Abstract
A 4-year-old female Appaloosa horse was referred to the Department of Comparative Pathology, Veterinary Faculty, with a history of colic and acute respiratory distress. At necropsy, gross lesions consisted of extensive firm, multinodular, hemorrhagic foci in fat tissues with yellow-brown discoloration. The most affected areas were peritoneal fat and perirenal, epicardial, and subcutaneous adipose tissues. Other findings were hepatic lipidosis and multiple 1–1.5 cm hemorrhagic foci scattered in both lungs. Histopathological examination revealed severe degeneration and necrosis of adipose tissue with dystrophic calcification. The necrotic fat was infiltrated by abundant foamy macrophages and multinucleated giant cells. Based on these histopathological changes, yellow fat disease, also called nutritional panniculitis, was diagnosed. In addition, the microscopic examination of lung and kidney sections stained with osmium tetroxide and oil red O revealed numerous lipid droplets within glomerular and alveolar septal capillaries. To the authors' knowledge, this is the first description of the development of fat embolism in horses affected by yellow fat disease.
The first report of yellow fat disease in 1957 described a disease in foals in New Zealand, characterized by muscular dystrophy and inflammation of the adipose tissue. 9 Yellow fat disease, or nutritional panniculitis, is a generalized disorder of fat deposits, characterized by extensive adipose cell degeneration and inflammation. During these processes, a progressive peroxidation of unsaturated fatty acids may occur. Ceroid pigment, which is responsible for the typical yellow discoloration of fat, is the final product of this peroxidation process. 6
A 4-year-old female Appaloosa horse with a previous history of colic with diarrhea for 8 days and acute respiratory distress was submitted to the Department of Comparative Pathology, Veterinary Faculty, University of Las Palmas de Gran Canaria, Spain for postmortem examination. No blood tests were performed. At necropsy, the animal was obese. The main necropsy lesion was diffuse, firm, multinodular, focally hemorrhagic fat with yellow-brown discoloration (Fig. 1). Most affected were peritoneal, perirenal, epicardial, and subcutaneous fat. Other findings were an abundant reddish serous fluid in the pericardial sac, swollen and yellow appearance of liver that floated in formalin, edema of ventral abdominal wall, and multiple 1–1.5 cm diameter red-black foci in both lungs. Ecchymoses and coalescing to diffuse (suffusive) serosal hemorrhages were observed in several segments of the small intestine (Fig. 2). Tissue samples were fixed in 10% neutral buffered formalin, trimmed, processed routinely, embedded in paraffin, sectioned at 5 μm, and stained with hematoxylin and eosin.
Microscopically, the adipose tissue had severe degeneration and necrosis with dystrophic calcification (saponification of fat). This necrotic fat had abundant foamy macrophages and multinucleated giant cells combined with a slight infiltrate of mononuclear leukocytes (Fig. 3). The kidneys had severe, diffuse medullary and cortical tubular necrosis, abundant deposits of granular eosinophilic pigment (presumed myoglobin) in distal and collecting tubules, slight infiltrate of interstitial lymphocytic, and dilatation of the glomerular capillaries (Fig. 4).
Histological evaluation of lung sections revealed numerous small hemorrhages, scattered along lung, within alveoli and the interstitium. Some of these were subpleural corresponding with the red spots observed grossly (Fig. 5). Other pulmonary lesions consisted of edema, dilatation of the blood vessels, and slight infiltrates of neutrophils. Clear spaces in the lumina of blood vessels and in the intima of pulmonary arteries and arterioles were suggestive of consistent lipid globules. The majority of the alveolar capillaries were dilated. Muscular samples showed severe degenerative changes, such as hydropic degeneration, myofiber fragmentation, and slight infiltrate of neutrophils, in the myofiber and also scattered in the interstitium (Fig. 6). These lesions were found in several locomotor muscles. In the liver, nearly all hepatocytes had moderate to severe lipid vacuolation without necrosis or significant inflammation. In addition, lung, liver, kidney, and adipose tissue samples were prepared with osmium tetroxide (OsO4) and oil red O (ORO) methods to detect fat emboli as described previously. 1 For the ORO method, frozen formalin-fixed tissues were used. Tissue sections were rinsed in distilled water and placed in absolute propylene glycol for 2 min and then stained in OrO solution for 16 hr. Sections were differentiated in 85% propylene glycol solution for 1 min, rinsed in 2 steps of distilled water, stained in Mayer's hematoxylin solution for 15–60 sec, and rinsed again thoroughly in several changes of distilled water for 3 min. Finally, tissues were coverslipped with warm glycerin jelly solution. For the OsO4 method, formalin-fixed tissues were cut no thicker than 2–3 mm to ensure an adequate reagent penetration. After washing in distilled water for 30 min, the tissues were placed in a glass jar with 1% aqueous solution of OsO4 for 2 hr, in continuous agitation. Tissues were then rinsed in running tap water for 30 min, immersed in 0.5% periodic acid until the dark osmicated tissues were uniformly cleared (30 min), and rinsed again in running tap water. The tissues were placed in distilled water for 5 min and then routinely processed in an automatic tissue processor. a Finally, embedded paraffin tissues were cut at 4 μm and stained with hematoxylin and eosin. As a result of these techniques, a high amount of intravascular lipid droplets were observed within veins, capillaries, and small arterioles in the kidney (Fig. 7) and lung sections (Fig. 8). The intravascular lipids appeared as single or multiple, well-defined, red (ORO) and black (OsO4) globules (circular spaces that displaced the erythrocytes in the pulmonary arteries and arterioles, alveolar septal capillaries, and glomerular capillaries).
