Abstract
Abortion and death caused by Francisella tularensis were well recognized in range flocks of domestic sheep in Idaho, Montana, and Wyoming in the first 6 decades of the 20th century. The current report describes 4 episodes of tularemia in 3 range flocks in Wyoming and South Dakota in 1997 and 2007 (1 flock was affected twice). Flock owners reported that ticks were unusually numerous and commonly present on sheep during outbreaks. Tularemia presented as late-term abortions (3 episodes) or listlessness and death in lambs and, to a lesser extent, ewes (1 episode). Lesions were multifocal pinpoint necrotic foci in tissues, particularly spleen, liver, and lung. An immunohistochemical procedure demonstrated F. tularensis, particularly in necrotic foci. The diagnosis was corroborated by bacterial isolation and, in individual cases, by serology, fluorescent antibody assay, and/or polymerase chain reaction detection of F. tularensis. Diagnosticians in endemic areas should include tularemia as a differential diagnosis when investigating late-term abortions or outbreaks of fatal illness in young lambs, particularly in years of high tick activity and when characteristic necrotic foci occur in spleen, liver, and lung.
Sheep are the only large animal species affected with epizootic tularemia caused by Francisella tularensis subsp. tularensis. 6,8 Following the first published account of tularemia in sheep in 1929, 13 reports documented the disease in flocks in northern Rocky Mountain states and Canadian provinces. 6–8,16 The flurry of published cases reflected increased awareness of the disease among wool growers, veterinarians, and physicians, in field research by personnel at the Rocky Mountain Laboratory in Hamilton, Montana, and in recognition of enzootic areas containing infected ticks (primarily Dermacentor andersoni, the Rocky Mountain wood tick). All or parts of these states and provinces fall inside the geographical range of A.II clades of F. tularensis subsp. tularensis and/or Dermacentor andersoni. 9 The morbidity in flocks was high, up to 50%, and mortality in young affected lambs approached 10%. 8,10,11 Transmission to wool growers and shearing crews occurred occasionally. 8 Wild cottontails (Sylvilagus spp.), jackrabbits (hares, Lepus spp.), and domestic sheep are donor/reservoir hosts of tularemia caused by F. tularensis subsp. tularensis. 6,19
