Abstract
A 1-year-old, intact, male dwarf rabbit (Oryctolagus cuniculus) was vaccinated against myxomatosis and rabbit viral hemorrhagic disease in February 1999, and a localized reaction appeared in the same anatomic site within a few days. No regression was observed after subcutaneous antibiotic treatment. The rabbit was kept under observation, and the swelling apparently disappeared in 3 months. The owner then decided to avoid any further subcutaneous drug administration. The referring veterinarian examined the animal on July 2006 for the sudden appearance of a nodular, 4.5 cm × 3.5 cm × 2.0 cm, subcutaneous mass located over the interscapular space. Fine-needle aspiration was performed, and a population of neoplastic spindle cells, rare pleomorphic multinucleated cells, and rare leukocytes were observed. The mass was surgically removed, fixed in 10% neutral buffered formalin, and routinely processed for histologic, histochemical, and immunohistochemical diagnostic investigation. The neoplastic tissue exhibited fascicles composed of malignant spindle-shaped cells with elongated to oval hyperchromatic nuclei and scant cytoplasm. Occasional multinucleated cells were also observed. The neoplastic cells were immunoreactive for vimentin but did not stain for smooth muscle actin, desmin, myoglobin, and cytokeratins (AE1/AE3). Moreover, the histochemical stain for aluminum was positive. The diagnosis was fibrosarcoma based on morphologic and immunohistochemical results. The histologic features of this neoplasm were remarkably similar to feline injection-site sarcoma.
Few reports have been published on cutaneous neoplasms in pet rabbits. 27 Cutaneous neoplasms in pet rabbits can be classified into 2 main groups: virus induced (Shope fibroma [Rabbit fibroma virus] and Shope papilloma [Cottontail rabbit papillomavirus]) and non-virus induced. 27 Nonviral malignant mesenchymal tumors in pet rabbits include liposarcoma, myxosarcoma, malignant peripheral nerve sheath tumor, fibrosarcoma, leiomyosarcoma, anaplastic sarcoma, osteosarcoma, hemangiosarcoma, and rhabdomyosarcoma. 27 The purpose of the present report is to present a case of a fibrosarcoma in a dwarf rabbit (Oryctolagus cuniculus) that originated at the injection sites of vaccine and antibiotics.

Focal alopecia is present in the interscapular area at the first presentation.
A 1-year-old, intact, male dwarf rabbit was vaccinated against myxomatosis and rabbit viral hemorrhagic disease (RHD) in February 1999 with a live attenuated vaccine and inactivated adjuvanted vaccine, respectively. Both vaccines were subcutaneously injected in the interscapular area; however, a portion of the anti-myxomatosis vaccine was also intradermally injected during needle extraction. A local reaction characterized by subcutaneous swelling and alopecia developed at the injection sites within a few days (Fig. 1). Despite local subcutaneous administration of enrofloxacin (10.0 mg/kg subcutaneously every 24 hr) for 5 days, the swelling did not regress. The rabbit was kept under clinical observation weekly, and the swelling apparently disappeared approximately 3 months later. However, a small nodule was palpated by the owner in the following months, and no further subcutaneous drugs were administered after the initial enrofloxacin treatment. The specific vaccine used to immunize the rabbit against myxomatosis could not be tracked. However, myxomatosis vaccines available on the Italian market in 1999 were generally nonadjuvanted, lyophilized, cell culture-derived, attenuated live virus. Furthermore, recommended administration was via intradermal injection in the external surface of the ear. Alternatively, subcutaneous injection was also possible for adequate immunization. Rabbit fibroma virus vaccine was also available at that time, and kaolin (40 mg/ml) was sometimes added as an adjuvant. In the rabbit of the current report, the vaccine was administered subcutaneously. 29 Some divalent vaccines against myxomatosis and RHD were also available. The vaccine against RHD usually was inactivated and adsorbed onto an aluminum hydroxide adjuvant.
