Abstract
Bordetella bronchiseptica frequently causes nonfatal tracheobronchitis, but its role in fatal pneumonia is less recognized. Our study evaluated histologic identification of cilia-associated bacteria as a method for diagnosis of B. bronchiseptica pneumonia. Cases of fatal bronchopneumonia were studied retrospectively, excluding neonates and cases of aspiration pneumonia, minor lung lesions, or autolysis. The study population comprised 36 canine and 31 feline cases of bronchopneumonia. B. bronchiseptica was identified in 8 of 36 canine and 14 of 31 feline cases based on immunohistochemistry (IHC) using serum from a rabbit hyperimmunized with pertactin, PCR testing (Fla2/Fla12), and/or bacterial culture data when available. Of these, IHC was positive in 4 canine and 7 feline cases, PCR was positive in 8 canine and 14 feline cases, and B. bronchiseptica was isolated in 2 of 5 canine and 3 of 9 feline cases tested. Examination of histologic sections stained with hematoxylin and eosin revealed bronchial cilia-associated bacteria in 4 of 36 canine and 5 of 31 feline cases; these were all positive by IHC and PCR. The presence of cilia-associated bacteria had been noted in the pathology report for only 2 of these 9 cases. Thus, the presence of cilia-associated bacteria seems frequently overlooked by pathologists, but is a diagnostically significant feature of B. bronchiseptica pneumonia. A specific diagnosis of B. bronchiseptica pneumonia is important because it suggests primary or opportunistic bacterial pneumonia rather than aspiration pneumonia, and because of the risk of animal-to-animal transmission of B. bronchiseptica, the availability of vaccines for disease prevention, and the potential zoonotic risk to immunocompromised pet owners.
Bordetella bronchiseptica is an important cause of respiratory disease in dogs and cats. Tracheobronchitis is the most common and well-recognized manifestation of this infection in dogs, leading to the self-limiting clinical condition known as canine infectious respiratory disease (CIRD) or kennel cough. Although some cases have concurrent viral infections including canine parainfluenza virus, canine respiratory coronavirus (Alphacoronavirus 1), canine adenovirus-2, or Canine distemper virus,14,26 B. bronchiseptica is the sole pathogen identified in other infections,6,26,30 and observations of natural disease 26 and experimental infections 11 suggest that it acts as a primary pathogen.
In addition to tracheobronchitis, B. bronchiseptica can cause pneumonia: it has been identified in 12–78% of dogs with lower respiratory tract infections3,19,26 and 5–13% of cats with lower respiratory tract disease,5,12 and seems particularly important as a cause of pneumonia in puppies 7–35 weeks of age 21 and kittens <12 weeks of age.5,31 Thus, the variability in prevalence in these studies most likely reflects the nature of the populations studied. Although pneumonia may be a less frequent outcome of infection than tracheobronchitis, it is nonetheless important because of greater severity of clinical disease and the potential for a fatal outcome. Finally, B. bronchiseptica is recognized as a zoonotic pathogen affecting humans, particularly those with immunosuppressive conditions or cystic fibrosis.8,33 Healthy cats or dogs, or those with signs of respiratory disease, have been implicated as the source of B. bronchiseptica infection in some cases of pneumonia in humans.16,22,32
Adherence to cilia is a recognized virulence attribute of B. bronchiseptica,2,7 dependent on bvg-regulated expression of filamentous hemagglutinin and pertactin, fimbriae, and adenylate cyclase toxin. 9 Although histologically visible cilia-associated bacteria (B. bronchiseptica) are described in experimental infections,7,28 this pathologic finding is not well described in the current literature. The finding of cilia-associated B. bronchiseptica bacteria is of potential value because it would allow diagnostic pathologists to directly suggest this pathogen as the cause of bronchopneumonia even if additional testing were not possible. Based on analysis of routine case material, we suspected that the histologically visible cilia-associated bacteria may be overlooked by veterinary pathologists. Thus, the objectives of our study were to 1) determine the prevalence of histologically visible cilia-associated bacteria in cases of fatal bacterial bronchopneumonia in dogs and cats, 2) determine whether the cilia-associated bacteria are B. bronchiseptica, and 3) compare the prevalence of detection of B. bronchiseptica based on microscopic observation of hematoxylin and eosin (HE)-stained slides, immunohistochemistry (IHC), PCR testing, and—where possible—bacterial culture.
