Abstract
A 3-year-old, intact, male Beagle dog developed clinical signs of pleurothotonus and altered head position to the right, neck pain, nystagmus, hyperreflexia of the left forelimb, and hyperextension of both forelimbs. Magnetic resonance imaging enabled a tentative diagnosis of thalamic neoplasia with incidental hydromyelia at the level of the second cervical vertebra. The animal was euthanatized due to the poor prognosis, and a necropsy was performed. A large, well-demarcated, nonencapsulated, and focally infiltrative mass was present in the approximate location of, and effacing, the pineal gland. The mass was composed of densely packed polyhedral neoplastic cells that exhibited epithelial characteristics, such as intercellular junctions, and contained carbohydrate granules and occasionally melanin granules. Immunohistology confirmed that neoplastic cells expressed neuron-specific enolase and, in a small proportion, cytokeratin. These combined findings led to the diagnosis of a papillary tumor of the pineal region, a tumor not previously described in dogs.
Primary pineal tumors are very uncommon in domestic animals. To date, pineal parenchymal tumors (i.e., pineocytomas and pineoblastomas) have been described in rats, with isolated reports in other species, including the cow, goat, horse, chicken, and cockatiel. 12,15 In humans, mixed pineocytoma/pineoblastomas are also reported, and recently, an additional variation has been characterized: papillary tumor of the pineal region (PTPR), a neoplasm that is thought to originate from the specialized ependymal cells of the subcommissural organ 3,5,13 or pineal gland. 8,9 The current report describes the clinical, histologic, immunohistologic, and ultrastructural characteristics of a tumor within the pineal region of a dog consistent with a diagnosis of PTPR. A 3-year-old, intact, male Beagle dog was first presented at the referral hospital with a 2-month history of neck pain, progressing to pleurothotonus to the right, sporadic nystagmus, altered head position to the right, hyperreflexia of the left forelimb, and hyperextension of both forelimbs.
Routine serum biochemistry, complete blood count, cerebrospinal fluid analysis, T3/T4 assay, and Toxoplasma and Neospora serology screening tests were performed with unremarkable results. A low-field magnetic resonance imaging scanner a was used to obtain scans in 3 planes of orientation (sagittal, transverse, and dorsal). The scans revealed a well-circumscribed mass that was hypointense on T1 weighting and hyperintense on T2 weighting (Fig. 1A-C). After administration of dimeglumine gadopentetate, b increased signal intensity on T1-weighted images was observed (Fig. 1D). The mass was located in the approximate location of the pineal gland. It was occupying the quadrigeminal cistern and causing caudal aqueductal stenosis, which had resulted in obstructive internal hydrocephalus (Fig. 2A). In addition, there was hydromyelia of the spinal cord at the level of the second cervical vertebra. A thalamic tumor was suspected, and the animal was euthanized due to the poor prognosis.
Necropsy was performed and did not yield any significant pathologic findings in any organ or parenchyma, apart from the brain. The brain was examined in detail after fixation in neutral buffered, 10% formalin solution for several days. Grossly, the brain exhibited a dark brown, marbled, soft neoplastic mass (cross section: 1.5 cm × 2.5 cm) extending over the entire length of the mesencephalon (rostral to caudal colliculi) within the thalamus and between the hippocampal gyri dorsolaterally, obliterating the pineal body and the commissure of the rostral colliculum and compressing the aqueduct (Fig. 2A, 2B). The neoplasm appeared well circumscribed and nonencapsulated.