Published descriptions of the histological changes in sheep with tularemia are cursory 7,8 ; early outbreaks were confirmed by animal inoculation and serology, and histological examination was rarely performed. The disease still occurs sporadically in range flocks in Wyoming and adjacent states. The following are 4 recent episodes of ovine tularemia investigated by the Wyoming State Veterinary Laboratory (WSVL).
Flock 1: Late-term abortions developed in a range flock of 3,000 ewes in Johnson County, Wyoming, in April 1997. Three to 5 aborted lambs were found daily during the outbreak, with a total of 20 ewes aborting (0.6% of flock). Two autolytic lamb carcasses weighing 1.0 and 1.5 kg, respectively, were submitted to the WSVL. Gross lesions were present in 1 of 2 lambs, consisting of 1-mm pinpoint white foci throughout both lungs. The principal histological finding in both lambs was multifocal splenic necrosis. Other changes were multifocal pulmonary necrosis in 1 lamb, corresponding to the white foci seen grossly, and hepatic and renal necrosis in the other. No bacteria were evident in foci of necrosis in hematoxylin and eosin or Gram preparations in this or subsequent cases, even when sections were examined after the location of bacteria was disclosed by immunohistochemistry (IHC; Fig. 1a, 1b). Fetal fluid was serologically negative for evidence of toxoplasmosis. No viruses were isolated from lung, liver, kidney, thymus, or spleen. Attempts to culture Chlamydia sp. were negative. An organism isolated from lung and liver of 1 lamb, and lung and abomasal contents of the other, was identified as a Brucella-like bacterium using a commercial identification program. a The isolate was submitted to the National Veterinary Services Laboratories (NVSL), b which established it was a non-Brucella sp. and identified it as F. tularensis subsp. tularensis. The identification was confirmed by the diagnostic and reference section of the Bacterial Zoonoses Branch at the Centers for Disease Control and Prevention (CDC) laboratory in Fort Collins, CO. c
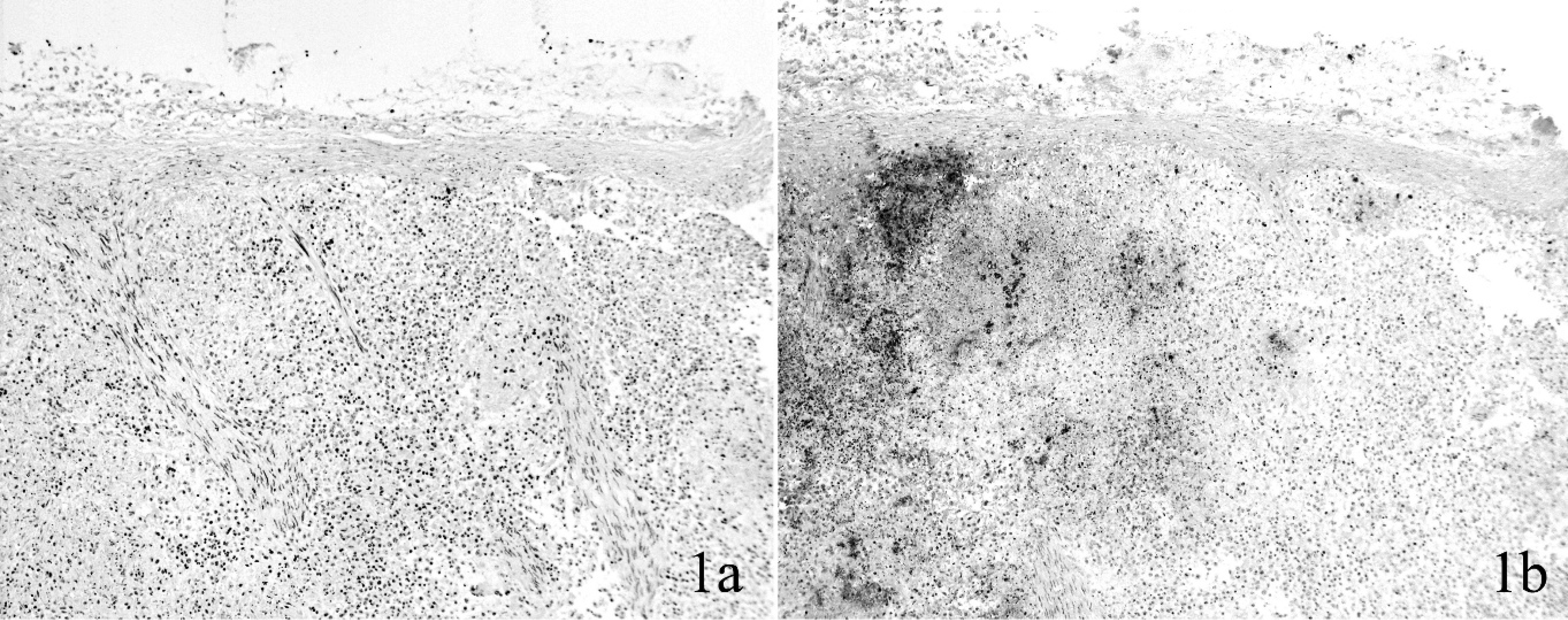
Spleen, fetal lamb; flock 1. Multifocal aggregates of Francisella tularensis are invisible in a Gram stain (

Spleen, fetal lamb; flock 1. Immunohistochemical preparation reveals a high proportion of the spleen's cross-sectional area contains antigen of Francisella tularensis (dark areas indicated by arrows). Because of the autolysis, the classical grain-of-millet lesion of tularemia was evident only microscopically. Immunohistochemistry. 40X.
Liver, fetal lamb; flock 1. In autolytic tissue, Francisella tularensis antigen is detectable, but individual organisms are not. Immunohistochemistry. 100X.
