Abstract
The aims of the current study were to assess the prevalence of von Willebrand disease (vWD) in dogs from the region of Botucatu, São Paulo State, Brazil, and to evaluate laboratory tests to diagnose this disease. The study included 350 dogs of various ages, different breeds, and both sexes. Dogs included in the study had no historical or clinical evidence of abnormal bleeding. von Willebrand factor antigen (vWF: Ag), buccal mucosal bleeding time, activated partial thromboplastin time, and factor VIII activity were evaluated in their ability to diagnose vWD. The prevalence of vWD in dogs was 1.43% in the Botucatu region of Brazil. Determination of vWF: Ag was the best laboratory test to diagnose vWD.
Keywords
Introduction
von Willebrand disease (vWD) is the most commonly inherited hemostatic abnormality in humans 10,22 and dogs. 3,4 The prevalence of vWD in men is 0.82–1.6%, although the incidence could be overestimated because of the difficulties in laboratory diagnosis of this disease. 42 Type I vWD is more frequently diagnosed (60–80%), followed by type II vWD (15–30%), and type III vWD (5–10%). 10 In dogs, vWD has been diagnosed in more than 54 breeds in the United States, with a higher prevalence in Doberman Pinschers, Airedale Terriers, and Scottish Terriers. 19,36 In the United Kingdom, vWD is more prevalent in German Shepherd Dogs, Golden Retrievers, Miniature Schnauzers, Pembroke Welsh Corgis, and Standard Manchester Terriers. 20 von Willebrand disease is inherited as an autosomal trait, which can be recessive or incompletely dominant. 16 Although acquired vWD has been associated with hypothyroidism, 1 recent studies did not find a correlation between vWD and hypothyroidism. 13,26
von Willebrand disease is caused by a quantitative or qualitative defect or by both defects in von Willebrand factor (vWF), a multimeric glycoprotein of high molecular weight. 8,10 This glycoprotein is necessary for the adhesion of platelets to exposed vascular subendothelial collagen in areas of high blood flow. 4 von Willebrand Factor functions by triggering and supporting platelet adhesion to the site of vascular injury 24,25 and by functionally stabilizing factor VIII in the plasma. 10,33 In dogs, clinical signs of vWD are similar to those in humans, 32 such as mucosal hemorrhage, prolonged bleeding following surgery, and excessive bleeding with tooth eruption. 33 In vWD type I, mild bleeding occurs when induced by injury. In vWD types II and III, a tendency toward severe bleeding exists. 4
In dogs, vWD is classified according to clinical severity, plasma vWF concentration, and the type of vWF multimeric structures. 4 Three types of vWD occur in dogs. 4,31 Type I is defined as a quantitative partial deficiency in vWF; type II involves a disproportional loss of high–molecular weight multimeric forms of vWF; type III is a severe quantitative deficiency in vWF. 4 Diagnosis vWD is based on the quantification of plasma vWF and tests of vWF-dependent platelet function, both in vivo and in vitro. 9,16
In veterinary medicine, hemorrhagic diathesis can have several causes and can be challenging to diagnose. Additionally, affected dogs might not always display clinical signs of bleeding, and excessive hemorrhage might only be evident after surgery or trauma. Proper laboratory diagnosis of vWD is necessary to decrease the incidence of the disease by excluding affected individuals from breeding programs. The objectives of this study were to determine the prevalence of vWD in dogs from the Botucatu region of Brazil and to evaluate various diagnostic tests in their ability to diagnose the disease.
Materials and methods
Animals
Three hundred and fifty dogs of various ages, breeds, and both sexes were selected for study. None of the dogs had a medical history or clinical evidence of excessive bleeding. All dogs were evaluated at the Veterinary Hospital of the School of Veterinary Medicine and Animal Science, São Paulo State University (Botucatu, São Paulo, Brazil).
Sample collection and storage
Blood was collected from the jugular vein in a single extraction. Vacuum tubes for platelet counts contained 10% ethylenediamine tetra-acetic acid. a Vacuum tubes for determination of activated partial thromboplastin time (aPTT), factor VIII (FVIII) activity, and vWF antigen (vWF: Ag) contained 3.2% sodium citrate. b Blood was collected in the sodium citrate tubes at a ratio of 1 part anticoagulant to 9 parts whole blood, and the tube was then placed in an ice bath. These latter blood samples were immediately centrifuged at 1,710 × g for 15 min, and the citrated plasma was collected, placed in Eppendorf tubes, and stored at −80°C until analyzed.
