Abstract
Mycobacterium bovis causes disease in numerous mammalian species including humans, thus making research, surveillance, and control important in the eradication of tuberculosis. Domestic cats are susceptible to multiple mycobacterial species including Mycobacterium bovis; however, their role in the epidemiology of bovine tuberculosis is not fully documented. The current study was an evaluation of the immune response in specific pathogen–free (SPF) cats stimulated with sensitinogen, a heat-killed M. bovis product, using the rapid test, multiple antigen print immunoassay (MAPIA), and bovine-purified protein derivative (bPPD) single skin test. Six cats were inoculated with sensitinogen subcutaneously on days 0 and 24; 2 noninoculated cats and 49 non-SPF cats were controls. Serial serum samples were collected during 135 days and assayed for M. bovis antibodies by rapid test and MAPIA. On day 123, bPPD skin test was performed and read at 48 and 72 hr. The bPPD test at 72 hr had a mean skin thickness of 0.3 mm for stimulated cats and 0.1 mm for controls. Rapid test identified 4 of 6 stimulated cats after bPPD injection. The MAPIA detected antibody against MPB83, 16/83, 16 kDa, and M. bovis culture filtrate (MBCF) antigens. All assays differentiated between stimulated and control cats; however, 7 of 49 non-SPF control cats had a reaction for either antigen MBCF or 16/83. These preliminary studies show potential for antemortem detection of M. bovis among domestic cats. Additional studies to better characterize virulent M. bovis infection in cats would be of value.
Keywords
Introduction
Domestic cats are susceptible to many mycobacterial species, and disease is classified into 3 categories: classic tuberculosis, leprosy, and opportunistic. 11,19,22 Multiple species of Mycobacterium are able to cause tuberculosis; however, the classic form of tuberculosis in cats is thought to be due to Mycobacterium bovis, which can produce clinical disease in a wide range of mammalian species including humans, thus making research, surveillance, and control of M. bovis important in the eradication of tuberculosis. 1–3,6,11,21,22,24
Antemortem diagnosis of M. bovis infection in potential reservoir species, especially cats, has been difficult. 8,19 Antemortem diagnosis of M. bovis infection within cattle uses the tuberculin skin test, the single intradermal comparative tuberculin test, and the gamma interferon assay; however, skin testing is reportedly inconsistent when used in domestic cats. 4,5,11,13,14,19,24 In an effort to increase sensitivity and specificity in antemortem assays, laboratories have expanded their research to include both cell-mediated and humoral immune responses with diagnostic tests such as enzyme-linked immunosorbent assay (ELISA), multiple antigen print immunoassay (MAPIA), and rapid test. The most recent immunodiagnostic tests developed for cats are the gamma interferon (IFN-γ) ELISA and enzyme-linked immunospot (ELISPOT) assay. 23 These blood assays incorporate the following antigens: avian-purified protein derivative (aPPD), bovine PPD (bPPD), positive mitogen (PMA/CA), and a cocktail of ESAT-6 and CFP10, which aid in predictability of M. bovis and M. microti infection, but are unable to reliably predict Mycobacterium avium infections. In other host species, MAPIA has identified MBP70 (cattle), MPB83 (cattle, deer, badgers), and ESAT-6 (elephants, nonhuman primates) as immunodominant antigens. 6,9,10,12,17,18,26 The rapid test and MAPIA, when used with the mycobacterial antigen cocktails, have been reported to have a higher sensitivity and specificity in Eurasian badgers (Meles meles) 9 and were able to maintain serologic activity longer than existing ELISA tests. 17 To date, the gold standard for diagnosis of feline tuberculosis still remains bacterial culture from biopsy or at time of necropsy. The present study was aimed at identifying antemortem methods to detect M. bovis infection by evaluating serologic assays, rapid test, and MAPIA, and the single skin test using bPPD in cats experimentally stimulated with sensitinogen, a heat-killed M. bovis strain AN-5 suspended in mineral oil. 7
Eight 6-month-old, specific pathogen–free (SPF), purpose bred domestic short hair cats were purchased from a commercial vendor. a All cats were vaccinated with a killed virus preparation at 5 months of age for protection against panleukopenia, rhinotracheitis, and calicivirus (FELOVAX PCTb) before purchasing. Institutional Animal Care and Use Committee (IACUC)-approved guidelines were implemented. Cats were group housed, monitored daily, housed at a temperature range of 22°C ± 2°C, humidity range of 40–50%, and were maintained on a commercially available cat food with water ad libitum within the animal containment facility at Michigan State University (MSU; Lansing, Michigan). In addition, serum was also collected from 9 deceased domestic cats received at the Diagnostic Center for Population and Animal Health (MSU) and from 40 SPF control cats that participated in a previous research project.
