoplastic hypercalcemia. 20 For these reasons, the finding of a serum PTH concentration within the low end of the reference interval in the dog in the present study, albeit unusual, does not preclude the diagnosis of paraneoplastic hypercalcemia. Likewise, although only a minority of the neoplastic cells showed positive staining with PTHrp immunohistochemistry, this did not exclude the diagnosis: tumors producing abundant PTHrp may not stain well for PTHrp because there is minimal storage and it is secreted on synthesis. 3 As further evidence in support of this, other tumors such as apocrine gland adenocarcinomas have been shown to be only weakly positive on PTHrp immunohistochemistry despite causing hypercalcemia. 7
Canine tumors associated with humoral hypercalcemia include lymphoma, apocrine gland adenocarcinoma, multiple myeloma, mammary adenocarcinoma, anaplastic carcinoma, malignant histiocytosis, and squamous cell carcinoma. 23 Although hypercalcemia can occur in association with benign processes such as granulomatous disease, reports of paraneoplastic hypercalcemia referable to benign neoplasia are very rare in both the veterinary and human literature. 12,13 Paraneoplastic hypercalcemia referable to a renal tumor has not been reported previously in the dog. In humans, humoral hypercalcemia of malignancy can be associated with both renal cell carcinomas and lymphoma. The former is more prevalent and is usually associated with increased serum PTHrp concentrations, whereas hypercalcemia referable to renal lymphoma is less common and is typically associated with calcitriol overproduction. 11 Other renal tumors, such as squamous cell carcinomas, have also been reported to cause humoral hypercalcemia in human beings. 24
In the human literature, angiomyxoma is described ultrasonographically as a hypoechoic or overtly cystic mass. However, differentiating myxoid soft-tissue tumors can be challenging. Computed tomography and magnetic resonance imaging (MRI) can be useful in making this distinction. Similar to reports in the human literature, the angiomyxoma in the present case adopted the tomographic appearance of a well-marginated mass, with an attenuation less than that of muscle (iso-attenuating to water) on precontrast images. 17 The relatively hypoattenuating characteristic of these tumors has been correlated with the myxomatous component of the angiomyxoma. 15 A swirled appearance of the tumor on postcontrast CT images, as has been reported in human beings, was not observed in the current case. 17 Angiomyxomas can also be confused with myxoid liposarcomas. For this reason, CT and MRI have been used to identify a fatty component to the tumor. 22 In the present case, lesional fat was not identified on CT, and thus liposarcoma was considered a less likely diagnosis. However, some liposarcomas may show little fat content (e.g., undifferentiated liposarcoma), while other abdominal tumors containing fat may not be liposarcomas. 10 The diagnostic imaging performed in the current case highlights the value of CT in differentiating different types of soft-tissue tumors. The vascular component seen in the early phase postcontrast and the hypoattenuation seen precontrast correlate well with respective vascular and myxoid components of this angiomyxoma.
Up to 50% of human angiomyxomas recur following surgical excision, which is suspected to be due to the inability to excise the tumor with clear surgical margins: the prefix aggressive is sometimes given to these tumors for this reason. 2 Chemotherapy and radiotherapy are not recommended, owing to the low mitotic activity of these tumors. Fortunately, the location of the tumor in the dog in the current study enabled complete excision via nephrectomy.
Acknowledgements
The authors are grateful to Drs. Thomas J. Rosol and Ramiro Toribio, Ohio State University, for performing the PTHrp immunohistochemistry and for interpreting the result.
Footnotes
a.
Combur 9 Test®, Roche Products Ltd., Welwyn Garden City, Hertfordshire, United Kingdom.
b.
Hema-Tek 2000, Bayer UK Ltd., Newbury, Berkshire, United Kingdom.
c.
Potassium chloride 20%, Martindale, Romford, United Kingdom.
d.
Dimazon 5%, Intervet/Schering-Plough Animal Health, Bray, Co. Wicklow, Ireland.
e.
EnVision™, Dako UK Ltd., Ely, Cambridgeshire, United Kingdom.
f.
Shandon Sequenza®, Thermo Fisher Scientific Inc., Waltham, MA.
g.
Sigma-Aldrich Co. Ltd., Dorset, United Kingdom.
h.
Vector Laboratories Ltd., Orton Southgate, Peterborough, United Kingdom.
i.
Novocastra Laboratories Ltd., Newcastle Upon Tyne, United Kingdom.