Laboratory assays
Platelet count. Platelets were counted in an electronic cell counter, c and platelet morphology was evaluated via Romanowsky-stained blood smears. 14 For inclusion in the study, dogs were required to have a count of >200,000 platelets/μl.
Buccal mucosal bleeding time. Buccal mucosal bleeding time (BMBT) was determined according to a technique modified from the veterinary literature. 23 The animal was placed in lateral or sternal recumbency, and the upper lip was everted. A standardized vertical incision was made with a lancet d perpendicular to the lip margin, directly above the maxillary canine tooth. Blood was absorbed with a round filter paper 1–2 mm below the incision until bleeding stopped. The bleeding time was recorded in seconds with a chronometer, extending from the time of the initial incision until bleeding ceased.
von Willebrand factor antigen. von Willebrand factor antigen was assessed by direct enzyme-linked immunosorbent assay (ELISA; a comparative semiquantitative test) with the use of anti-canine vWF antibody e according to the manufacturer's guidelines. Control samples were kindly supplied by Dr. James Catalfamo.
Coagulation tests. Activated PTT and FVIII were only determined in animals that had ≤70% vWF: Ag. Activated PTT was determined with a commercial kit. f Factor VIII was determined by evaluating the capacity of the patient's citrated plasma to correct the aPTT from FVIII-deficient plasma. g As a control for aPTT, citrated plasma from 8 animals and a plasma pool (4 animals) were used. All control plasma samples had >70% vWF: Ag and were considered free of vWD. The control plasma samples also provided a reference interval for the aPTT test (14.67 ± 2.19 sec). The reference interval adopted for FVIII activity was 56–180%. 37 The vWD status of the study animals was classified according to vWF: Ag results as follows: negative for vWD (>70%), suspect for vWD (50-70%), and vWD affected (<50%). 39
Statistical analyses
von Willebrand factor antigen was compared with BMBT, aPTT, and FVIII with the use of Pearson's correlation coefficient. This statistical test was also used to compare aPTT with FVIII. Comparisons for vWF: Ag were done among breeds and between sexes for vWD-negative and vWD-suspect animals, excluding breeds represented by only 1 individual. Analysis of variance was followed by Tukey's test. For vWD-suspect animals, the variables BMBT, aPTT, and FVIII were evaluated according to the methodology also used for vWF: Ag. For vWD-affected animals, the values obtained for vWF: Ag, BMBT, aPTT, and FVIII were not compared among breeds or between sexes because there were only 5 animals (5 different breeds with 1 male each). von Willebrand disease-suspect and -affected animals were compared for the parameters vWF: Ag, BMBT, aPTT, and FVIII by the Wilcoxon nonparametric test. All analyses were done with the use of commercial software. h Significance level was P = 0.05.
Results
The analysis of vWF: Ag in vWD-negative animals indicated no statistical differences between sexes. The greatest statistical differences among breeds were found in Siberian Huskies and Pugs (Table 1). Akita, Alaskan Malamute, American Fox Terrier, Border Collie, Chow Chow, Dalmatian, Doberman Pinscher, Dogue de Bordeaux, English pointer, English Springer Spaniel, Fila Brasileiro, Giant Schnauzer, Golden Retriever, Irish Setter, and Pekingese dogs were not included in Table 1 because only 1 individual was represented for each breed. No statistical differences for vWF: Ag, BMBT, and aPTT were found for sex or breed in vWD-suspect dogs. Factor VIII was significantly different in certain breeds, but no differences were found regarding sex (Table 2). Basset Hound, Boxer, Doberman Pinscher, German Shepherd Dog, German Spitz, Giant Schnauzer, Golden Retriever, Labrador Retriever, Lhasa Apso, Miniature Dachshund, Terrier Brasileiro, Pembroke Welsh Corgi, and Yorkshire Terrier dogs were not included in Table 2 because these breeds were represented by only 1 individual each.