Material and methods
Case selection, histopathology, and bacterial culture
A search of the archive of the Animal Health Laboratory, University of Guelph (Guelph, Ontario, Canada) revealed 290 canine cases and 146 feline cases coded as lung inflammation in the 5-year period beginning September 2007. To focus the study on cases in which infectious pneumonia rather than aspiration pneumonia was the cause of death, exclusion criteria were as follows: no recorded pathologic diagnosis of bronchopneumonia or bacterial pneumonia, only minor lung lesions that were not considered a probable cause of death, unavailability of paraffin-embedded lung tissue, autolysis that interfered with histologic evaluation, neonates (<2 weeks of age), pulmonary neoplasia, blastomycosis, aelurostrongylosis, or clear evidence of aspiration pneumonia such as a clinical history of megaesophagus. The remaining 36 canine cases and 31 feline cases were subjected to analysis. Histologic sections stained with HE were reviewed (K Taha-Abdelaziz, JL Caswell) without knowledge of the culture, PCR, or IHC data for the presence or absence of cilia-associated bacteria on the apical surface of the airway epithelium. Gram, Warthin–Starry, and periodic acid–Schiff (PAS)/Alcian blue–stained sections were examined from a subset of cases, but the objective assessment of the presence of cilia-associated bacteria was based only on the HE-stained sections.
Bacterial culture data were available from 29 canine and 18 feline cases. Historically in such cases, identification of B. bronchiseptica was based on cell characteristics in Gram-stained preparations, growth on MacConkey agar after 48 h and on Salmonella–Shigella agar; colony morphology; positive catalase, urease, oxidase, and citrate tests; and negative fermentation-oxidation, nitrate, and dextrose tests. 20
Pertactin purification and polyclonal rabbit serum
Antiserum to B. bronchiseptica pertactin was raised in a rabbit for use in IHC. Pertactin was purified from a Stainer–Scholte broth culture of the swine B. bronchiseptica isolate KM22. 18 Bacterial cells were pelleted, resuspended in Tris-buffered saline, and lysed as described, 25 with the addition of phenylmethylsulfonyl fluoride a (PMSF) to 1 mM. The lysate was centrifuged at 15,000 × g for 30 min at 4°C. The supernatant was decanted and dialyzed 4 times against 6 L each of Tris-buffered saline. Proteins were precipitated at 4°C for 1 h following the addition of ammonium sulfate to 40% saturation. Precipitates were collected by centrifugation for 20 min at 12,000 × g and resuspended in 0.025 M bis-Tris buffer–0.05 M NaCl (pH 6.5), followed by overnight dialysis at 4°C against the same buffer, using a 50-kDa cutoff membrane. The following day, the dialysate was treated with 1 mM PMSF and centrifuged at 31,000 × g for 30 min at room temperature. The supernatant was applied to a regenerated 2.6 × 17 cm diethylaminoethyl-sepharose CL-6B column, b and fractions were eluted using 0.15 M NaCl. Material eluted in the first protein peak was concentrated with an ultrafiltration device. c A portion of the concentrate was evaluated by sodium dodecyl sulfate–polyacrylamide gel electrophoresis on a 7.5% gel as described. 27 The predominant band of ~70 kDa, presumably comprised of pertactin, was excised from the gel and electroeluted as described. 1 The electroeluted material was concentrated and re-equilibrated with PBS over an ultrafiltration device. c
Purified pertactin (0.5 mL, ~60 μg) was emulsified by sonication with an equal volume of adjuvent d and injected intradermally into a Bordetella-free New Zealand White rabbit at 25 sites along the back. Approximately 5 weeks later, the rabbit was euthanized and exsanguinated, and the serum obtained was evaluated by Western blot. Use of rabbits in this research was approved by the Institutional Animal Care and Use Committee of the National Animal Disease Center, following the “Guide for the Care and Use of Laboratory Animals.” Reactivity of the serum with a ~70 kDa protein from wild-type KM22 and lack of reactivity with a KM22-derived pertactin mutant 17 confirmed the specificity of the polyclonal antibodies present. This pertactin antiserum and pre-immunization serum obtained from the same animal were subsequently used for IHC.
