Abstract
Polymerase chain reaction (PCR) for antigen receptor rearrangement is a sensitive technique for detecting lymphocyte-proliferative disorders, but it tends to produce false-positive results, a phenomenon termed pseudoclonality. Heteroduplex analysis, which is useful to distinguish clonal reactions from pseudoclonal ones in dogs, can be applied to avoid misdiagnosis and determine the reliability of results. In the current study, PCR for antigen receptor rearrangement was used to identify clonal proliferation of lymphocytes in duodenal and lymphoid tissue from dogs presenting with chronic vomiting and enlarged peripheral lymph nodes typical of multicentric lymphoma, and the test results were verified with heteroduplex analysis. In the case of almost all of the duodenal samples, even without a histologic diagnosis of lymphoma, a distinct band similar to that observed in the case of lymphoma was obtained for both B- and T-cell clonality. All of the bands obtained from the nonneoplastic duodenum disappeared following heteroduplex analysis of the PCR product, whereas the distinct bands from the lymphoma remained. In the lymph node samples, the pseudoclonal bands that disappeared in the heteroduplex analysis were detected mainly in B cells. In conclusion, heteroduplex analysis with PCR for antigen receptor rearrangement is a suitable tool for diagnosing canine lymphoma and decreasing the possibility of misdiagnosis of pseudoclonality.
Keywords
In recent years, molecular techniques have become available for the diagnosis of canine lymphocyte-proliferative disorders. 1 Polymerase chain reaction (PCR) for antigen receptor rearrangement (PARR) is a method used for the detection of clonal lymphocyte populations, phenotyping of lymphocytes, detection of malignant lymphocytes in blood, and diagnosis of small-cell lymphocytosis. 2,13 It is reportedly useful for the diagnosis of canine alimentary lymphoma. 4,7 This technique is highly sensitive but tends to produce false-positive results, a phenomenon termed pseudoclonality, possibly because a single copy is not amplified during PCR because of the immunologic diversity of the target genes: immunoglobulin heavy chain (IgH) and T-cell receptor gamma (TCRγ). To avoid diagnostic errors, it is necessary to determine the reliability of the results of PARR. Single-strand conformation polymorphism is useful for verifying the results of conventional PARR, 6 but the procedure involved is relatively complicated and time consuming. On the other hand, heteroduplex analysis is reportedly useful to distinguish clonal reactions from pseudoclonal ones in cats 9 and dogs. 11 In the current study, PARR was used to identify clonal proliferation of lymphocytes in duodenal and lymphoid tissues from dogs presenting with signs typical of multicentric lymphoma, and the test results were verified with heteroduplex analysis.
The duodenal mucosa was endoscopically biopsied from 30 dogs (ages 1–13 years) suffering from chronic vomiting. The tissue specimens were stored at −20°C until use. Some of the tissue samples were fixed in 4% formaldehyde solution for histologic diagnosis. Fine-needle aspiration (FNA) was used to obtain cell samples from 8 dogs with enlarged lymph nodes, and the FNA samples were dried on glass slides. The tissue and FNA samples were processed for preparation of genomic DNA by incubation in a universal reagent a at 80°C for 10 min. The tissue residue was precipitated by brief centrifugation, and the supernatant was used immediately for PCR. The FNA smears were scratched from the air-dried glass slides, immersed in the universal reagent, a and incubated at 80°C for 10 min. Polymerase chain reaction was performed in a total volume of 20 μl containing 1× PCR buffer, 2.5 mM deoxynucleotide triphosphates, 4 μM each forward and reverse primers, and 0.1 unit of Taq DNA polymerase. b The primers used in the present study are listed in Table 1. After initial denaturation at 95°C for 120 sec, 35 cycles of amplification were carried out at 94°C for 25 sec, followed by annealing for 20 sec and extension at 72°C for 20 sec. The annealing temperature for each primer set was similar to that used in previous studies. 2,12 Following PCR, the contents of each tube were divided into 2 aliquots, and 1 aliquot of the PCR product was processed for heteroduplex analysis. The PCR products were incubated at 95°C for 5 min and then reannealed at 4°C for 30 min. Both the intact and the denatured PCR products were applied on 12% nondenatured polyacrylamide gel c and electrophoresed in 0.5× Tris–borate–ethylenediamine tetra-acetic acid buffer at 100 V for 1.5 hr. After electrophoresis, the gel was stained with a highly sensitive fluorescent stain d for 30 min and visualized by transillumination.
