Abstract
Three assays were evaluated for their ability to detect antibodies against Porcine reproductive and respiratory syndrome virus (PRRSV) in porcine muscle transudate (“meat juice”) samples. Samples were derived from 91 pigs inoculated with PRRSV isolate VR-2332 and 46 age-matched controls. Serum and muscle (Musculus longissimus dorsi) samples were collected from randomly selected animals euthanized at ∼14-day intervals from 28 to 202 days postinoculation. Serum samples were assayed at a dilution of 1:40, and muscle transudate samples were assayed at 5 dilutions (1:2, 1:5, 1:10, 1:20, 1:40) using a commercial PRRSV antibody enzyme-linked immunosorbent assay (ELISA). In addition, muscle transudate samples were tested using an indirect fluorescent antibody test (IFAT) at 5 dilutions (1:2, 1:5, 1:10, 1:20, 1:40). Attempts to assay muscle transudate samples for neutralizing antibodies using a modified fluorescent focus neutralization assay were unsuccessful. Receiver operator characteristic (ROC) curve analyses were used to estimate cutoff thresholds and the associated diagnostic sensitivities and specificities for ELISA and IFAT at each dilution. For ELISA, muscle transudate samples at the ROC-optimized cutoffs were >95% sensitive and 100% specific at each dilution. At a cutoff dilution of ≥1:5, the IFAT diagnostic sensitivity and specificity of muscle transudate was estimated at 63.3% and 100%, respectively. These findings validated the use of muscle transudate samples in PRRSV surveillance programs based on ELISA antibody testing.
Keywords
Introduction
Unrecognized prior to the 1980s (Keffaber KK: 1989, Reproductive failure of unknown etiology. Am Assoc Swine Pract Newsletter 1:1-10), Porcine reproductive and respiratory syndrome virus (PRRSV; order Nidovirales, family Arteriviridae, genus Arterivirus) had become endemic by the mid-1990s in most areas of the world where swine are raised, and it is currently the most costly infectious disease confronting U.S. swine producers. 19 , 25 Despite advances in basic and applied research, the control of clinical losses on farms and prevention of the spread of PRRSV between farms has been problematic. 11 Faced with ongoing PRRS losses, the general consensus in North America is that the elimination of PRRSV is the best long-term solution. 3
Successful disease elimination programs are based on accurate, cost-effective, and timely detection of infected animals and herds. Historically, such efforts have relied on down-the-road or on-farm serum testing of individual animals. More recently, muscle transudate (“meat juice”) samples (i.e., fluid recovered from frozen muscle as it thaws) have proven to be a useful alternative to serum in epidemiological studies and surveillance for infectious agents. 2 , 14,16 , Muscle samples are easily collected at slaughter, less costly to collect than serum, and completely avoid the biosecurity risks that on-farm visits present. The performance parameters of diagnostic assays (i.e., diagnostic sensitivity and specificity) are critical to the accurate interpretation of test results. Diagnostic performance has not been reported for PRRSV antibody assays testing muscle transudates. Therefore, the objective of this study was to estimate the diagnostic sensitivity and specificity of a commercial PRRSV enzyme-linked immunosorbent assay (ELISA), an indirect fluorescent antibody test (IFAT), and a modified fluorescent focus neutralization (FFN) assay. Tests were evaluated using muscle transudate samples collected from animals of known PRRSV status in a longitudinal experimental study.
Materials and methods
Experimental design, animal care, and animal-handling procedures were approved by the Biosafety (#04-I-028-A) and Animal Care and Use (#9-04-5751-S and #7-05-5933-S) Committees at Iowa State University. The project was conducted according to Guide for the Care and Use of Agricultural Animals in Agricultural Research and Teaching. 9
Fourteen-day-old (n = 165) PRRSV-negative, healthy, Large White/Landrace cross piglets (50% barrows and 50% gilts) were received at the Iowa State University Livestock Infectious Disease Isolation Facility (Ames, IA) 1 week prior to the beginning of the experiment. Pigs were derived from 17 litters farrowed on day 162 of a 1,000-day production calendar. Individual age-matched pigs were randomly assigned to treatments by blindly selecting ear tags (pigs were ear tagged upon arrival). Tag numbers 1 to 109 (n = 109) were assigned to the PRRSV-inoculated group; numbers 110 to 165 (n = 56) were assigned to the negative control group. Pigs were divided equally by gender between the infected and control groups. Even-numbered tags were assigned to males, and odd-numbered tags were assigned to females.
On day 0, blood samples were collected, and pigs were intramuscularly (IM) inoculated in the neck just behind and below the ear with 1 ml of PRRSV (1 × 104.0 TCID50) diluted in minimum essential medium (MEM). a Negative-control pigs were sham inoculated with MEM by the same route. Following inoculation, animals were euthanized at approximately 14-day intervals for up to 202 days postinoculation (DPI). Five pigs (3 PRRSV inoculated; 2 negative control) were euthanized at each sampling point from DPI 28 through DPI 112; thereafter, through DPI 202, approximately 15 pigs (10 PRRSV inoculated; 5 negative control) were euthanized per sampling. Serum and muscle (Musculus longissimus dorsi) samples were collected from each pig at the time of euthanasia. Among the 165 animals, 28 pigs were excluded from the current study because they were too small to provide adequate-sized muscle samples (i.e., prior to DPI 28), died unexpectedly, or were among a group that was rechallenged with PRRSV on DPI 193. In total, paired serum and muscle transudate samples were available from 137 pigs. This subset consisted of 91 PRRSV-infected pigs and 46 negative-control pigs.
Porcine reproductive and respiratory syndrome virus
Virus strain. The North American prototype PRRSV, American Type Culture Collection [ATCC] VR-2332, b was used in the current study. The specific virus isolate used was derived from a plaque-cloned virus recovered from the serum of a pig inoculated with VR-2332. 5
Virus propagation. To avoid the effects of cell passage, virus was inoculated into a 21-day-old pig. On DPI 7, the pig was euthanized and the serum harvested. For virus propagation, serum was inoculated onto 24-hr-old confluent MARC-145 cells, a clone of the MA-104 cell line considered to be highly permissive to North American (type 2) PRRS viruses. 12 MARC-145 cells were prepared in 162-cm 2 flasks c containing MEM growth medium: MEM a supplemented with 10% fetal bovine serum (FBS), a 50 μg/ ml of gentamicin, a 100 IU/ml of penicillin, a 100 mg/ml of streptomycin, a and 0.25 μg/ml of amphotericin B. a After 24 hr at 37°C in a humidified 5% CO2 incubator, the MEM growth medium was discarded and the flasks inoculated with 5 ml of serum. After 2 hr at 37°C in a humidified 5% CO2 incubator, the inoculum was discarded and 50 ml of maintenance medium was added: 50 ml of MEM supplemented with 5% FBS, 50 μg/ml of gentamicin, 100 IU/ml of penicillin, 100 μg/ml of streptomycin, and 0.25 μg/ml of amphotericin B. Thereafter, cells were examined daily for cytopathic effect (CPE). When 75% CPE was observed, the flask was freeze thawed (-80°C/25°C), and cell lysates were harvested and stored at −80°C.