Sections from representative brain areas and the neoplasm were prepared and routinely embedded in paraffin wax. Three- to 5-μm sections were prepared and stained with hematoxylin and eosin. Selected tumor sections were also stained with Masson Fontana to highlight melanin and with periodic acid-Schiff with and without diastase treatment to identify carbohydrates, such as glycogen, or used for immunohistological examination. Immunohistology was performed using antibodies for vimentin (mouse anti-swine vimentin; clone V9), c glial fibrillary acidic protein (rabbit anti-glial fibrillary acidic protein), c melan-A (mouse anti-human melan A; clone A103), c synaptophysin (mouse anti-cow synaptophysin; clone SY-38), c cytokeratin (mouse anti-human-cytokeratin; clone AE1/AE3), c S-100 protein (rabbit anti-cow S-100 protein), c neurofilament (mouse anti-human neurofilament protein; clone 2F11), c and neuron-specific enolase (mouse anti-human neuron specific enolase; clone BBS/NC/VI-H14) c that have been shown to cross-react with canine tissues, according to previously published protocols. 10 Transmission electron microscopy was performed on formalin-fixed neoplastic tissue after postfixation in 4% paraformaldehyde/2.5% glutaraldehyde in a sodium cacodylate buffer and secondary fixation in osmium tetroxide.
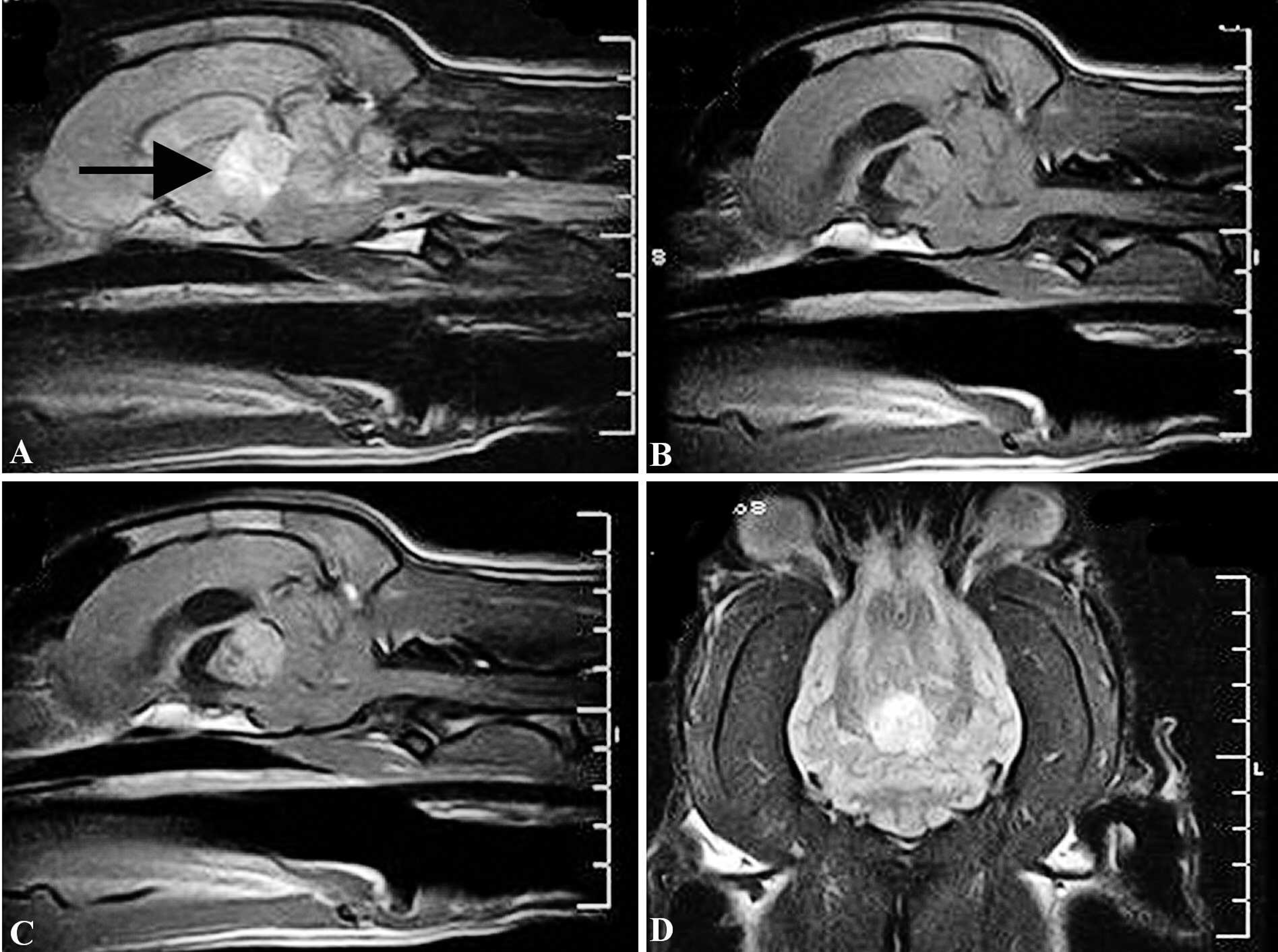
Magnetic resonance imaging (MRI) of the brain.
