Abstract
The objective of the current study was to compare the diagnostic performance of a direct isolation method for Erysipelothrix spp. with a broth-based enrichment technique. Samples were obtained from three sources: 1) experimentally inoculated pigs, 2) porcine tissue samples submitted to the Iowa State University Veterinary Diagnostic Laboratory (Ames, IA), and 3) tissues from condemned carcasses at an abattoir. Culture plates from direct isolation and broth-based technique were evaluated for growth at 24 and 48 hr. Results indicated that the broth enrichment method was markedly more sensitive for the isolation of Erysipelothrix spp. To the authors' knowledge, this is the first comparison of direct culture and broth-based enrichment methods for the isolation of Erysipelothrix spp. Interestingly, in several samples, a Gram-positive bacterium with almost identical growth characteristics to Erysipelothrix spp. was detected and identified as a Vagococcus sp. through 16S ribosomal RNA gene sequencing. The results of this study indicate that the broth-based enrichment method should be used for the isolation of Erysipelothrix spp. from clinical samples with a history suggestive of erysipelas and that Vagococcus spp. is potentially an important differential diagnosis.
Keywords
Erysipelothrix spp. are facultatively anaerobic, Grampositive small rods with worldwide distribution. Strains have been isolated from many domestic and wild species, including reptiles, amphibians, fish, and humans. The genus Erysipelothrix contains four species: E. rhusiopathiae (serotypes 1a, 1b, 2, 4, 5, 6, 8, 9, 11, 12, 15, 16, 17, 19, 21, and N), E. tonsillarum (serotypes 3, 7, 10, 14, 20, 22, and 23), Erysipelothrix sp. strain 1 (serotype 13), and Erysipelothrix sp. strain 2 (serotype 18). 16–18
Erysipelothrix spp. are the causative agent of erysipelas in swine, sheep, fish, reptiles, and birds. Three clinical presentations of swine erysipelas are recognized. These include acute infection commonly associated with serotype 1a and subacute infection and chronic infection, which are both typically associated with serotype 2. The additional serotypes (3–26, N) have minimal clinical significance in swine. 21 It is estimated that 30–50% of healthy pigs harbor E. rhusiopathiae in tonsils and lymphoid organs. These subclinically infected pigs are thought to be the source for acute erysipelas outbreaks due to shedding of the organism in urine, feces, saliva, and nasal secretions. 12,21
Erysipelothrix spp. typically appear on artificial agar media as very small colonies after 24–48 hr of incubation at 37°C. 5,21 Specimen contamination can obscure colony growth on artificial media, resulting in unrewarding and inconsistent isolation. To address potentially contaminated specimens, microbiology manuals describe the isolation of Erysipelothrix spp. using blood agar plates with sodium azide added to inhibit contamination. 2 In 1965, a liquid Erysipelothrix selective enrichment method was developed, commonly referred to as “Wood's Erysipelothrix selective broth.” The enrichment method was found useful when attempting Erysipelothrix spp. isolation from feces and other contaminated material, including intestinal lymphoid tissue, urine, nasal secretions, and decomposing animal tissue. 20 In addition, selective agar media including sodium azide crystal violet (SACV), also known as “Packer's medium,” 13 nalidixic acid medium, 1 and a modified blood azide agar 15 have also been described to aid in the isolation of Erysipelothrix spp. 4,5 A previous study 8 reported no significant difference in isolation frequency between blood azide agar and SACV medium.
In a survey conducted in March 2008, all 10 Veterinary Diagnostic Laboratories (VDLs) surveyed in the Midwest were using direct culture as the standard method for isolation of Erysipelothrix spp. from case submissions suspected to be swine erysipelas (Joann Kinyon, personal communication, 2008). The objective of the current study was to compare the diagnostic sensitivity of the direct isolation method for Erysipelothrix spp. with a broth-based enrichment technique.
The technique for sample preparation used in this investigation was based on methods previously described. 3 Briefly, the outside of the tissue specimens was seared with a heated spatula to remove surface contaminants, the specimen was incised using a sterile scalpel blade, and a sterile swab was inserted for collection of a tissue for culture.
Experimental design.