Virus titration. Virus titrations were performed on confluent monolayers of MARC-145 cells in 96-well plates. c Cell monolayers were prepared by adding 200 μl of a solution containing 4 × 10 5 cells per ml suspended in MEM growth medium to each well, after which plates were placed for 24 hr in a 37°C, humidified, 5% CO2 incubator. Each sample was serially diluted 10-fold in MEM, and then 4 wells were inoculated with 100 μl of stock virus at each dilution. Thereafter, plates were incubated at 37°C in a humidified 5% CO2 incubator for 2 hr, the inoculum was discarded, and 200 μl of maintenance medium was added to each well. Wells were examined for CPE for up to 2 DPI. At the end of 2 days, the cells were fixed with aqueous 80% acetone solution and stained with fluorescein isothiocyanate-conjugated monoclonal antibody SDOW17-F. d
Euthanasia protocol
Pigs to be euthanized were first IM administered (1.0 ml per 22.5 kg of body weight) a solution composed of 250 mg of tiletamine and 250 mg of zolazepam e reconstituted with 2.5 ml of xylazine f (100 mg/ml) and 2.5 ml of ketamine e (100 mg/ml). When recumbent, pigs were intravenously administered (1.0 ml/kg of body weight) a solution containing 0.2 mg of butorphanol, e 2.0 mg of xylazine, g and 2.0 mg of ketamine. e When a surgical plane of anesthesia was reached, pigs were weighed and then exsanguinated.
Samples and sample processing
Serum and muscle tissue were collected from each pig at the time of euthanasia. Blood samples were collected using a single-use blood collection system h and then centrifuged at 1,000 × g for 10 min. Serum was harvested, aliquoted into 2.0-ml cryovials, i and stored at −80°C until tested.
At necropsy, 2 sections (left and right) of the M. longissimus dorsi were collected from the lumbar area. The following measures were taken to eliminate virus cross-contamination of samples at necropsy: 1) negative-control animals were necropsied before inoculated pigs; 2) carcasses were washed with a disinfectant solution j ; 3) necropsy instruments were washed, immersed in methanol, and flamed between each tissue collection; 4) specimens were immediately placed in sterile plastic bags k ; 5) specimens were placed on wet ice immediately after collection, frozen (-20°C), and stored frozen until assayed; and 6) latex gloves were changed between each pig.
While still frozen, the muscle sample from the left side was halved and each half processed separately, resulting in 2 samples per pig. One-half was placed in a sterile plastic bag. The remaining half was minced in a sterile meat grinder l while still frozen and then placed in a plastic bag. Thereafter, intact and ground muscle samples were held at 4°C for 8 to 12 hr, after which the transudate was collected into a 50-ml centrifuge tube. e The pH and quantity of fluid were recorded, and the samples were then clarified (1,000 × g for 10 min), aliquoted into 6 snap-cap tubes, e and stored at −80°C until assayed. For testing, serum and muscle transudate sample tubes were randomized, renumbered, and submitted as 1 set of samples.
Antibody assays
Enzyme-linked immunosorbent assay. Serum and muscle transudate samples were tested for the presence of PRRSV-specific antibodies using the HerdChek PRRS 2XR ELISA m in a laboratory meeting the requirements of ISO 17025 and designated an A2LA accredited provider of PRRSV ELISA testing. g Four serum standards (high positive, medium positive, low positive, and negative) were run on each test plate. Serum standard test results (i.e., ELISA sample-to-positive [S/P] ratios) were analyzed through statistical process control charting to ensure acceptable variation prior to approval of test results. The same PRRS 2XR ELISA test kit lot was used for all samples tested. Serum samples were assayed at a dilution of 1:40, as recommended by the manufacturer. Muscle transudate samples were assayed at 5 dilutions (1:2, 1:5, 1:10, 1:20, 1:40) using buffer solution provided with the ELISA kit as the diluent. ELISA results were expressed as S/P values.
Indirect fluorescent antibody test. The IFAT was performed as previously described. 18 , 23 In brief, MARC-145 cells at a concentration of 4 × 10 5 cells per milliliter (150 μl per well) suspended in MEM h supplemented with 10% heat-inactivated FBS, n were grown in 96-well plates. i Diluted PRRSV (ATCC VR-2332) was added to alternative rows of cells. For the current study, each sample was assayed by a semiquantitative IFAT at 5 dilutions (1:2, 1:5, 1:10, 1:20, 1:40). Staining was performed using fluorescein isothiocyanate-conjugated goat antiswine IgG. The IFAT response was recorded as the highest dilution providing specific cytoplasmic fluorescence. Positive and negative controls were included on each plate.
Fluorescent focus neutralization assay. Porcine reproductive and respiratory syndrome virus-neutralizing activity in serum and muscle transudate samples was measured using a modified FFN assay as previously published. 22 In brief, 2-fold dilutions (1:4-1:512) of heat-inactivated pig serum and muscle transudate samples were tested with the homologous virus (ATCC VR-2332). Neutralizing titer was reported as the last dilution that showed 90% or greater reduction in the number of fluorescent foci.
Statistical analysis
Data were analyzed using MedCalc 9.2.1.0° and Sigma-Plot 10. p Results were expressed in descriptive parameters and analyzed by correlation analysis and analysis of variance, with specific contrasts performed using a 2-tailed Student's t-test.
Receiver operator characteristic (ROC) curve analyses were used to estimate diagnostic sensitivities and diagnostic specificities by cutoff using the cumulative data and without considering the effect of time postinoculation. The ROC analyses were conducted on the basis of known PRRSV infection status of the animals. Test accuracy was assessed on the basis of the estimated area under the curve generated by plotting test sensitivity (y-axis) against 1-specificity (x-axis) for all possible cutoff thresholds. Essentially, the ROC analysis evaluated all possible values of diagnostic sensitivity and specificity and identified the optimized cutoff, that is, the point at which both sensitivity and specificity were maximized. In addition, specific cutoffs and their effect on sensitivity and specificity were considered in the analysis.