Histologically, the neoplastic mass showed focal infiltrative growth dorsally into the white matter of the hippocampal commissure (Fig. 2C). The mass was composed of lobules of closely packed, relatively monomorphic polyhedral cells, which were occasionally arranged in palisading, papillary, and pseudorosette patterns within a delicate, pale eosinophilic, fibrillar stroma (Fig. 2D). Individual cells were approximately 10 μm in diameter, with pale eosinophilic cytoplasm, distinct cell borders, and a central, large (8 μm), round, hypochromatic nucleus (Fig. 2E). The cells often contained dark brown granules that were positive by Masson Fontana stain (melanin; Fig. 2F) and appeared period acid-Schiff positive (Fig. 2G). The period acid-Schiff reaction also identified diastase-resistant carbohydrates in numerous pigment-free cells (Fig. 2G). In addition, scattered round cells with abundant melanin were seen (melanoma-crophages; Fig. 2F). Mitotic figures were rare. Multifocally, variably sized necrotic areas containing detached neoplastic cells were present. The immediately adjacent neuroparenchyma of the hippocampal commissure exhibited compression and mild vacuolation of the white matter. Phosphotungstic acid hematoxylin failed to demonstrate cilial basal bodies (blepharoblasts), which may be observed in some cases of ependymoma. Immunohistology identified a strong diffuse cytoplasmic staining for neuron-specific enolase in neoplastic cells (Fig. 2H). Scattered individual and groups of neoplastic cells exhibited moderate cytokeratin expression (Fig. 2I). A small proportion of neoplastic cells exhibited weak nuclear S-100 protein expression. Neoplastic cells were negative for vimentin, neurofilament, synaptophysin, glial fibrillary acidic protein, and melan-A.
Ultrastructural examination was limited due to the presence of marked fixation artifacts; however, it was obvious that neoplastic cells exhibited electron-lucent nuclei and a mitochondria-rich cytoplasm. Occasional desmosomes were observed between adjacent cells (Fig. 3A). Melanin pigment was evident within neoplastic cells, and melaninladen macrophages were recognized (Fig. 3B).
Given the anatomical site of the neoplasm, a diagnosis of pineal parenchymal tumor (pineocytoma, pineoblastoma) was considered. However, based on the light and electron microscopical findings, the diagnosis of a PTPR was considered more appropriate. As differential diagnoses, both metastatic carcinoma and metastatic melanoma were considered; however, the expression of cytokeratin and the presence of intercellular junctions, in conjunction with the expression of neuron-specific enolase and the lack of melan-A/S-100 protein expression, ruled out these diagnoses, 8 particularly because the clinical and gross postmortem examination did not provide any evidence of a primary tumor in another location.