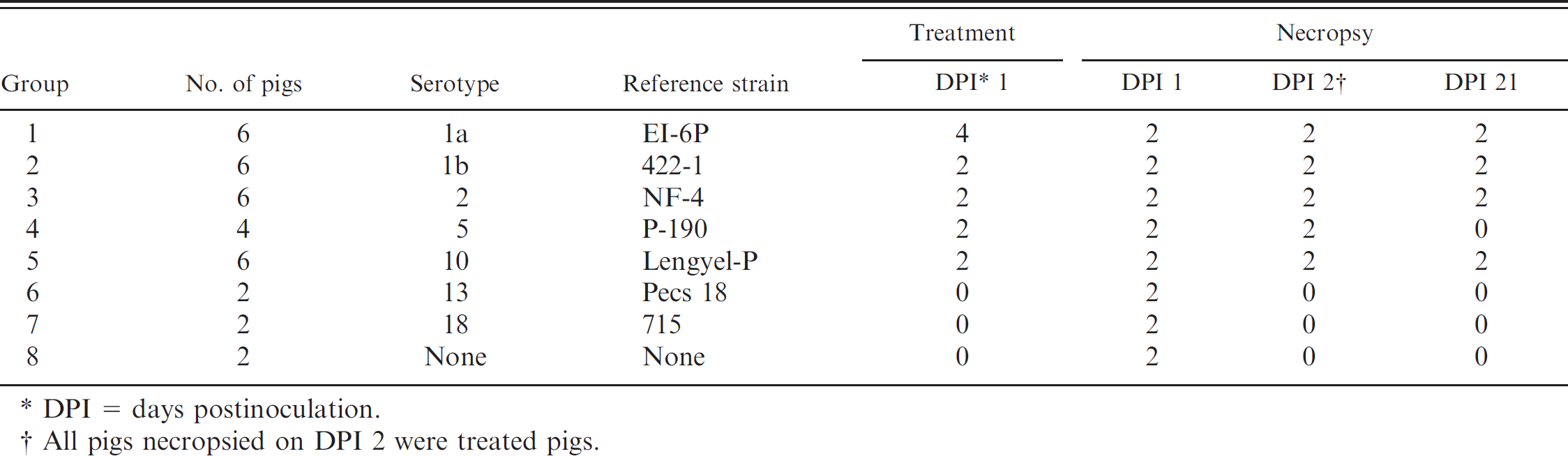
DPI = days postinoculation.
All pigs necropsied on DPI 2 were treated pigs.
For the direct culture, the swabs were cultured on agar plates, including trypticase soy agar containing 5% sheep blood (BA) a and colistin-nalidixic acid agar containing 5% sheep blood (CNA). a Plates were incubated aerobically at 35°C and examined at 24 and 48 hr postinoculation. 3,6 Suspect colonies with the characteristic appearance similar to Erysipelothrix spp. were subcultured on BA, incubated for 24 hr, and then biochemically confirmed using standard laboratory methods. 16,21 Isolates confirmed as Erysipelothrix spp. were saved in brain heart infusion (BHI) broth containing 50% glycerol b and frozen at −80°C for future evaluation.
The Erysipelothrix spp. selective broth base was prepared as described and stored at 5°C for a maximum of 2 weeks prior to use. 20 The Erysipelothrix spp. selective medium, SACV, was prepared as previously described. 13
Tissue specimens were homogenized using a stomacher, c and 300 μl of resulting liquid tissue homogenate supernatant was added to a tube containing 3 ml of Erysipelothrix spp. selective broth. Incubation was conducted at 35°C for 24–48 hr. At both time points, a 100-μl subculture from the Erysipelothrix spp. selective broth was made onto a BA plate, a CNA plate, and a SACV plate. Inoculated plates were incubated at 35°C and observed at 24, 48, and 72 hr for colonies characteristic of Erysipelothrix spp. Suspect colonies were subcultured on a BA plate, incubated for 24 hr, and then biochemically confirmed using standard laboratory methods. 16,21 Erysipelothrix spp. serotyping was performed as previously described. 11,23 Homologous positive controls were used with each test. Reactions were recorded after 24 hr. 22 Gram stain, cell morphology, motility, oxidase and catalase activity, and H2S production on triple sugar iron agar (TSI) medium were used to confirm Erysipelothrix spp. 16
Data obtained from isolation attempts using different laboratory media and tissue comparison were assessed to detect statistical differences between direct and enrichment culture methods. A 2-sample test (t-test) on the proportions utilizing the R version 2.7.2 d statistical package was used. A P-value of <0.05 was considered significant.