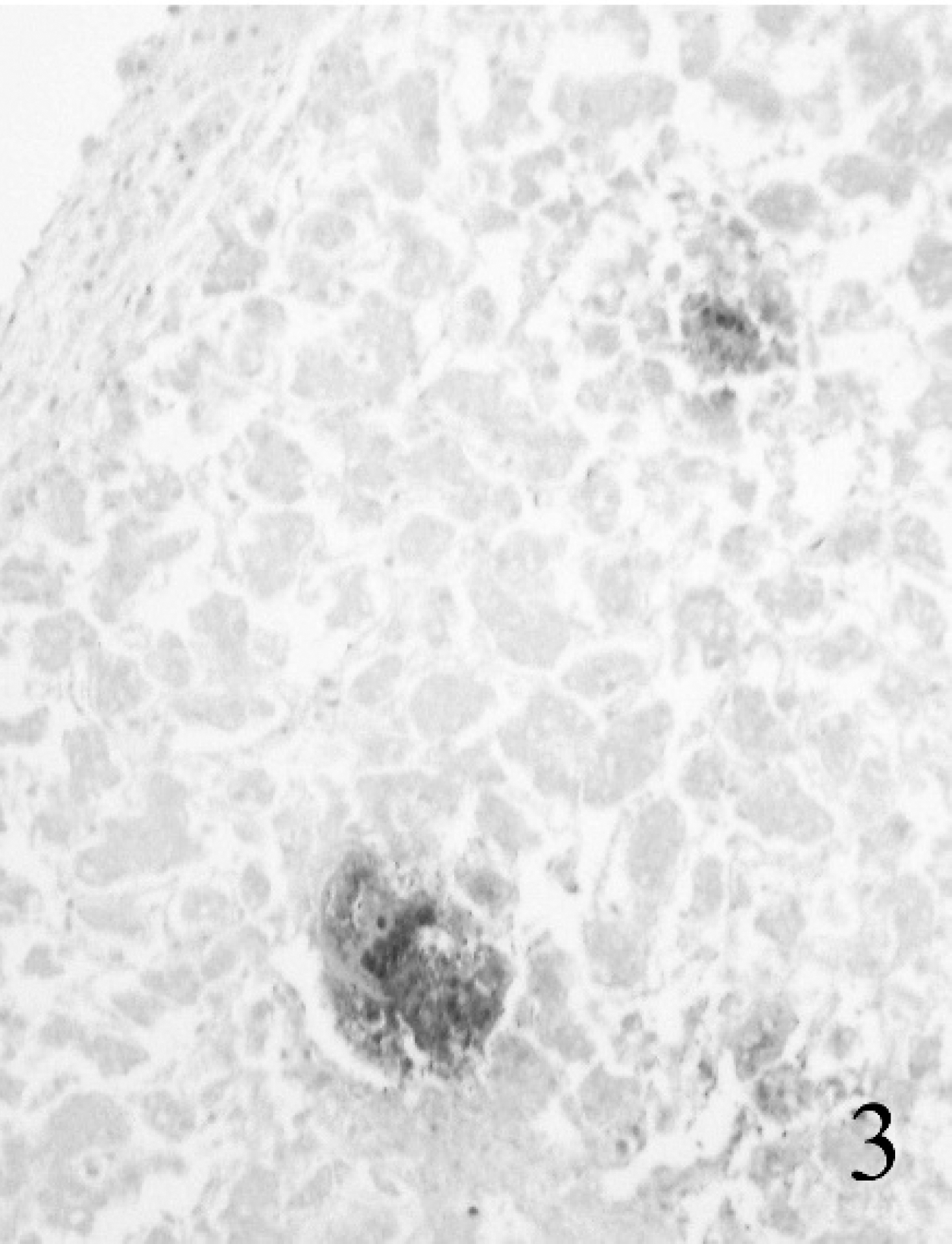
For IHC, tissue sections were cut at 4–5 μm, mounted on glass slides, d deparaffinized, rehydrated, and treated for 5 min with proteinase K e following a 5-min protein-blocking step. Francisella tularensis was detected using an automated immunostaining machine, f a commercial-source monoclonal antibody at a dilution of 1:1,500 for 30 min, g an autostainer visualization system, h and a commercial permanent chromogen. i Levamisole was included in the chromogen solution to counteract nonspecific staining. j The positive control tissue was formalin-fixed lung from a culture-positive foal that died of tularemia. The negative control was the same tissue and procedure, but with the primary antibody omitted. Intracellular and extracellular antigen was present in necrotic foci in spleen, lung, kidney, and liver (Figs. 2, 3), and in skeletal muscle and small intestine, which were without lesions. Intracellular and extracellular bacteria circulated in blood vessels (Fig. 4a, 4b).