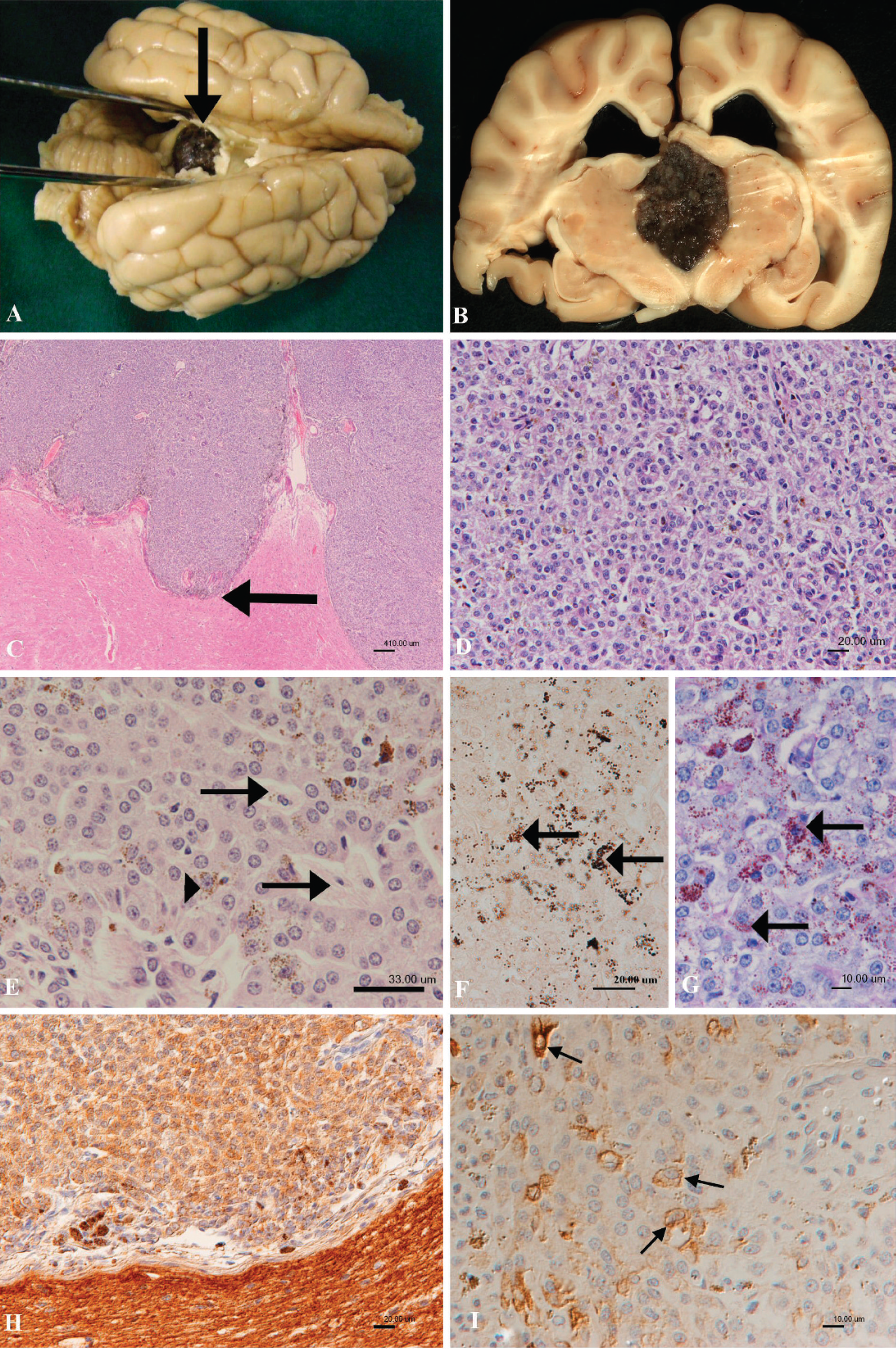
A, brain after formalin fixation. Dorsal view, showing the dark brown-pigmented tumor located between the cerebral hemispheres, obscuring the pineal gland and the midbrain (arrow).