Immunohistochemistry
Sections from paraffin-embedded tissue were deparaffinized, rehydrated, and subjected to heat-induced epitope retrieval at 110°C for 4 min using modified citrate buffer, pH 6.1. e IHC was done with a commercial kit. e Tissue sections were blocked by incubation with endogenous dual enzyme blocker for 10 min at room temperature then washed with Tris phosphate buffered saline (PBS)–0.1% Tween for 5 min before incubation with serum-free protein block for 30 min. Tissue sections were incubated with a 1:3,000 dilution of serum from a rabbit immunized with purified pertactin, as described below, for 2 h at room temperature or overnight at 4°C. 24 A 1:3,000 dilution of serum from the same rabbit, obtained prior to immunization, was used as a negative control. Sections were washed several times with Tris PBS–0.1% Tween, followed by incubation with peroxidase-labeled goat anti-rabbit immunoglobulin e for 30 min at room temperature. Tissue sections were washed several times with Tris PBS–0.1% Tween prior to incubation with chromogen, f according to manufacturer’s directions, and counterstained with Mayer hematoxylin. The positive control was a case with a positive B. bronchiseptica culture and positive immunolabeling. The negative controls were processed similarly, but the pertactin antiserum was substituted with preimmunization serum (obtained from the same rabbit before injecting pertactin) and, in some cases, as an additional control by omission of the primary antibody.
PCR
Sections of formalin-fixed, paraffin-embedded (FFPE) lung tissue were prepared. To prevent cross-contamination, gloves were used, equipment was cleaned with isopropanol between blocks, a new blade or segment of a blade was used for each block, sections for the facing of each block were discarded, and forceps used to handle paraffin sections were immersed in alcohol between blocks. For each case, six 10-μm-thick sections were deparaffinized with 1 mL of xylene for 5 min at room temperature with shaking. DNA extraction was carried out according to the manufacturer’s protocol. g The quality and quantity of DNA was based on absorbance h at 260 nm.
A 100-bp region upstream of the B. bronchiseptica geneencoding flagellin, flaA, was amplified by PCR using the primers Fla2 (5′-AGGCTCCCAAGAGAGAAGGCTT-3′) and Fla12 (5′-AAACCTGCCGTAATCCAGGC-3′; Fla2/Fla12). 29 Both B. bronchiseptica and Bordetella parapertussis are expected to be positive with this PCR test, but the latter is not known to infect dogs or cats. In addition to the Fla2/Fla12 PCR, a 307-bp portion of the glyceraldehyde 3-phosphate dehydrogenase gene was amplified as a positive control using the following primers: 5′-AGCCTTCTCCATGCTGGTGAAGAC-3′ and 5′-CGGAGTCAACGGATTTGGTCG-3′. Each reaction mixture consisted of 10 μL of master mix, i 0.5 μL of 50 µM of each forward and reverse primer, 1 μg of DNA, and PCR-grade water added to a final volume of 20 μL. Cycling conditions included denaturation for 10 min at 95°C, followed by 35 cycles of 30 s at 95°C, 30 s at 55°C and 45 s at 72°C, with a final elongation step of 72°C for 5 min, and maintenance at 4°C. The PCR products were analyzed by gel electrophoresis using 5 μL of gel dye j in a 1% agarose gel in Tris–borate–ethylenediamine tetra-acetic acid buffer. B. bronchiseptica DNA (described below) was used as a positive control. The products of 2 independently run Fla2/Fla12 PCRs were sequenced after extraction from the gel to confirm the amplified sequence. To determine the lower limit of detection of the Fla2/Fla12 PCR, DNA was extracted from 1.5 × 108 colony-forming units (CFU) of B. bronchiseptica. Serial 10-fold dilutions of DNA were tested by PCR, and the lower limit of detection corresponded to 15 CFU.