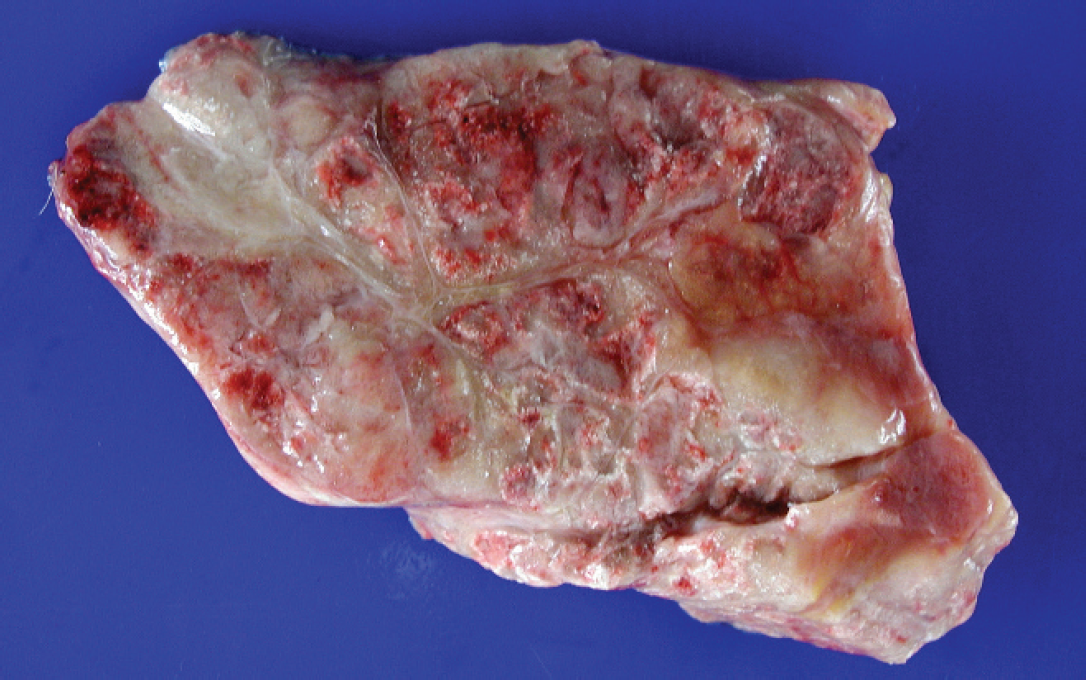
Peritoneal fat. Note the firm and irregular surface of the abdominal fat with multinodular hemorrhagic foci.