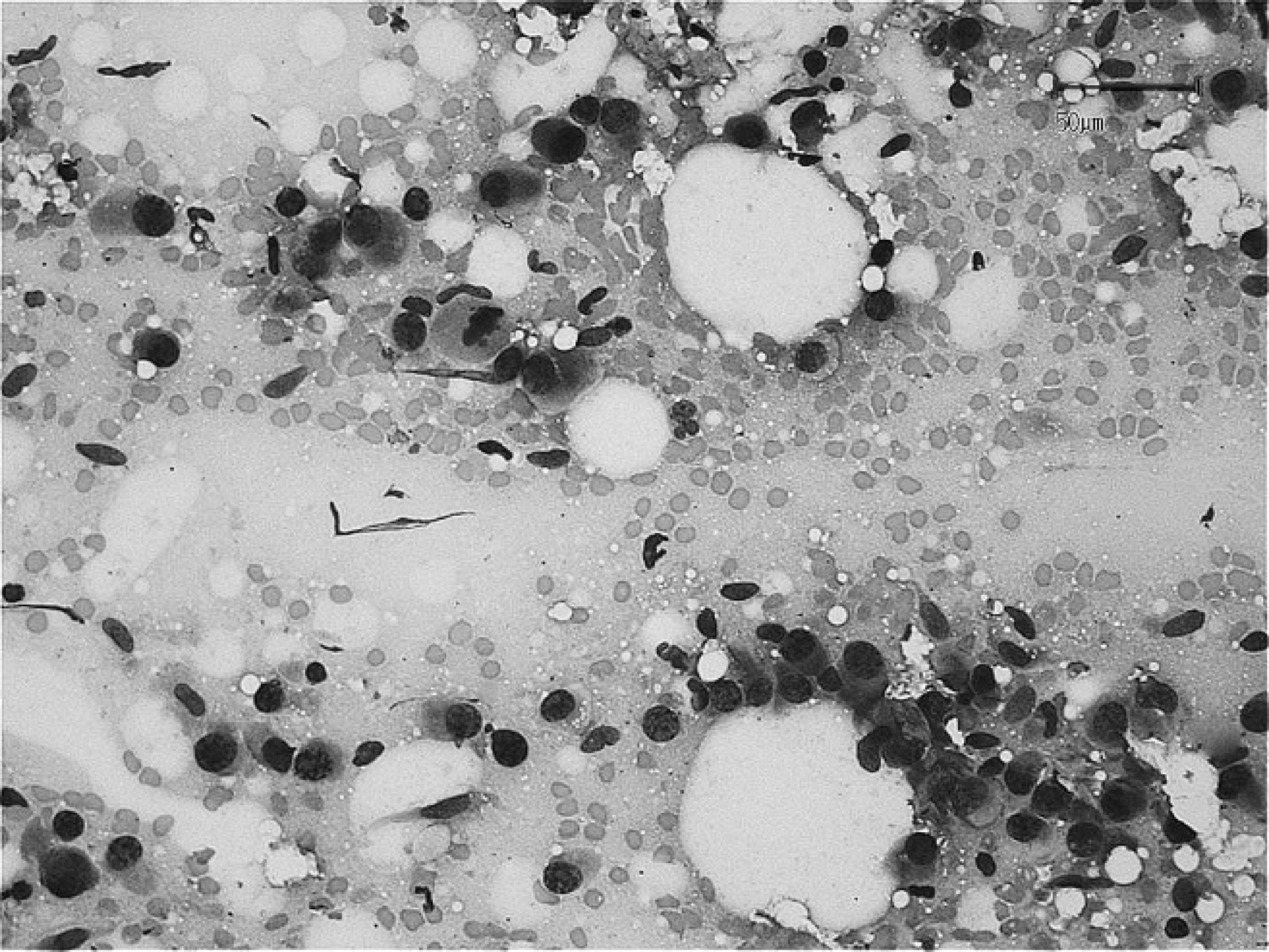
Spindloid cells demonstrate features of malignancy and atypical mitoses. Wright-Giemsa stain. Bar = 50 μm.
The rabbit in the present case was examined on July 2006 by the referring veterinarian when it was 8 years old and had sudden development of a subcutaneous nodule located in the right interscapular-scapular area. After physical examination, a fine-needle aspiration of the subcutaneous nodule was performed for cytologic examination.
The fine-needle aspirates were of good quality but had low to moderate cellularity. The nucleated cell population consisted of many spindloid cells admixed with rare lymphocytes and disrupted heterophils. The spindloid cells had indistinct cell borders and a moderate amount of basophilic cytoplasm that occasionally contained small clear vacuoles (Fig. 2). An intercellular, eosinophilic, delicate matrix was rarely present between cells that were organized into small cohesive groups. The nuclei were fusiform to ovoid with coarse to finely granular chromatin and 1 or more occasionally prominent nucleoli. Nuclei were paracentrally to peripherically located. Rare multinucleated cells were also observed. Anisocytosis and anisokaryosis were noticeable, and mitotic figures were infrequent. A moderate number of erythrocytes, strands of nucleoprotein, and disrupted cells were scattered throughout the preparations. The background appeared eosinophilic and amorphous. The cytologic findings were suggestive of mesenchymal neoplasia, and the most likely cytologic diagnosis was of fibrosarcoma. The differential diagnosis included giant cell tumor of soft tissue, histiocytic sarcoma, malignant fibrous histiocytoma, rhabdomyosarcoma, and anaplastic sarcoma. Complete surgical excision and histologic examination were recommended for a definitive diagnosis.