Lot number 0401 sensitinogen c was administered at a dose of 0.4 ml subcutaneously over the lateral body wall to 6 of the 8 cats on days 0 and 24 to induce a cell-mediated hypersensitivity to M. bovis. The 2 remaining cats that were not injected served at controls. Approximately 4 ml of blood was collected by jugular venipuncture approximately every 30 days. Immediately after venipuncture, blood samples were clotted at 4°C for 1 hr, centrifuged at 5,000 × g for 5 min, and serum was then separated into sterile tubes and frozen at −20°C until all samples were collected for the entirety of the study.
On day 123, approximately 2 weeks before the last blood draw, the right pinna of each cat was measured with dermal calipers to the nearest 0.05 mm, and 0.1 ml of bPPD d at a concentration of 1.0 ± 0.1 mg/ml was injected intradermally to the right inner pinna. Pinnae were monitored for heat, reddening, and thickness. Pinna thickness was measured by dermal calipers at 48 and 72 hr after bPPD injection.
The rapid test, performed at a commercial laboratory, e is a lateral flow, blue latex bead signal-based, qualitative antibody detection assay that uses a nitrocellulose membrane impregnated with a cocktail of selected M. bovis antigens (ESAT-6, CFP10, Acr1, MPB83). Thirty microliters of test serum and 3 drops of diluent buffer were added to the test well, and the result of the reaction was read by visual evaluation after 20 min. 15 An antibody-positive sample was indicated by a visible band at both the test and control lines, whereas an antibody-negative sample was indicated by a visible band at the control line but no band at the test line. 15
Bovine-purified protein derivative (bPPD) single skin test results (in millimeters). *
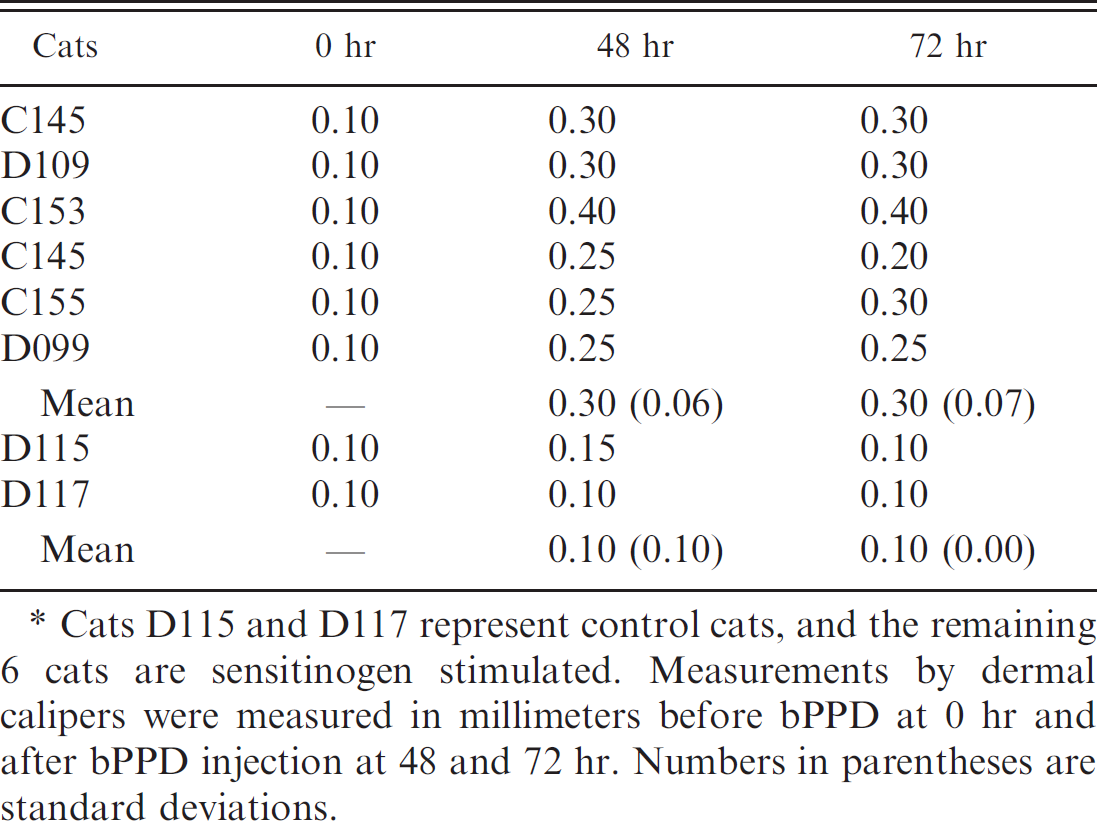
Cats D115 and D117 represent control cats, and the remaining 6 cats are sensitinogen stimulated. Measurements by dermal calipers were measured in millimeters before bPPD at 0 hr and after bPPD injection at 48 and 72 hr. Numbers in parentheses are standard deviations.
