Abstract
Accurate diagnosis of Mycobacterium bovis infection (bovine tuberculosis [bTB]) in live animals is notoriously problematic. The aim of the present study was to evaluate the diagnostic potential of 2 new serologic tests (multiantigen print immunoassay [MAPIA] and lateral flow immunoassay rapid test [RT]) in comparison with mycobacterial culture of tracheal washes for determining M. bovis infection status in a free-ranging population of wild meerkats (Suricata suricatta). During a longitudinal study lasting 2.5 years, 240 individually identifiable meerkats were each sampled up to 8 times under anesthesia every 3 months. Diagnostic accuracy was determined through Bayesian and maximum likelihood estimations of sensitivity, specificity, and likelihood ratios for each diagnostic test when used independently and in parallel to classify the disease status of individual meerkats in the absence of a gold standard. Culture of tracheal washes was highly specific (0.99; 95% confidence interval [CI] = 0.77, 1.00) but of low sensitivity (0.36; 95% CI = 0.24, 0.50) for diagnosing M. bovis-infected individuals. The longitudinal nature of the study with repeated sampling of the same individual animals served simultaneously to improve chances of detecting infection and increase confidence in a negative result in individual animals repeatedly testing negative. Although MAPIA and RT were individually of limited diagnostic use, interpreting the results of these 2 tests in parallel produced estimates of sensitivity (0.83; 95% CI = 0.67, 0.93) and specificity (0.73; 95% CI = 0.62, 0.82) high enough to usefully inform decision making when determining exposure to bTB in wild meerkats and potentially other species in which bTB poses a diagnostic challenge.
Introduction
Accurate diagnosis of Mycobacterium bovis infection (bovine tuberculosis [bTB]) in live animals is notoriously problematic. 15 Pathogenesis of this disease may vary within as well as between species, resulting in different routes of excretion and transmission patterns. 4 Prolonged incubation periods, up to several years in some species, such as African buffalo (Syncerus caffer), 16 indicate that apparently healthy animals may be carrying and excreting pathogenic mycobacteria. Furthermore, mycobacteria may be shed only intermittently by infected animals, limiting the sensitivity of tests based on detecting M. bovis in excretions at a single time point. 11,34
Serologically based assays for the detection of bTB have many potential advantages over mycobacterial culture in terms of cost, ease of application, and speed of result. 14 However, the development of serologic tests for M. bovis infection with sufficiently high sensitivity and specificity to be of practical use in the field has been hampered by several factors. Delayed seroconversion, 24 cross-reactivity with environmental mycobacteria, 40 and the inherent trade-off between sensitivity and specificity in tests with moveable cutoff points 39 have all contributed to the difficulties in establishing a satisfactory serologic test for bTB. One possible solution is to test for a panel of antibodies to multiple mycobacterial proteins, some of which may be produced early in the infection in detectable amounts. 1,32 This is important because ideally a serologic test should be able to correctly identify infected animals before they become infectious or show clinical signs, if it is to be of any use in management of disease. Two new serologic tests based on this premise have been developed with apparent improved success in the early diagnosis of bTB, namely the multiantigen print immunoassay (MAPIA) 33 and a lateral flow immunoassay rapid test (RT). 8
Natural infection with M. bovis has been present in a study population of free-living meerkats (Suricata suricatta) in the Kalahari Desert of South Africa for several years. 18 To better understand the epidemiology of bTB in meerkats, and in particular to elucidate the role of specific social interactions in transmission of this infectious disease, accurate and simple tests to determine exposure to and shedding of M. bovis are needed.
The aim of the present study was to evaluate the diagnostic potential of MAPIA and RT in comparison with mycobacterial culture of tracheal washes in a population of wild meerkats with naturally occurring M. bovis infection. This was achieved through determination of the sensitivity, specificity, and likelihood ratios for each of the tests when used independently and in parallel (whereby the 2 serologic tests were run concurrently with a positive diagnosis requiring that only 1 test result be positive) to classify the disease status of individual meerkats in the absence of a gold standard.
Materials and methods
Study site and population
Data and samples were all collected at the Kalahari Meerkat Project in the Northern Cape, South Africa (26°58′S, 21°49′E). This is an area consisting of dry riverbed, semiarid herbaceous flats, and sparsely grassed dunes surrounded by ranchland on which cattle, sheep, goats, and ostriches (Struthio camelus) are farmed. Approximately 300 wild meerkats living in 14 social groups have been habituated to close observation (<1 m) facilitating individual identification and enabling relatively easy capture for sampling. 12 Meerkats were individually identified by small marks of hair dye, confirmed when necessary by scanning subcutaneous microchips. All research protocols were approved by the Research Ethics Committee at the University of Pretoria, South Africa, and permission to conduct the research was granted by Northern Cape Nature Conservation Service.
Sample collection
Meerkats were sampled in the mornings (between 6:00
A cohort of meerkats was sampled approximately every 3 months between December 2005 and December 2007 (8 sampling sessions). Cohort size varied from 33 to 154 (median = 73). During this period, 581 sets of samples were collected from 258 different meerkats. The number of meerkats included in subsequent analyses varied because not every diagnostic test was performed on all meerkats and some analyses included only meerkats tested multiple times. An explanation of the number of meerkats included in each test is given where appropriate.