To evaluate the effect of time on diagnostic sensitivity, the estimated diagnostic sensitivity for each DPI was modeled by moving average (3 data points) in a 5-parameter logistic model with a derivation of the equation expressed as y = d + a − d/[1 + (x/c) b ] g , where a is the estimated response at zero DPI, b is the slope, c is the midrange DPI, d is the estimated response at infinite DPI, and g is asymmetry.
Thereafter, diagnostic sensitivity and specificity estimates from the 5-parameter logistic model were used to estimate the probability of correctly classifying a herd as PRRSV negative or positive over a range of population prevalences (0-40%) and for a range of ELISA S/P cutoffs (0.1, 0.2, 0.3, 0.4) using a herd sample size of either 5 or 10 muscle transudate samples. Calculations were performed using HERDACC version 3 software q and were based on a theoretical population of 1,000 animals. Positive herds were assumed to have become infected with PRRSV approximately 98 days earlier.
Results
Blood was collected from all pigs upon arrival to the Iowa State University animal holding facilities and prior to PRRSV inoculation on DPI 0. Porcine reproductive and respiratory syndrome virus-negative status was confirmed by quantitative reverse transcription polymerase chain reaction (qRT-PCR) on these samples. All PRRSV-inoculated pigs were confirmed to be productively infected on the basis of positive PRRSV qRT-PCR results on DPI 7 and 14. All negative control animals remained free of PRRSV infection throughout the observation period on the basis of ELISA testing.
Analysis of serum: Porcine reproductive and respiratory syndrome virus enzyme-linked immunosorbent assay (ELISA) and fluorescent focus neutralization (FFN) antibody response by day postinoculation (DPI).*
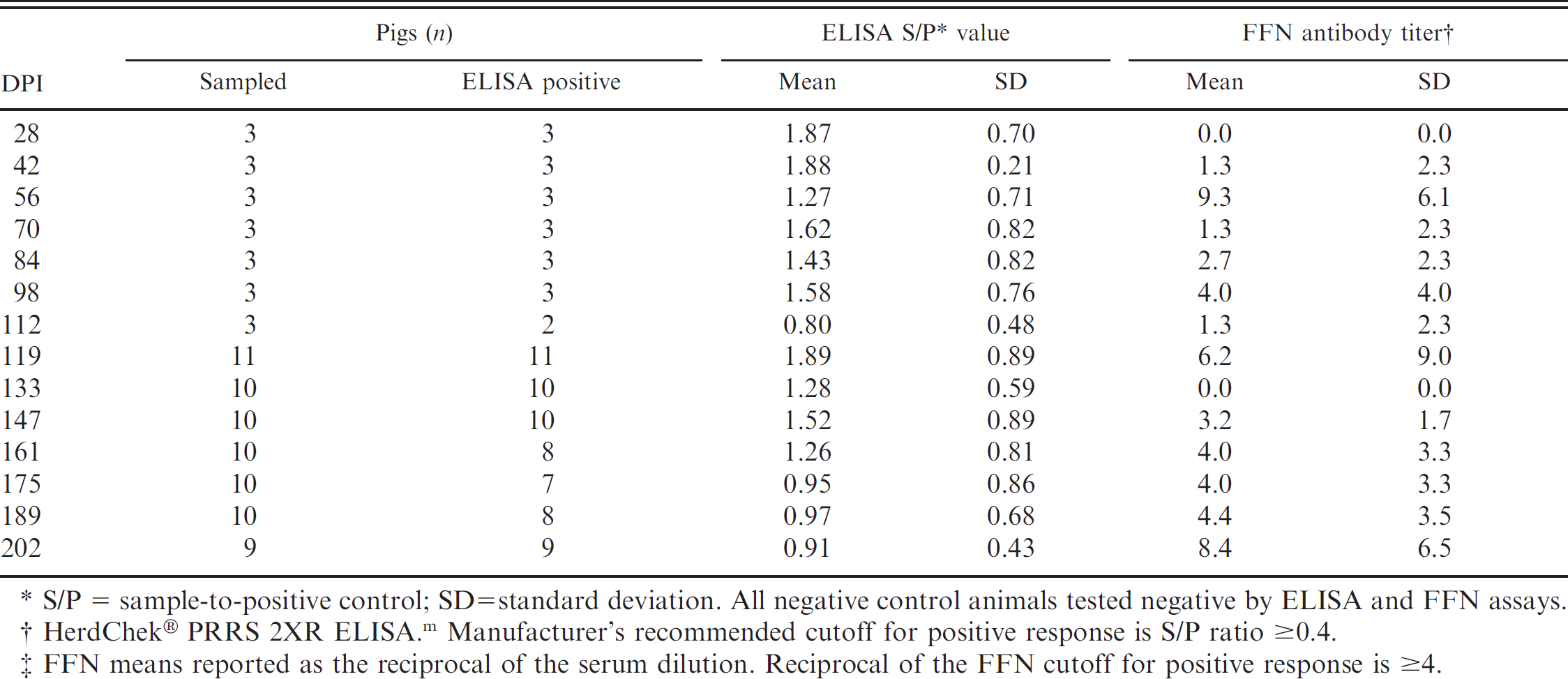
S/P = sample-to-positive control; SD = standard deviation. All negative control animals tested negative by ELISA and FFN assays.
HerdChek® PRRS 2XR ELISA. m Manufacturer's recommended cutoff for positive response is S/P ratio ≥0.4.
FFN means reported as the reciprocal of the serum dilution. Reciprocal of the FFN cutoff for positive response is ≥4.