Lung, fetal lamb; flock 1. Francisella tularensis antigen (arrowheads) in circulating cells (
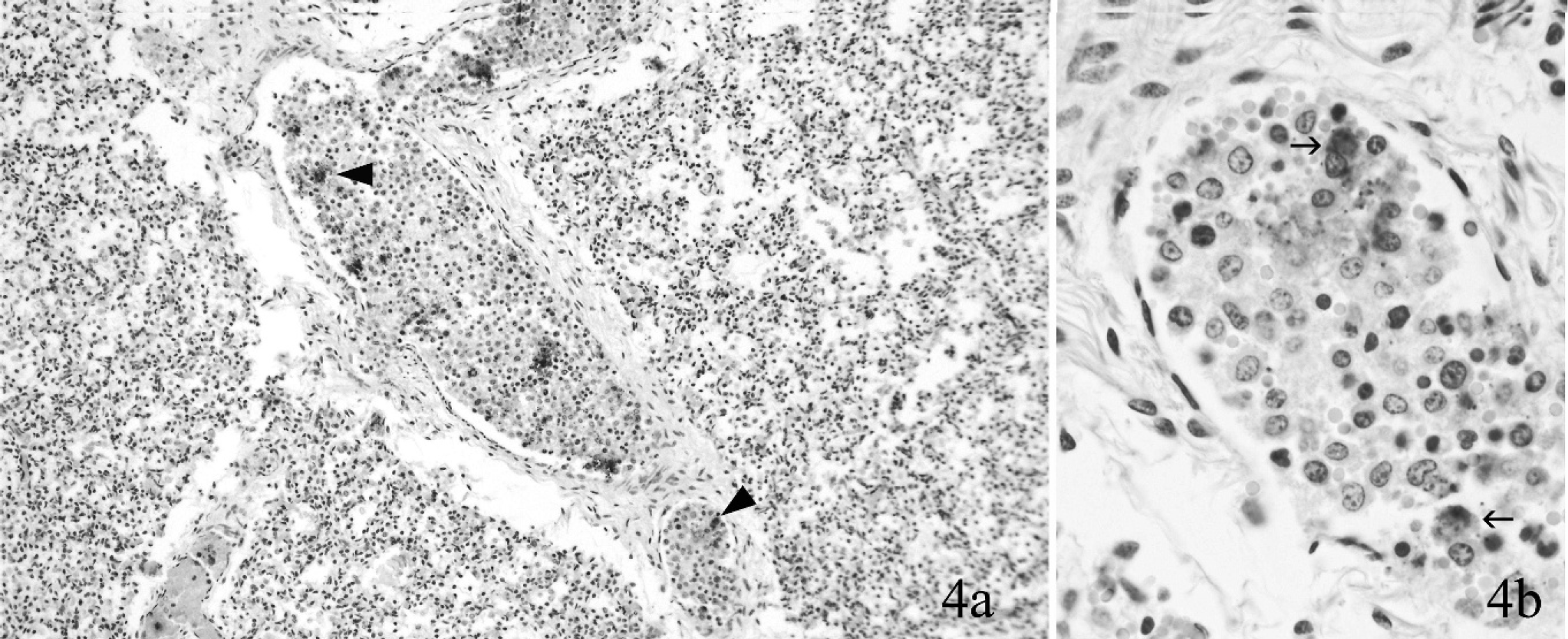
Spleen, neonatal lamb; flock 3. Typical lesion of tularemia in spleen with minimal autolysis. Note a central area of necrosis (N) invested by a zone of macrophages and neutrophils. In addition to extracellular Francisella tularensis, there are aggregates of intracytoplasmic bacteria in macrophage-like cells. Immunohistochemistry. 200X.
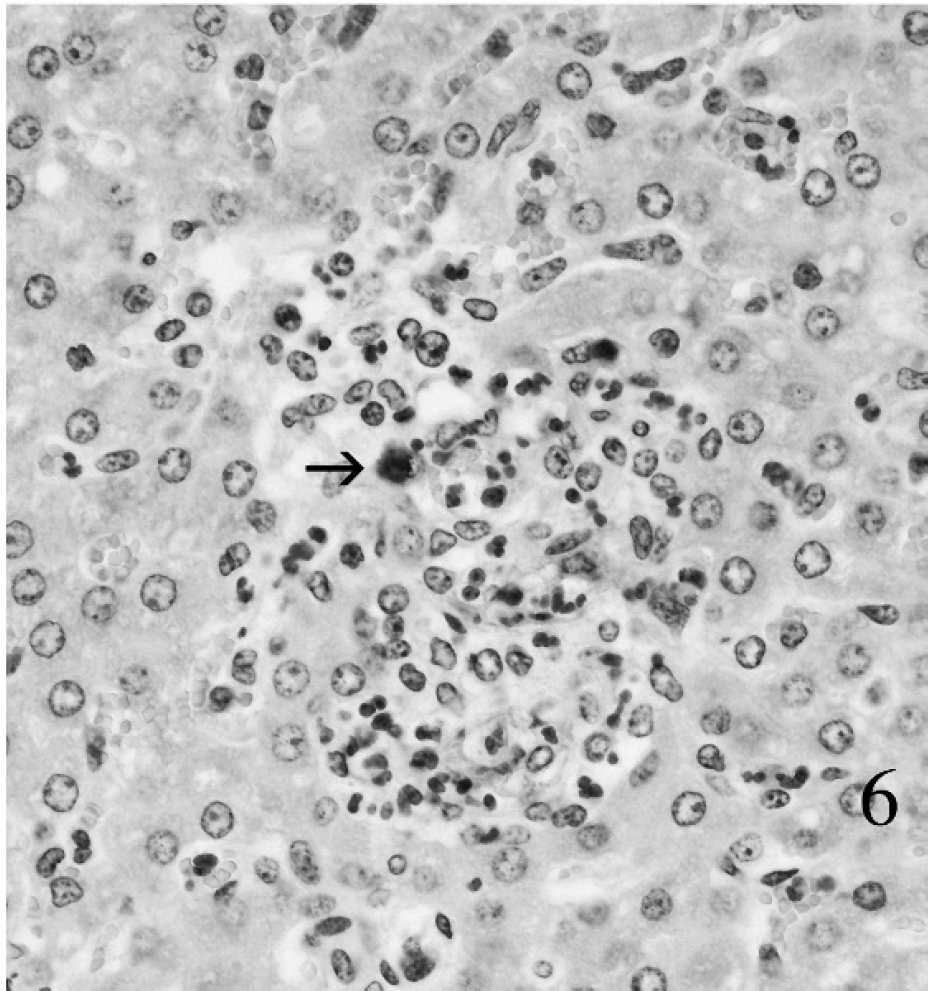
Liver, postnatal lamb; flock 3. Typical early hepatic lesion of tularemia. Necrosis is minimal. There is infiltration by neutrophils and macrophages. Some macrophages contain intracytoplasmic bacteria (arrow). Immunohistochemistry. 400X.
Tularemia reappeared on the ranch 10 years later in April 2007. There were no abortion storms in the intervening period. During the second episode, 396 of 840 coming 2-year-old ewes (47%) aborted. The ewes had recently been moved from a low tick area in Bighorn Basin to a sagebrush range heavily infested with wood ticks. Losses began abruptly 30–45 days prior to lambing, when 60 ewes aborted over 2 days. Abortions continued for 2 weeks. There was no illness in the ewes, according to the owner. He reported a recent population explosion among cottontail rabbits, followed by a die-off among cottontails and jackrabbits, resulting in numerous carcasses on pastures used by the sheep. An adjacent 60,000-acre prairie dog town containing black-tailed prairie