Abstract
An adult Brown Swiss cow was presented to the Large Animal Hospital of the Centre Hospitalier Universitaire Vétérinaire de l'Université de Montréal due to a postpartum downer cow syndrome. The animal had severe and generalized swelling of all 4 limbs and was in shock, as demonstrated by hypotension, dehydration, hypothermia, altered mental status, and abnormal blood parameters. It died rapidly, and necropsy revealed a generalized, severe cellulitis caused by Streptococcus dysgalactiae subsp. dysgalactiae, bronchopneumonia, and lesions of disseminated intravascular coagulation (DIC) in the kidneys. The portal of entry of the bacteria in the subcutaneous tissue was not found, as there was no history of skin trauma or mastitis. The pure growth of large quantities of an invasive Streptococcus species, associated with hypotension, coagulopathy, and renal failure, are supportive of streptococcal toxic shock-like syndrome in a bovine.
Cellulitis is defined as a poorly circumscribed infection of soft tissues that tends to dissect through tissue planes. 19 It is generally caused by a skin or mucosal wound that can be minute and difficult to detect. 18 Various aerobic and anaerobic bacteria can be isolated from lesions of cellulitis. Streptococcal and staphylococcal cellulitides are well-known and may be life-threatening, as they can result in complications like septic shock (septicemia) or toxic shock syndrome. 6 Toxic shock syndrome (TSS) is a multisystemic disease characterized by rapid onset of fever, hypotension, and multi-organ failure, which often leads to death. 6 Severe streptococcal cellulitis associated with septicemia or toxic shock syndrome is rare, especially in veterinary medicine, but outbreaks due to Streptococcus canis have been described in dogs and cats, and Streptococcus agalactiae and Streptococcus pneumoniae were respectively cultured from a bottlenose dolphin and a kitten with cellulitis and septicemia. 9,11,20,21 Cellulitis has rarely been described in the bovine species. 1,12,13 In this species, it is often associated with Clostridium spp. (malignant edema, blackleg) and Arcanobacterium pyogenes. 10,19 The present report describes an unusual, severe, and generalized subcutaneous cellulitis with toxic shock-like syndrome, caused by Streptococcus dysgalactiae subsp. dysgalactiae in a cow.
A 10-year-old Brown Swiss cow was presented as an emergency case to the Veterinary Teaching Hospital (VTH) of the Centre Hospitalier Universitaire Vétérinaire de l'Université de Montréal due to postpartum recumbence that was nonresponsive to primary treatment (downer cow syndrome). She calved 6 days prior to presentation and was found recumbent the following morning. She had been treated at the farm with intravenous and subcutaneous calcium solutions and milk was withheld. She was unable to stand on her own and was lifted with a hip lifter and a sling once a day by the owner. Her appetite and general condition deteriorated markedly during the following days, and a generalized swelling of the limbs developed. She was given dexamethasone and ketoprofen by the referring veterinarian but had not received any antibiotics. Due to the worsening of the condition, the decision was made to bring her in emergency to the Veterinary Teaching Hospital.
On presentation at the hospital, the cow was depressed and in lateral recumbency. She was hypothermic (37.5°C), tachycardie, and tachypneic. Mucous membranes were pale with a prolonged capillary refill time (>4 seconds). She was 7% to 10% dehydrated, had a poor pulse quality, and intermittent cardiac arrhythmias. Conjunctivae were injected, and pupillary light reflexes were slow. The most striking finding was that all 4 limbs were markedly edematous and cold to the touch, and she had subcutaneous emphysema and oedema on the right side, more pronounced over the neck and flank areas. No evidence of fracture or luxation was found on physical examination. The uterus appeared normal by transrectal examination. The udder was filled with milk, and there was no evidence of mastitis on visual examination of the milk.
Principal blood parameters from the cow upon arrival at the hospital, with reference ranges.