Serum antibody assays
Serum enzyme-linked immunosorbent assay. Using the manufacturer's recommended cutoff (S/P ≥ 0.4), all pigs were seronegative for PRRSV antibody at DPI 0 (mean S/P = 0.04; range: 0.00-0.23), and all negative-control pigs tested negative throughout the study (mean S/P = 0.02; range: 0.00-0.09). ELISA results for serum samples collected at the time of euthanasia from 91 PRRSV-inoculated animals (DPI 28–202) are reported in Table 1. From DPI 28 to 98, 100% of pigs were ELISA positive, with S/P means ranging from 1.27 to 1.88. From DPI 112 to 189, 87.5% of pigs were positive, with S/P means by DPI ranging from 0.80 to 1.89. At DPI 202, 9 of 9 pigs tested ELISA positive (mean S/P = 0.91). Cumulatively, serum samples from 91.2% (83/91) of PRRSV-inoculated pigs were ELISA positive at euthanasia. The results of the ROC analysis on the cumulative ELISA data are reported in Table 2. Using the manufacturer's recommended cutoff (S/P ≥ 0.4), diagnostic sensitivity and specificity were estimated at 91.2% (95% confidence interval [CI]: 83.4, 96.1) and 100.0% (95% CI: 92.3, 100.0), respectively. Using the infection status of the animals as the basis for the analysis, the ROC analysis determined that the optimized cutoff was S/P ≥ 0.3. At the ROC-optimized cutoff, diagnostic sensitivity and specificity were estimated at 93.3% (95% CI: 86.2, 97.5) and 100.0% (95% CI: 92.3, 100.0), respectively.
Serum fluorescent focus neutralization assay. Serum FFN results are reported in Table 1. Based on a cutoff of ≥ 1:4, all pigs were seronegative for PRRSV at day 0, and all negative-control pigs remained negative throughout the study. Neutralizing antibodies were first detected on DPI 42, with the peak response at DPI 56 (mean FFN antibody titer: 1:9.3). At DPI 202, 8 of 9 pigs tested FFN positive. Overall, PRRSV-specific neutralizing antibodies were detected by FFN in 59.3% (54/91) of inoculated pigs. Analysis of ELISA S/P values versus log2 transformed FFN antibody titers estimated the coefficient of correlation (r)at 0.136.
Analysis of serum and muscle transudate: receiver operating characteristic analysis of cumulative Porcine reproductive and respiratory syndrome virus enzyme-linked immunosorbent assay (ELISA) sample-to-positive control (S/P) data.
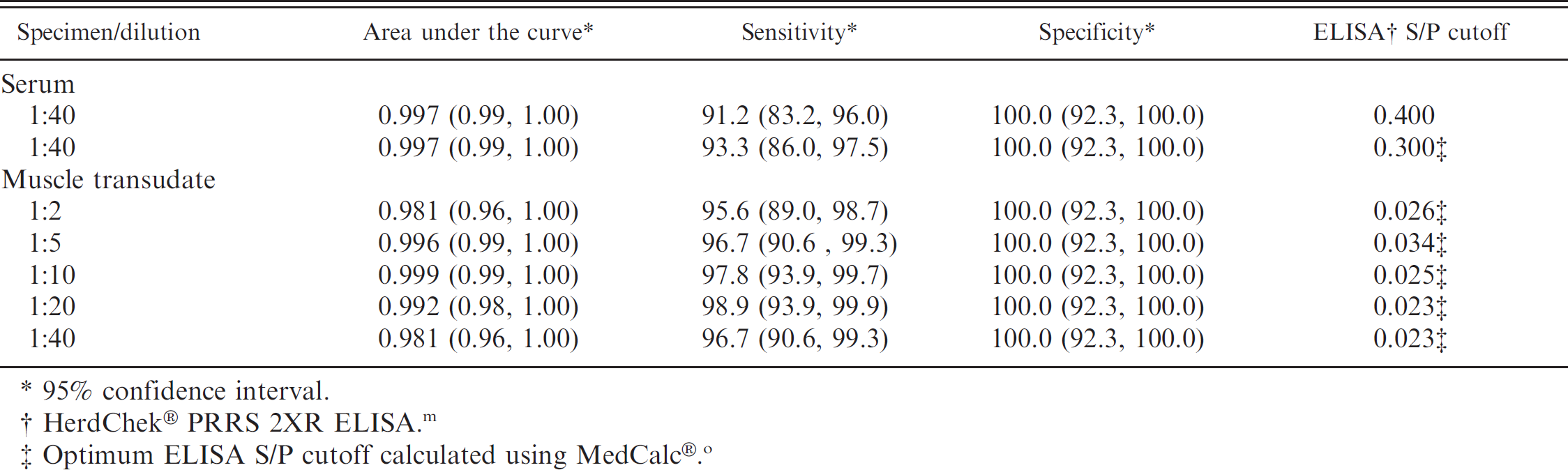
95% confidence interval.
HerdChek® PRRS 2XR ELISA. m
Optimum ELISA S/P cutoff calculated using MedCalc®. o
95% confidence interval (CI).
Diagnostic specificity estimated at 100% (95% CI: 93.6, 100) for all dilutions and cutoffs evaluated.
HerdChek® PRRS 2XR ELISA. m
Muscle transudate antibody assays
Muscle transudate characteristics. M. longissimus dorsi muscle specimens from individual pigs weighed an average of 249 g, with a range by DPI of 74 g (DPI 28) to 465 g (DPI 202). Muscle transudate pH ranged from 6.5 to 7.0 in 133 of 137 (97%) samples, with the exception of 4 samples in which the pH was ≤6.0. The volume of muscle transudate recovered from muscle tissue averaged 10% of the weight of the specimen. Approximately 10% of the transudate samples were untestable at a 1:2 dilution because of coagulation.