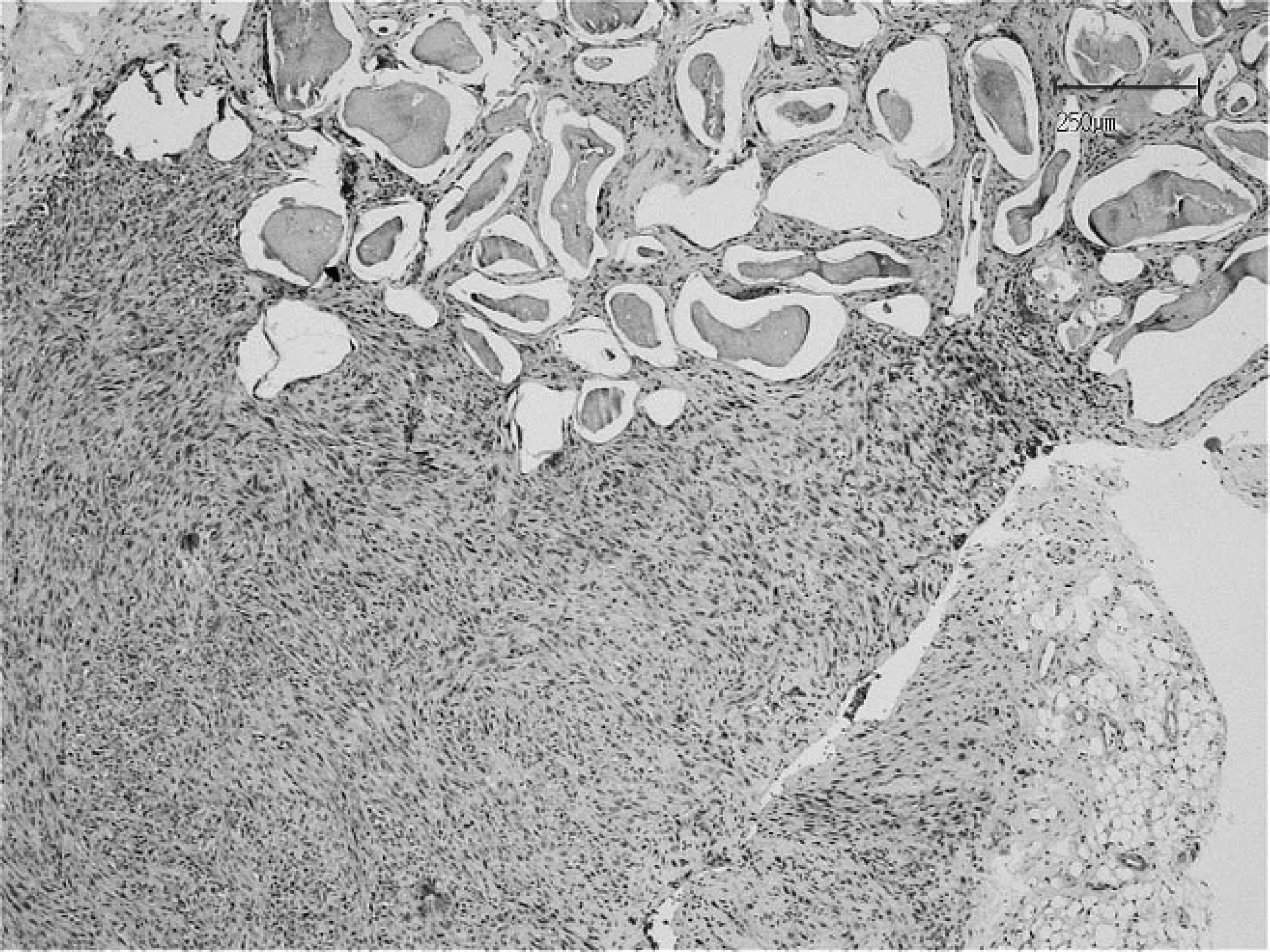
Pseudomultilacunar and solid areas are present within a section of fibrosarcoma. Hematoxylin and eosin stain. Bar = 250 μm.
The mass measured 4.5 cm × 3.5 cm × 2.0 cm and was surgically excised with wide margins, immediately fixed in 10% neutral buffered formalin, routinely processed, and embedded in paraffin. Tissue sections were prepared for histologic, histochemical, and immunohistochemical investigation. Histologic examination was performed according to the standard criteria of the World Health Organization. 12 However, the general diagnostic criteria describing the feline postvaccinal and injection-site fibrosarcomas as reported in international peer-reviewed journals were also considered, 3,9,13 as well as specific references describing disease in rabbits. 4,11,21 Histologic examination revealed an unencapsulated, nodular, subcutaneous mass. On one side of the mass, the neoplastic cells were arranged around irregularly shaped lacunae of variable in diameter that contained a pale basophilic amorphous material (Figs. 3, 4). Neoplastic tissue was composed of interwoven fascicles of malignant ovoid to spindle-shaped cells with a low mitotic rate (0–4 mitotic figures per 400× field). Neoplastic cells exhibited moderate to marked pleomorphism and ranged from 10 to 60 μm in diameter. Nuclei were large, round to oval, hyperchromatic, and centrally to eccentrically located. Prominent single to multiple nucleoli were observed along with a moderate amount of eosinophilic cytoplasm. Multinucleated cells with large, bizarre nuclei were occasionally scattered throughout the tissue sections. Rare neoplastic fibroblasts were observed within vascular lumens, suggesting hematogenous metastasis (Fig. 5). Multifocal necrosis was observed with postnecrotic formation of a central cavity. Small, peripheral, nodular aggregates of lymphocytic were occasionally present. The histologic findings supported a diagnosis for fibrosarcoma; however, giant cell fibrosarcoma, histiocytic sarcoma, malignant fibrous histiocytoma, myxosarcoma, and rhabdomyosarcoma were considered in the differential diagnoses. Clinical and histopathologic investigations supported the diagnosis of fibrosarcoma and included the typical features of injection-site fibrosarcoma.