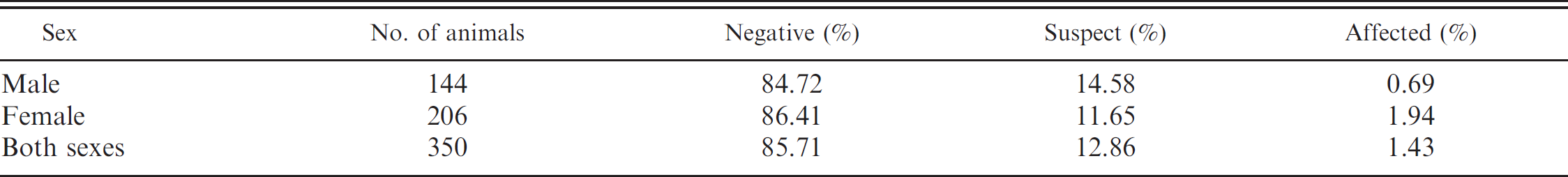
On the basis of 350 samples tested, as presented in Tables 1, 2, and 4, 300 (85.71%) dogs were vWD-negative, with a vWF: Ag value of 155.99 ± 84.86% (x ± standard deviation) and a range of 70.66–619.80%; 45 (12.86%) dogs were vWD-suspect, with a vWF: Ag value of 61.11 ± 5.05% and a range of 51.16–69.55%; and 5 (1.43%) dogs were vWD-affected, with a vWF: Ag value of 33.49 ± 11.52% and a range of 24.15–48.70%. von Willebrand disease–affected dog breeds included Doberman Pinscher, Golden Retriever, Miniature Poodle, Yorkshire Terrier, and mixed breed (Table 4). The incidence of vWD for these breeds was 33.33%, 33.33%, 1.64%, 7.14%, and 1.75%, respectively. The sex of vWD-affected dogs was 0.69% (1/146) male and 1.94% (4/204) female, with no statistical difference between sexes. Most of the tested animals were adults, and none showed hemorrhagic tendencies during blood collection.
Results of von Willebrand factor antigen determination (mean and standard deviation [SD]) in dogs that were test-negative for von Willebrand's disease.
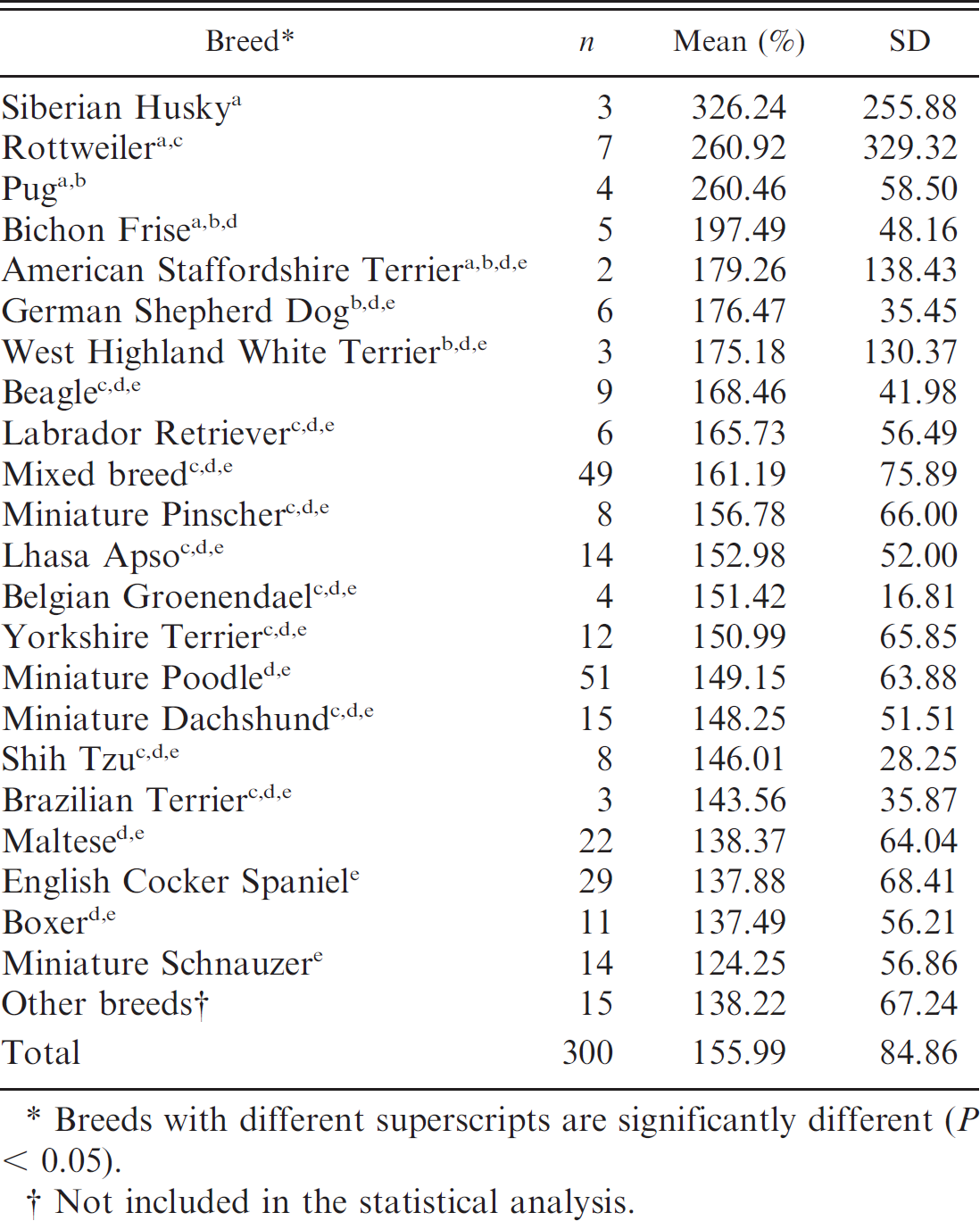
Breeds with different superscripts are significantly different (P < 0.05).