The primers used in the current study. *
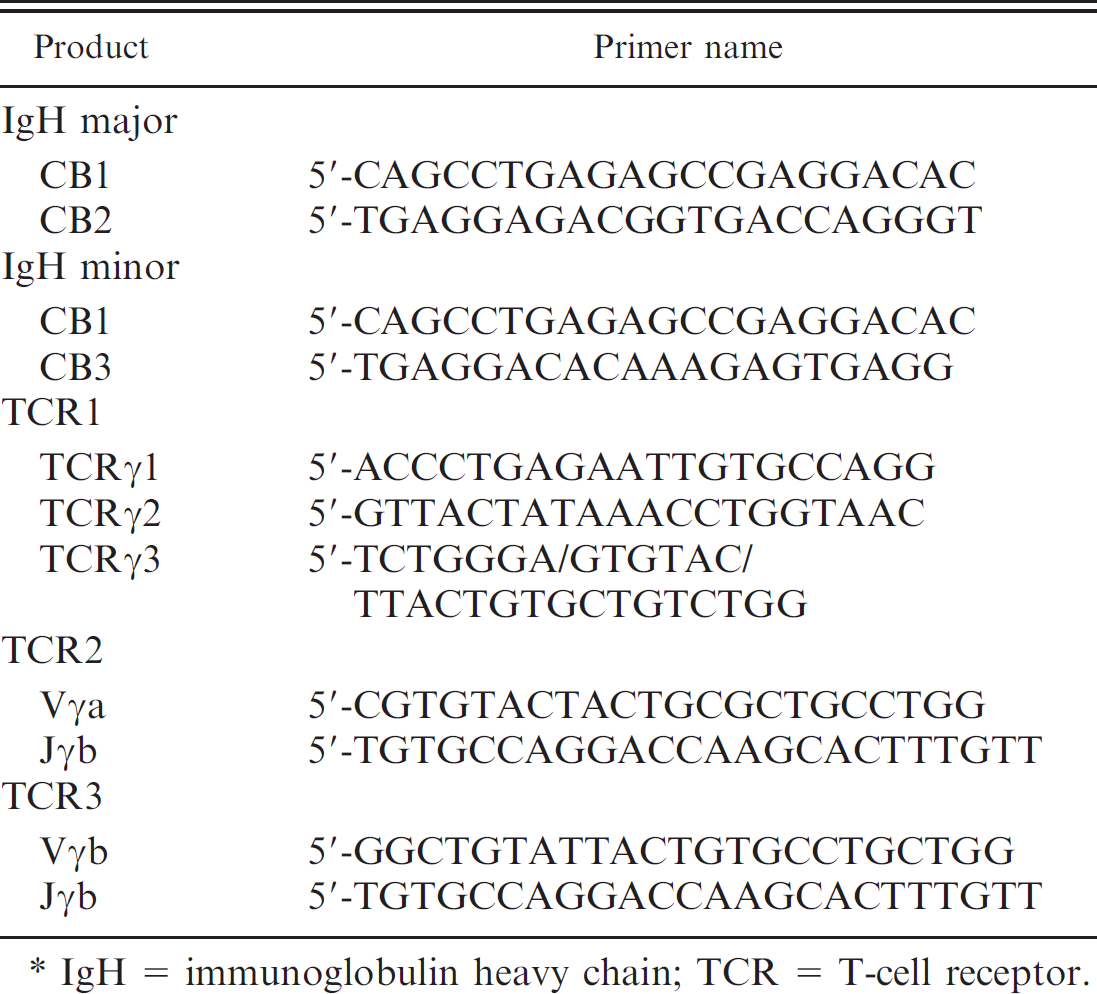
IgH = immunoglobulin heavy chain; TCR = T-cell receptor.