dogs (Cynomys ludovicianus ludovicianus) had recently been depopulated by a plague-like syndrome. Two markedly autolytic fetal lambs were submitted at the start of the outbreak. No infectious agents, IHC evidence of F. tularensis, or lesions were found in either lamb (data not shown). Subsequently, a placenta and 2 additional autolytic fetal lambs weighing 1.2 and 2.2 kg, respectively, were submitted. Gross lesions were absent. Histological lesions comprised acute multifocal necrosis and/or inflammation in spleen and lung. One of 2 fetuses had hepatic necrosis. No bacteria, viruses, or Chlamydia sp. were isolated from multiple tissues of either lamb. Fetal pleural fluid was examined for phase 1 and 2 antigens of Coxiella burnetii with negative results. k Examination of IHC preparations of multiple tissues for antigens of F. tularensis revealed myriads of positively stained organisms in foci of necrosis and/or inflammation, which were most severe in spleen. Serum was collected 1 month later from ewes and tested by a F. tularensis microagglutination assay. 19 Using a cutoff of 1:128 or greater to establish a diagnosis of exposure to F. tularensis subsp. tularensis, 15 of 16 ewes were serologically positive (titers of 1:128–1:512). None of the personnel working with the flock in 1997 or 2007 reported a tularemia-like illness, and the flock was therefore not examined for serological evidence of infection. Losses ended coincident with treatment of the flock with injectable oxytetracycline.