Results
The possible involvement of B. bronchiseptica in each case was investigated by histopathology, by IHC using serum containing polyclonal antibody to pertactin, by the Fla2/Fla12 PCR, and by retrospective data on bacterial culture. Of the canine bronchopneumonia cases, 4 of 36 were positive for B. bronchiseptica with both IHC and PCR; culture data were available for 2 of these cases and both were positive for B. bronchiseptica. For 4 additional canine cases, the Fla2/Fla12 PCR was positive but the other tests were negative (including IHC in all cases, and culture in 3 of the cases; Fig. 1, Supplemental Table 1, available online at http://vdi.sagepub.com/content/by/supplemental-data). For these cases, PCR testing was repeated on new sections of FFPE tissue, with the same result. In the HE-stained histologic sections, cilia-associated bacteria were observed in the 4 cases that were positive for B. bronchiseptica by IHC and PCR, but were evident neither in the 4 cases that were positive only by PCR nor in the 28 other cases of bronchopneumonia (Fig. 1, Supplemental Table 1).

Diagnostic test results for Bordetella bronchiseptica in 36 dogs and 31 cats with bronchopneumonia. The data show the number of cases that tested positive (+) or negative (–) for B. bronchiseptica by Fla2/Fla12 PCR, by immunohistochemistry (IHC) for pertactin, and by observation of cilia-associated bacteria in hematoxylin and eosin–stained histologic sections (CAB). Bacterial culture data were available for 29 of 36 dogs and for 18 of 31 cats. Culture-positive refers to those cases in which B. bronchiseptica was isolated, of the total number in which culture was attempted.
Of the feline bronchopneumonia cases, 7 of 31 were positive for B. bronchiseptica based on IHC and PCR; culture data were available for 4 of these cases and 3 were positive for B. bronchiseptica. Seven additional feline cases were positive for B. bronchiseptica by Fla2/Fla12 PCR but not by other tests (including IHC in all cases, and culture in 5 of the cases), and the positive results were confirmed as above by repeating the PCR test (Fig. 1, Supplemental Table 1). In the HE-stained histologic sections, cilia-associated bacteria were observed in 5 of the 7 cases that were positive for B. bronchiseptica by IHC and PCR, but were evident neither in the 7 cases that were positive only by PCR nor in the 17 other cases of bronchopneumonia (Fig. 1, Supplemental Table 1).
Retrospective bacterial culture data
Bordetella bronchiseptica was isolated from 2 of 29 canine and 3 of 18 feline cases of bronchopneumonia in which bacterial culture of lung tissue was attempted. Cilia-associated bacteria were detected histologically in all 5 of these cases, and in none of the B. bronchiseptica culture–negative cases (Supplemental Tables 1, 2, available online at http://vdi.sagepub.com/content/by/supplemental-data). Bordetella bronchiseptica was the only pathogen identified in 3 cases; concurrent pathogens included Streptococcus canis in a canine case, and Mycoplasma felis and feline herpesvirus in a feline case. Of the 29 canine cases with aerobic bacterial culture data, 6 had no growth, and other isolates were Escherichia coli (n = 10), S. canis (n = 8), Pasteurella canis (n = 4), Enterococcus sp. (n = 5), Staphylococcus pseudintermedius (n = 4), Klebsiella pneumoniae (n = 3), Actinomyces sp. (n = 1), Citrobacter sp. (n = 1), Enterobacter sp. (n = 1), Pasteurella multocida (n = 1), Prevotella sp. (n = 1), Staphylococcus aureus (n = 1), and Streptococcus equi subsp. zooepidemicus (n = 1); 15 of 29 cases had more than 1 isolate. Of the 18 feline cases submitted for culture, 3 had no growth and other isolates were E. coli (n = 5), P. multocida (n = 4), Pasteurella dagmatis (n = 3), Streptococcus sp. (n = 3), Enterococcus sp. (n = 2), Enterobacter sp. (n = 1), Moraxella sp. (n = 1), P. canis (n = 1), Prevotella sp. (n = 1), Staphylococcus sp. (n = 1), and S. canis (n = 1); 9 of 18 cases had more than 1 isolate.