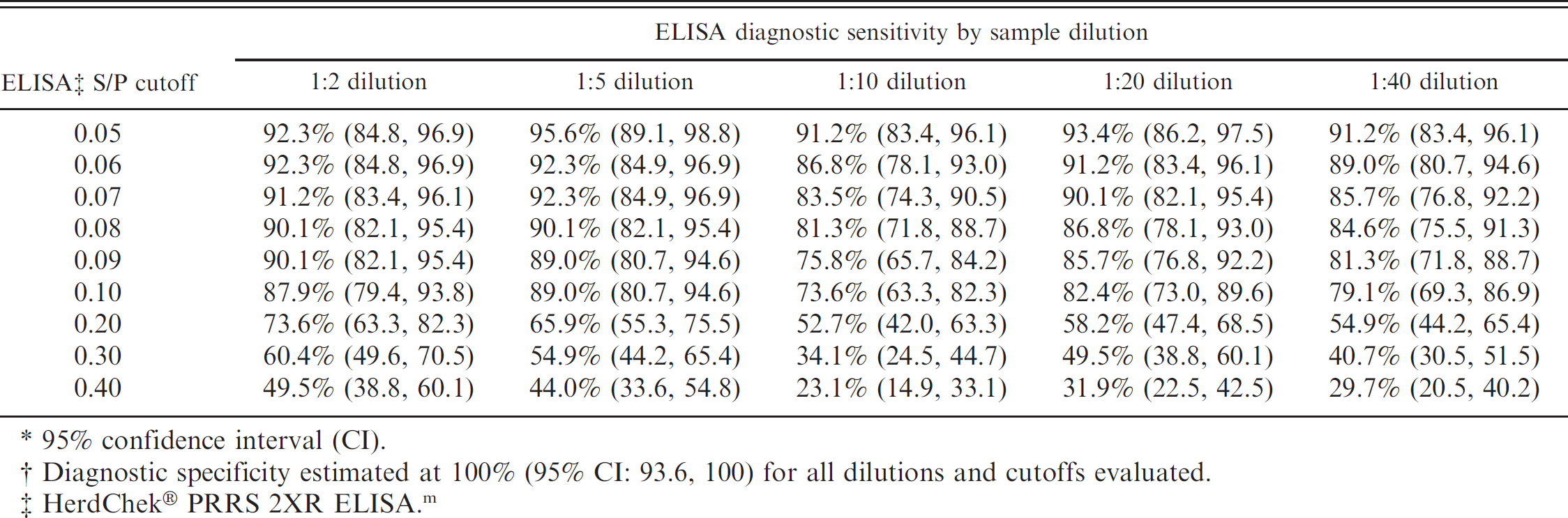
Muscle transudate ELISA results. Transudate from intact and ground muscle specimens were assayed independently by ELISA. The results were compared for each sample dilution (1:2, 1:5, 1:10, 1:20, 1:40) to determine if either protocol produced a larger harvest of ELISA-detectable antibody. Although transudate from ground muscle specimens tended to produce slightly higher ELISA S/P values, no statistically significant difference was detected (paired Student's t-test) in the ELISA S/P values between intact and ground meat specimens using P = 0.05 as the criterion. Because the results were not different, the means of the ground and intact muscle transudate ELISA results were calculated for each pig and used in subsequent statistical analyses. The association between serum ELISA and muscle transudate ELISA values was evaluated by correlation analysis for each muscle transudate dilution. The correlation coefficients (r) ranged from 0.64 (1:2 dilution) to 0.80 (1:5 dilution). The cumulative ELISA diagnostic performance for muscle transudate was evaluated by ROC analysis for each dilution (Table 2). Diagnostic sensitivities for a range of S/P cutoff values (0.05-0.40) is presented for all dilutions (1:2, 1:5, 1:10, 1:20, 1:40) in Table 3. In all cases and for all cutoff values, diagnostic specificity was estimated at 100%.
To evaluate the effect of time postinoculation on ELISA performance, the calculated diagnostic sensitivities of meat transudate dilutions 1:5 and 1:10 were analyzed in a 5-parameter logistic regression model by DPI at 4 S/P cutoffs (Fig. 1). The 1:5 and 1:10 dilutions were selected for analysis because they did not exhibit the sample-handling problems (coagulation) associated with the 1:2 dilution, and they provided a stronger S/P response than other dilutions (1:20, 1:40). As shown in Figure 1, the 1:5 dilution provided better diagnostic sensitivity than the 1:10 dilution for any S/P cutoff. At a 1:5 dilution, the model predicted a diagnostic sensitivity of 96.3%, 86.3%, and 77.8% for S/P cutoffs 0.1, 0.2, and 0.3, respectively, for DPI 28 to 112 (Table 4). The cutoff of 0.4 resulted in an estimated diagnostic sensitivity of 73.5% at DPI 28, with subsequent declines in sensitivity at each sampling point thereafter.
Muscle transudate indirect fluorescent antibody test results. All negative control samples tested <1:5 by IFAT. In the inoculated group, 74.4% of the samples showed specific fluorescence at a dilution of 1:2, 63.3% at a dilution of 1:5, and 43.3% at 1:8. Using PRRSV inoculation to define status, an ROC analysis of the cumulative data estimated the optimized cutoff at ≥1:5, with an associated diagnostic sensitivity of 63.3% (95% CI: 53.0, 73.6) and specificity of 100% (95% CI: 92.3, 100.0). To evaluate the effect of time postinoculation on diagnostic sensitivity, the IFAT diagnostic sensitivity was modeled by moving average in a 5-parameter logistic regression model by DPI. As shown in Table 4, a cutoff of ≥1:5 resulted in an estimated diagnostic sensitivity of 100% at DPI 28, with subsequent declines in sensitivity at each sampling point thereafter.
Muscle transudate fluorescent focus neutralization results. The first step in the FFN assay is heat inactivation of samples. For muscle transudate samples, this step produced total or partial clotting of the samples. In addition, unacceptably high background levels were observed in seronegative samples. Therefore, it was not possible to produce acceptable results with this assay.