The experimental protocol was approved by the Iowa State University Institutional Animal Care and Use Committee. Thirty-two 2-week-old conventional pigs were purchased from an isolated herd free of major swine pathogens, including swine influenza virus, porcine reproductive and respiratory syndrome virus, and Mycoplasma hyopneumoniae. Erysipelothrix rhusiopathiae vaccination was not used in the breeding stock on the source farm. Upon arrival at the research facility, serum samples were collected and tested by an in-house enzyme-linked immunosorbent assay (courtesy Dr. J. Huchoppa) for the presence of anti-Erysipelothrix spp. antibodies, and all pigs were found to be negative. The pigs were randomized by weight and divided into 8 groups of 2–6 pigs each (Table 1). Each group was housed in a separate room with identical dimensions, feed and water delivery systems, and environmental controls. Pigs were monitored and allowed to acclimate prior to inoculation (day 0) for 15 days. At inoculation, the pigs were 4 weeks old.
Erysipelothrix spp. strains used for inoculation (Table 1) were selected based on their importance to the U.S. swine industry: serotypes 2 (41.2%), 1a (21.1%), 1b (15.1%), and 5 (9%) historically are the most common strains isolated from pigs in the United States. 23 Erysipelothrix tonsillarum (serotype 10) has been noted to cause clinical erysipelas in swine when they are inoculated via the intravenous route. 22 Erysipelothrix sp. strains 1 (serotype 13) and 2 (serotype 18) were chosen in order to have representative serotypes from all described species of Erysipelothrix. The inocula were prepared in BHI broth b supplemented with 5% fetal bovine serum (FBS) b and plated on BHI agar b containing 5% FBS. Pretrial standard plate counts were performed to determine log phase growth within 30–60 min after harvest and to estimate bacterial concentrations that were subsequently adjusted to log10 7 colony-forming units/ml. Purity of the inocula was assured by culture and identification by standard methods. 16 The pigs were inoculated using 3 routes. Each pig received 2 ml of the respective inoculum intramuscularly into the right neck, 2 ml intravenously in the left ear vein, and 0.1 ml intradermally in the left flank. One day postinoculation, selected pigs (Table 1) were treated with 1.5 ml penicillin e (intramuscular into the right neck) in order to evaluate treatment effect on isolation success. At necropsy, tissues, including tonsil, lung, heart, kidney, spleen, liver, skin, and blood, were collected. Fresh tissues were collected aseptically and cultured immediately. Representative samples from each pig were stored in sterile specimen bags and frozen at −80°C for future evaluation by enrichment.
Comparison of successful Erysipelothrix spp. isolation from different laboratory media.*
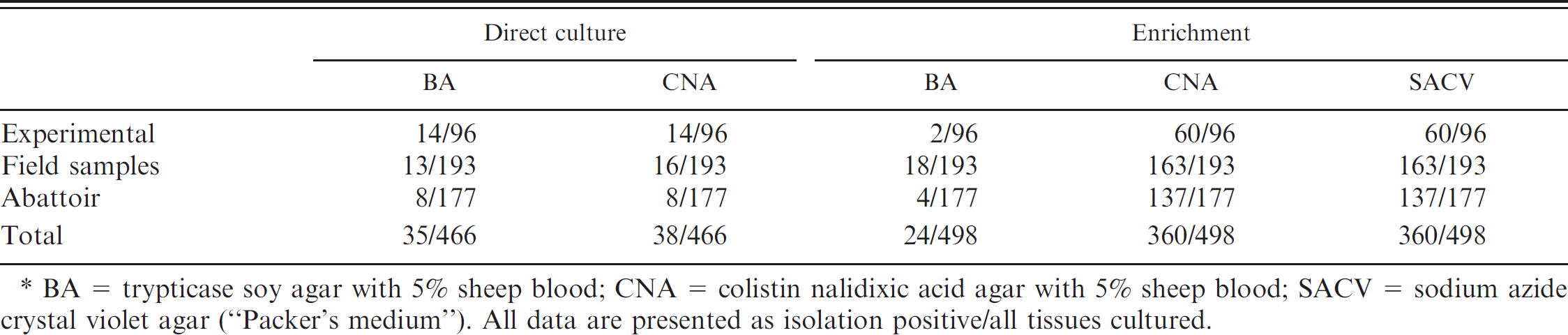
BA = trypticase soy agar with 5% sheep blood; CNA = colistin nalidixic acid agar with 5% sheep blood; SACV = sodium azide crystal violet agar (“Packer's medium”). All data are presented as isolation positive/all tissues cultured.