Not included in the statistical analysis.
No statistical differences were present between BMBT values of vWD-negative and -suspect dogs. However, statistical differences were found between BMBT values of vWD-negative and vWD-affected animals and between vWD-suspect and -affected animals. In this study, no correlation was found between vWF: Ag and BMBT of vWD-affected or -suspect animals. All aPTT and FVIII values found in this study were within the adopted reference values. 37 No statistical differences were found for aPTT and FVIII values between vWD-suspect and -affected animals. Additionally, there was no correlation between vWF: Ag and aPTT or between vWF: Ag and FVIII of vWD-affected or -suspect dogs. In contrast, a correlation was present between aPTT and FVIII values for vWD-suspect dogs, but not for vWD-affected dogs.
Discussion
Siberian Husky, Rottweiler, Pug, Bichon Frise, American Staffordshire Terrier, West Highland White Terrier, Miniature Pinscher, Belgian Groenendael, and Shih Tzu dogs were negative for vWD, with at least 2 individuals tested per breed. These breeds generally had the highest vWF: Ag concentrations (Table 1), indicating a possible difference in vWF gene expression among breeds. The BMBT values obtained for all vWD-negative dogs ranged from 30 to 195 sec and were within the reference intervals proposed by several authors. 2,5,11,15,30
The laboratory test of choice for initial diagnosis of vWD is determination of vWF: Ag; however, BMBT, aPTT, and FVIII also have a limited value for the diagnosis of vWD. The overall prevalence of vWD-affected dogs is similar to a published prevalence of 0.82–2% 10,42 for humans. The breed predisposition of Doberman Pinscher and Golden Retriever to have vWD has also been reported by other authors. 6,18,38
Results of von Willebrand factor antigen (vWF: Ag), buccal mucosal bleeding time (BMBT), activated partial thromboplastin time (aPTT), and factor VIII (FVIII) activity of von Willebrand disease–suspect dogs (mean ± standard deviation).
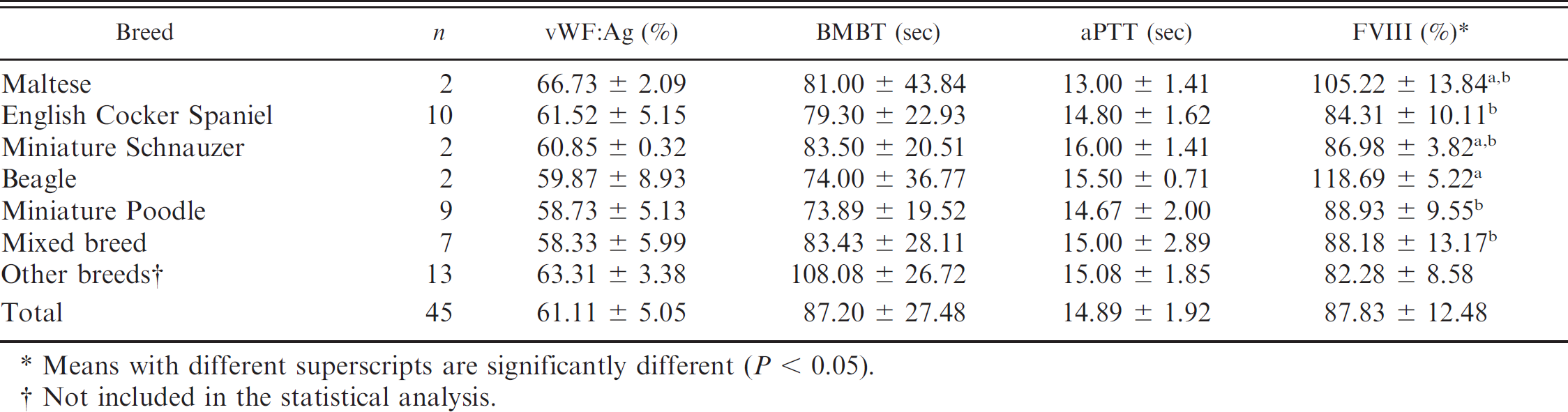
Means with different superscripts are significantly different (P < 0.05).