For conventional PARR, the distinct bands of IgH major (120 bp) and TCRγ (90 bp) were detected for all of the duodenal tissue samples of histologically diagnosed lymphocytic–plasmacytic enteritis (Fig. 1). These bands disappeared or smeared after the heteroduplex analysis (Fig. 1). In one case, the extremely faint band was observed in TCR2 after the heteroduplex (Fig. 1). No pseudoclonal bands were observed with the primer sets of IgH minor and TCRγ3 (data not shown). In the case of alimentary lymphoma, the band of the same size as the bands for lymphocytic–plasmacytic enteritis did not disappear after the heteroduplex analysis (Fig. 2). In one case of alimentary lymphoma, although the double bands were detected in the intact PCR product, one band disappeared and the other remained even after heteroduplex analysis (Fig. 2). Among the 30 duodenal tissue samples, 4 samples of histologically diagnosed alimentary lymphoma were positive for TCRγ. When PARR was performed by using the lymph node samples of cytologically diagnosed multicentric lymphoma, the IgH major band remained after the heteroduplex analysis (Fig. 3). Similar results were observed for both monoclonal and biclonal lymphoma. The positive bands never disappeared following the heteroduplex analysis. The pseudoclonal bands were detected mainly in the case of IgH and rarely in the case of TCRγ when the FNA samples from the lymph node were used for PARR (data not shown).
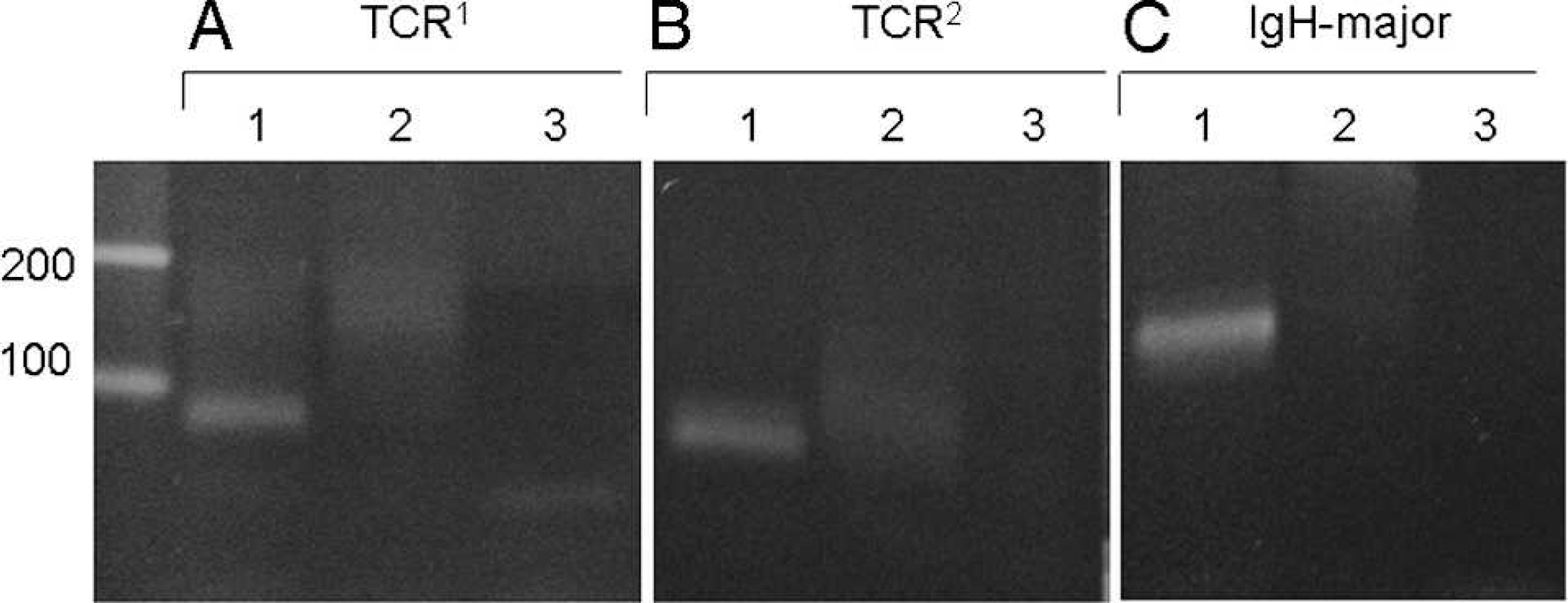
Polymerase chain reaction (PCR) for antigen receptor rearrangement of duodenal tissue excised from a dog without histologically diagnosed lymphoma.