Field samples from pigs submitted to the Iowa State University Veterinary Diagnostic Laboratory (ISU-VDL) with a history suggestive of swine erysipelas, such as acute septicemia (fever, lethargy, and decreased feed intake), rhomboid skin lesions, or chronic changes (swollen joints, lameness), were included in the investigation. In addition, tissue specimens including lesions suggestive of erysipelas that resulted in carcass condemnations were collected by the veterinary inspector-in-charge at a regional abattoir. Tissues from the harvest facility were collected, placed into individual specimen bags, labeled, and then frozen at −20°C prior to transfer to the laboratory. All tissues collected from field samples and abattoir condemnation were placed into individual specimen bags and stored at −80°C for direct enrichment.
Table 2 summarizes the positive isolation results categorized by plate media. Using direct culture, there was no significant (P = 0.07) difference between the recovery of Erysipelothrix spp. from BA (35/466, 8%) or CNA (38/466, 8%). There was no significant difference (P = 0.71) in the total number of isolations when CNA media was added to the enrichment method protocol. Both CNA and SACV resulted in 360 of 498 (72%) tissue specimens being positive, a value that was significantly (P < 0.05) higher than that associated with positive isolations from BA plates (24/498, 4.8%). Although the use of CNA plate was not previously described in the enrichment method, this media was included to see if it could increase the speed of isolation. When CNA media was included, Erysipelothrix spp. were isolated in many cases as soon as 12 hr postinoculation from the Erysipelothrix selective broth and by 24 hr in every positive isolation case regardless of the tissue sample. Previous literature 8 has described the growth of Erysipelothrix spp. on SACV medium as taking as long as 48 hr. In addition, CNA medium is commercially produced and readily available. The addition of the CNA medium allowed for faster isolation and diagnosis of swine erysipelas; however, it did not increase the sensitivity of the enrichment method.
It is common for diagnostic laboratories to receive tissues from animals that have been previously treated with antibiotics. The isolation success for the different tissues using direct or enrichment culture methods in treated and nontreated pigs is summarized in Table 3. For all tissue samples (from treated and nontreated pigs), direct culture resulted in 14 out of 96 (15%) positive isolations of Erysipelothrix spp. The direct culture method for all tissue samples (treated and nontreated) resulted in 14 out of 96 samples being positive for Erysipelothrix spp. isolation. Using enrichment culture, 51 out of 96 (51%) of the tissue samples were positive for Erysipelothrix spp. Enrichment culture resulted in more positive isolation results for tissue samples from both the treated and untreated pig groups, and the isolation rate was particularly improved in the treated group. Interestingly, 13 out of 36 (36%) tissues obtained from experimentally inoculated pigs that were also treated with antibiotics were positive using enrichment culture. This may indicate that although antibiotics alleviate clinical signs, the organism is incompletely cleared from the body. In comparison, when using the direct method, Erysipelothrix spp. isolation was not successful (0/36) on any of the tissues from the experimentally inoculated and treated pigs. In addition, the choice of tissue to culture has minimal effect when using enrichment methods; however, the use of spleen tissue resulted in slightly more positive isolations. Recovered isolates were identical to the inoculum administered to the pigs based on serotyping. Erysipelothrix sp. strain 1 was not cultured from any specimens using either direct or enriched culture. These findings may indicate that the pig is not the ideal host for Erysipelothrix sp. strain 1. Noninoculated control animals were negative for Erysipelothrix spp. by both culture methods (data not shown).