Not included in the statistical analysis.
Absolute number (n = 350) and relative percentage of dogs that were negative for, suspect for, or affected with von Willebrand's disease.
The incidence of vWD reported in Yorkshire Terriers (7.14%) was higher than the overall mean in the current study (1.43%). Perhaps because only 14 dogs were tested and only 1 individual had decreased vWF: Ag. The incidence of vWD in Miniature Poodles and mixed breed dogs in this study was similar to that reported previously because these breeds are more common in Brazil.
For vWD-suspect dogs, additional laboratory testing would be required to definitively identify them as vWD-negative or -affected individuals. In addition, the sire, dam, and siblings could also be tested and might reveal additional information regarding the familial incidence of vWD, if present. A high incidence of vWD-suspect dogs was found in this study for Basset Hounds, Doberman Pinschers, Giant Schnauzers, Golden Retrievers, and Pembroke Welsh Corgis that also have a predisposition for vWD. 20 Because few individuals of these breeds were tested, more vWD-suspect dogs might have been found.
A higher prevalence of vWD-affected females was found in this study in contrast to a higher incidence of vWD in males in previous reports. 7,39 However, the difference of vWD between sexes tends to decrease as the sample size increases because diseases inherited in an autosomal pattern affect males and females equally. 20 However, a higher percentage of females has been reported 38 because postsurgery bleeding is usually more common following ovariohysterectomy, parturition, and estrus.
The BMBT testing was done without sedation or anesthesia, but the procedure was well tolerated, as previously reported. 15,30 All BMBT values in this study were within reference intervals reported for dogs. 15 The BMBT values for vWD-affected dogs were not increased, possible because vWF: Ag concentrations were not markedly decreased (Table 4) and a slightly different technique was used. Previous studies used a device to make the incision followed by direct pressure on the maxillary or mandibular mucosae with manual pressure to ensure high BMBT values for vWD-affected dogs. 2,5,15,30 In this study, the BMBT was not very sensitive in the detection of vWD in dogs and may not be prolonged with slight decreases in plasma vWF activity when the platelet function is adequate. 30 However, the use of a lancet instead of a template-bleeding device could be responsible for the lack of sensitivity of the BMBT in this study. The incision made by a lancet does not provide control over the depth and length of the incision and, thus, is not appropriate for this test.
A correlation between vWF: Ag and BMBT was found for vWD-affected Doberman Pinschers 5 but not for Greyhounds. 30 This observation could be the result of a difference in the multimeric structure of vWF. Greyhounds with a low plasma vWF concentration can have a higher concentration of high molecular weight multimers compared with Doberman Pinschers. 30 The absence of a correlation between vWF: Ag and FVIII observed in the current study has been reported for vWD-negative dogs 27 ; however, previous authors 37 have found a positive correlation in dogs with vWD.
Results of von Willebrand factor antigen (vWF: Ag), buccal mucosal bleeding time (BMBT), activated partial thromboplastin time (aPTT), and factor VIII (FVIII) activity tests of dogs with von Willebrand's disease.