Serologic tests
Multiantigen print immunoassay (MAPIA). Nitrocellulose strips printed with a panel of 12 mycobacterial antigens of the Mycobacterium tuberculosis–complex b were used to test meerkat sera for antibodies using MAPIA. The panel consisted of 8 purified recombinant proteins (ESAT-6, CFP10, Acr1, 38 kDa protein, MPB59, MPB64, MPB70, and MPB83), 2 protein fusions (CFP10/ESAT-6 and Acr1/MPB83), and 2 native antigens (bovine purified protein derivative [PPDB] and M. bovis culture filtrate [MBCF]). The MAPIA was performed using a modified version of methods previously described. 33 Strips were prewetted in multichannel troughs and then blocked for 1 hr with blocking solution (BS; phosphate buffered saline c with 0.05% Tween 20 d and 1 % bovine serum albumin d ) before incubation for 1 hr at room temperature with serum samples diluted 1:40 in BS. After washing, strips were incubated for 1 hr at room temperature with alkaline phosphatase-conjugated protein A e diluted 1:1,000 in BS, followed by another washing step. Preliminary studies (K.P. Lyashchenko, unpublished data) indicated that binding of conjugated protein A provided a positive visual readout in samples known to contain antibody. The data were confirmed in the current study where MAPIA results are in agreement for both negative and positive samples (Figs. 1, 2). Meerkat antibodies bound to immobilized antigens were visualized with BCIP (5-bromo-4-chloro-3′-indolyphosphate) and NBT (nitro-blue tetrazolium chloride). d Strips were washed twice in distilled water then allowed to dry overnight before being examined. Appearance of a purple band at the level of bound antigen indicated presence of antibody in the meerkat serum being tested. Presence of 1 or more purple bands of any intensity was considered a positive test result, except for a single band at the level of MBCF, which was considered a negative test result. This was because studies using MAPIA in European badgers (Meles meles) suggest low specificity of this band, which is likely to be due to components of MBCF being common to environmental mycobacteria that sensitize badgers and are responsible for cross-recognition of MBCF. 23
Case definition. To elucidate the relative reactivity to different MAPIA antigens of meerkats with and without bTB, MAPIA results were compared with results from mycobacterial culture of pooled tissues (lung, spleen, and lymph nodes) collected subsequently from the same individual animals during detailed postmortem examination. This comparison using culture results as the gold standard reference test allowed a best estimate of each meerkat's true disease status to be made. Cadavers of 57 meerkats from which serum had previously been obtained for MAPIA were available for postmortem examination and so the analysis of MAPIA reactors was based on a sample size of 57.
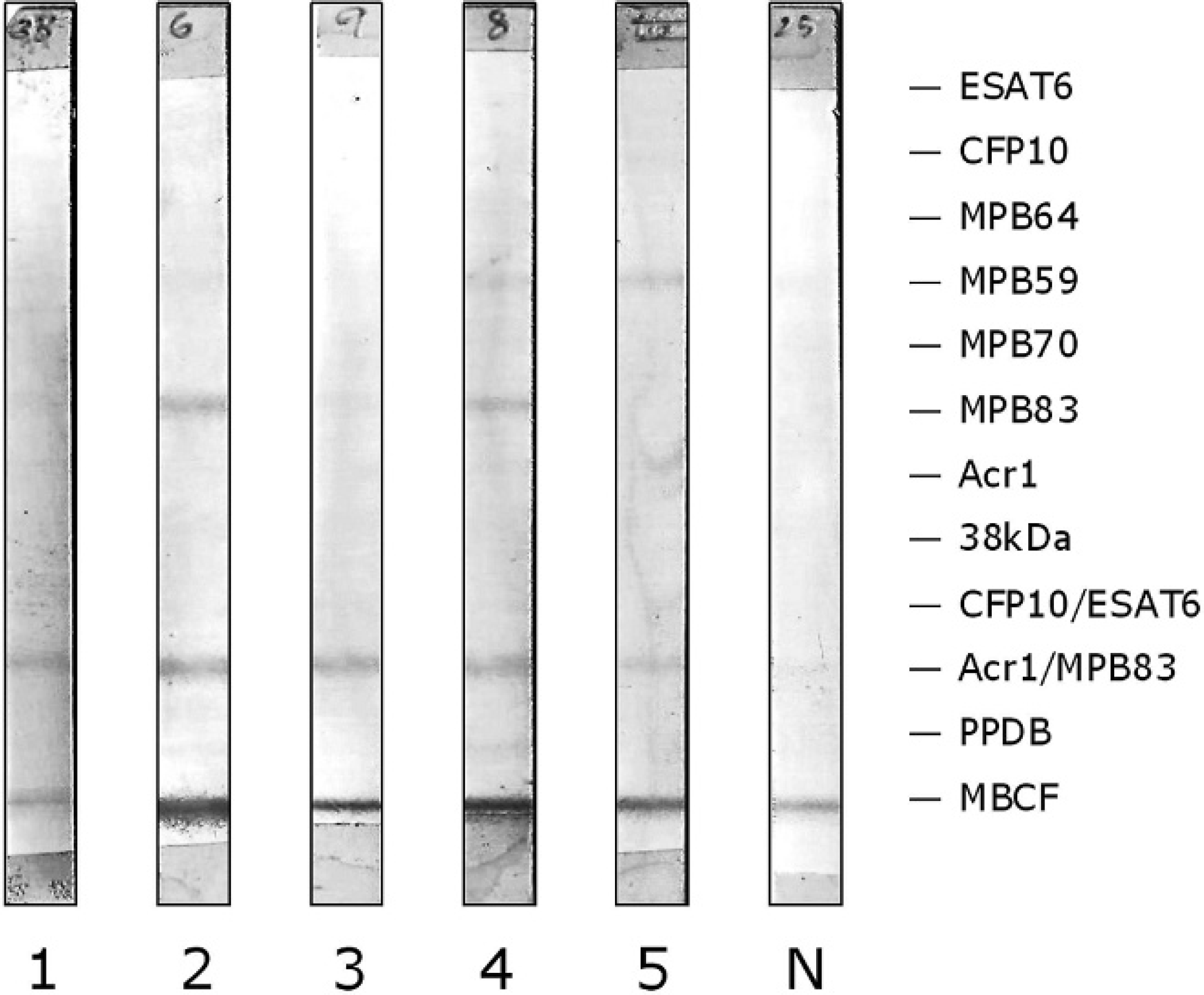
Antibody responses to multiple Mycobacterium tuberculosis-complex antigens in a meerkat with Mycobacterium bovis infection revealed by multiantigen print immunoassay (MAPIA) of sequential serum samples. Antigens are indicated in the right margin, with a band visible on each strip where antibody is present. Samples were collected at the following time points: 1 = 0 months (December 2005); 2 = 8 months (August 2006); 3 = 10 months (October 2006); 4 = 15 months (March 2007); 5 = 17 months (May 2007). N = negative control.