Field sample culture results are summarized in Table 4. A total of 193 tissue specimens from 89 individual pigs were submitted for culture. Erysipelothrix spp. were isolated from 16 out of 193 (8%) of the submitted tissues by direct culture and from 163 out of 193 (84%) of the submitted pigs by the enrichment culture method, which was a significant (P < 0.05) improvement in isolation rate. Increased sensitivity through enriched culture is a great benefit for veterinarians and producers who expect an accurate diagnosis when submitting specimens to a VDL. In addition, successful isolation allows for antimicrobial susceptibility profiles to be conducted, availability of isolates for autogenous vaccine production, and further characterization through serotyping and pulsed-field gel electrophoresis. These options are not available when only polymerase chain reaction (PCR) diagnostics are utilized.
Table 4 also summarizes the results obtained with direct and enrichment culture methods on tissue specimens that were condemned and collected at the abattoir. By the direct method, 8 out of 177 (5%) of the tissues were classified as positive. In comparison, 137 out of 177 (77%) of the tissues were confirmed as positive by enrichment culture. Kidney was the tissue sample in which Erysipelothrix spp. were most frequently isolated by direct culture, and skin was the tissue most commonly determined to be positive by the enrichment culture method. The culture results from the condemned pig tissues provided the veterinary inspector-in-charge at the abattoir with diagnostic evidence that carcasses condemned for swine erysipelas are frequently harboring Erysipelothrix spp.; these results also provided producers with a documented reason for the condemnations so they could use that information to implement appropriate prevention and control strategies.
Comparison of direct and enrichment culture methods in pigs experimentally inoculated with different Erysipelothrix spp.*
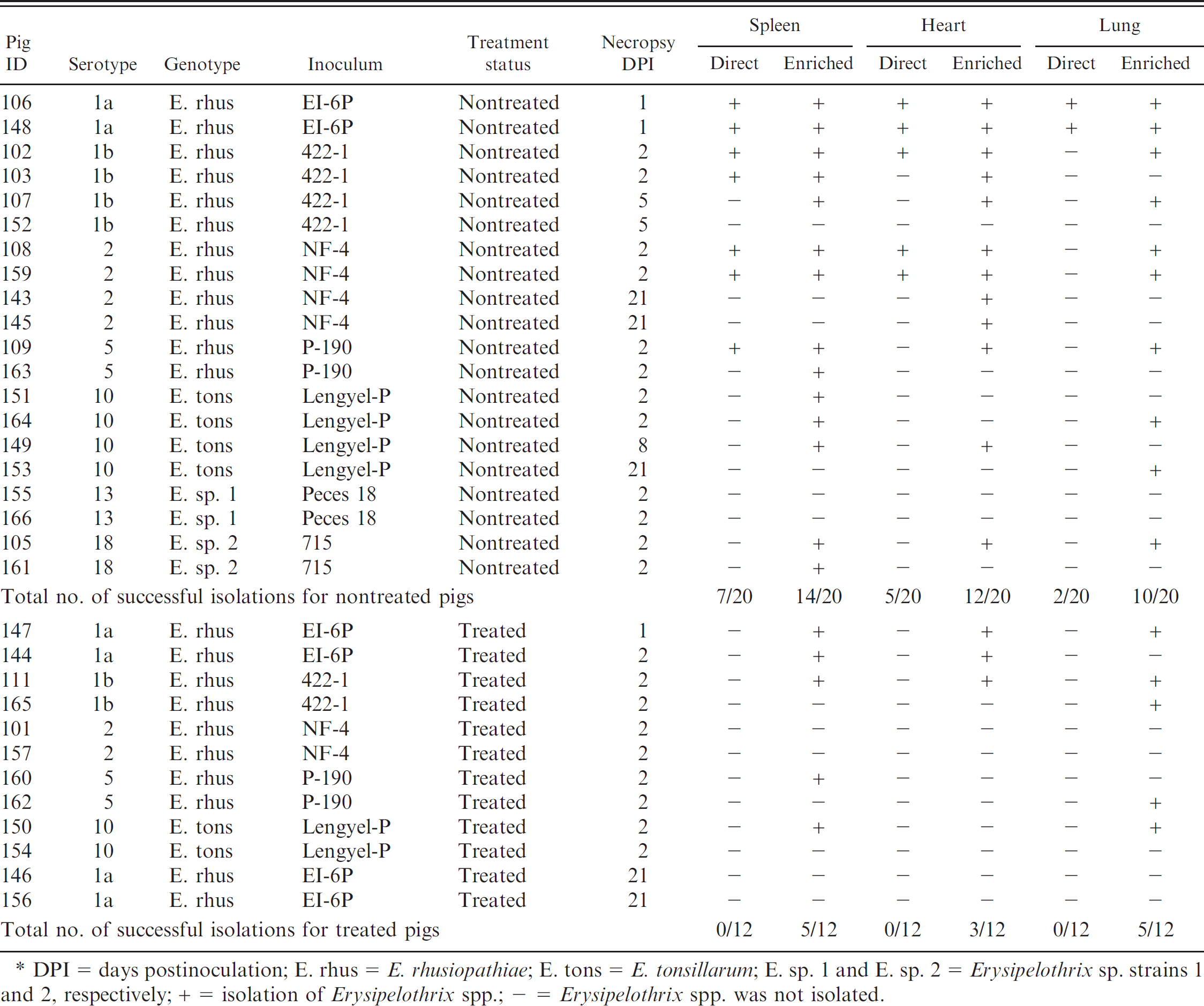
DPI = days postinoculation; E. rhus = E. rhusiopathiae; E. tons = E. tonsillarum; E. sp. 1 and E. sp. 2 = Erysipelothrix sp. strains 1 and 2, respectively; + = isolation of Erysipelothrix spp.; − = Erysipelothrix spp. was not isolated.