The MAPIA, performed at a commercial laboratory, e used purified Mycobacterium tuberculosis–complex antigens (ESAT6, CFP10, MPB64, MPB59, MPB70, rMPB83, 16 kDa, and 38 kDa), fusion proteins (E6/P10 and 16/83), native antigens (bPPD), and M. bovis culture filtrate (MBCF) at a concentration of 0.05 mg/ml. f The antigen-imprinted nitrocellulose membrane was cut into strips and processed as described previously. 17 Test strips were then incubated with serum samples diluted 1:40 in blocking solution for 1 hr at room temperature. Antibody/antigen reaction bands were visualized by using protein A–horseradish peroxidase conjugate and substrate 3,30,5,50-tetramethyl benzidine. g , h The most prominent bands noted as positive results were also analyzed semiquantitatively by densitometry. 17
Two representative sensitinogen-stimulated cats were sedated with a cocktail of ketamine hydrochloride (11 mg/kg) and acepromazine (0.01 mg/kg) before euthanasia by intracardiac injection of sodium pentobarbital (200 mg/kg). These two sensitinogen-stimulated cats were euthanized, and full necropsies were performed. Two large ulcerated granulomas were noted grossly on each cat. These lesions correlated with the site of the sensitinogen injection. Tissue samples collected included skin at the sensitinogen injection site, submandibular and retropharyngeal lymph nodes, and tonsil. Harvested tissues were fixed in 10% neutral buffered formalin, routinely processed, sectioned, and stained with hematoxylin and eosin. Light microscopy revealed chronic granulomatous dermatitis with multifocal fibrosis, necrosis, and mineralization in both cats. The tonsil of one cat was determined to have a mild multifocal tonsillitis. Regional lymph nodes of both cats had reactive lymphofollicular hyperplasia, but no inflammation.

Multiantigen print immunoassay (MAPIA) results for a representative sensitinogen-stimulated cat and control cat, C147 and D117, respectively. Mycobacterium tuberculosis complex antigens are indicated to the right of the test strips; a visible band indicates antibody presence and a positive reaction. The MAPIA strips are organized into groups of 6 strips per cat with each strip representing a sampling time point: 1 = day 0, 2 = day 24, 3 = day 51, 4 = day 80, 5 = day 109, and 6 = day 135. Day 135 readings were collected 12 days after bovine-purified protein derivative (bPPD) injection for skin testing. Among the stimulated cats, Mycobacterium bovis culture filtrate (MBCF), 16/83, 16 kDa, and rMPB83 were the most reactive.