Rapid test (lateral flow immunoassay). A commercially available lateral flow immunoassay f was used to examine for IgM and IgG antibodies to a cocktail of M. tuberculosis-complex antigens. 1 The disposable RT device consisted of a plastic cassette containing a strip of nitrocellulose membrane impregnated with test antigen (MPB83, ESAT-6, CFP10) and laminated with several pads made of glass fiber and cellulose. 31 Meerkat serum (30 μl) and 3 drops of sample diluent buffer were added sequentially to the sample pad. Antigen-conjugated blue latex particles bound with antibody (if present in serum sample) to form a colored immune complex that was visible as a blue band in the test window. The test was left to run for 20 min, during which time a similar blue band appeared in the control area of the device, irrespective of the presence of specific antibody in the test sample. A visible band in the test area, in addition to the control line, was considered an antibody-positive result, whereas presence of the control line only was considered a negative result. Positive samples were subjectively scored as faint (+), moderate (++), or strong (+++), depending on the strength of the test band.

Rapid test (lateral flow immunoassay) results of sequential serum samples from a meerkat with Mycobacterium bovis infection. In the test window, the upper line (labeled C) is an internal positive control indicating mobility of the serum sample and diluent across the membrane. The lower bands of reactivity (labeled T) visualized on samples 1–5 indicate a positive reaction to the test antigens. Samples were collected from the same meerkat and at the same time points as in Figure 1. Relative test band strengths are given in brackets: 1 = 0 months (+); 2 = 8 months (++); 3 = 10 months (+++); 4 = 15 months (++); 5 = 17 months (+++). N = negative control. Tracheal washes collected from the same animal at time points 1 and 5 cultured positive for M. bovis.
Mycobacterial culture
Tissues collected during postmortem examination of 57 meerkats were processed for culture as previously described. 5 Tracheal washes and submandibular lymph node aspirates from each animal were cultured together to produce a single culture result for each animal at each sample point. (Preliminary tests in which the 2 sample types were cultured separately resulted in a high degree of false-negative results due to rapid desiccation of the extremely small sample volume of lymph node aspirates.) Only 1 decontamination method (2% sodium hydroxide at a 1:1 ratio by volume) was used for the tracheal washes. After standing for 10 min at room temperature, decontaminated samples were centrifuged at 1,650 × g for 10 min then neutralized with distilled sterile water and centrifuged again. The pellet that remained after pouring off the supernatant was inoculated onto solid mycobacteria-selective culture media (Lowenstein-Jensen with pyruvate and Lowenstein-Jensen with glycerol). Media were incubated at 37°C for 10 weeks and examined weekly for growth. The organisms from colonies growing on the mycobacteria-selective media were confirmed as acid-fast bacilli by Ziehl-Neelsen staining and identified initially as members of the M. tuberculosis complex and then as M. bovis by polymerase chain reaction. 35 A positive control (bovine lung tissue with culture-confirmed M. bovis infection) and a negative control (culture-negative bovine tissue from a bTB-free animal) were included in each batch of samples at all stages.
Statistical analyses
Evaluation of the performance of 3 diagnostic tests with each test used independently. The sensitivity and specificity of the each of the 3 diagnostic tests (RT, MAPIA, and culture) were estimated in the absence of a gold standard via Bayesian modeling. 6 This analysis allowed for dependency between the 2 serologic tests (RT and MAPIA) but assumed these tests were conditionally independent of culture. 41 All 3 tests were performed on samples collected from 110 meerkats. For each meerkat, serum and tracheal washings were collected on the same date. Data were analyzed using WinBUGS software 30 to run a Markov chain Monte Carlo model with uniform (0, 1) priors on all parameters. Convergence was monitored using 2 overdispersed chains, each initiated by random sampling from the prior for each unknown parameter. Estimates of sensitivity and specificity for each test were generated from 20,000 posterior samples collected after thinning the chains every 50 iterations after a burn-in of 1,000 iterations. Convergence was assessed by visual checking of trace plots of both chains for each parameter.
Assessment of agreement between serologic tests RT and MAPIA. The degree to which the 2 serologic tests RT and MAPIA agreed in their results beyond that which would be expected by chance alone was assessed through calculation of a kappa coefficient. This calculation was carried out on the same data set of 110 meerkats used to evaluate the performance of the diagnostic tests. Kappa was judged according to the following guidelines: <0.20 = slight agreement; 0.21–0.40 = fair; 0.41–0.60 = moderate; 0.61–0.80 = substantial; >0.80 = almost perfect agreement. 27
Evaluation of the performance of 3 diagnostic tests with 2 tests used in parallel and 1 test applied independently. A maximum-likelihood procedure was used to estimate the performance of the 3 tests when 2 were used in parallel and 1 in isolation in the absence of a gold standard reference test. 19 This represented “field use” of tests as used to classify the infection status of meerkats into 3 categories: negative, exposed, and infectious. The serologic tests RT and MAPIA were performed in parallel with meerkats testing positive on at least 1 of these 2 tests being classified as exposed to tuberculosis. Meerkats testing positive for M. bovis on culture of tracheal wash samples were classified as infectious. Meerkats testing negative on all 3 tests were classified as negative. A Newton-Raphson technique using TAGS programming 37 of R software (R Development Core Team: 2007, R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. http://www.R-project.org) was used to evaluate the performance of tests on 403 sets of samples from 240 meerkats in 14 social groups. Samples came from meerkats ranging in age from 3 months to 9 years and included meerkats in all 3 categories of infection (negative, exposed, infectious). For the purposes of this analysis, meerkats were allocated into 1 of 2 subpopulations based on social group bTB history: meerkats from groups with current or historical cases of bTB (confirmed by culture of M. bovis or microscopic identification of granulomatous lesions containing acid-fast bacilli) were allocated to population 1 (high disease prevalence; 7 groups) whereas meerkats from groups without a history of bTB or of unknown disease status were allocated to population 2 (low disease prevalence; 7 groups). The Newton-Raphson algorithm included 2 assumptions: 1) diagnostic tests were conditionally independent (this assumption was met by using RT and MAPIA in parallel to give a single serologic result, the conditionally independent test being culture); and 2) test diagnostic values were constant when applied to different populations.