For further identification, the 16S ribosomal RNA (rRNA) gene was amplified, sequenced, and analyzed as described. 7 Amplified products were purified with a commercial kit f and sequenced bi-directionally at the DNA Sequencing and Synthesis Facility at Iowa State University (Ames, IA) using the BigDye terminator chemistry. g The forward and reverse sequences were assembled into a consensus sequence and edited with a software. h Finally, the consensus 16S rRNA sequence was compared with those available in the GenBank i using the Basic Local Alignment Search Tool to find the most likely match. 7 All but 16 isolates were identified as Erysipelothrix spp. using previously described laboratory methods. 16 Twelve out of 16 of the isolates were confirmed to be E. rhusiopathiae by 16S rRNA PCR. Interestingly, 16S rRNA PCR identified the other 4 isolates (4/16) as Vagococcus spp. Vagococcus spp. appeared at 12–18 hr on CNA as small, transparent colonies with weak alpha or no hemolytic pattern very similar to Erysipelothrix spp. Upon Gram staining, the organism was Gram positive and appeared as coccobacilli or as short rods. Biochemically, Vagococcus spp. were nonmotile, catalase-negative, and produced hydrogen sulfide gas on TSI medium similar to Erysipelothrix spp. Vagococcus spp., however, produced a greater amount of hydrogen sulfide that is visible throughout the entire media, not just along the stab line. Vagococcus spp. were identified in tissues from three separate field cases. In all three cases, Vagococcus spp. growth was identified using the enrichment method followed by subculture on CNA plates. In addition, Vagococcus spp. were isolated from a kidney specimen obtained from a harvest facility and was present on both CNA and SACV media using the enrichment method. While the significance of Vagococcus spp. has yet to be determined in swine, they have been described as an emerging disease of rainbow trout, 14 seal, and harbor porpoise, 9 and it has been isolated from pigs 19 and a swine manure storage pit. 10 Additionally, Vagococcus spp. need to be considered as a potential rule-out or contaminant that are not inhibited by the enrichment method.