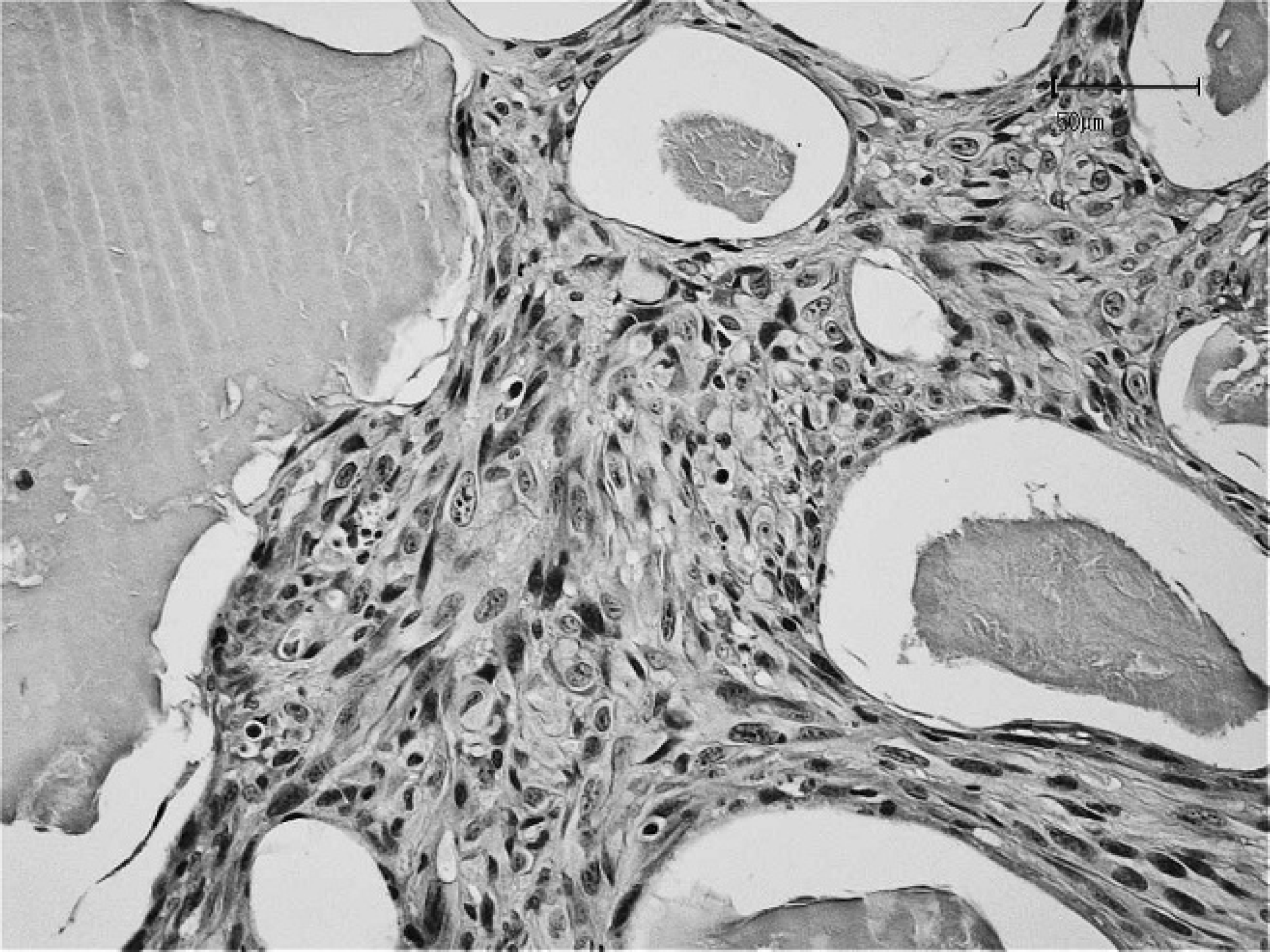
Amorphous material is present in the lacunae apart form the malignant fibroblasts at higher magnification. Hematoxylin and eosin stain. Bar = 50 μm.