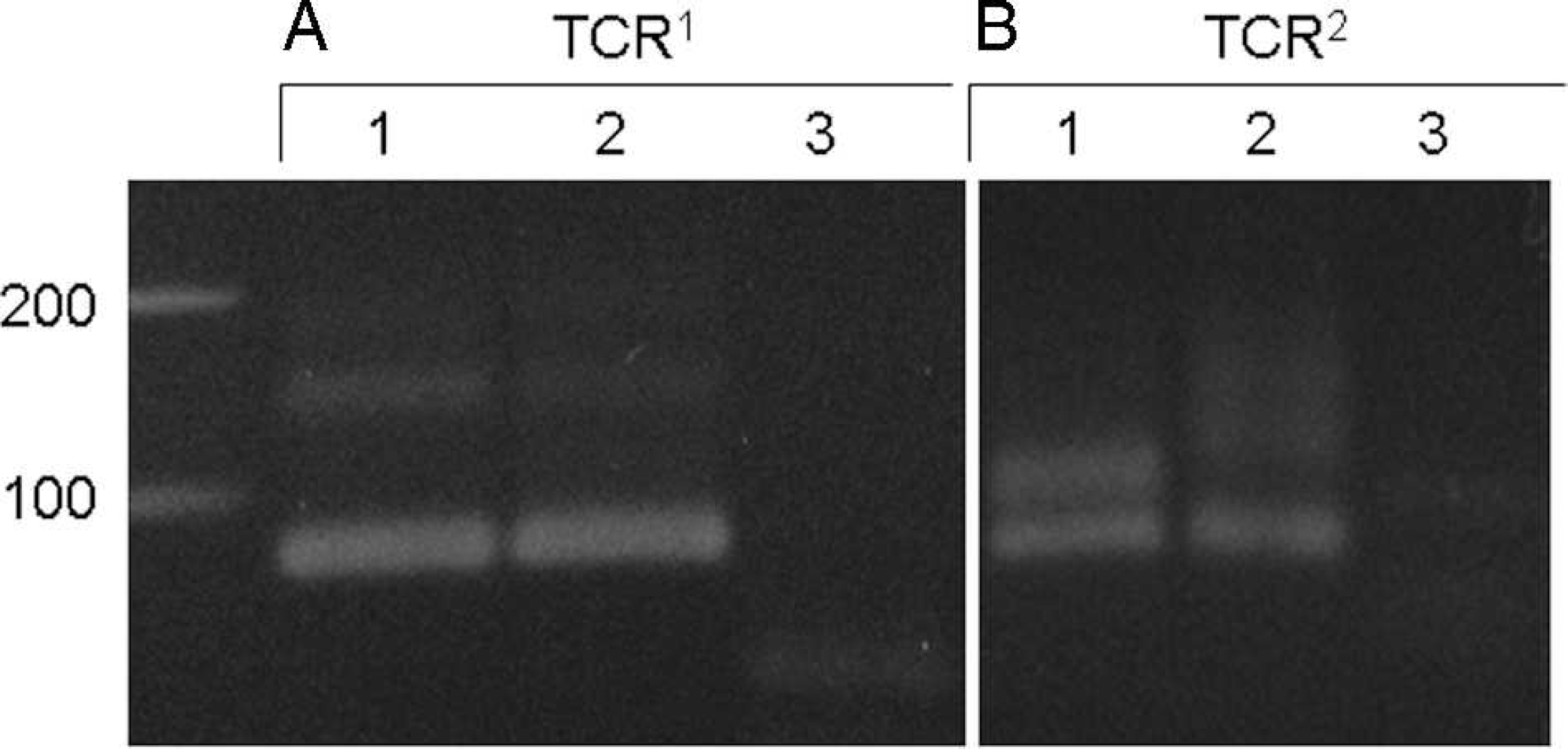
Polymerase chain reaction (PCR) for antigen receptor rearrangement for T-cell receptor gamma (TCRγ) in duodenal tissue excised from dogs with histologically diagnosed lymphoma.