. Proportion of serum antibody reactors to protein antigens of the Mycobacterium tuberculosis-complex detected by multiantigen print immunoassay in 57 wild meerkats.
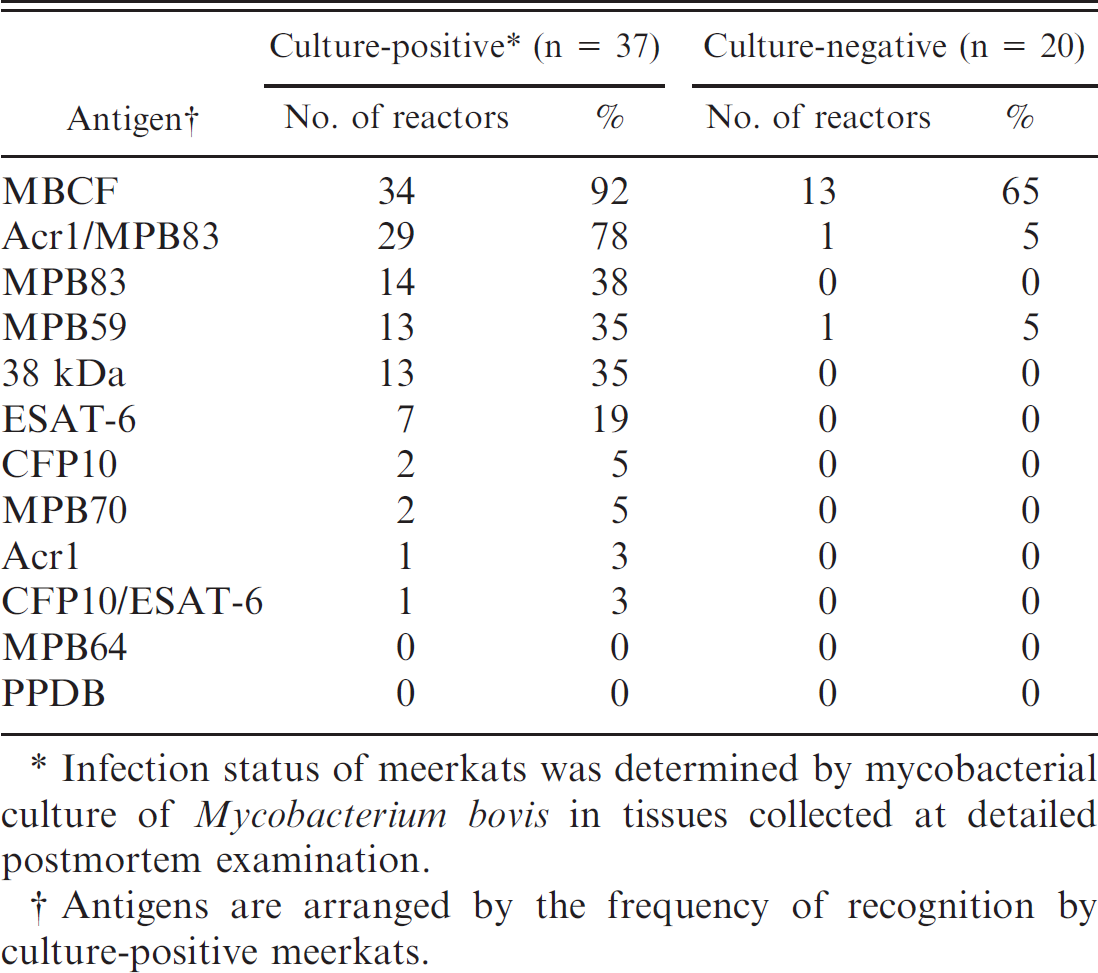
Infection status of meerkats was determined by mycobacterial culture of Mycobacterium bovis in tissues collected at detailed postmortem examination.
Antigens are arranged by the frequency of recognition by culture-positive meerkats.
To allow a comparison of the usefulness of serology and culture in ruling a diagnosis of M. bovis infection in or out, likelihood ratios were calculated for positive and negative results for each test (Table 5). The likelihood ratio of a positive test (the ratio of the true positive rate to the false-positive rate) was calculated as:
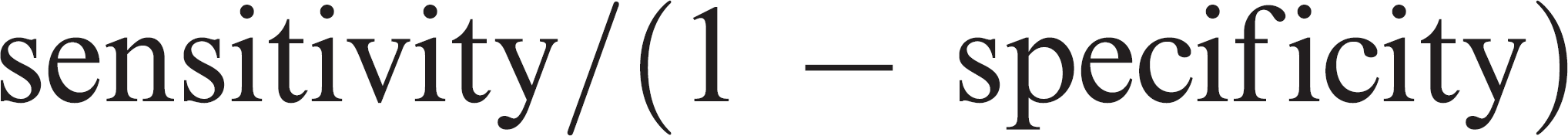
and the likelihood ratio of a negative test (the ratio of the false-negative rate to the true negative rate) as:
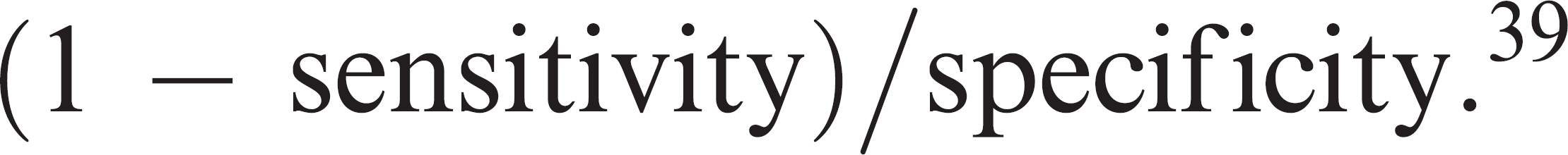
Results
Multiantigen print immunoassay
Of the 57 meerkats whose “true” disease status was determined by detailed postmortem examination and culture of tissues, 37 tested positive for bTB and 20 tested negative (Table 1). Antibodies against 1 or more antigens in the MAPIA panel were detected in 37 (100%) of the culture-positive meerkats and in 13 (65%) of the culture-negative meerkats. Further, MBCF and Arc1/MPB83 were the antigens most frequently recognized in meerkats with bTB (in 92% and 78% of cases, respectively; Table 1). Although MBCF elicited stronger antibody responses than did Arc1/MPB83 (Fig. 1), it was far less specific, being reactive with 65% (13/20) of sera from culture-negative meerkats compared with 5% (1/20) of sera reactive with Arc1/MPB83 in this group (Table 1). Two antigens, MPB64 and PPDB, showed no serum reactivity in infected meerkats, whereas 9 antigens went unrecognized by culture-negative meerkats (Table 1). Some tuberculous meerkats that were tested on multiple occasions over 2 years showed evidence of progression in reactivity to MAPIA antigens; a commonly observed progression began with MBCF and Acr1/MPB83 being visible in 1 serum sample, followed by MPB83 and then MPB59 in subsequent samples from the same animal (Fig. 1). However, such progression was not evident in all cases.