PCR testing
The Fla2/Fla12 PCR was positive in 8 of 36 canine cases and 14 of 31 feline cases. The amplicons of 2 randomly selected cases were sequenced, and a BLAST query (http://blast.ncbi.nlm.nih.gov/Blast.cgi) revealed 100% identity with other sequences from B. bronchiseptica.
Reported clinical findings
Of the 8 dogs with evidence of B. bronchiseptica pneumonia, 4 were ≤11 weeks of age. The reported clinical signs included lethargy, inappetence, cough, shallow breathing, hyperpnea, and dyspnea. The duration of clinical signs, when specified, was 2–3 days (except for 1 dog with a 20-day history of kennel cough). One case involved multiple animals. Three dogs had been recently purchased, 1 died in a boarding kennel, 1 developed signs 2 days after vaccination, and 1 died <1 day after anesthesia (Supplemental Tables 1, 2).
Of the 14 cats with evidence of B. bronchiseptica pneumonia, 10 were ≤7 months of age, and 3 cases involved multiple animals. The reported clinical signs included lethargy, inappetence, tachypnea, dyspnea, sneezing, fluid draining from the laryngeal cavity, pyrexia, weight loss, and vocalization. The duration of clinical signs, when specified, was 0–4 days. Death without observed clinical signs occurred in 2 cases. Potential risk factors mentioned in the history included recent purchase, parturition, anesthesia, or upper respiratory tract infection.
Pathologic findings
Lesions were similar in cats and dogs with evidence of B. bronchiseptica pneumonia and are described together (Supplemental Tables 1, 2). The gross anatomic lesions were usually most severe in the cranial and middle lung lobes; however, some cases were reported to mainly involve the caudal lobes. The affected tissue was red, wet, heavy, and firm. Nine cases (5 dogs, 4 cats) had frothy, mucoid, or purulent content in the trachea and bronchi. Three dogs had subpleural or pulmonary hemorrhages. Five cats and 1 dog had serosanguinous or fibrinous pleural effusion.
Histologically, cilia-associated bacteria were mainly located in bronchi, rarely present in medium to large bronchioles and bronchial glands, and absent from terminal bronchioles. Cilia-associated bacteria appeared as short bacilli that intermingled with or obscured the cilia at the apical surface of bronchial epithelium; their basophilic staining contrasted with the eosinophilic cilia (Fig. 2A–F). The bacteria usually appeared as localized “carpet-like” aggregates covering 2–5 epithelial cells (Fig. 2A), or as confluent growth affecting the entire circumference of the bronchus, or rarely as individual bacteria that were difficult to reliably differentiate from granules of mucus (Fig. 2D, Supplemental Fig. 1A–C, available online at http://vdi.sagepub.com/content/by/supplemental-data). The bacteria were gram negative and stained black with Warthin–Starry, on a subset of cases tested (Supplemental Fig. 1D–F). Of the 9 cases with histologically visible cilia-associated bacteria, this finding was only mentioned in the pathology report of 2 cases.