Principal blood parameters of this animal are listed with reference ranges in Table 1. Venous blood gas analysis indicated metabolic acidosis (pH 7.27; bicarbonate 15.7 mequ/liter) with hyperchloremia (115 mequ/liter) and increased lactic acid concentration (7.5 mmol/liter), suggestive of hypoperfusion. Compensatory respiratory alkalosis was also present (PCO2 33.7 mmHg). Complete blood count showed a marked inflammatory response with increased fibrinogen (10 g/liter), presence of immature neutrophils (0.1 × 109/liter), numerous neutrophils with toxic changes, and lymphopenia (1.7 × 109/liter). She was also severely thrombocytopenic (platelets 29 × 109/liter), and mildly hemoconcentrated (hematocrit 56.6%). Total protein was low to normal (72 g/liter) considering her hydration status. Potassium was mildly elevated (5.4 mmol/liter), as well as blood urea nitrogen (11.0 mmol/liter) and creatinine (167 μmol/liter), suggesting impaired renal function. There was mild hyperbilirubinemia (19.4 μmol/liter) but gamma-glutamyl transferase (GGT) and lactate dehydrogenase (LDH) were within normal range (32 international units [U]/liter and 11.20 U/liter, respectively). Creatine Phosphokinase (CK) and aspartate amino transferase (AST) were markedly elevated (19,521 U/liter and 1,045 U/liter, respectively), indicating probable muscle damage.
A fine needle aspirate of the swollen subcutaneous tissues was taken, and slides were Gram stained and examined for the presence of bacteria. Numerous Gram-positive cocci were present in the aspirated fluid. Despite aggressive support therapy (fluids, broad-spectrum antibiotics, intranasal oxygen), her vital signs, mental status, pulse quality, and hydration status did not improve, and the animal died less than 2 hours after admission. A complete necropsy was performed the following day.
The animal had mild to moderate postmortem changes. The skin was carefully examined, and several superficial decubitus erosions were found. Large amounts of yellowish to serosanguineous exudate were diffusely present in the subcutaneous tissues. The dependent parts (limbs and ventral abdomen) were more severely affected. Some isolated, scattered, necrotic, and hemorrhagic foci were found in the muscles of the coxofemoral region as well as in thigh muscles on both sides. These foci were located deep in the muscles and did not appear to be associated with the overlying cellulitis. The cerebral gyri and cerebellar folia were mildly flattened with small amounts of clear gelatinous fluid distending the subdural space (edema). The lungs were heavy, wet, and the interlobular septae were diffusely and moderately distended by clear gelatinous fluid. A moderate, generalized interlobular emphysema was also observed. Scattered groups of lobules were atelectatic in the antero-ventral portion of the lungs (about 20% of the whole parenchyma). Some wedge-shaped, necrohemorrhagic foci were found in the renal cortex, bilaterally.
Tissues collected at postmortem were fixed in 10% neutral buffered formalin, routinely processed and stained with hematoxylin, eosin-phloxine, and saffron (HPES) and, for the subcutaneous tissues, with a standard Gram coloration. Sections of subcutaneous tissue from the dorsum and from the lateral right thigh areas were taken and examined. Lesions were similar at both sites. Myriad Gram-positive cocci, sometimes arranged in pairs or in chains, were found throughout the subcutaneous tissue, associated with marked congestion and edema, some hemorrhages, and mild to moderate fibrinosuppurative inflammation (Figs. 1, 2). Necrosis of adipose tissue and collagen degeneration were present but not prominent, and neutrophils were often degenerate. Some vascular thromboses and a single neutrophilic vasculitis were seen. Large bacterial colonies were found in the lumen of some vessels. The cocci were sometimes present in the perimysium of the superficial muscles underlying the inflamed subcutaneous tissues, eliciting little inflammatory response (mainly degenerate neutrophils, serum, and fibrin). Their presence was also associated with necrosis and degeneration of individual myocytes and with some interstitial hemorrhages. Deep necrotic muscle foci were not examined histologically as they were assumed to be secondary to the prolonged decubitus. Emphysema and alveolar and interstitial edema were obvious on histologic examination of the lungs. In the atelectatic lobules, the epithelium of bronchi and bronchioli was generally necrotic. Their denuded submucosa was covered by fibrin, and their lumens were filled with necrotic debris and neutrophils. The alveoli of these lobules contained abundant, homogenous eosinophilic material (serum), fibrin clots, and degenerate leukocytes that were sometimes oat-shaped. Some small-sized and medium-sized vessels were thrombotic. In the kidneys, multiple thromboses of glomerular capillaries and of some interstitial vessels were found, as well as wedge-shaped lesions of ischemic infarction. In the brain, there was moderate distension of Virchow-Robin spaces and leptomeninges by an optically clear material (edema).