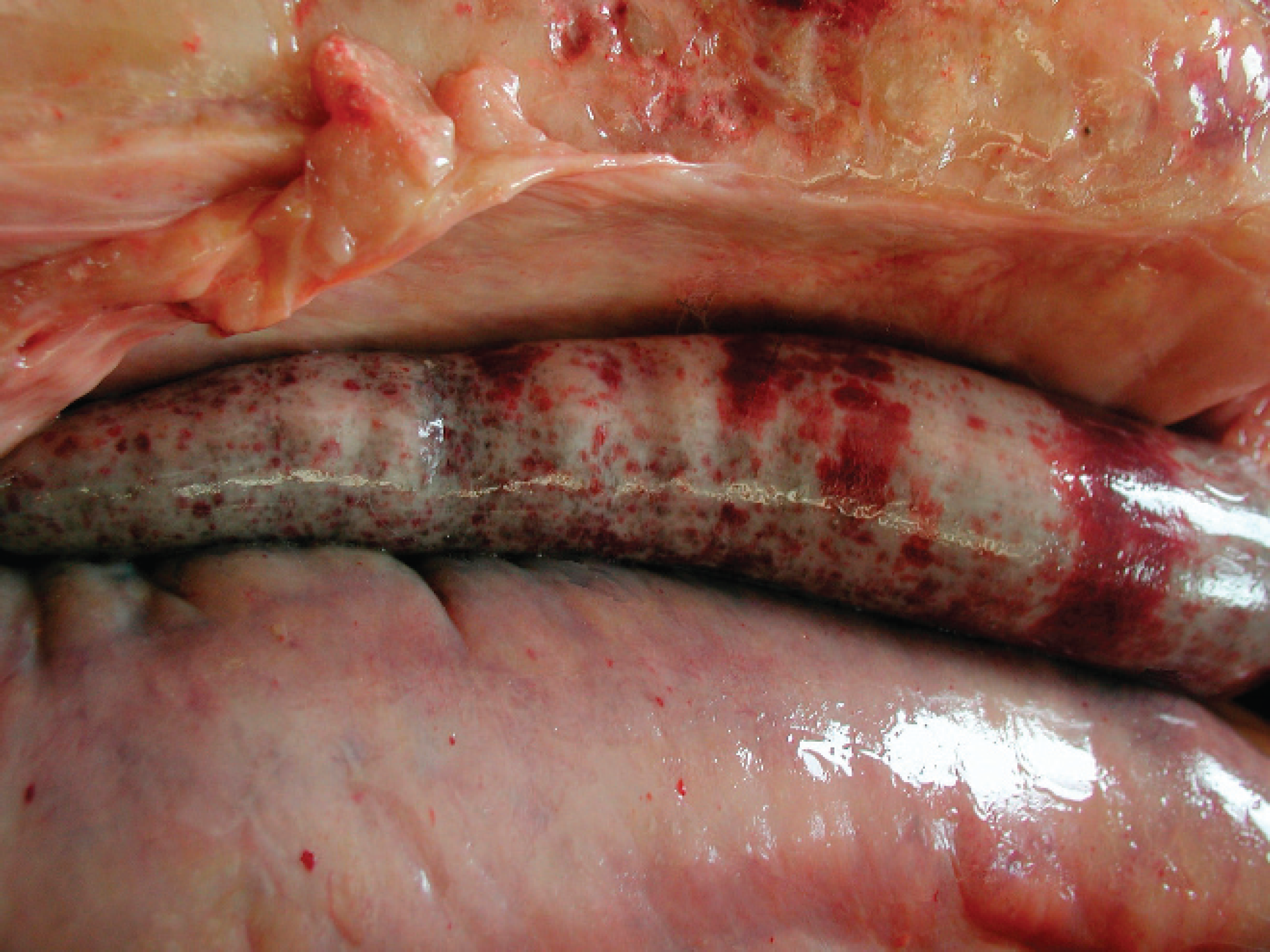
Intestine and abdominal wall with peritoneum. There is coalescing to diffuse serosal hemorrhage of the intestine, which may be due to severe injury to endothelial cells in the capillary beds.