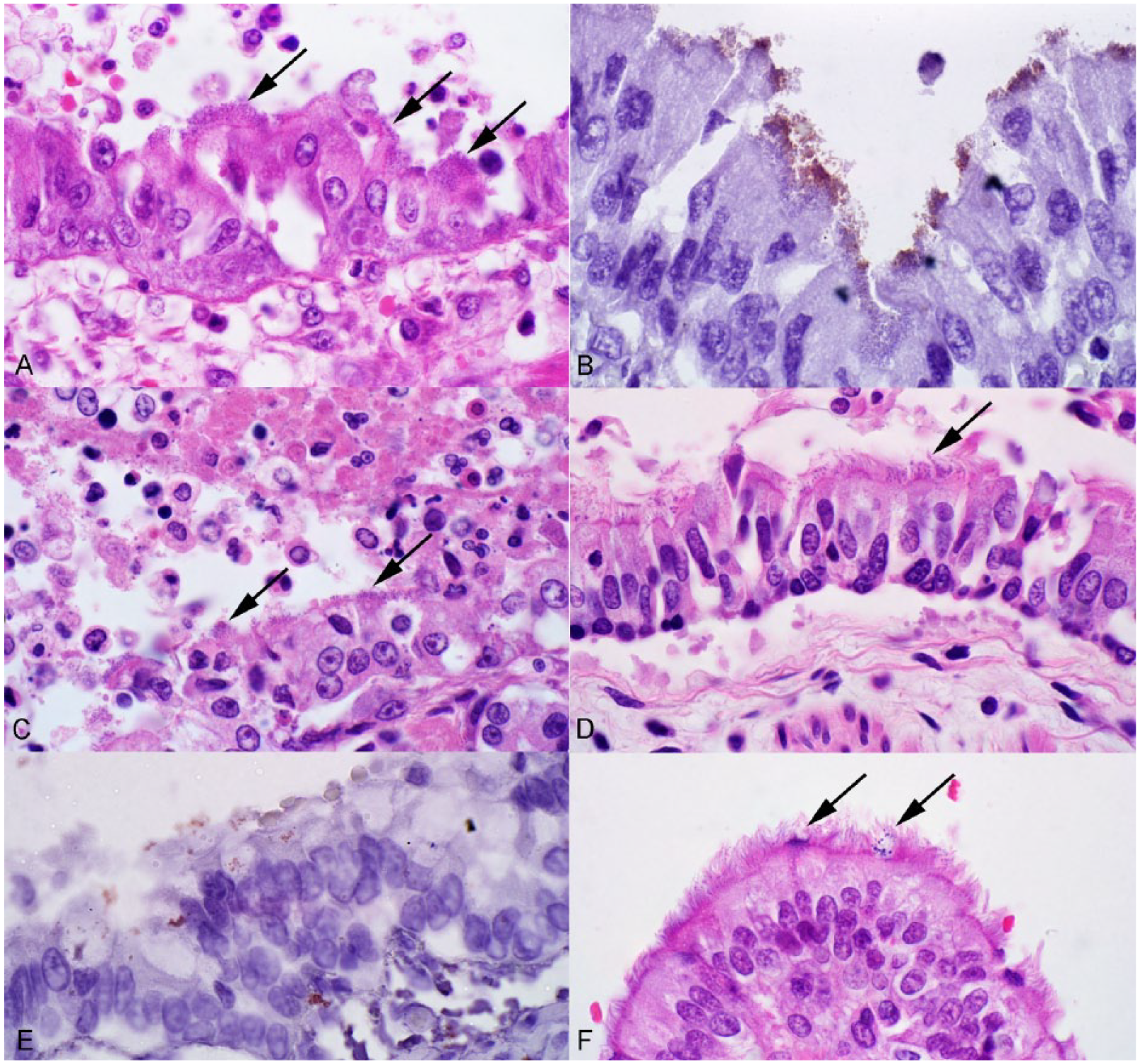
Bordetella bronchiseptica pneumonia in dogs (A, B, D–F) and a cat (C).
Attenuation of bronchial or bronchiolar epithelium was noted in 6 of 8 canine and 9 of 14 feline cases. This lesion was patchy so that most of the affected airways had some normal and some thinned epithelium. Bacteria were often absent or inconspicuous near these areas of erosion, although present elsewhere in the same airway. The bronchial and bronchiolar lumens contained neutrophils, macrophages, and fibrin (Fig. 2C). Low numbers of neutrophils infiltrated the epithelium. The lamina propria contained many macrophages and low numbers of neutrophils.
Alveoli were filled with neutrophils and macrophages, and in some cases with fibrin and erythrocytes. Often, these leukocytes appeared necrotic, retaining their round shape but having pyknotic nuclei and hypereosinophilic cytoplasm. Intra- and extracellular tiny coccobacilli were infrequently observed in alveoli in HE-stained sections and were confirmed to be B. bronchiseptica by Gram stain and IHC (Supplemental Fig. 2C, 2D, available online at http://vdi.sagepub.com/content/by/supplemental-data). The intra-alveolar B. bronchiseptica bacteria were inconspicuous, in part because of their small size. Hyaline membranes or type II pneumocyte proliferation were not frequent. Mononuclear cells within alveolar septa were more prominent than normal. Thrombosis was present in 5 cases and focal necrosis of inflamed lung tissue was noted in 4 cases, but bacterial pathogens other than B. bronchiseptica were also isolated from all such cases for which culture data were available.