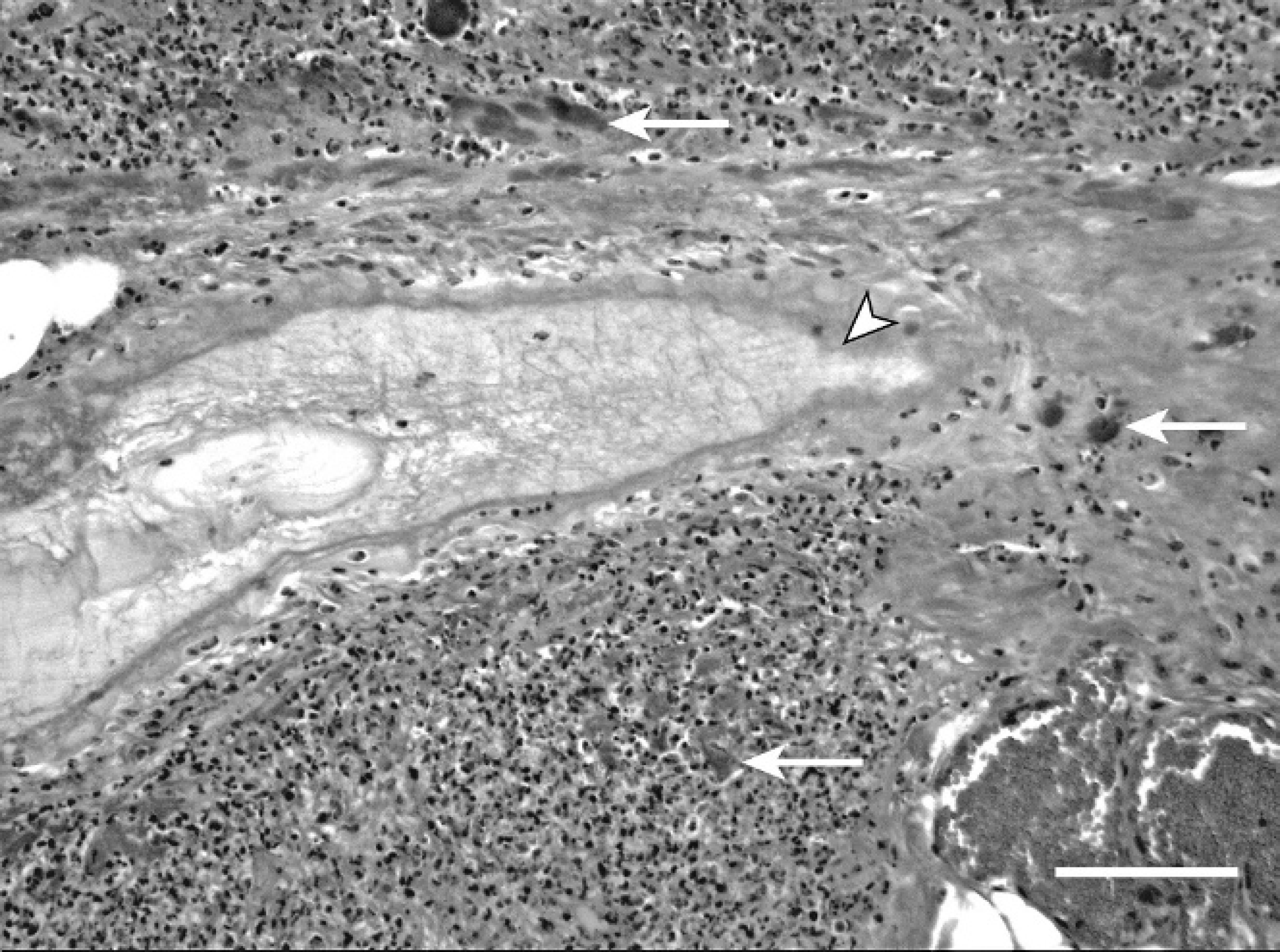
Subcutaneous tissue, dorsum; a moderate amount of fibrin, acidophilic proteinaceous material, and degenerate neutrophils is present between collagen fibers. The wall of a lymphatic vessel is necrotic (arrowhead). Note multiple large bacterial microcolonies lying free in the tissue (arrows). Hematoxylin-phloxine-eosin-saffron. Bar = 100 μm.