Flock 2: In April 1997, the carcass of a 2-kg moderately autolytic fetal lamb from a flock in Johnson County, Wyoming, was submitted for examination. Gross lesions were absent. Histologically, there was multifocal necrosis in spleen, small intestine, and diaphragm, with multifocal periportal hepatitis and single-cell necrosis of hepatocytes. Immunohistochemically, F. tularensis antigen was abundant in necrotic foci. There was no serological evidence of toxoplasmosis. Attempts to isolate viruses and Chlamydia sp. from lung, liver, kidney, thymus, and spleen were negative. As in flock 1 in 1997, a Brucella sp.–like organism was isolated from lung, liver, and stomach contents. The NVSL identified it as a non-Brucella sp. b The CDC identified it as F. tularensis subsp. tularensis. c
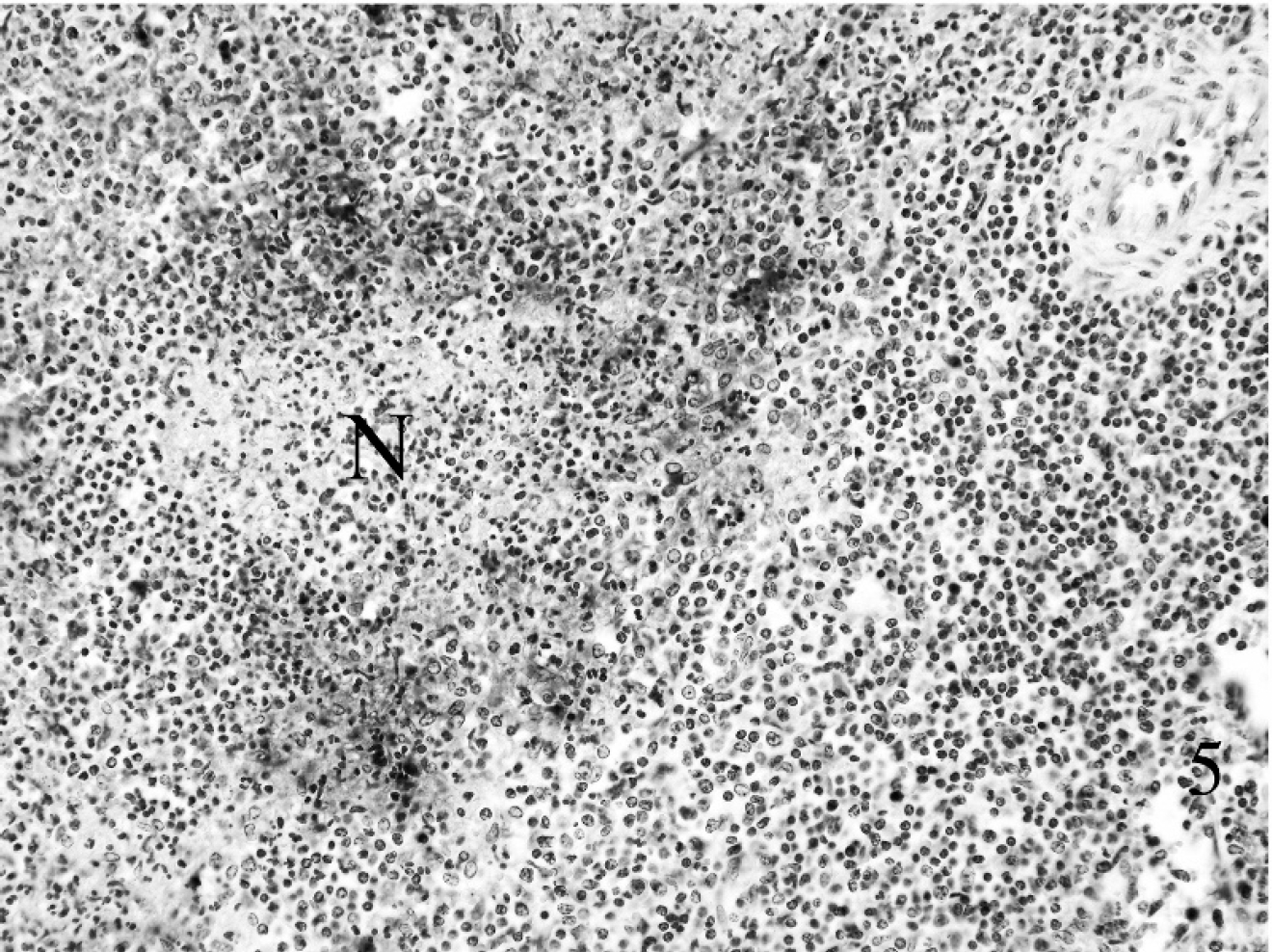
Flock 3: In early May 2007, a producer in Butte County, South Dakota, reported illness and death in 2-month-old lambs shortly after they were turned out on a greasewood pasture. Tularemia was suspected as individual sheep were parasitized by hundreds of wood ticks, and fatal tularemia in 2 adjacent riparian corridors had been recently confirmed in foals, calves, and a cat by the WSVL (data not shown). There was also a die-off of jackrabbits and pronghorn during the winter of 2006–2007. The flock consisted of 275 Targhee ewes and 425 lambs. Total death loss was 70 lambs and 15–20 ewes (12.1% total death loss; 16.5% among lambs). Clinical signs were weakness and death. Two lambs were anemic (hemoglobin of 5.5 and 4.0 g/dl; hematocrit of 17.7% and 13.1%). Gross lesions in a lamb that was examined postmortem were pallor, swollen mesenteric lymph nodes, and pulmonary congestion. Serum samples from 2 acutely sick lambs and a range of fresh and fixed tissues from a lamb with typical signs were submitted. Francisella tularensis was detected using a fluorescent antibody assay 19 in 1 of 2 samples of spleen, but not in samples of liver, lung, and bronchial or mesenteric lymph nodes. Histological lesions consisted of multifocal necrosis and inflammation in spleen, liver, lung, and mesenteric lymph nodes. Intralesional F. tularensis were detected immunohistochemically in these 4 tissues (Figs. 5, 6). The organism was cultured from mesenteric lymph node, but not from samples of liver, lung, or bronchial lymph node. Examination of tissues by polymerase chain reaction (PCR) using a Laboratory Response Network protocol l was positive. One of 2 serological samples from sick lambs was positive on the microagglutination test (1:128). 19 Losses ended shortly after animals were moved to a pasture with fewer ticks and were treated using oxytetracycline and multiple topical insecticides. Two family members developed an acute febrile syndrome during the outbreak. They were treated successfully with antimicrobials. They were tested serologically for tularemia while acutely ill, with negative results; no convalescent samples were taken.