The results of conventional PARR may need verification to determine whether a band is derived from a single copy of the rearranged gene for IgH or for TCRγ, especially when duodenal tissues are used. Heteroduplex analysis is reportedly helpful to identify whether the bands obtained in conventional PARR of feline TCRγ are clonal or pseudoclonal. 9 Heteroduplex analysis is a simple procedure developed for detecting a mutation in a gene. 8 The target genes of PARR, IgH, and TCRγ have a complementarity-determining region (CDR) for the complex diversity of immunity. 12 In PARR, the PCR products may not always be derived from a single copy of the genes, because the forward and reverse primers were designed outside of the CDR, which shows sequence homology in many segments of the V or J regions. 2,12 Various-sized PCR products corresponding to each rearranged gene would be amplified in cases of lymphocytic–plasmacytic enteritis, in which many types of the lymphocyte are distributed in intestinal villi, because the size of the CDR is different in several amino acids. If migration distance is short on electrophoresis, it may be difficult to distinguish PCR products that have several base-pair differences. In the present study, electrophoresis was completed within 90 min to shorten all procedures of clonality analysis. This may be a reason why the distinct bands of IgH and TCRγ were detected in a conventional PARR.
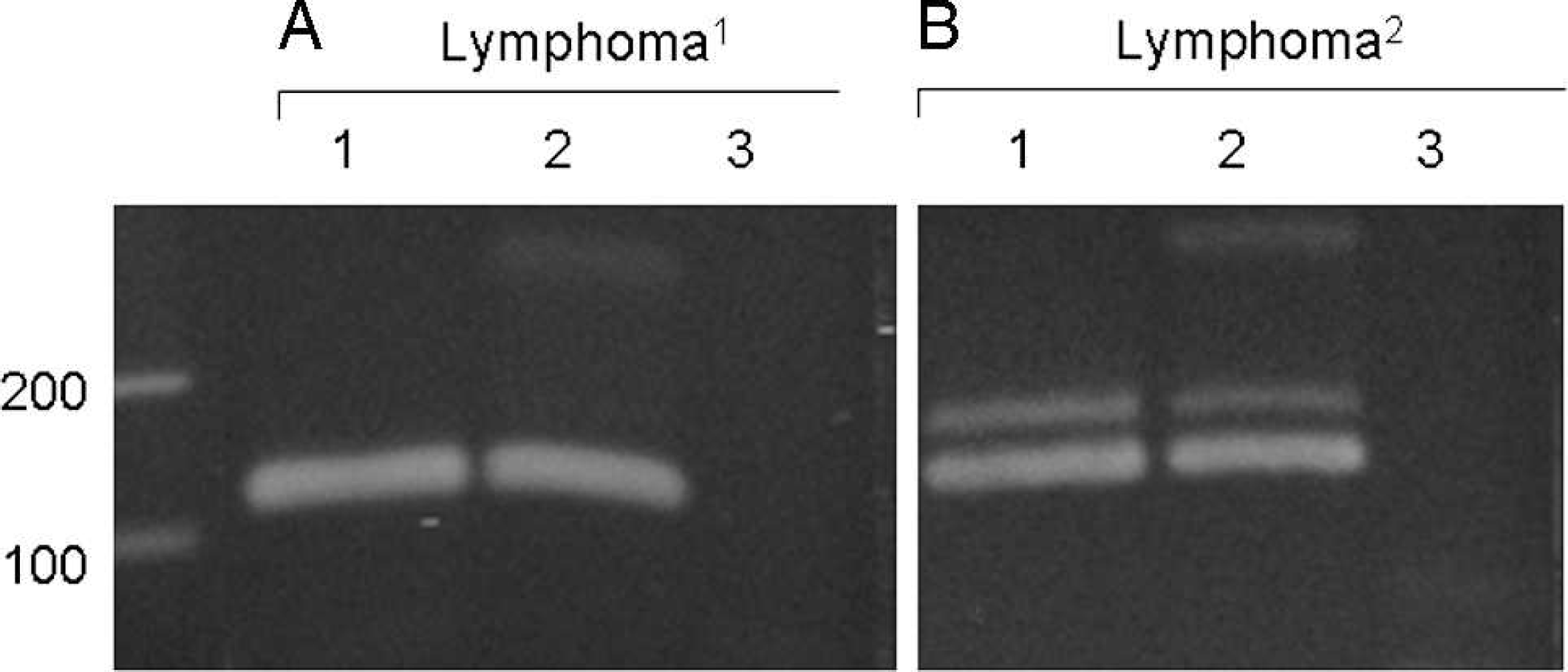
Polymerase chain reaction (PCR) for antigen receptor rearrangement for immunoglobulin heavy chain (IgH) in lymphoid tissue. The primer set for IgH-major was used.