Mean and standard deviation were calculated for the numerical data collected. 20 The 2-sample t-significance test was used to assess statistical difference between tuberculin skin test results for the stimulated and control cats. The t-statistic obtained from the data was compared to the t-distribution critical values table using the smallest degrees of freedom and a P value of 0.05 for a 1-sided test and 0.025 for a 2-sided test. 20 Repeated measures analysis of variance (ANOVA) was performed on SAS 9.1 software for both tuberculin skin test and MAPIA densitometry data. i
In all cats, thickness of the pinna before the bPPD injection was 0.1 mm (Table 1). Change in pinna thickness for sensitinogen-stimulated cats ranged from 0.15 to 0.3 mm, whereas change in pinna thickness for control cats ranged from 0 to 0.05 mm. Pinnae of sensitinogen-stimulated cats after the skin test were grossly thickened and reddened, and were warm to the touch; the controls had no notable change. Using the 2-sample t-test, the t-statistic was 7.08 with degrees of freedom equal to 1 resulting in a value of P < 0.05. Using the repeated measures ANOVA for determining significance of pinna skin thickness over time shows an F value of 9.67 and a P value of 0.0191, and an F value of 6.49 and a P value of 0.0408 when looking at effects of sensitinogen stimulation over time. Quantifiable measurements from dermal calipers yielded more than twice the increase in thickness of pinnae and P < 0.05, suggesting a significant difference between stimulated and control groups at 72 hr after bPPD injection. Previous reports on the intradermal test in domestic cats suggested poor prediction of disease in naturally infected animals. 11,19 In contrast, the current study correctly identified all 6 sensitinogen-stimulated cats by the bPPD single skin test. It should be recognized, however, that this experimental model using a killed M. bovis preparation may not fully reflect the dose and complexity of the delayed-type hypersensitivity responses induced during a virulent M. bovis infection. Therefore, the diagnostic performance of the skin test in cats with naturally acquired M. bovis infections may be different from that observed among the sensitinogen-stimulated cats. Limitations of intradermal testing in cats echo concerns seen within bovine tuberculosis control and eradication programs for cattle, such as cross-reactivity from other Mycobacterium spp. and the potential effects on subsequent antemortem assays. 23
Multiple antigen print immunoassay densitometry readings for antigens 16/83 and Mycobacterium bovis culture filtrate (MBCF) from days 0–135. *
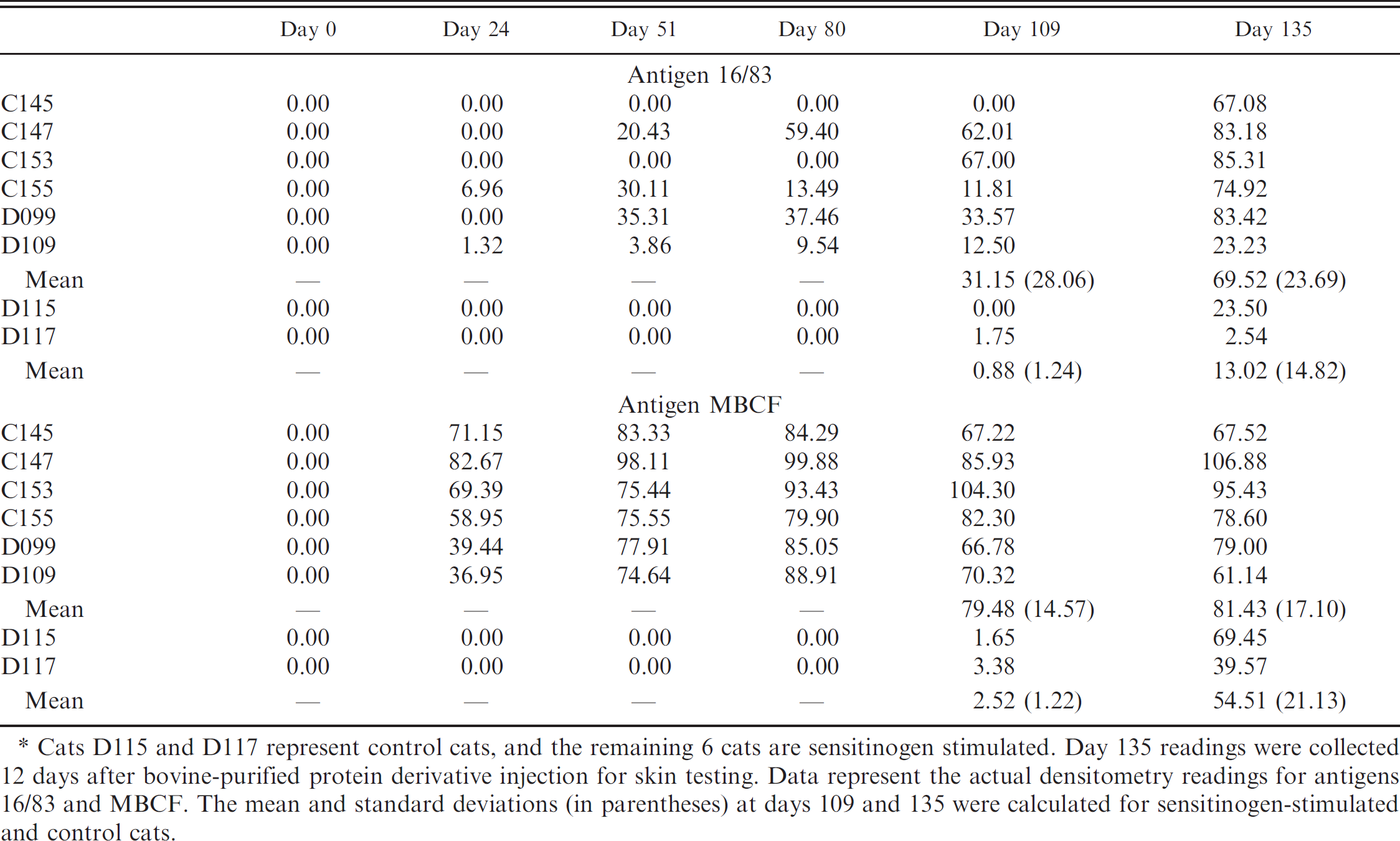
Cats D115 and D117 represent control cats, and the remaining 6 cats are sensitinogen stimulated. Day 135 readings were collected 12 days after bovine-purified protein derivative injection for skin testing. Data represent the actual densitometry readings for antigens 16/83 and MBCF. The mean and standard deviations (in parentheses) at days 109 and 135 were calculated for sensitinogen-stimulated and control cats.