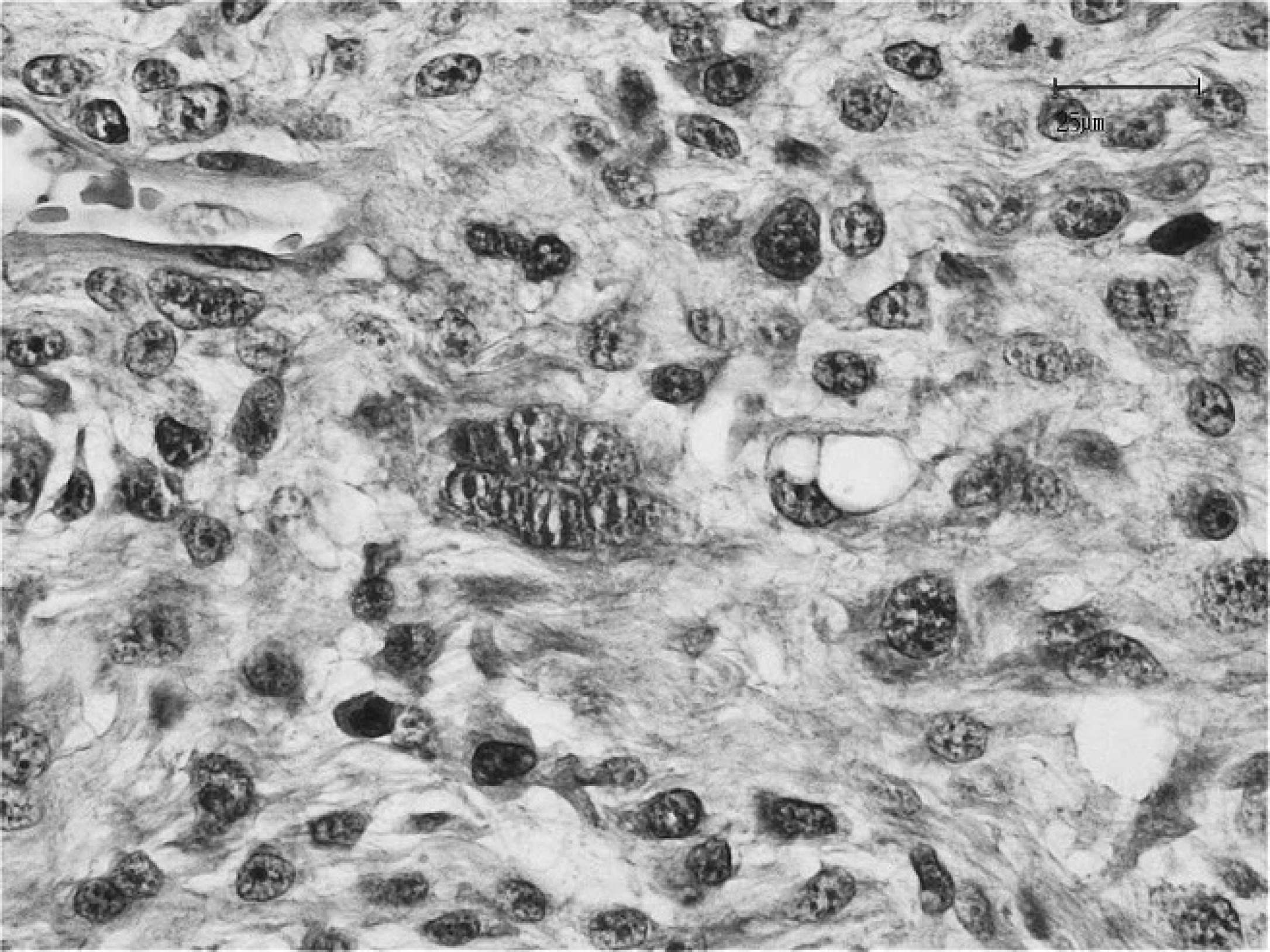
Atypical neoplastic fibroblasts are present within a solid area of the neoplasm. Notice the presence of 2 spindle-shaped cells in the vascular space at the upper left corner. Hematoxylin and eosin stain. Bar = 25 μm.
Replicate serial sections from the paraffin-embedded tissue blocks were immunostained for cytokeratin (AE1/AE3), a vimentin, a smooth muscle actin, b desmin, b myoglobin, b cluster of differentiation (CD)3, a and CD79α a using a commercial kit. c Mayer's hematoxylin solution was used as a counterstain. Positive and negative control tissue sections were run in parallel. Negative controls were produced by substituting the primary antibody with 5% bovine serum albumin in phosphate buffered saline. All tissue sections were evaluated by light microscopy. Immunohistochemical investigations (data not shown) demonstrated that the neoplastic cells were positive for vimentin and negative for cytokeratin (AE1/AE3), desmin, alpha smooth muscle actin, and myoglobin. Lymphoid aggregates were CD3 positive and CD79α negative.