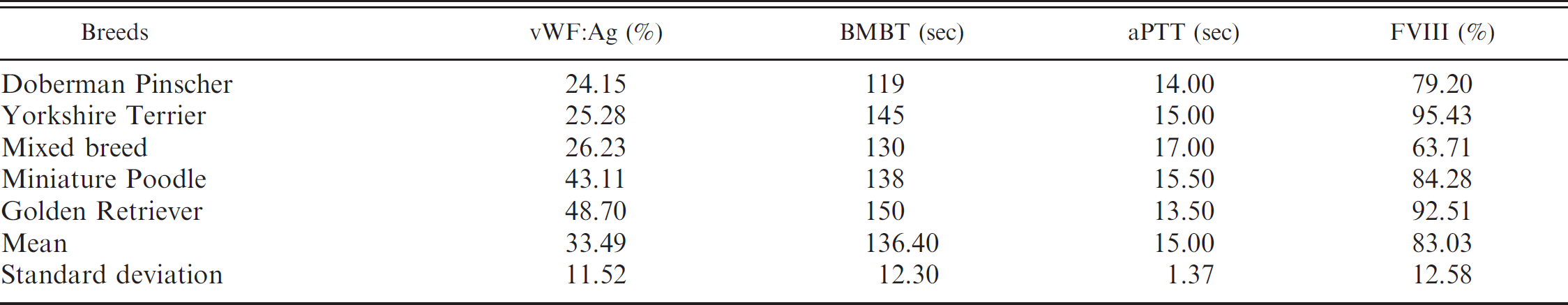
In the present study, results for aPTT and FVIII did not fluctuate for vWD-affected and -suspect dogs. This observation differs from previous reports, wherein binding of FVIII to vWF prolonged the half-life of FVIII in the circulation, and decreased vWF concentration was associated with decreased FVIII activity. 17,29,34 However, other researchers state that aPTT can be within the reference intervals in vWD-affected dogs. 18 In a previous study, all vWD-affected dogs had reduced FVIII activity, but not to the same degree as the decrease in vWF: Ag and not severe enough to prolong the aPTT. 20 A prolonged aPTT and moderate decrease in FVIII activity has been observed in some dogs with reduced vWF: Ag 7,40 and was most likely due to the lower vWF: Ag concentration in animals with type II vWD, compared with the vWD-affected animals in this study.
The 5 vWD-affected dogs in the current study probably had type I vWD, which wouldexplain the lack of prolongation in the aPTT and decreased FVIII activity. Thus, aPTT testing alone diagnoses few dogs with vWD. 21 In humans, type I vWD has a decrease in FVIII activity that is proportional to the reduction in vWF: Ag, although some patients will have normal FVIII activity. 9,41 Human patients with vWD types II and III have a marked decrease in FVIII activity, 12,28 demonstrating that the response of FVIII activity and aPTT is different in dogs and humans.
In this study, a correlation was present between aPTT and FVIII for vWD-suspect dogs, but not for vWD-affected dogs, probably because of the small number of animals that had vWF: Ag values <50% (5 animals). It is important to note that a single normal measurement of vWF: Ag does not necessarily exclude vWD, especially in dogs that only have a slight decrease in vWF: Ag. Some vWD-affected dogs might not have laboratory abnormalities when tested because vWF: Ag might increase with exercise, pregnancy, endotoxemia, azotemia, liver disease, and other illnesses. 35
The prevalence of vWD in dogs without clinical evidence of hemorrhage is 1.43% in São Paulo State. Furthermore, no evidence of sexual predisposition exists, and Doberman Pinschers and Golden Retrievers have a higher incidence of the disease. The test of choice for initial diagnosis of vWD is the determination of vWF: Ag. Buccal mucosal bleeding time, aPTT, and FVIII determination have a limited value for the diagnosis of vWD.
Acknowledgements
The authors thank Instituto Brasileiro de Diagnósticos e Especialidades Veterinárias (PROVET Moema).
Footnotes
a.
Vacutainer®, BD, Franklin Lakes, NJ.
b.
Vacuette®, Greiner Bio-one, Kremsmüenster, Österreich, Austria.
c.
CELL-DYN 3500 R, Abbott Laboratories, Abbott Park, IL.
d.
Feather Blood Lancets®, Feather Safety Razor Co. Ltd., Kita-ku, Osaka, Japan.
e.
Sheep anti-canine VWF®, Research Diagnostics Inc., Concord, MA.
f.
Helena aPTT SA (ellagic acid), Helena Laboratories Corp., Beaumont, TX.
g.
Factor VIII Deficient Plasma, Helena Laboratories Corp., Beaumont, TX.
h.
SAS software for Windows v. 9.1.3, SAS Institute Inc., Cary, NC.