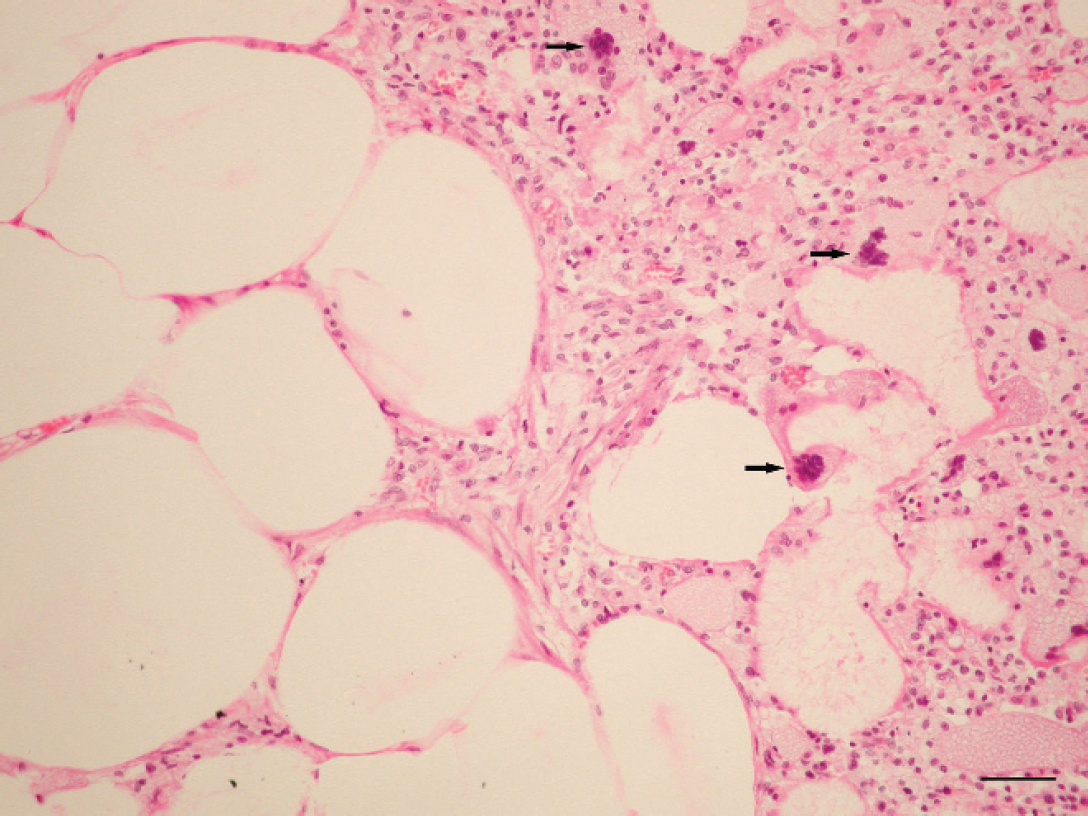
Fat tissue. Note the degeneration and necrosis of adipose tissue with a mixed inflammatory infiltrate with abundant numbers of macrophages and giant cell (arrows). Hematoxylin and eosin. Bar = 50 μm.
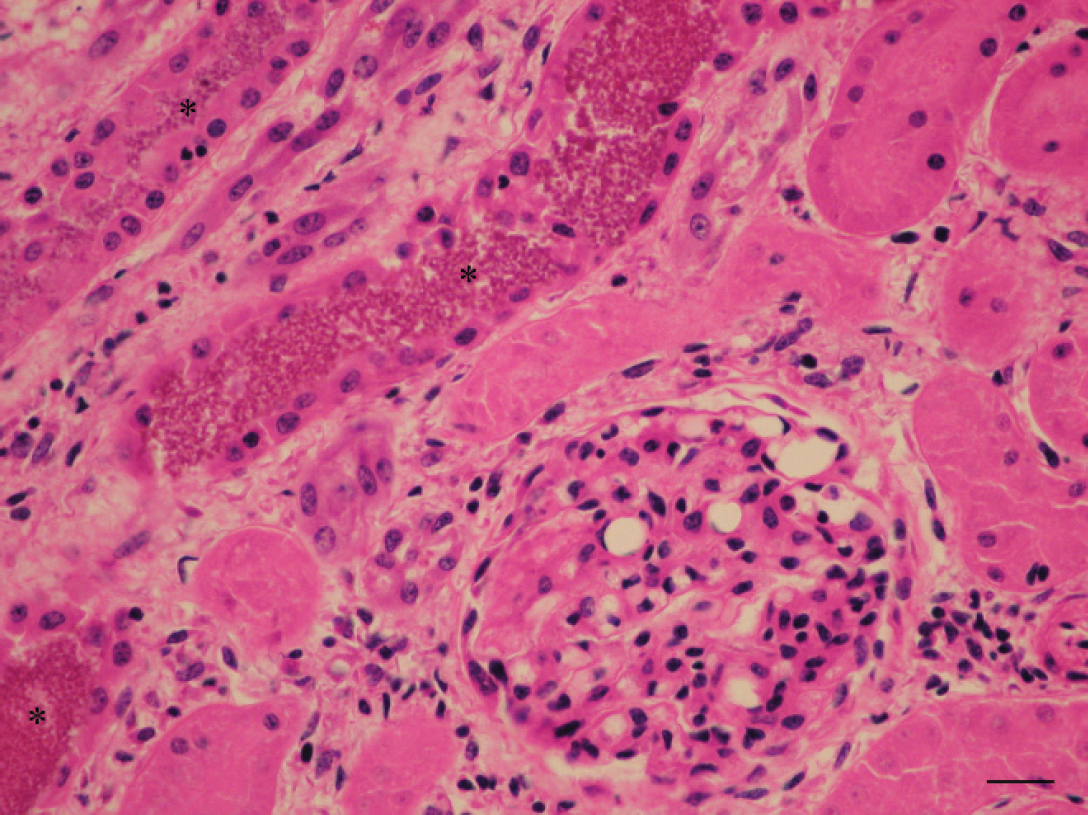
Kidney. Deposits of granular eosinophilic pigment (asterisk), tubular degeneration, and dilatation of the glomerular capillaries. Hematoxylin and eosin. Bar = 25 μm.