Comparison of direct and enriched culture methods on field and abattoir samples.*
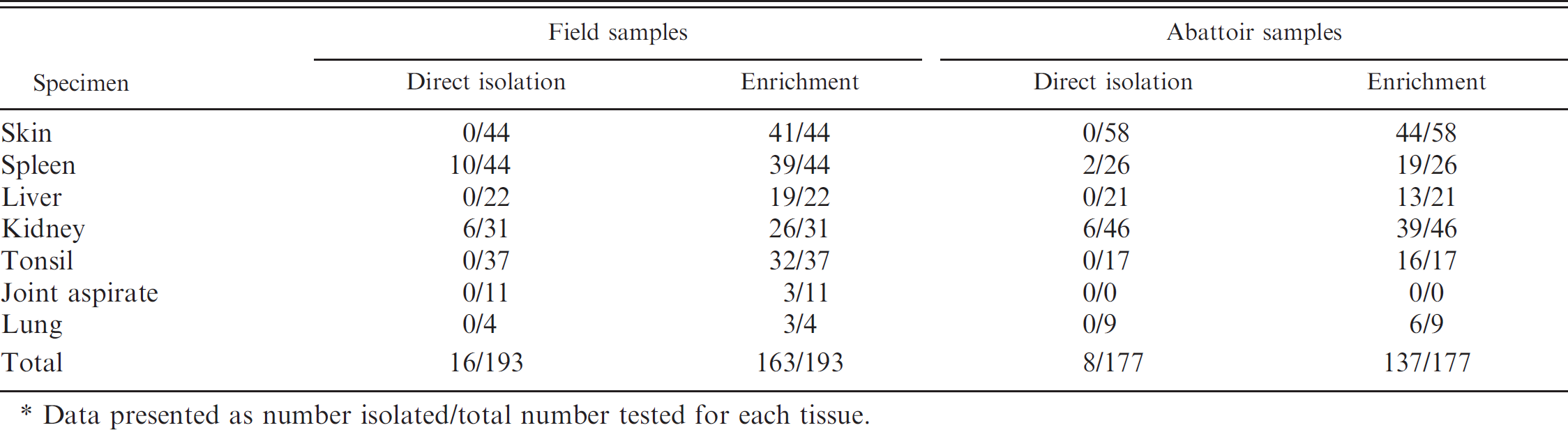
Data presented as number isolated/total number tested for each tissue.
Erysipelothrix spp. have been important bacterial pathogens in the swine industry for over 100 years, and swine erysipelas continues to be one of the leading causes of swine carcass condemnations at harvest worldwide. Methods for accurate diagnosis and applicable treatment are becoming increasingly important. Diagnostic assays continue to be improved with advancements in PCR technology; however, these tests are not readily available in all VDLs.
Historically, the ISU-VDL has not conducted both direct culture and broth-based enrichment methods for isolation of Erysipelothrix spp. from suspect swine erysipelas cases. In several of these cases, Erysipelothrix spp. were not routinely isolated in spite of clinical signs and lesions consistent with systemic bacterial infection. Adoption of the enrichment method has substantially improved the quality of diagnostic capabilities for the ISU-VDL. Unlike with PCR, the availability of the isolates allows the laboratory to conduct antimicrobial sensitivities and further characterization of the isolates; this information can then be provided to clients, who may consider the potential use of the isolates in autogenous vaccines if desired.
Acknowledgements. Funding for this study was provided by Pork Check Off Dollars from the National Pork Board, the Iowa Livestock Health Advisory Council, and Schering-Plough Animal Health. The authors thank Dr. Howard Lindaman for assistance in procuring samples, Dr. Abby Patterson for critical review of the manuscript, and the ISU-VDL Bacteriology section for assistance with the laboratory work.
Footnotes
a.
Thermo Fischer Scientific Remel products, Lenexa, KS.
b.
BD, Sparks, MD.
c.
Seward, Bohemia, NY.
d.
R Foundation for Statistical Computing, Vienna, Austria.
e.
Sigma-Aldrich, St. Louis, MO.
f.
QIAquick PCR Purification kit, Qiagen, Valencia, CA.
g.
Applied Biosystems, Foster City, CA.
h.
DNASTAR, Madison, WI.