Lungs were positive by direct fluorescent antibody staining for bovine herpesvirus-1 (infectious bovine rhinotracheitis [IBR]). Moderate numbers of Mannheimia haemolytica and Pasteurella multocida were isolated from the atelectatic parts of the lungs. A very large quantity of Lancefield group C S. dysgalactiae subsp. dysgalactiae was recovered from the subcutaneous tissue in pure culture. The routine bacterial culture was negative in samples of liver, kidney, and brain. The anaerobic culture of the subcutaneous tissue was negative.
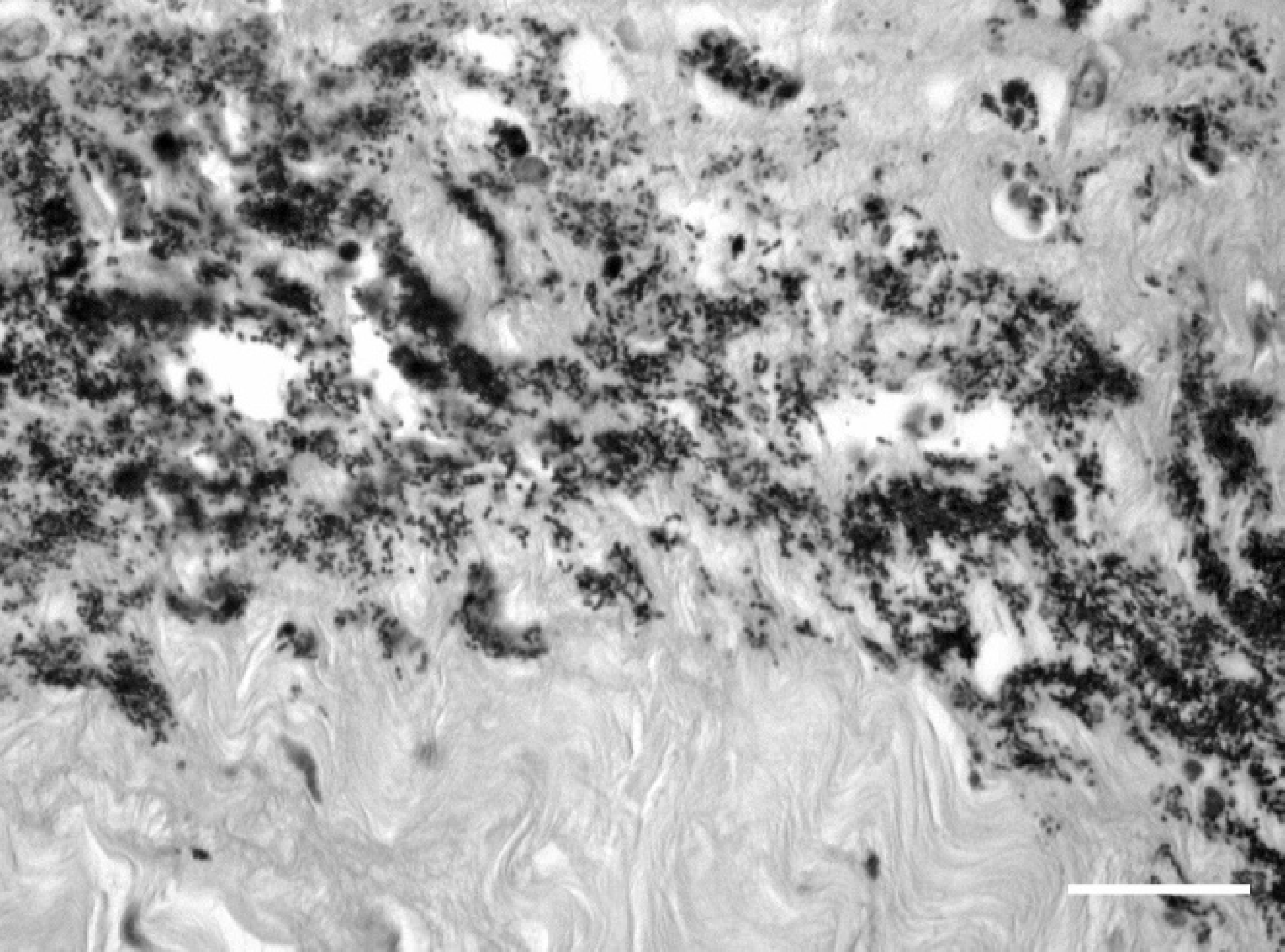
Subcutaneous tissue, dorsum; myriad gram-positive cocci are arranged in pairs and chains in the edematous subcutaneous tissue. Gram stain. Bar = 20 μm.