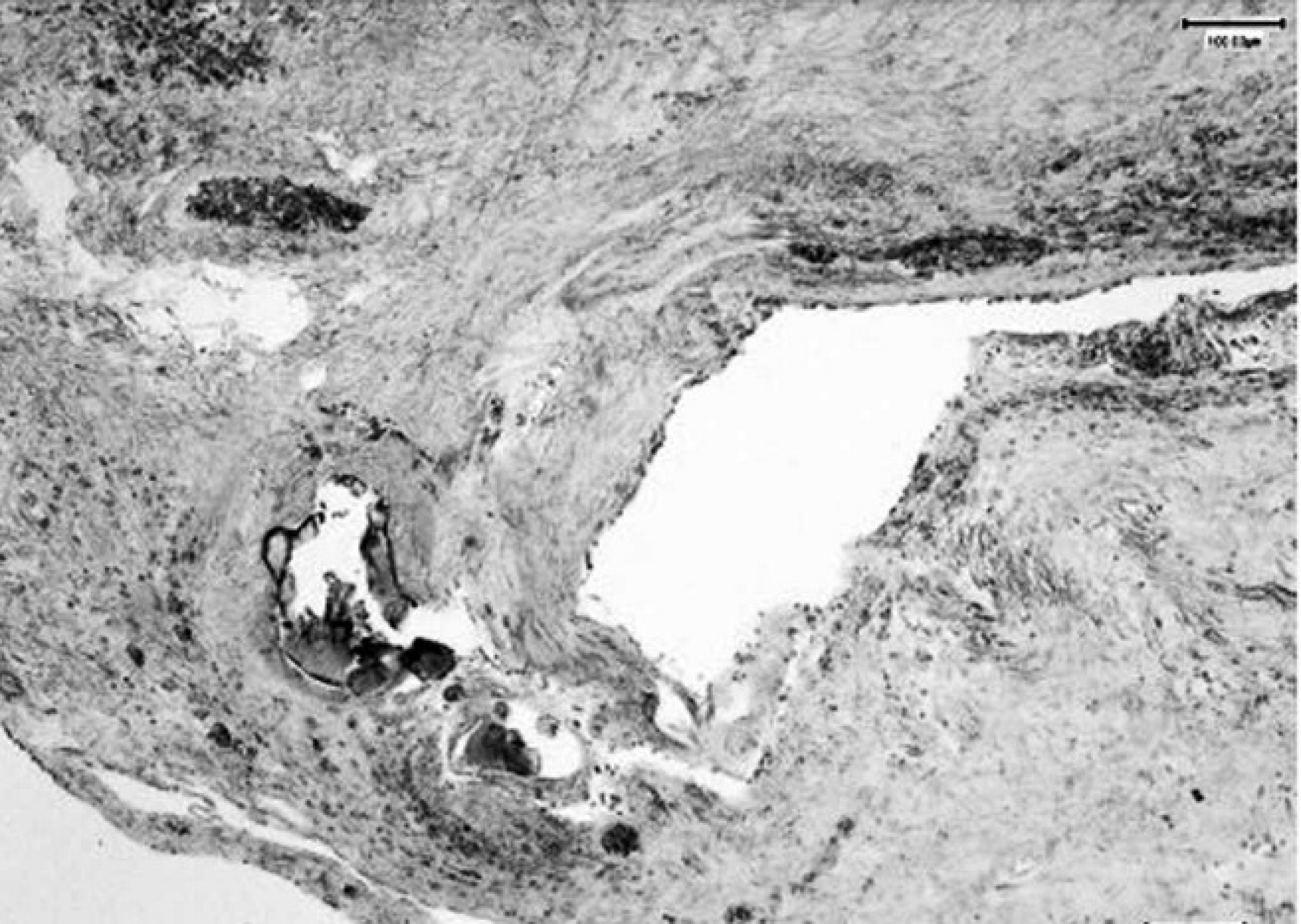
Foreign material embedded in the neoplastic tissue red stained by the aurintricarboxylic acid method, indicating the presence of aluminum deposits. Bar = 100 μm.
Histochemistry was performed to identify and evaluate aluminum deposits in the tissue sections by the aurintricarboxylic acid method. A buffer solution was prepared as follows: 38 ml of 5 M ammonium chloride were added to 38 ml of 5 M ammonium acetate and 10 ml of 6 N chloride acid. The solution (10 ml) was added to 2 g of a sodium salt of aurintricarboxylic acid, and the previously prepared buffer solution was added to obtain a total volume of 100 ml. Serial tissue sections were deparaffinized and rehydrated by adding the same buffer solution and incubating for 5 min at 75°C. Subsequently, the tissue sections were rinsed in cold distilled water. The slides were then incubated for 3 sec with a previously prepared solution of 50-ml buffer and 22 ml of 1.6 M ammonium carbonate. Finally, the tissue sections were rinsed in distilled water, counterstained with a 1:10,000 aqueous solution of methylene blue dye, rinsed again in distilled water, and mounted with a coverslip. The red deposits of aluminum were primarily localized within the lacunar area (Fig. 6).
Two months after surgical excision, the neoplasm recurred at the site of origin and measured 12 cm × 8 cm × 4 cm (Fig. 7). The owner refused any further treatment, and the dwarf rabbit was euthanized in November 2006 due to rapid progression of the neoplasm and poor general condition. Necropsy was not permitted.