. Cross-classified observed results of 3 tests for tuberculosis performed on samples collected from 110 live, wild meerkats.*
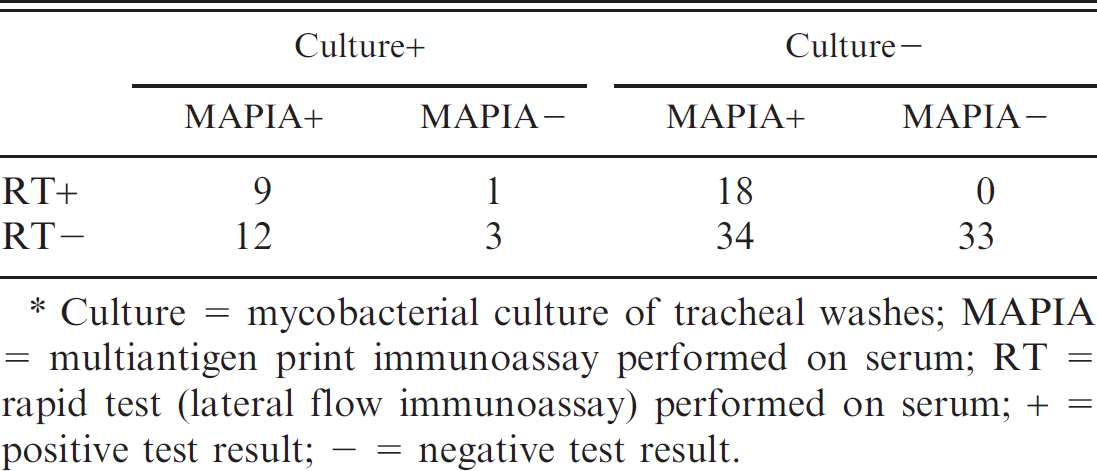
Culture = mycobacterial culture of tracheal washes; MAPIA = multiantigen print immunoassay performed on serum; RT = rapid test (lateral flow immunoassay) performed on serum; + = positive test result; – = negative test result.
Lateral flow immunoassay (rapid test)
A positive RT result was obtained in 25% (28/110) of meerkats tested (Table 2). A general trend of increased RT band strength over time was seen in infected meerkats tested on several occasions (Fig. 2). Antibody was detectable by RT from 0 to 14 months before culture-positive tracheal washes were obtained from the same animal, suggesting that RT was in some cases able to identify infected meerkats well in advance of the shedding stage of the disease.
Assessment of agreement between serologic tests MAPIA and RT
The kappa coefficient (K = 0.26; 95% CI = 0.15, 0.38) revealed a fair agreement between the results of MAPIA and RT when applied to samples from 110 meerkats.
Evaluation of the performance of 3 diagnostic tests with each test used independently
The cross-classified results of RT, MAPIA, and culture performed on samples collected from 110 meerkats appear in Table 2. Tracheal washes from 25 of 110 meerkats (23%) cultured positive for M. bovis, whereas 28 (25%) tested positive using RT and 73 (66%) were positive on MAPIA. Estimates for the sensitivity and specificity of these 3 tests, determined by Markov chain Monte Carlo simulation, appear in Table 3. These values represent the performance of each test when used independently with no prior knowledge of true disease status. The 2 serologic tests differed markedly in their performance; RT showed low sensitivity but high specificity (indicating that this test fails to detect many infected animals but is good at correctly excluding noninfected meerkats), whereas MAPIA was found to have high sensitivity but low specificity (suggesting that this test rarely misses cases of infection but is relatively poor at identifying noninfected animals as negative). Because of the contrasting performance characteristics of these 2 tests they were evaluated in parallel in future analyses (see the following sections) to minimize the chances of missing any cases of disease.
. Bayesian estimates (median and 95% probability interval) for sensitivity and specificity of 3 tests for tuberculosis in 110 wild meerkats.*
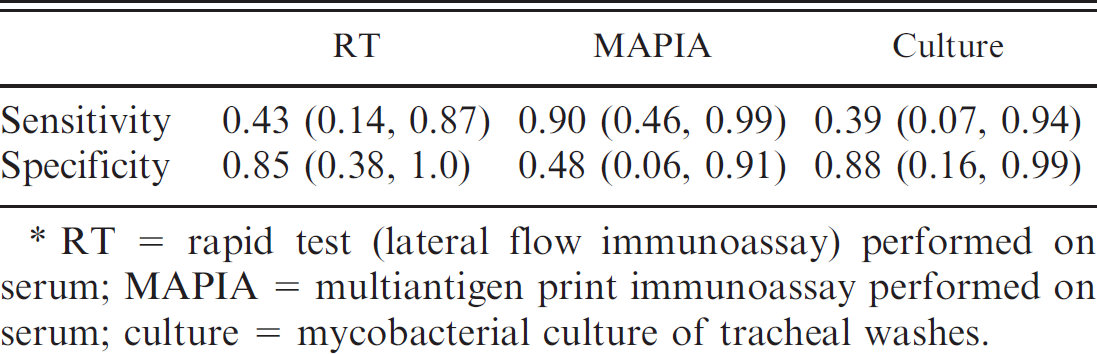
RT = rapid test (lateral flow immunoassay) performed on serum; MAPIA = multiantigen print immunoassay performed on serum; culture = mycobacterial culture of tracheal washes.