The rapid test identified 4 of 6 stimulated cats after injection of bPPD; all other cats were uniformly negative. The rapid test antigen cocktail (ESAT-6, CFP10, Acr1, MPB83) used in the current study was originally designed for species other than feline. The current cocktail of antigens may contribute to the low reactivity seen in the present experiment, as the only antigen identified by MAPIA as a potential reactive antigen in the sensitinogen-stimulated cats was MPB83. Reformulation of the rapid test to include 16/83, 16 kDa, and MBCF may enhance the results of the rapid test. Recent studies have proven the rapid test to be useful in detecting M. bovis infections in llamas (Llama glama), elk (Cervus elaphus), white-tailed deer (Odocoileus virginianus), wild boar (Sus scrofa), Eurasian badgers, brushtail possums (Trichosurus vulpecula), and meerkats (Suricata suricatta); however, validation of the rapid test for domestic cats has yet to be achieved. 6,9,10,12,16,17,26
Multiple antigen print immunoassay identified 16/83, 16 kDa, MPB83, and MBCF as major antigens involved in the humoral immune response in sensitinogen-stimulated cats. Visually all sensitinogen-stimulated cats reacted consistently to antigen MBCF and less consistently to 16/83, 16 kDa, and MPB83 antigens (Fig. 1). The experimental controls had no reaction band early in the experiment, and by day 109, a faint band was noted and quantified by optical densitometry. Sensitinogen-stimulated and control cats were group housed, allowing for social interactions such as grooming between individuals. All sensitinogen-stimulated cats developed ulcerative granulomas at the site of injection, and it is possible that control cats were exposed to sensitinogen during grooming and contributed to a mild immune response before administration of bPPD or this could be simply due to nonspecific responses. The non-SPF controls were mostly unremarkable with 5 of 49 reacting to MBCF and 2 of 49 reacting to 16/83. Using densitometry, semiquantitative estimates of band signal intensities for sensitinogen-stimulated cats were compared for antigens 16/83 and MBCF (Table 2). Using the repeated measures ANOVA, significance was determined for MBCF band intensity over time, days 0–109 (F = 51.98, P = 0.0042), and when looking at effects of sensitinogen stimulation over time (F = 49.35, P = 0.0045). Using the repeated measures ANOVA, a lack of significance was determined for the 16/83 band intensity over time; days 0–109 (F = 0.59, P = 0.6930), and when looking at effects of sensitinogen stimulation over time (F = 0.55, P = 0.7142). A repeated measures ANOVA was not determined for days 0–135 due to overlap in values between sensitinogen-stimulated and control cats indicating no statistical significance. The mean optical density for MBCF reactions obtained before the bPPD skin test within sensitinogen-stimulated cats was significantly greater than the mean for the control cats. In addition, antigens ESAT-6 and CFP10, commonly used for stimulation in the IFN-γ assays are also cited to cross-react with nontuberculosis mycobacteria such as M. kansasii, M. marinum, and M. gordonae. 25 These findings suggest that antigens 16/83, 16 kDa, and MPB83 play a major role in the humoral immune response of domestic cats stimulated with sensitinogen.