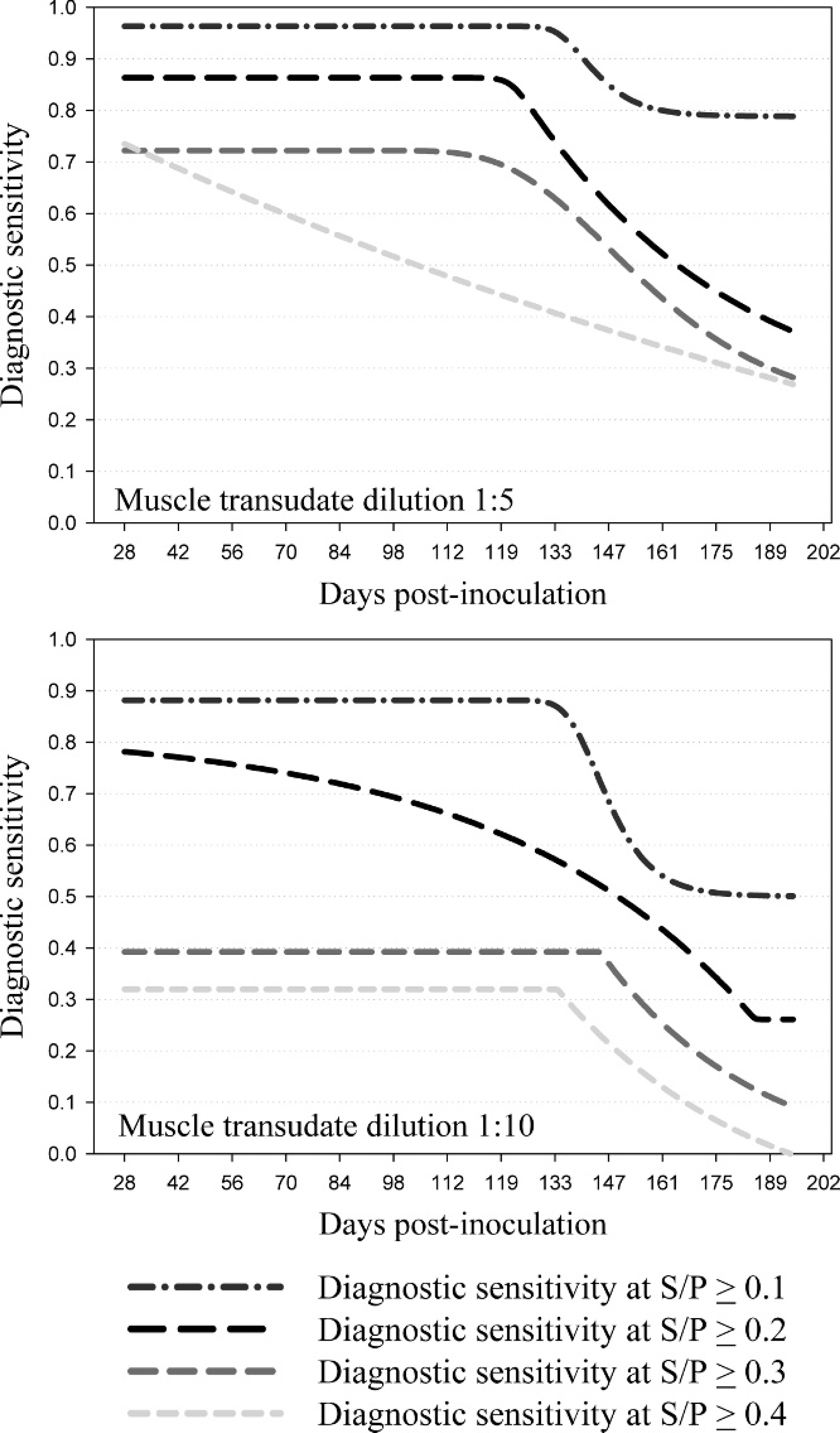
Analysis of muscle transudate: predicted Porcine reproductive and respiratory syndrome virus enzyme-linked immunosorbent assay diagnostic sensitivity by day postinoculation at 4 sample-to-positive control (S/P) cutoff levels as estimated in a 5-parameter logistic regression model.
Analysis of herd-level diagnostic sensitivity and specificity
The probability of classifying a population of 1,000 animals as PRRSV negative or positive using either 5 or 10 muscle transudate samples was calculated using HERDACC version 3 software q for a range of ELISA S/P cutoffs and across a range of in-herd prevalences (0-40%). Positive results at zero prevalence provide an estimate of the probability of false-positive herd classifications for the parameters described. The analysis was based on the estimates derived from this study; that is, the diagnostic sensitivity of the ELISA was assumed to be 96.3%, 86.3%, 77.8%, and 51.7% at S/P cutoffs of 0.1, 0.2, 0.3, and 0.4, respectively, and the diagnostic specificity of ELISA was assumed to be 99.5%. Since independent estimates of the diagnostic specificity of the PRRS ELISA on meat juice were not available, this assumption was based on the sensitivity estimates derived from this study and a presumed misclassification error rate of 5 samples per thousand for true-negative samples. As shown in Table 5, in-herd prevalence, ELISA cutoff, and the number of samples per herd dynamically affect the likelihood of detecting PRRSV-positive herds. Among true PRRSV-negative herds (zero prevalence), 2.5% were misidentified as infected with a sample size of 5 and 4.9% with 10 samples.
Analysis of muscle transudate at 1:5 dilution: predicted Porcine reproductive and respiratory syndrome virus enzyme-linked immunosorbent assay (ELISA) and indirect fluorescent antibody test (IFAT) diagnostic sensitivity by cutoff by day postinoculation (DPI) as estimated in a 5-parameter logistic regression model (%).