Evaluation of the performance of 3 diagnostic tests with 2 tests used in parallel and 1 test applied independently
Some prior information on the likely disease status of individual meerkats was known because of the longitudinal design of this study. This information was used to inform the interpretation of subsequent test results where meerkats were sampled at multiple time points. The serologic tests RT and MAPIA were performed in parallel with meerkats testing positive in at least 1 of these 2 tests being classified as exposed to tuberculosis. Meerkats were classified as exposed from their first seropositive test result onward. Similarly, because of the low sensitivity of single tracheal wash culture results (Table 3), meerkats were classified as infectious from their first positive culture result onward. This was considered acceptable because of the relatively high specificity of culture (Table 3) and because mycobacteria may be shed intermittently from tuberculous animals. 11,34 Using these assumptions, the cross-classified results of tests to determine the infection status of 240 meerkats, each sampled between 1 and 8 times, are presented in Table 4. Meerkats in groups with a history of bTB (population 1) showed a culture-positive rate of 17.7% (50/282) whereas meerkats from groups without a history of bTB or of unknown status (population 2) had a lower culture-positive rate of 2.5% (3/121). Estimates of the sensitivity and specificity of the tests for diagnosing bTB exposure and infection in meerkats, calculated using a Newton-Raphson procedure, appear in Table 5. Increasing the sample size and incorporating information from previous test results made little difference to the estimate of culture sensitivity but did increase the estimate of culture specificity from 0.88 to 0.99 (Tables 2, 4). By interpreting the results of RT and MAPIA in parallel, estimates of sensitivity and specificity were produced that were high enough to usefully inform decision-making when determining exposure to bTB in wild meerkats (Table 5).
. Cross-classified results of tests used to classify infection status of 403 wild meerkats in groups with high (population 1) and low (population 2) prevalence of tuberculosis.*

Serologic tests RT (rapid test; lateral flow immunoassay) performed on serum and MAPIA (multiantigen print immunoassay) performed on serum were performed either singly or in parallel with meerkats testing positive on at least 1 of these 2 tests being classified as exposed to tuberculosis. Meerkats testing positive for Mycobacterium bovis on mycobacterial culture of tracheal wash samples were classified as infectious. Culture = mycobacterial culture of tracheal washes; + = positive test result; – = negative test result.
Discussion
The purpose of this research was to determine the accuracy of 3 diagnostic tests in detecting M. bovis infection status using samples collected from anesthetized wild meerkats during a longitudinal study of tuberculosis epidemiology. Bayesian methods were used to estimate test performances in the absence of a gold standard reference test. Although the RT and MAPIA were individually of limited diagnostic use, when used in parallel they were sufficiently sensitive and specific to allow reasonably confident classification of infection status.
MAPIA was found to be highly sensitive (0.90) but of limited specificity (0.48) in detecting antibodies to M. bovis in meerkats (Table 3). Poor specificity may have been due to meerkats being infected with nontuberculous mycobacteria (J.A. Drewe, unpublished observations) or closely related bacteria resulting in a cross-reactive response. 23,42 Seroreactivity of badgers to antigens of environmental mycobacteria was found to be widespread in one study, 40 and it is feasible that a similar process occurs in meerkats. Antibodies to 7 antigens of the M. tuberculosis-complex (MPB83, 38 kDa protein, ESAT-6, CFP10, MPB70, Acr1, and fusion protein CFP10/ESAT-6) were detected only in meerkats culturing positive for M. bovis (Table 1) suggesting that a serologic test examining for antibodies to only these proteins may be of higher specificity than the current test based on a wider range of antibody responses.
. Maximum-likelihood estimates (95% confidence interval) of the sensitivity, specificity, and associated likelihood ratios of tests used to classify the infection status of 240 meerkats.*
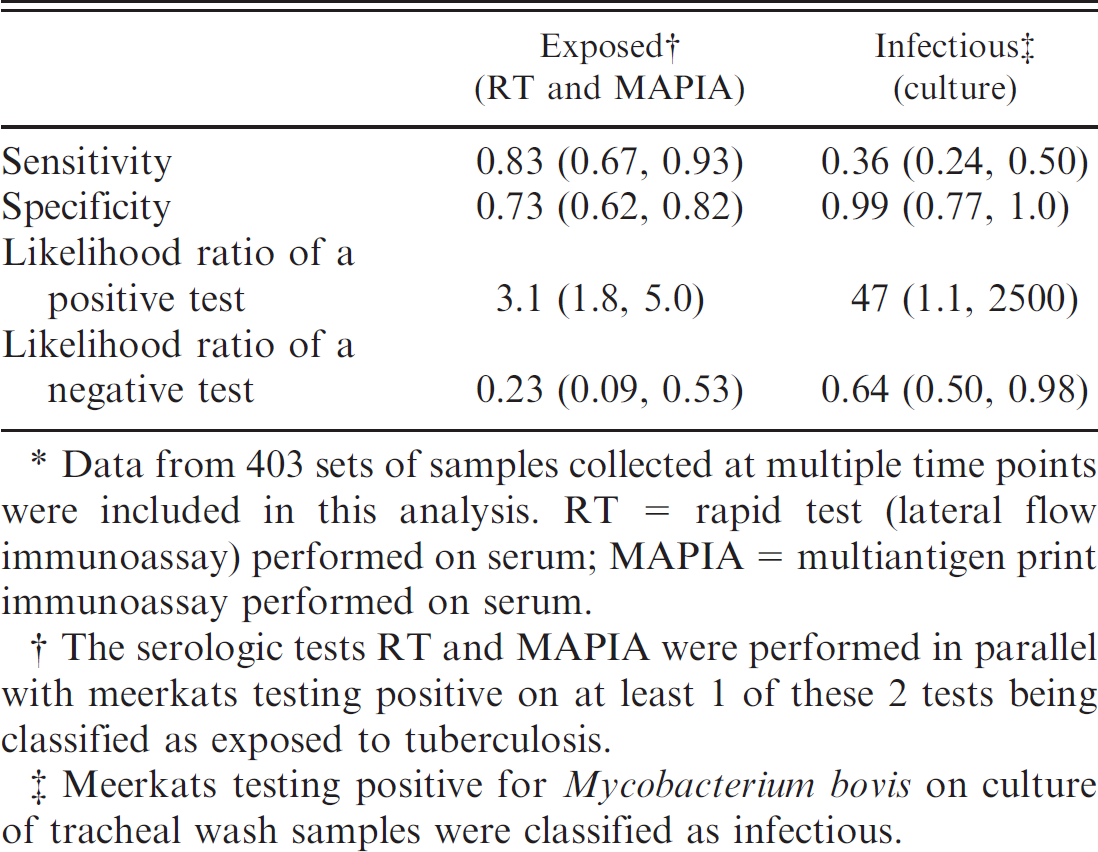
Data from 403 sets of samples collected at multiple time points were included in this analysis. RT = rapid test (lateral flow immunoassay) performed on serum; MAPIA = multiantigen print immunoassay performed on serum.