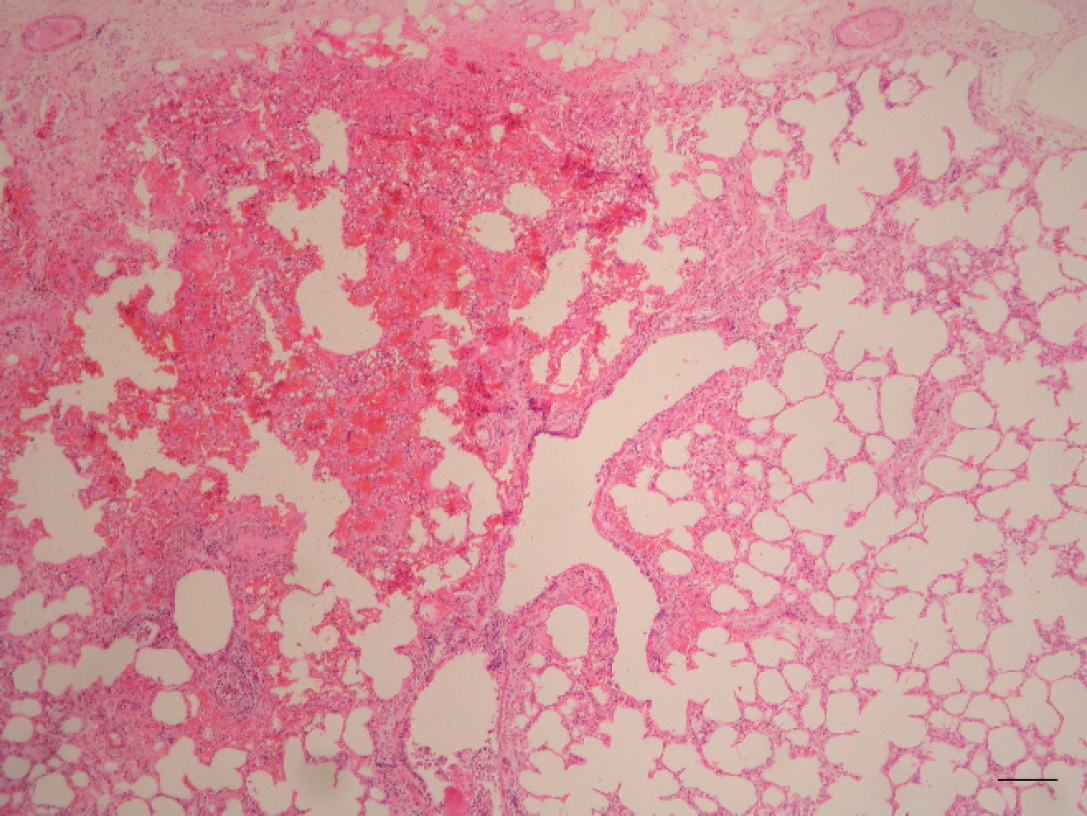
Lung. Hemorrhagic subpleural foci. Hematoxylin and eosin. Bar = 250 μm.
Because of the lesions seen, including necrosis of adipose tissues, hepatic lipidosis, and degeneration and fragmentation of muscle fibers, the present case was diagnosed as yellow fat disease. The outstanding feature of the disease in this case is the presence of fat embolism.