Few studies have been published concerning neoplasms in rabbits. In a recent retrospective study of cutaneous neoplasms in pet rabbits, 7 spontaneous fibrosarcomas were reported that originated primarily on the limbs, 27 and few fibrosarcomas developed in laboratory rabbits, as reported in a different study. 28 The subcutaneous intrascapular fibrosarcoma in the dwarf rabbit of the current report had histologic features that were remarkably similar to feline injection-site sarcoma. The primary cause of the neoplasm is difficult to establish, because vaccine and antibiotics were administered in the same anatomic site, although dual origin could be hypothesized. The presence of multilacunar spaces containing amorphous material and multifocal aluminum deposits within the neoplasm supports the presumptive pathogenic role of xenobiotic substances. Although adjuvants may have a contributory role in the genesis of sarcoma, a previous study reported that both aluminum-adjuvanted and non-adjuvanted vaccines were also associated with development of sarcoma. 15 Chronic inflammation and subcutaneous injection of enrofloxacin might also have been responsible for sarcoma development. In fact, neoplastic transformation of mesenchymal cells involved in connective tissue repair secondary to chronic inflammation has been suggested by other authors. 11,17 A previous study also suggested that inflammation may have been responsible for the development of primary intraocular sarcoma in some cats after ocular trauma or chronic uveitis. 3 Finally, reactions due to intramuscular injection of saline solution may induce inflammation and fibrosis in laboratory animals, such as rats, mice, and rabbits. 23
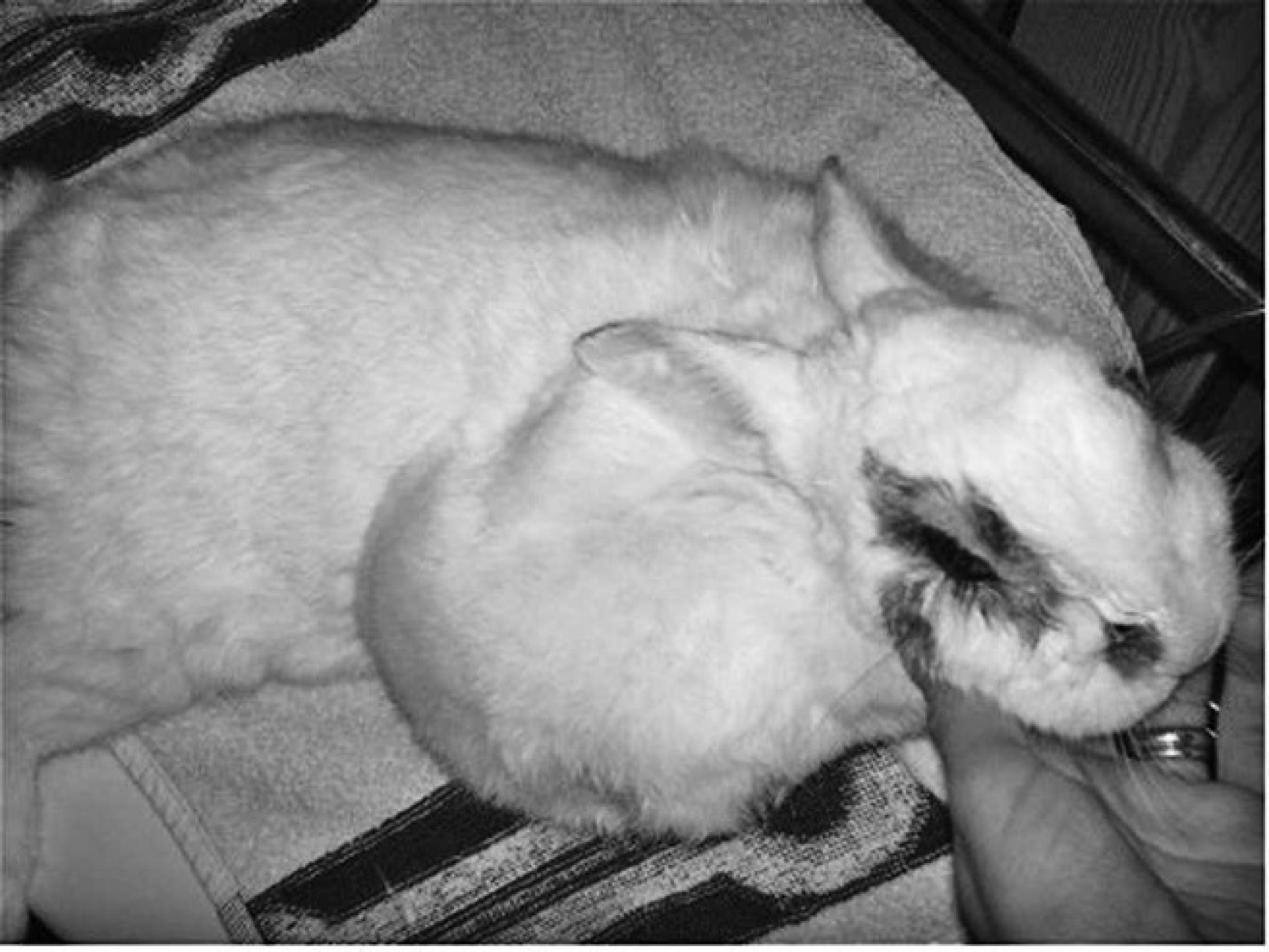
Gross photograph of the enlarged mass involving the subcutis of the interscapular region and right shoulder after surgical excision of the primary neoplasm.