The serologic tests RT and MAPIA were performed in parallel with meerkats testing positive on at least 1 of these 2 tests being classified as exposed to tuberculosis.
Meerkats testing positive for Mycobacterium bovis on culture of tracheal wash samples were classified as infectious.
Immune responses to mycobacterial infections in humans 2 and animals 31,43 have been shown to vary depending on stage of infection, with differential expression of antigens occurring over time and with severity of infection. 17 Magnitude of humoral immune response was associated with severity of disease due to M. bovis infection in a study of racing dromedary camels (Camelus dromedarius), 43 although because of the small sample size of infected camels (n = 3), further observations are needed to validate this observation. Progression in antibody production to different antigens was noted in captive Asian elephants (Elephas maximus) infected with M. tuberculosis and tested using MAPIA. 31 However, in the current study of meerkats, progression in reactivity to different antigens over time was not observed consistently enough to be a useful determinant of the stage of infection with M. bovis. This may indicate a difference in immune response in this species or it may be that the meerkats tested were not at sufficiently diverse stages of infection for detecting such an effect.
Although MBCF was the antigen most commonly recognized by tuberculous meerkats, it is unlikely to be of practical diagnostic use because of its very low specificity of 0.35 (eliciting a response in 13/20 culture-negative meerkats; Table 1). The most sensitive and specific antigen, and therefore the one of most diagnostic use, was fusion protein Acr/MPB83. A response to this antigen occurred in 78% (29/37) of culture-positive and 5% (1/20) of culture-negative animals (Table 1), representing a sensitivity of 0.78 and specificity of 0.95. This broadly reflects the findings of a study of reindeer (Rangifer tarandus) infected with M. bovis in which Acr/MPB83 was 1 of 4 antigens recognized in 100% (11/11) of infected animals, although few disease-free animals were tested. 42 In cattle, MPB70 and MPB83 proteins are predominantly reactive, 21 whereas ESAT-6 is the serodominant antigen recognized by elephants 31 and MPB83 is the most immunodominant antigen in tuberculous badgers. 28 It thus appears that antigen recognition in tuberculous animals varies between species, and species-specific diagnostic tests may need to be developed to reflect this in order to be of practical diagnostic value in the field.
The RT showed reasonably high specificity (0.85) but low sensitivity (0.43) in detecting antibodies to M. bovis in meerkats (Table 3). These results broadly agree with RT performance in a study of 1,464 badgers where the test was 93% specific and 49% sensitive against culture for M. bovis at necropsy. 8 Low sensitivity appears to be a common feature of serologic tests for M. bovis infection in many species, including badgers, 22 possums (Trichosurus vulpecula), 7 and cattle. 44 Detection of early stages of infection (before onset of clinical signs) is particularly difficult because of an inverse relationship between the courses of cell-mediated and humoral immunity, the latter being preferentially detected in cases of advanced disease. 38 Although the results of RT and MAPIA showed only fair agreement beyond that which would be expected by chance alone (K = 0.26), agreement in positive test results of meerkats sampled multiple times tended to occur in those samples collected closest to death from bTB. This suggests that agreement between RT and MAPIA was occurring during the late stages of disease during the period associated with enhanced humoral immunity. 38 Despite the low sensitivity of culture (Table 3), only 40% (10/25) of culture-positive meerkats also tested positive using RT (Table 2), limiting the usefulness of RT as a sole test for exposure to M. bovis in wild meerkats.
Although sensitivity of the RT in the current study was improved by parallel testing alongside MAPIA (Table 5), sensitivity of serologic tests may also be increased through interpretation at the group rather than the individual animal level. In a field trial of the “Brock” test enzyme-linked immunosorbent assay in 1,982 badgers, a sensitivity of only 0.41 was achieved at the individual level but this rose to 0.73 at the population level. 10 In the current study, interpreting serologic results at the social group rather than at the individual meerkat level considerably increased RT sensitivity from 0.43 to 0.91 (meerkats from 10/11 culture-positive groups tested positive with RT). However, a trade-off for this increased sensitivity was a marked loss of precision in estimates of prevalence of infection.