Yellow fat disease, or nutritional panniculitis, is a well-described disease in many species, with young horses and ponies being especially predisposed. 6–8,11,16 The 2 main causes associated with this disease are 1) high intake of polyunsaturated fatty acids that accumulate in the adipose tissue 16 and 2) vitamin E deficiency, which promotes oxidation of these acids. 6
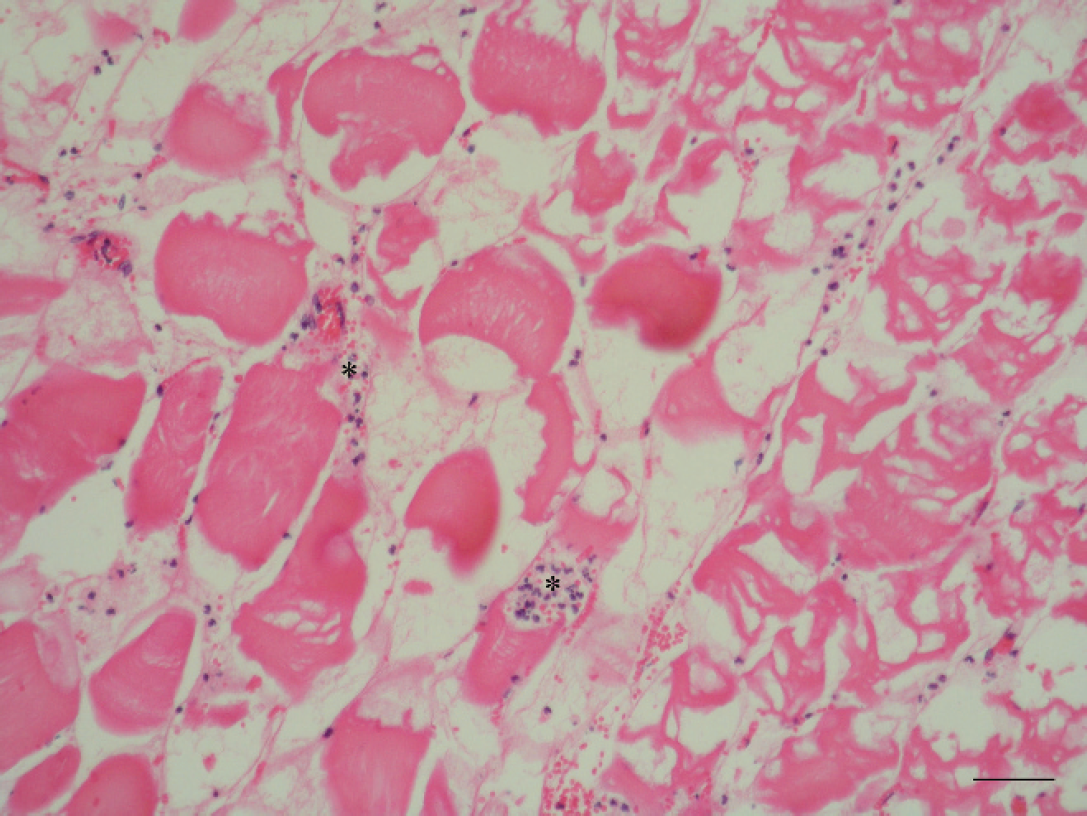
Muscle. Hydropic degeneration, myofiber fragmentation, and slight infiltrate of neutrophils in the myofiber and in the interstitium (asterisks). Hematoxylin and eosin. Bar = 25 μm.