Collectively, the outbreaks underscore reported features of tularemia in sheep. 8,11 Principal manifestations were late-term abortion and death in young lambs, little or no disease in adults, and losses in March–May. Tick-induced anemia and tick paralysis may have been a complicating factor in 1 flock (No. 3). Fetal lambs were autolyzed and bacteria were not evident in conventional or Gram-stained sections. Efforts to culture the organism from many tissues were unsuccessful, presumably because of a combination of autolysis, the organism's specific growth requirements, and competition by postmortem bacterial contaminants. 12 The organism was isolated and initially misidentified in the 2 1997 episodes. The proprietary program a used at that time identified Francisella tularensis as a Brucella sp.; this has since been corrected by the manufacturer. The disease should be suspected when pathologists see gross or microscopic evidence of disseminated focal necrosis in tissue (Fig. 2). 6 Outbreaks in flocks tend to occur in years when tick populations, particularly of Dermacentor andersoni, are unusually abundant, and shortly after sheep are moved to tick-infested pastures. Tularemia may occur recurrently in particular flocks or geographic areas, as in the human population. 19
Suspected or confirmed tularemia in other species, such as cottontails and jackrabbits (flocks 1 and 3) and domesticated species including cats, cattle, and horses (flock 3) may occur coincidentally, alerting wool growers and veterinarians to the disease. Transmission from sheep (or ticks on sheep) to people is well documented and used to be relatively common. In 1 retrospective study, 183 cases of tularemia occurred in sheep industry workers in the western United States, at least 7 of which were fatal. 8 Human tularemia in Wyoming is now rare (0–6 cases annually; 1996–2007), m but in the past human illness was associated with sheep; 2 of 79 human cases identified in Wyoming in 1940 were sheep-associated. Johnson County, where flock 1 is located, is the Wyoming county that historically had the most cases among wool growers and related workers (1931–1952; 13 cases). 8 Prompt confirmation of tularemia in flocks is important, as mortality rates in humans infected with F. tularensis are 5–15% (untreated) and <2% (treated). 6 A human antitularemia vaccine was used successfully for many years, but availability was limited to laboratory personnel at recurrent risk, and it is now no longer available. 6 The best way to prevent transmission from sheep to people is to follow sensible sanitary precautions on ranches and in veterinary clinics, and by safe laboratory practices. The World Health Organization recently published detailed laboratory guidelines for the diagnosis and safe handling of F. tularensis in carcasses and tissues. 19
Immunohistochemistry is a useful diagnostic adjunct when tularemia is suspected. Francisella tularensis was found primarily in macrophages and neutrophils and was absent in parenchymal cells. 20 No validation study was undertaken to establish whether the commercial antibody used in the described IHC procedure cross-reacts with antigens of closely related bacterial agents. The method has been used at the WSVL in cases where tularemia was a possibility; there is excellent concordance with positive isolation of F. tularensis. The method demonstrates F. tularensis—like organisms in tissue and strengthens a suspicion of tularemia, rather than being used to confirm a diagnosis.
Francisella tularensis has fastidious growth requirements, and protocols for enhanced recovery have been published. 15,19 Francisella tularensis can be difficult to culture from field specimens, particularly carcasses. 11 The authors currently use Columbia agar with sheep blood for routine isolation of organism from abortion cases in sheep. Chocolate agar is used when tularemia is suspected. The optimal temperature for growth is 35°C, and the characteristic small colonies take 2–4 days to appear.