Immunohistochemistry for B. bronchiseptica pertactin labeled coccobacilli at the apical surface of epithelial cells lining the bronchi (Fig. 2B, 2E). In contrast to the extensive labeling within bronchi, immunolabeled bacteria were infrequent on the surface of the bronchiolar epithelium. Apical labeling of epithelial cells of bronchial glands was noted in 1 cat (Supplemental Fig. 2A, 2B). Immunolabeling of the cytoplasm of leukocytes in alveoli and bronchial exudate was noted in a few cases (Supplemental Fig. 2C, 2D).
Discussion
Bronchopneumonia is an important cause of mortality in dogs and cats. In cases of bronchopneumonia, the histologic lesions often do not indicate a specific causative pathogen. However, our study demonstrates the value of careful examination of multiple histologic sections of pneumonic lung for detection of cilia-associated bacteria affecting the large airways of dogs and cats with bronchopneumonia.
The study population was selected from an archive of cases submitted from both primary care veterinary clinics and from a university referral hospital. Exclusion criteria were intended to focus the study population on cases that died of opportunistic bacterial pneumonia. Cases with mild lung lesions or an obvious alternative cause of death were excluded, as were cases with clear evidence of aspiration pneumonia because B. bronchiseptica is not recognized as a cause of aspiration pneumonia. These exclusion criteria were intended to refine the study population to reflect those cases in which careful assessment for cilia-associated bacteria is most warranted. In this population, 8 of 36 (22%) canine and 14 of 31 (45%) feline cases of bacterial pneumonia had evidence of B. bronchiseptica infection.
The major objective of our study was to establish the value of examining the bronchial epithelium in HE-stained histologic sections for the presence of cilia-associated bacteria, which suggests a diagnosis of B. bronchiseptica pneumonia. In all cases where cilia-associated bacteria were observed in HE-stained histologic sections, B. bronchiseptica was identified in the lung tissue by PCR testing, and B. bronchiseptica was isolated from all of those cases that had culture data available. Furthermore, IHC was used to colocalize B. bronchiseptica antigen with histologically observed bacteria, confirming the identity of the observed bacteria. A caveat is that cases without evidence of B. bronchiseptica infection did have tiny basophilic granules in association with cilia and the apical surface of airway epithelial cells; these granules stained red-pink with Gram stain. The granules were interpreted to be mucus based on their location and similarity to granules in goblet cells, and magenta staining with PAS/Alcian blue. Their presence was considered nonspecific, whereas confluent basophilic aggregates or mats of cilia-associated material were indicative of B. bronchiseptica infection, particularly if bacillus-shaped structures could be discerned. Thus, the finding of aggregates of cilia-associated bacteria in this study population was specific for B. bronchiseptica.
Cilia-associated bacteria were not observed in all cases of B. bronchiseptica infection. Specifically, 2 cats tested positive by IHC and PCR but cilia-associated bacteria were not apparent histologically. In these cases, the IHC staining was not associated with the cilia of airway epithelial cells but was instead amid the exudate in the lumen. Thus, for the present study population, careful examination of HE-stained histologic sections identified cilia-associated bacteria in all cases in which cilia-associated bacteria were identified by IHC, and in all culture-positive cases.
Four canine and 7 feline cases tested positive by PCR but were negative by IHC, and cilia-associated bacteria were not observed histologically. Culture data from 7 of these cases were available but B. bronchiseptica was not isolated. We considered the possibility that these may represent a false-positive PCR test, but the measures taken to prevent cross-contamination during sample processing and the confirmation of the PCR findings using new sections of the FFPE tissue made this possibility unlikely. Autolysis with sloughing of bronchial epithelium is a potential reason for failure to identify B. bronchiseptica infection using IHC and histopathology, but the histologic appearance ruled out this possibility. Furthermore, although the presence of cilia-adherent bacteria is variable among samples of lung from the same case, all tests in these cases were performed on the same paraffin block. Of the cases that were positive by PCR but not by other tests, 1 had a history of kennel cough, and 3 developed pneumonia after recovery from anesthesia. The positive PCR tests in these cases may not reflect established infection in the lung of these animals; instead, this may represent aspiration of B. bronchiseptica from the upper respiratory tract or introduction into the lung during endotracheal intubation. Thus, adherence to cilia may be of less importance for pathogenesis in these cases. Because B. bronchiseptica infection is prevalent in the upper respiratory tract of healthy dogs and cats,15,26 the findings suggest that microscopic observation of cilia-adherent bacteria by histopathology or IHC could be of value to differentiate transient introduction versus established infection of the lung with B. bronchiseptica.