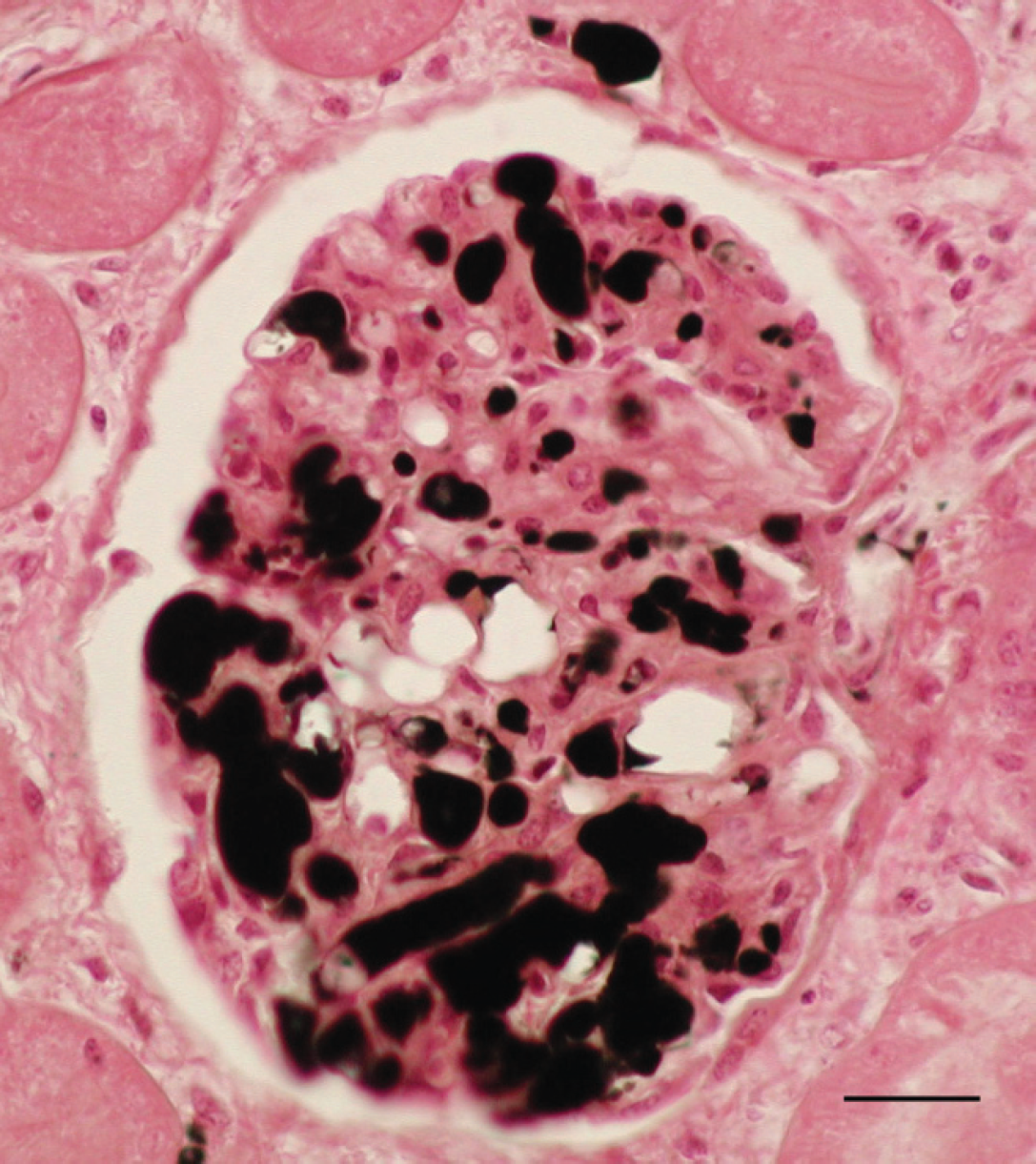
Kidney. The glomerular capillaries are distended with fat globules, and a small amount of fat is present in afferent vessels. Osmium tetroxide. Bar = 25 μm.
Clinical signs include anorexia, subcutaneous edema, tachycardia, and episodes of diarrhea, sometimes with fibrin. Most clinical signs are associated with the inflammation of the fat tissue. The lesions that characterize this disease are a widespread disorder of fat with extensive adipocytes degeneration and an associated inflammatory reaction in the fat, as well as severe hepatic lipidosis and muscular degeneration. 5 Very similar lesions were found in the present case. The horse showed an acute episode of colic with diarrhea, ecchymoses, and suffusions on the intestinal serosa. During the adipose necrosis, a progressive peroxidation of unsaturated fatty acids may occur. 7 This oxidation of fat was suggested by an intense rancid odor of the fat, observed during necropsy. With respect to the etiology, the cause of the disease was undetermined because no information about the diet was available.