Among pineal parenchymal tumors, pineocytomas are expansile, highly cellular neoplasms with lobular architecture that are composed of polygonal cells, which form pseudorosettes and may form irregular fibrillary zones referred to as pineocytomatous rosettes. Cells in the fibrillary zones express synaptophysin, neuron-specific enolase, and occasionally neurofilament protein. 12 Pineoblastomas lack the lobular architecture and exhibit greater cell pleomorphism, multifocal necrosis, and locally invasive behavior. 11,12
The clinical signs in the presented case can be explained by progressive obstructive hydrocephalus with increased intracranial pressure, as well as the compression of structures surrounding the tumor, such as the thalamus, the rostral to caudal colliculi, and, dorsally, the white matter of the hippocampal commissure. Neck pain has been described as a consequence of meningeal compression. 18 The dog sporadically showed nystagmus, which could be explained by interruption of the thalamic relay pathways from the vestibular nuclei and cerebellum. In humans, the most common clinical signs associated with tumors of the pineal gland are due to pressure on the roof of the midbrain (quadrigeminal plate). Ocular paresis is common. At a later stage, pyramidal and cerebellar signs contribute. In addition, obstructive effects on cerebrospinal fluid flow have been described. 6
As in the present case, clinical pathology parameters were all expected to be within normal limits in cases of primary pineal tumors. On magnetic resonance imaging studies, the neoplasm was well defined, and dimeglumine gadopentetate accumulated within and enhanced the signal generated from tumor tissue. This effect is due to gadolinium leakage from the focally disrupted blood brain barrier within the neoplasm. 16,19 The location of the mass in the pineal region was clearly demonstrated by magnetic resonance imaging, which also showed that it had compressed the quadrigeminal cistern and had caused caudal aqueductal stenosis and subsequent obstructive internal hydrocephalus. These findings are comparable to those seen in human patients with primary tumors of the pineal gland. 4,7 The hydromyelia that was observed at the level of the second cervical vertebra in the present case was considered an incidental finding.
In the present case, the diagnosis of a PTPR, a tumor not previously described in the dog, is based on histologic, immunohistological, and ultrastructural findings. Papillary tumors of the pineal region in humans constitute a rare class of tumors that manifest in children and young adults and are thought to arise from the specialized ependymal cells of the subcommissural organ 3,5,13 or pineal gland. 8,9 These neoplasms are usually relatively large (2.5–4 cm) and well-circumscribed and show a low T1 and increased T2 signal, as well as contrast enhancement on magnetic resonance imaging. To date, 38 human PTPR cases have been published, 3,8,14 and the diagnosis is based on the tumor site, its papillary growth, and epithelial morphology, in combination with its immunoreactivity for cytokeratin and, in some cases, focal glial fibrillary acidic protein expression. 