Culture of tracheal washes collected from anesthetized live meerkats was found to be highly specific (0.99) but of low sensitivity (0.36) for diagnosing individual meerkats shedding M. bovis (Table 5). A similarly low sensitivity of tracheal wash culture as a diagnostic tool for tuberculosis was reported in a study of possums, 25 in which only 36% (9/25) of tracheal washes collected from tuberculous animals cultured positive for M. bovis. Low sensitivity in the current study may be due to limited pulmonary excretion of mycobacteria from tuberculous meerkats. Although excretion via the respiratory tract appears to occur frequently in meerkats, other routes such as suppurating skin wounds have been found to be important in some. 18 Furthermore, mycobacteria may be shed intermittently from tuberculous animals. 11,34 Other factors that may have contributed to the low tracheal wash culture sensitivity in the current study include imperfect sampling technique (tracheal washes may have sampled only airways rather than lung tissue), temperature fluctuations during storage and transport, small sample volume, and loss of viable mycobacteria during the decontamination stage. 13 Despite the low degree of confidence in a negative tracheal wash culture result (likelihood ratio of a negative test = 0.64; Table 5), the longitudinal nature of the study with repeated sampling of the same meerkats served to simultaneously improve chances of detection of infection and increase confidence in a negative result if individual animals repeatedly tested negative.
The use of RT and MAPIA in parallel in this study produced estimates of sensitivity and specificity that were considered high enough to usefully determine exposure to bTB in wild meerkats (Table 5). Interpretation of parallel testing was based against performance of RT when used singly, as MAPIA was not performed on every meerkat because of practical limitations. The increase in sensitivity (from 0.43 when RT was evaluated singly to 0.83 when RT and MAPIA were evaluated in parallel) was offset to some extent by the reduction in specificity (from 0.85 when RT was evaluated singly to 0.73 with parallel testing). The likelihood ratio of a positive test result of 3.1 indicated that a positive serologic result was more than 3 times as likely to be found in an infected meerkat as opposed to one without bTB. Conversely, the likelihood ratio of a negative test result of 0.23 suggested that a negative serologic test result was likely to be found just under a quarter as often in an infected meerkat as opposed to one without bTB. The reciprocal of this, being more intuitive, has been cited as the true likelihood ratio of a negative test. 3 In this case, 1/0.23 = 4.4 (i.e., a negative serologic test result was more than 4 times as likely to occur in an uninfected meerkat as opposed to one with bTB). The use of RT and MAPIA in parallel thus allowed a diagnosis of bTB exposure status to be made on an individual animal basis with a certainty of between 75% and 81%.
The sensitivity and specificity of tests quoted in the current article relate to the study population of wild meerkats in the southern Kalahari. An element of caution is necessary if the same tests were to be applied to populations of meerkats in other settings, such as zoos, where the course of infection may vary and tests may perform differently. Although diagnostic test sensitivity and specificity are unlikely to change with disease prevalence, 9 variation in test performance may be expected in the presence of concurrent infections, 26 altered immune response due to malnutrition, or genetic differences. Differences in sampling technique and storage and processing of samples may also be expected to affect performance of diagnostic tests. 36 Because of this potential for variation, the most practical value of test performance calculations is likely to come from consideration of likelihood ratios. Since the likelihood ratios of positive and negative results are derived from test sensitivity and specificity only, they are unaffected by disease prevalence and are therefore stable expressions of test performance. 39 When used in conjunction with disease prevalence (the pretest probability of disease), likelihood ratios can be used to estimate the post-test probability of disease given a positive or negative test result. 20 For example, in the current study the apparent prevalence of exposure to M. bovis was estimated to be 23% during a cross-sectional survey of 154 meerkats (using the RT and MAPIA in parallel). Applying the likelihood ratios in Table 5 to Fagan's nomogram 20 revealed the post-test probability of disease in a meerkat testing positive to be approximately 60%, whereas the chances of a meerkat being disease-free after a negative test were around 95%. Far more confidence could be thus ascribed to negative result, and the main value of these serologic tests therefore appears to lie in ruling out disease.
In conclusion, MAPIA, RT, and mycobacterial culture of tracheal washes all provided valuable information to allow reasonably confident classification of M. bovis infection status in free-living wild meerkats. All 3 tests exhibited diagnostic limitations when used independently, expressed either as low sensitivity (culture and RT) or limited specificity (MAPIA). The longitudinal nature of the study with repeated sampling of the same individual meerkats served to improve diagnostic accuracy of culture, especially in meerkats repeatedly testing negative. Although MAPIA and RT were individually of limited diagnostic use, interpreting the results of these 2 tests in parallel produced estimates of sensitivity and specificity that were high enough to usefully inform decision making when determining exposure to bTB in wild meerkats and potentially other species in which bTB poses a diagnostic challenge.
Acknowledgements
Permission to work in the Kalahari was granted by the owners and managers of the Reserve and surrounding farms, Northern Cape Nature Conservation Service and Tim Clutton-Brock. Rob Sutcliffe provided invaluable daily assistance with anesthesia and sampling of meerkats. Tom Flower, Dave Bell, and several Meerkat Project volunteers helped collect additional samples. Tiny Hlokwe and Annah Macholo at Onderstepoort Veterinary Institute facilitated mycobacterial culture. Martin Haupt of the University of Pretoria supplied logistical support. Konstantin Lyaschenko at Chembio Diagnostics Systems kindly supplied the MAPIA strips. Martin Vordermeier at the Veterinary Laboratories Agency provided the RT devices. T. J. McKinley at Cambridge Infectious Diseases Consortium (CIDC) offered statistical advice. Main funding was provided by the Department for Environment, Food and Rural Affairs (defra) and the Higher Education Funding Council for England (HEFCE) through CIDC. Subsidiary funding came from the Jowett Fund (UK) and Northern Cape Department of Agriculture and Land Reform (South Africa).
Footnotes
a.
Isofor, Safe Line Pharmaceuticals, Johannesburg, South Africa.
b.
MAPIA, Chembio Diagnostic Systems, New York, NY.
c.
Gibco, Paisley, UK.
d.
Sigma-Aldrich, Gillingham, UK.
e.
Rockland, Gilbertsville, PA.
f.
BovidTB STAT-PAK, Chembio Diagnostic Systems, New York, NY.