The presence of the faint band after the heteroduplex was considered to be caused by pseudoclonality (Fig. 1). Compared with the band from lymphoma, the intensity of the remaining band after heteroduplex was extremely low. Even if a small number of tumor cells exist in the tissue, the specific PCR product was predominantly amplified compared with nonspecific products. 2 The faint band may be derived from a heteroduplex complex consisting of different PCR products with a few differences of base pairs.
In the current study, the pseudoclonal bands were distinguished by using heteroduplex analysis, which is beneficial for recognizing lymphocyte clonality in canines as well as felines. Recently, PARR with GeneScanning or real-time PCR has also been applied for high-sensitivity analysis of lymphocyte clonality; however, this method would require specific and expensive equipment. 5,14 In the present study, it was possible to obtain reliable PARR results promptly by using simplified genome preparation and heteroduplex analysis after conventional PCR. Because the universal reagent a used in the present study is commercially available and a particular apparatus is not necessary for heteroduplex electrophoresis, all procedures were completed within 5–6 hr. A therapeutic strategy for lymphoma can be determined quickly if phenotyping could be completed in a day. The band of TCRγ pseudoclonality, which disappeared after the heteroduplex analysis, was observed in all of the duodenal tissue and a few lymphoid tissue samples. For IgH, pseudoclonality was observed in both the duodenal epithelium and the lymph nodes. Because many types of immunocompetent cells migrate to the small intestine, 10 it is possible that a subset of T or B cells detectable by the primer sets used for the PARR is distributed in the intestinal epithelium. The results of conventional PARR for diagnosing canine alimentary lymphoma may be less accurate when intestinal epithelium is used, because the duodenal epithelium showed pseudo-clonality. Although histology is the main method for diagnosing lymphoma, it may be difficult to apply for distinguishing lymphoma from inflammatory bowel disease. 7 In such cases, PARR may be an effective method for the differential diagnosis of alimentary lymphoma. Because T-cell lymphoma has a short survival time compared with B-cell lymphoma, 3 phenotypic classification of lymphoma is essential to the prognosis. For example, when dogs with histologically diagnosed enteritis showed TCRγ clonality, their survival time was significantly less than that of dogs negative for TCRγ, which means that PARR could overcome the limitation of a histological diagnosis. 7 The combination of PARR and heteroduplex analysis is a simple procedure that can be performed by using conventional PCR and electrophoresis apparatus. Therefore, the methods together would be a suitable tool for diagnosing canine lymphoma and decreasing the possibility of misdiagnosis of pseudoclonality.
Footnotes
a.
DNAzol® Direct, Molecular Research Center Inc., Cincinnati, OH.
b.
TaKaRa Taq™ Hot Start Version, Takara Shuzo, Tokyo, Japan.
c.
Polyacrylamide gel, Embi Tec, San Diego, CA.
d.
GelStar™, Cambrex Bio Science Rockland Inc., Rockland, ME.