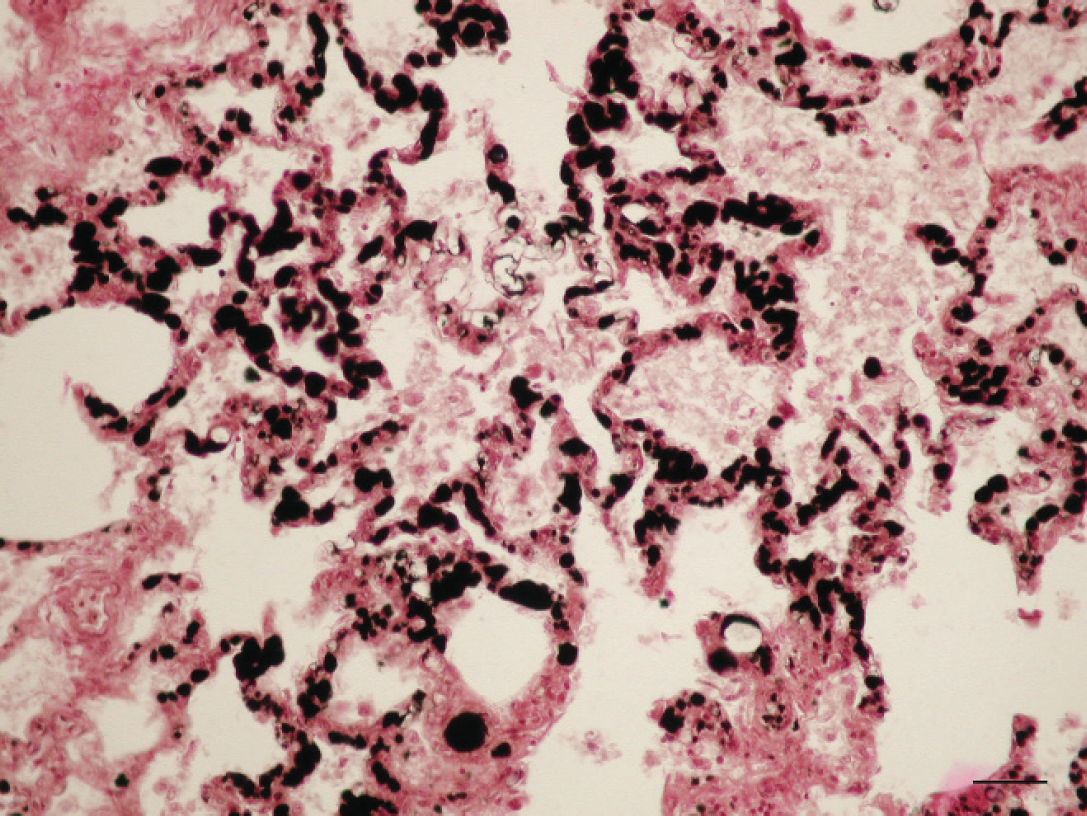
Lung. Alveolar septal capillaries are totally occluded by fat globules. Osmium tetroxide. Bar = 25 μm.
Fat embolism is an unusual complication described in veterinary medicine associated mainly with traumatic processes, such as bone fractures and fat and soft-tissue injury, and nontraumatic diseases, such as acute pancreatitis and diabetes mellitus. 4,10,13,14 Two mechanisms have been proposed for the pathogenesis of fat emboli: 1) direct entry of fat emboli into the bloodstream after trauma and 2) the generation of fat emboli from plasma lipoprotein disruption and coalescence of lipid at the intravascular gas bubble interface. 15 In the present case, the transport of fat emboli in the systemic circulation might be due to the disorder in the adipose tissue that characterizes yellow fat disease. Therefore, this fat disorder could promote a high mobilization of fatty acids into the systemic circulation exceeding their capacity of incorporation into lipoproteins and consequently causing fat emboli. The hepatic lipidosis found in the present case suggests an increased entry of lipids into the liver as a result of this increased mobilization of lipids from adipose tissue. The movement of fat vacuoles first causes the occlusion of small blood vessels, with the capillaries of the lungs and kidneys being the most affected, as seen in the present case. 14 Numerous small hemorrhages were found in the lungs of the horse, which might be due to the highly toxic action of free fatty acids, especially in capillaries. It is well known that free fatty acids can produce a toxic and inflammatory reaction resulting in pulmonary edema, hemorrhage, and microatelectasis. 2,3,12 This fact might explain the acute respiratory distress presented by the horse that led to its death. In conclusion, these findings show that massive pulmonary and renal fat emboli may be secondary to yellow fat disease. This discovery constitutes the first report of fat embolism in horses affected by this nutritional panniculitis.
Footnotes
a.
Leica TP1050 Automatic Tissue Processor, Leica Microsystems Nussloch GmbH, Nussloch, Germany.