Experimentally, sheep are readily infected, yet paradoxically they resist disease when inoculated experimentally. 3 Factors that may explain the periodicity of tularemia in range flocks are cyclical fluctuations of lagomorphs and rodent populations and/or infected ticks, concurrent disease, emergence of more virulent strains of F. tularensis, and tick-induced anemia. Weather stress and poor nutritional condition were speculated to predispose flocks to outbreaks in the past. 6,10 None of the flocks examined here was in poor nutritional condition at the start of outbreaks. The weather was not unseasonably severe for Wyoming and western South Dakota at the time the 1997 and 2007 outbreaks occurred. The owner of flock 1 attributed the abundance of wood ticks in 2007 to a recent 2-week period of warm spring weather.
Serology is of value in corroborating infection in sheep. Past studies reported seroprevalence rates of 45.7% and 23% among healthy sheep in endemic areas. 7 The high cutoff used with the agglutination test (≥1:128) to identify previously infected animals is a result of a high cross-reactivity with related infectious agents.
There is limited information on how wool growers should treat sheep when confronted with tularemia. Before the advent of antibiotics, the only option was to move sheep to less tick-infected ground, if available. Historically, the best results in affected flocks have been seen with oxytetracycline, which remains the recommended treatment in veterinary textbooks, 10 along with the use of effective topical insecticides. The use of oxytetracycline appears to have been effective in curtailing losses in the 2 flocks affected in 2007. Other antibiotics, such as streptomycin, gentamicin, ciprofloxacin, doxycycline, and chloramphenicol, are effective in treating the disease in people 19 and may be of value in animals.
Few cases of tularemia in sheep in the United States and Canada have been reported in the veterinary literature since the initial reports between 1929 and the 1960s. This parallels a decline in confirmed cases of human tularemia since its peak in 1939 4 as a result of an awareness of risks in handling sick lagomorphs and rodents, and the advent of effective antibiotics and acaricides. Although listed as a cause of abortion in sheep, the current edition of the AAVLD's diagnostic handbook for livestock abortion has no account of tularemia. 5 A standard English language textbook of diseases in sheep does not mention tularemia as a cause of abortion or acute death in lambs as a tick-borne infection or as a regional problem in North America. 1 This contrasts with older veterinary textbooks, which described the syndrome in detail. 11 Combined with the decline in the sheep industry, reluctance among range flock owners to submit samples to diagnostic laboratories, and reduced skinning of dead sheep to salvage pelts, this may explain the disappearance of tularemia as a recognized disease syndrome in sheep.
Tularemia should be part of a differential diagnosis when diagnosticians are presented with sheep with compatible signs from tularemia-endemic areas. Tularemia is a risk to laboratory personnel, given the long, fascinating, and sometimes tragic history associated with laboratory-acquired tularemia. 2,4,6,7 The presence of pinpoint foci of necrosis in the spleen and other organs from sheep with a suggestive history should prompt diagnosticians to investigate the possibility of tularemia. Immunohistochemistry, bacterial isolation on F. tularensis–appropriate media, and PCR can help corroborate clinical and morphological suspicions of the disease.
Footnotes
a.
MicroLog, Biolog Inc., Hayward CA.
b.
Mycobacteria and Brucella section, National Veterinary Services Laboratories, Ames, IA.
c.
Bacterial Zoonoses Branch, Division of Vector-Borne Infectious Diseases, National Center for Infectious Diseases, Centers for Disease Control and Prevention, Fort Collins, CO.
d.
Superfrost® Plus, VWR International Inc., West Chester, PA.
e.
Proteinase K, Dako North America Inc., Carpinteria, CA.
f.
Autostainer Plus, Dako North America Inc., Carpinteria, CA.
g.
Francisella tularensis antiserum (#240939) Becton, Dickinson and Company, Sparks, MD.
h.
LSAB 2 AP, Dako North America Inc., Carpinteria, CA.
i.
Bio-RED, Biopath Lab Inc., Oklahoma City, OK.
j.
Levamisole Solution, Dako North America Inc., Carpinteria, CA.
k.
Texas Veterinary Medical Diagnostic Laboratory, Texas A&M University, College Station, TX.
l.
Laboratory Response Network, Centers for Disease Control and Prevention, Atlanta, GA.
m.
Dr. Dan O'Leary, Dr. Karl Musgrave, and Clay Van Houten, personal communications, Wyoming Department of Health, Cheyenne, WY.