As with any drug, vaccines can have side effects, although problems in rabbits are very unusual. Skin reactions are occasionally reported at the sites of injection. A study on the safety of combined intradermal vaccination against myxomatosis and RHD in dwarf rabbits using a reconstituted vaccine of live homologous myxomatosis component SG33 strain and an inactivated RHD component in the adjuvant AG88 strain produced a localized reaction at the vaccination site. This localized reaction was frequently observed 2 to 3 days after vaccination in young animals (7 weeks old) but observed less frequently in adults. The reaction consisted of a local rash that usually disappeared from 2 to 3 days to 1 week after vaccination. 16 However, in a laboratory trial of 6 experimental homologous inactivated myxomatosis vaccines prepared using 6 different adjuvants, all of the vaccine lots were acceptable on safety assessment, independent of the adjuvant. 2 None of the intradermally inoculated rabbits exhibited localized reactions at the inoculation site or general reactions, even when the vaccine was administered in an excessive amount or after repeated doses.
Injection-site fibrosarcomas are well described in cats 7,17 and have been recently reported in ferrets 19 and dogs. 20,24 The first potential relationship between sarcoma development and subcutaneous injections of xenobiotics in cats was reported in 1991. 10 Although pharmaceutical compounds may be involved, such reactions are primarily associated with the use of inactivated virus vaccines, such as those protective against Feline leukemia virus and Rabies virus, that contain aluminum-based adjuvants. 6,11 However, inflammation secondary to the injection of xenobiotics, 17 nonabsorbable sutures, 1 or microchip implantation in the subcutis 20,25,26 are now considered potential causes of fibrosarcoma. Previous studies of injection-site sarcomas have suggested that local inflammation caused by potentially irritating injected substances (e.g., adjuvants, antibiotics) or implanted material (e.g., suture material, microchip) may predispose tissue to tumor development. However, other factors, such as anatomic site of injection, 13,15 frequency of injection in the same anatomic sites, 15 genetic predisposition, growth factors, oncogenes (e.g., aberrant expression of c-jun and p53), and the effects of different growth factor and receptor activation (such as the platelet-derived growth factor), should also be considered. 8,14,22 Notably, inflammation is a common event secondary to the administration of an anti-myxomatosis vaccine. 2
Vaccine-induced reactions are considered to have additive effects in cats, and the risk for development of neoplasia increases with the number of injections in the same anatomic area or tissue. 15 In cats, the probability of developing a sarcoma in the interscapular area is higher compared with other locations. 15 Moreover, the risk increases from 1.27- to 1.75-fold if 2 or 3 subcutaneous injections are administered in the same area, respectively. 15 In the rabbit of the present report, multiple injections in the interscapular area might be considered as an additional tumorigenic factor. Different studies have indicated that the time required for development of a neoplasm in the injection site may range from 3 months to 3 years or more. 3 However, these intervals may be longer depending on the anatomic site chosen for the injection. 13,15 Based on the data available for cats, development of a large tumor approximately 7 years after the first examination in the rabbit of the current report may not be unexpected. A previous study reported that a sarcoma developed at a vaccination site in one cat more than 5 years (71 months) after inoculation. 9
Based on histopathologic findings, the neoplasm in the dwarf rabbit in the present study was classified as an injection-site fibrosarcoma and shared similarities with injection-induced fibrosarcomas in cats. Neoplastic fibroblasts, multinucleated cells, and perilesional infiltrates of lymphocytes are common histopathologic findings in feline injection-site fibrosarcomas. Lymphocytes infiltrating the fibrosarcomas associated with feline vaccine-associated sarcomas 3,8 and microchip implant in a dog 25 were T cells.
The typical lesions for Shope fibromatosis differ with regard to anatomic location, macroscopic appearance, and microscopic features from those described in the current study. 3,27 Finally, the association between vaccination and fibrosarcoma development in cats was made only after millions of doses of vaccine had been given over approximately a 5-year period. 18 Because rabbits are increasing in popularity as a companion animals, they can be expected to live longer (natural life span range: 5–10 years) than wild rabbits and rabbits raised for meat and fur. 5 Therefore, more cases of sporadic or induced neoplasms may be expected in pet rabbits in the future.
Acknowledgements. This work was supported by the EU Network of Excellence, EPIZONE (contract no. FOOD-CT-2006-016236).
Footnotes
a.
Dako Denmark A/S, Glostrup, Denmark.
b.
Ventana Medical System SA, Illkirch, France.
c.
EnVision™ Detection Kit Peroxidase/DAB, Dako Denmark A/S, Glostrup, Denmark.