14 The biologic behavior of PTPRs is variable and may correspond to World Health Organization grades II or III, but precise histologic grading criteria remain to be defined. 14 Although macroscopically indistinguishable from pineocytomas, PTPRs are histologically distinct from pineal parenchymal tumors, exhibiting a loose papillary to more densely cellular arrangement of neoplastic cells with rosettes, pseudorosettes, and tube formation, often with necrotic areas and vacuolated tumor cells that occasionally contain periodic acid-Schiff-resistant carbohydrates. 3,8 Ultrastructural features suggest ependymal differentiation and some secretory function. 8
In the present case, the growth pattern and light and electron microscopy features, together with cytokeratin expression, are suggestive of an epithelial origin. The presence of intracytoplasmic diastase-resistant carbohydrates and a total lack of synaptophysin expression, the latter of which is a consistent feature and diagnostic criterion for pineal parenchymal tumors in both humans and animals, 15,17 is compatible with the diagnosis of a PTPR. In considering the diagnosis of PTPR, several antigens in the present case were expressed that were consistent with results of a multicenter study of 31 human PTPRs, in which neuron-specificenolase expression was observed in 100% of cases (17 tested) and cytokeratin in 100% of cases (30 tested) and S-100 protein expression in 92% of cases (24 tested). 3 However, in the present case, neoplastic cells were negative for vimentin and synaptophysin expression, whereas in the study, vimentin expression was observed in 85% (21 tested) and synaptophysin in 12% (25 tested). 3 Glial fibrillary acidic protein was usually negative in human PTPRs, as in the present case. 3,5,8,13
Based on the growth pattern of the described tumor, a differential diagnosis of papillary ependymoma must be considered. However, the tumor showed no communication with the ventricles or the mesencephalic aqueduct, which suggests that it did not have any connection to the pre-existing ependyma but had originated from the specialized ependymocytes from within the pineal gland or the subjacent subcommisural organ, which are the putative cell of origin of PTPRs. 8,9,14 As such, a neoplasm arising from this cell population of specialized ependymocytes is recognized as a separate entity to those originating from the ependyma, the latter of which consistently express glial fibrillary acidic protein with coexpression of vimentin. 1
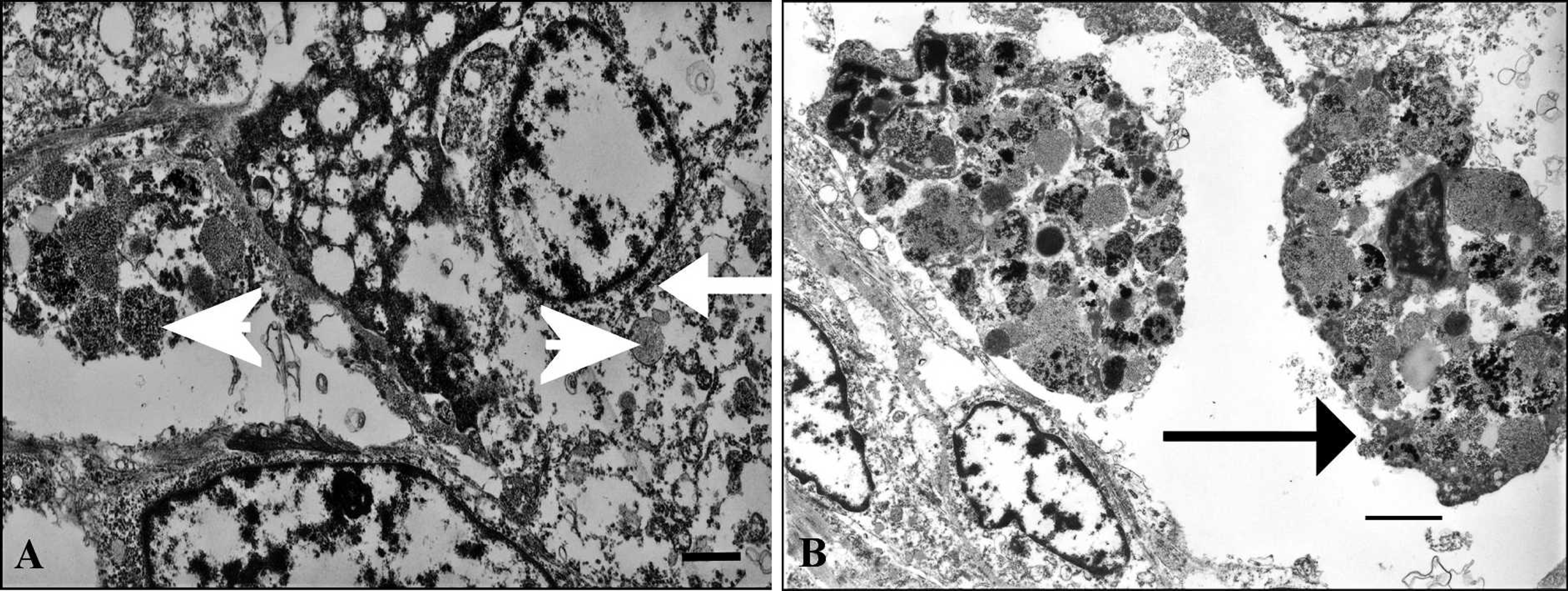
Transmission electron microscopy.
The presence of melanin pigment in a proportion of neoplastic cells was observed in the present case without other features of melanocytic cells, such as melan-A and S-100 protein. In the dog, melanin pigmentation of the pineal body is frequently seen and considered normal. 2 In the present case, it is therefore most likely an incidental finding and unrelated to the neoplastic process.
Footnotes
a.
VetMR Grande, 0.375 Tesla field, Esaote Europe BV, Reading, Berkshire, United Kingdom.
b.
Magnevist®, Schering Health Care Ltd., Burgess Hill, West Sussex, United Kingdom.
c.
Dako UK Ltd., Ely, Cambridgeshire, United Kingdom.