Apart from the observation of cilia-associated bacteria, the histologic lesions of bronchopneumonia in these cases did not indicate a specific bacterial pathogen. Thus, careful examination of large airways for the presence of cilia-associated bacteria is of value in identifying the specific cause, particularly when fresh tissue is not available for culture and other testing is not available. Anecdotal evidence and the fact that cilia-associated bacteria were only mentioned in 2 pathology reports in this series suggests that this diagnostically useful lesion is overlooked by veterinary pathologists. A specific diagnosis of B. bronchiseptica pneumonia suggests primary or opportunistic bacterial pneumonia rather than aspiration pneumonia, and a specific diagnosis may be of value because of the risk of animal-to-animal transmission of B. bronchiseptica,4,10 the availability of vaccines for disease prevention, and the risk for infection of immunosuppressed humans.
Necrosis of bronchial or bronchiolar epithelium was noted in 68% of cases with evidence of B. bronchiseptica infection. Both tracheal cytotoxin and dermonecrotic toxin may contribute to lesions of the respiratory epithelium during Bordetella infections in other hosts, 23 but the possible role of these or other toxins in the lesions observed in our study is unknown. Underlying viral infections may also have caused epithelial necrosis. Felid herpesvirus was detected in 2 cats in this study, and Feline calicivirus in another. However, we did not systematically test for viruses because testing of postmortem cases is expected to underestimate the true prevalence of concurrent viral infection compared to testing samples obtained at the onset of disease, and because the likelihood of concurrent viral infection is already known.13–15,26
The prevalence data reported in our study are constrained by the relatively low number of cases, and we expect that the prevalence of B. bronchiseptica infection would vary considerably depending on the age of animals examined, the nature of the animal populations, and the details of inclusion and exclusion criteria. This limitation does not affect the major conclusions of the study with respect to the value of identifying cilia-associated bacteria. Although the PCR assay appeared to be more sensitive than IHC or histopathology, the study was not designed to formally compare test sensitivity.
In this study, B. bronchiseptica was identified in 22 of 67 cases of fatal bronchopneumonia in dogs and cats. Cilia-associated bacilli were observed in HE-stained histologic sections in 9 cases, and B. bronchiseptica was detected by IHC in 11 cases, by PCR in 22 cases, and by bacterial culture in 5 of the 47 cases with available data. The majority of animals were <7 months of age, but adults were also affected. The cilia-associated bacteria formed basophilic, gram-negative, argyrophilic clusters at the apical surface of epithelial cells lining the bronchi and rarely the large bronchioles, and immunohistochemical colocalization confirmed their identity as B. bronchiseptica. Care is needed to distinguish individual bacteria from granules of mucus, but the diagnosis is straightforward when the bacteria form large clusters or confluent mats. The presence of cilia-associated bacteria was only mentioned in the pathology report for 2 of these 9 cases, suggesting that this diagnostically significant lesion is overlooked by veterinary pathologists. Because the cilia-associated bacteria are inconsistently distributed among sections of lung and may be subtle, it is recommended to carefully examine the epithelium of large bronchi from multiple samples of lung tissue affected by bronchopneumonia in cats and dogs.
Footnotes
Authors’ contributions
K Taha-Abdelaziz and JL Caswell contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; and drafted the manuscript. LL Bassel, ML Harness, ME Clark, and KB Register contributed to acquisition, analysis, and interpretation of data. All authors critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Notes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Natural Sciences and Engineering Research Council of Canada (NSERC; RGPIN 227845 to JLC).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