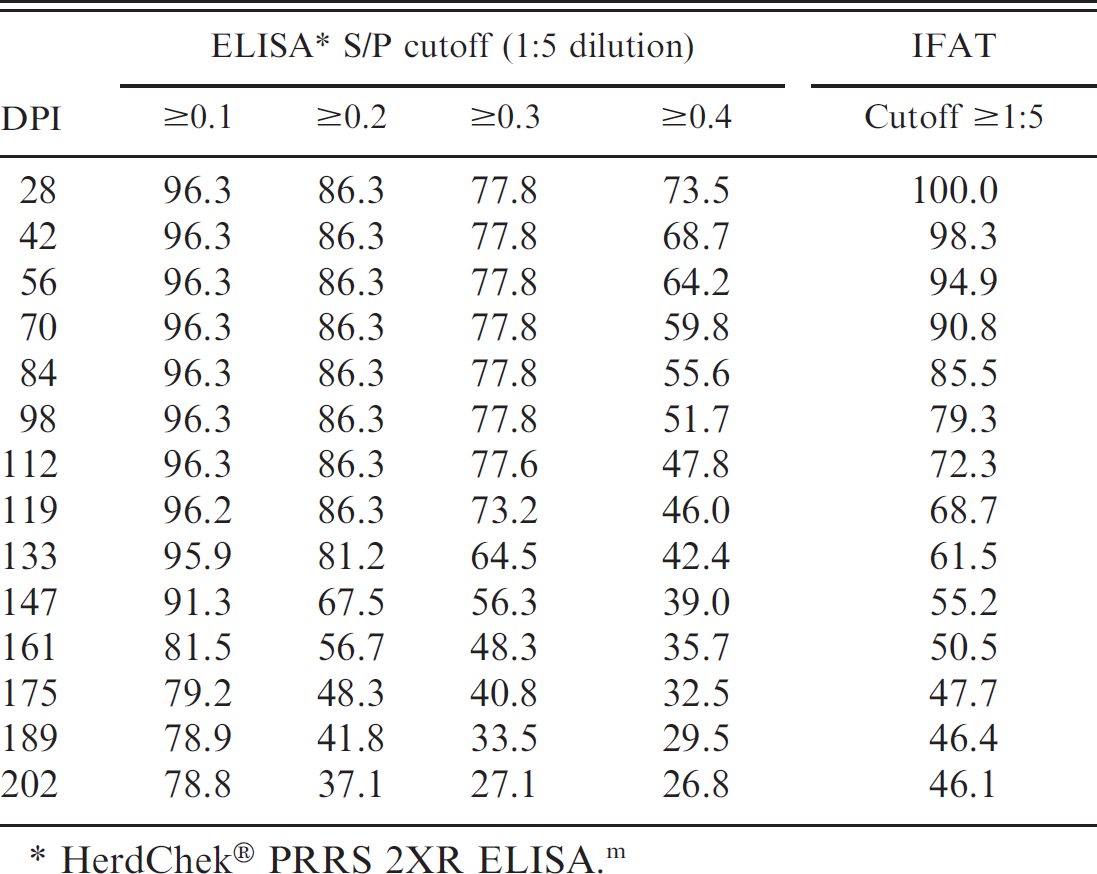
HerdChek® PRRS 2XR ELISA. m
Probability of detecting 1 or more Porcine reproductive and respiratory syndrome virus (PRRSV)-positive samples in a swine population as a function of prevalence, number of muscle transudate samples tested, and enzyme-linked immunosorbent assay (ELISA) cutoff.*
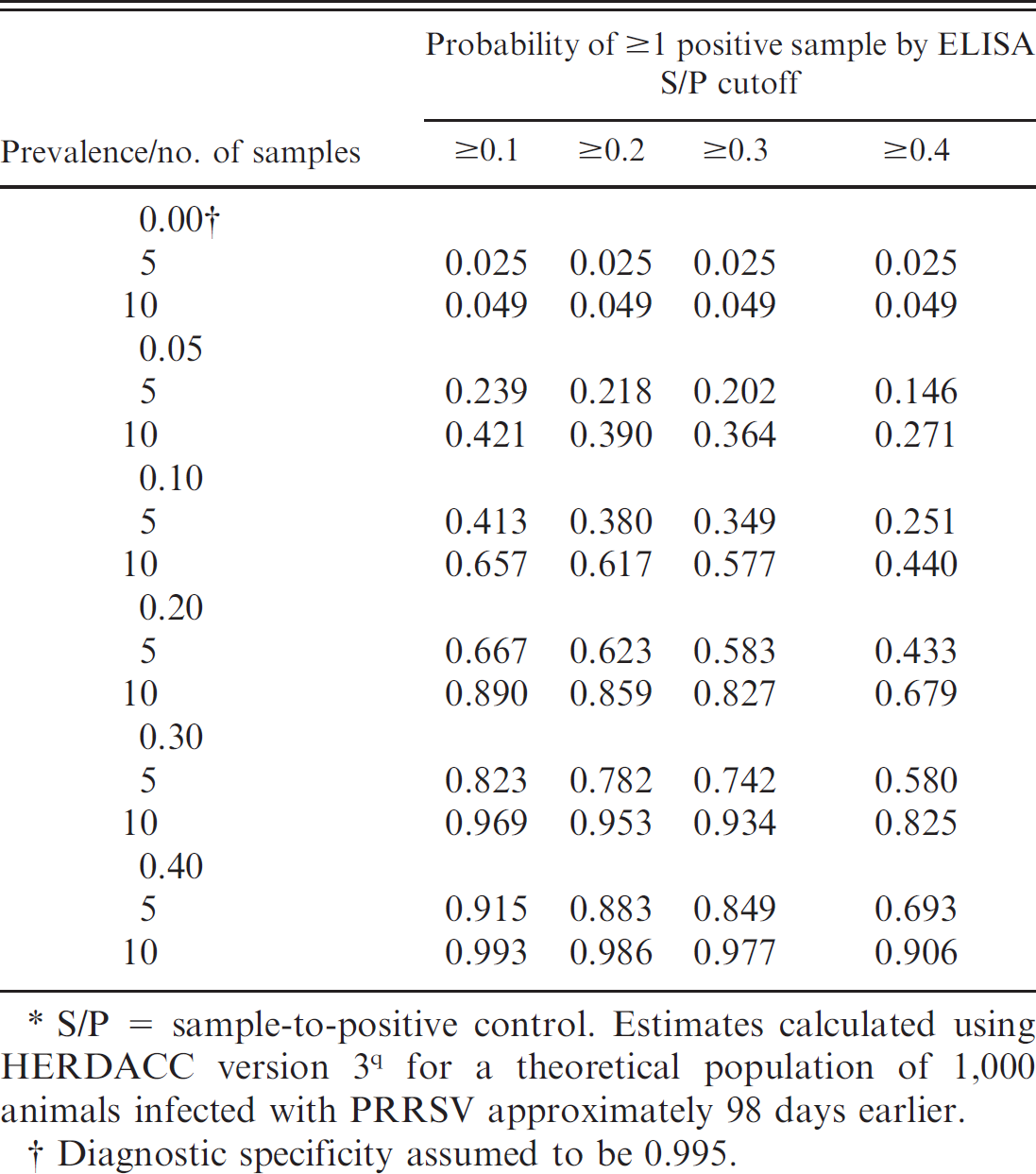
S/P = sample-to-positive control. Estimates calculated using HERDACC version 3 q for a theoretical population of 1,000 animals infected with PRRSV approximately 98 days earlier.
Diagnostic specificity assumed to be 0.995.
Discussion
Muscle transudate (meat juice) is a fluid mixture of lymph, serum, and intracellular liquid. 20 Muscle transudate has been shown to be an effective and practical sample for surveillance of salmonella, 20 , 21 Suid herpesvirus 1 (commonly known as Pseudorabies virus), 14 Yersinia enterocolitic, 21 Trichinella spiralis, 2 and PRRSV. 16 Porcine muscle specimens and herd identifiers are easily collected at slaughter, and transudate is readily recovered by thawing the frozen sample. Diaphragmatic muscle (M. diaphragma crura) is commonly collected for recovery of muscle transudate, 7 , 20 , 21 but M. sterno-mastoideus has also been used. 8 , 16 The possible effect of muscle type on the concentration of total antibody or antibody isotype in transudate has not been evaluated; thus, the selection of muscle is dictated by practical considerations (e.g., the ease with which the specimen may be collected and its value). Musculus longissimus dorsi samples were used in the present study because the large size of the muscle made it possible to recover an adequate volume of muscle transudate from small pigs and because the pigs were precluded from entering the food chain.