The identification of immunodominant antigens 16/83, 16 kDa, MPB83, and MBCF in domestic cats had not been elucidated before this experiment and identifies the need for an experiment using virulent M. bovis strains as there are inherent limitations of using a heat-killed product such as sensitinogen. Documentation on the humoral and cell-mediated immune responses for sensitinogen versus a live M. bovis strain are lacking; however, one can speculate that antigens that are actively secreted especially during replication or those easily destroyed during heat processing will likely be represented at reduced or absent responses. In the present study, the only available option due to low numbers of known feline cases of natural M. bovis infection and standard animal care guidelines was to evaluate the antigenic profile of sensitinogen-stimulated cats. In addition, this experiment did not include cats infected with nontuberculous mycobacteria. Future research comparing the antigenic profiles of cats infected with virulent M. bovis and nontuberculous mycobacteria are needed to optimize these potential diagnostic assays.
The epidemiology of feline tuberculosis, its relationship to bovine tuberculosis, and formulation of practical management strategies has been hindered by lack of reliable methods of antemortem detection of mycobacterium infections in cats. The 3 immunodiagnostic assays for bovine tuberculosis evaluated in the present study show the potential of detecting specific immune responses in experimentally sensitinogen-stimulated SPF cats. The preliminary results show promise for the development of a serologic antemortem assay for domestic feline M. bovis infection. All tests (bPPD single skin test, MAPIA, and rapid test) would greatly benefit from a controlled study to determine the sensitivity and specificity of these tests within domestic cats infected with virulent M. bovis infection.
Acknowledgements
The authors would like to acknowledge Janet Hengesbach, Jean Gaymer, Danielle Ferguson, and the MSU animal containment facility staff for help in animal care and restraint, and RoseAnn Miller for help in data analysis.
Footnotes
a.
Harlan Laboratories Inc., Indianapolis, IN.
b.
FELOVAX PCT, Fort Dodge Laboratories Inc., Fort Dodge, IA.
c.
National Veterinary Service Laboratory, U.S. Department of Agriculture, Ames, IA.
d.
Synbiotics Corp., San Diego, CA.
e.
Chembio Diagnostic Systems Inc., Medford, NY.
f.
Linomat IV, Camag Scientific Inc., Wilmington, DE.
g.
Sigma-Aldrich, St. Louis, MO.
h.
Kirkegaard & Perry Laboratories, Gaithersburg, MD.
i.
SAS Institute Inc., Cary, NC.