In the current study, muscle transudate and serum samples from 91 pigs inoculated with PRRSV isolate VR-2332 and 46 age-matched negative controls were tested in 3 PRRSV antibody assays. Samples were collected from randomly selected animals euthanized at approximately 14-day intervals from 28 to 202 DPI. Serum samples were assayed at a dilution of 1:40, and muscle transudate samples were assayed at 5 dilutions (1:2, 1:5, 1:10, 1:20, 1:40) on a commercial PRRSV ELISA. r Likewise, muscle transudate samples were tested on an IFAT at 5 dilutions (1:2, 1:5, 1:10, 1:20, 1:40). Attempts to assay muscle transudate samples for neutralizing antibodies using a modified FFN assay were unsuccessful.
An ROC analysis of the cumulative serum ELISA data estimated the diagnostic sensitivity and specificity of the test at 91.2% (95% CI: 83.4, 96.1) and 100.0% (95% CI: 92.3, 100.0), respectively. This estimate is somewhat lower than the sensitivity of 97.2% (95% CI: 94.7, 98.7) and specificity of 100% (95% CI: 99.1, 100) previously reported. 10 An explanation for the difference in estimates is that the serum samples used in the present study included a substantial number of specimens from convalescent and chronic stages of the infection (i.e., up to 202 DPI). As reported previously, 1 , 13 , 15 , 24 the serum neutralizing antibody response appears ≥4 weeks following inoculation. In agreementwith previous studies, 1 , 15 the PRRSVFFN assay wasless sensitive than ELISA. Neutralizing antibodies were first detected on DPI 42, with the peak response at DPI 56 (mean FFN antibody titer: 1:9.3). At DPI 202, 8 of 9 pigs tested FFN positive. Overall, PRRSV-specific neutralizing antibodies were detected by FFN in 59.3% (54/91) of inoculated pigs.
The overarching objective of the current study was to arrive at recommendations for testing muscle transudate for the presence of PRRSV antibodies. An underlying assumption of the discussion is that muscle transudate samples will be used in the surveillance of PRRSV infection, not in routine diagnostics. For surveillance, diagnostic specificity is paramount because false-positive responses are costly in terms of both follow-up costs and erosion of producer confidence in the program.
A comparison of results by DPI showed that transudate from ground muscle samples produced slightly higher ELISA S/P ratios than transudate from intact muscle samples, but no statistically significance difference was detected. Thus, these results justify the current practice of collecting transudate from intact muscle samples.
Muscle transudate samples tended to coagulate at the lowest dilution tested (1:2); therefore, it is advisable to use assay samples at dilutions >1:2. For all muscle transudate dilutions tested (1:2, 1:5, 1:10, 1:20, 1:40), ELISA diagnostic specificity approached 100%, suggesting that nonspecific reactions are not the major consideration in selecting the optimal testing dilution. A comparison of ELISA diagnostic sensitivity by DPI showed that the 1:5 dilution was more sensitive over a longer period of time postinoculation than dilutions of 1:10 (Fig. 1), 1:20, or 1:40. Therefore, the 1:5 dilution appears to be the best choice for testing muscle transudates under the conditions described in the current study. For muscle transudate assayed at a dilution of1:5, the ROC-optimized ELISA S/P cutoff of 0.034 resulted in a diagnostic sensitivity of96.7% (Table 2). However, this cutoff value is too close to the S/P value ofzero to accommodate the normal variability in test response expected under routine diagnostic testing conditions. For this reason, diagnosticians may consider alternative ELISA S/P cutoffs of 0.1, 0.2, and 0.3. These cutoffs appear to provide acceptable diagnostic sensitivity when assaying muscle transudate at a 1:5 dilution (Table 4). In addition, the use of higher cutoffs may be justified by the fact that muscle transudate sampling for surveillance is generally done on a population basis. Therefore, the loss of assay sensitivity is offset by the fact that samples are collected repeatedly over time from the same production sites and that multiple samples are taken from each population (Table 5).
The PRRSV IFAT was run on muscle transudate samples because it has been used extensively as a confirmatory test. 13 The IFAT is considered to be highly specific, but diagnostic sensitivity is affected by a variety of factors, including the extent to which the PRRSV isolate used in the assay differs antigenically from the isolate that infected the pig. 6 To control for this source of variability, the homologous virus (ATCC VR-2332) was used in the IFAT. At a cutoff of ≥1:5, the cumulative diagnostic sensitivity of the test was 63.3% and the specificity of the test was 100%. An evaluation on the basis of DPI showed that IFAT is diagnostically sensitive early in the infection but loses sensitivity over time (Table 4). Therefore, in addition to the other factors that affect IFAT performance, time postinfection will be a consideration in the interpretation of IFAT-negative results on meat transudate samples.
Overall, these data support the use of muscle transudate samples collected at slaughter for surveillance and provide performance estimates for using the PRRSV ELISA and IFAT assays described herein. Particularly in the case of the PRRSV ELISA, a stronger S/P response in positive samples would be desirable to improve the ability of the test to discriminate between negative and positive muscle transudate samples. Future research should focus on this objective. For the ELISA, this might be achieved by increasing the incubation time to increase antibody-antigen binding or by diluting the positive control. Both would serve to increase the S/P response in positive samples; the former by increasing the sample response (i.e., ↑ S), and the latter by decreasing the positive control response (i.e., ↑ P). This approach has been used to adapt other ELISAs to samples containing low levels of antibody (e.g., oral fluids). 4
Acknowledgements
This project was funded in part by an Advanced PRRS Research Award provided by Boehringer Ingelheim Vet-medica Inc., St. Joseph, Missouri, and by the National Research Initiative of the USDA Cooperative State Research, Education and Extension Service, grant 2004-35605-14197.
Footnotes
a.
Sigma-Aldrich, St. Louis, MO.
b.
American Type Culture Collection, Manassas, VA.
c.
Corning Inc., Corning, NY.
d.
Rural Technologies Inc., Brookings, SD.
e.
Fort Dodge Animal Health, Fort Dodge, IA.
f.
LLOYD Laboratories, LLOYD Inc., Shenandoah, IA.
g.
Boehringer Ingelheim Vetmedica Inc., Health Management Center, Ames, IA.
h.
Kendall Co., Mansfield, MA.
i.
Fisher Scientific Co., Hanover Park, IL.
j.
Pharmacal Research Laboratories Inc., Naugatuck, CT.
k.
Nasco International Inc., Fort Atkinson, WI.
l.
LEM Products Inc., Harrison, OH.
m.
IDEXX Laboratories Inc., Westbrook, ME.
n.
Atlanta Biologicals Inc., Lawrenceville, GA.
o.
MedCalc® software, Mariakerke, Belgium.
p.
Systat Software Inc., San Jose, CA.
r.
Invitrogen Corp., Carlsbad, CA.