Chromosomal DNA from the S. dysgalactiae strain was tested using polymerase chain reaction (PCR) for the presence of eleven superantigen genes (speA, speC, speG, speH, speI, speJ, speK, speL, speM, ssa, and smeZ) with primers specific for each gene. Positive controls for each superantigen were included by using chromosomal DNA from the sequenced Streptococcus pyogenes strains SF370. MGAS315, MGAS5005, MGAS8232, and the strain 90226. No bands were detected for the S. dysgalactiae strain, nor was mitogenic activity present in the supernatant of this strain for human lymphocytes (data not shown).
Toxic shock syndrome (TSS) is a life-threatening, clinical entity characterized by invasive staphylococcal or streptococcal infection, hypotension, and multi-organ failure. The pathogenesis of this syndrome has clearly been linked to the expression of superantigens by some strains of Staphylococcus aureus and Streptococcus spp. 6 Superantigens are proteins that bypass the normal pathway of antigen recognition by their ability to bind nonspecifically to major histocompatibility complex II and T-cell receptor, thus activating up to 25% of an individual's T cells. 7 This overwhelming activation induces the release of major quantities of proinflammatory cytokines, especially TNFα, IL-1, IL-6, and IFNγ, that cause clinical signs characteristic of TSS. 6,7,17
A definitive streptococcal TSS diagnosis requires 1) the isolation of a group A Streptococcus from a normally sterile site and 2) hypotension as a major clinical sign with at least 2 of the following signs: renal or liver impairment, coagulopathy, adult respiratory distress syndrome (ARDS), extensive tissue necrosis (necrotizing fasciitis), or erythematous rash. 17 This case definition is essentially based on bacterial culture and clinical signs, but since its publication in 1993, it is generally admitted that superantigens must also been identified in the bacterial strain to conclude to a real TSS case. Moreover, although the classic cases described in the 1990s involved exclusively group A streptococci (GAS), β-hemolytic groups B, C, and G streptococci have also been involved in several human and animal cases of TSS and/or necrotizing fasciitis. 5,20 For example, in 1 study, 21 human isolates of invasive S. dysgalactiae subsp. equisimilis were screened for the presence of 6 superantigens genes often found in cases of GAS TSS. 5 Fifty percent of the strains were positive for at least 1 of them, suggesting that this bacterial species has the potential to cause TSS by the same mechanisms as GAS.
Suspected animal cases of streptococcal TSS are rare, and the identification of superantigens has not been attempted in most of them. 4,11,20 Only 1 potential virulence gene found in some human strains of S. pyogenes causing TSS (emm1.0 gene) was identified in 1 of 7 dogs that suffered from S. canis-associated TSS; in this study, no superantigens genes could be demonstrated. 9 As well, no superantigens have been detected to date in the strains of Streptococcus suis involved in the human outbreak of streptococcal TSS in China in 2005, which was clearly swine-associated. 16 It was then not unexpected that no superantigens genes could be identified in our case. We could hypothesize that the animal strains of Streptococcus spp. possess superantigens variants that could not be detected by the PCR tests developed for human strains. However, the proliferation experiment performed on our strain revealed no sign of mitogenic activity on human lymphocytes, thus suggesting that there were no superantigens in this sample or that the superantigens present had no activity on human cells, which was unlikely.
On the other hand, TSS could possibly be mediated by other mechanisms. The M protein of streptococci has recently been demonstrated to play a major role in the development of TSS, by forming complexes with intravascular fibrinogen, triggering the activation of polymorphonuclear leukocytes that release enzymes and toxic oxygen metabolites that cause vascular damage and hypercoagulability of the blood. 2 Other exotoxins and exoproteins of streptococci have also been shown or may have the potential to play a role in the virulence of some strains. 8
In addition to the culture of an invasive Streptococcus spp. in the subcutis of this cow, she also presented clinical signs and lesions that suggest TSS. Although direct or indirect arterial pressure was never measured, hypotension and hypoperfusion were assumed based on pale mucous membranes, slow capillary refill time, poor pulse quality, increased lactic acid, and the altered mental status of the animal. Hypotension ensues as fluid volume in the blood vessels decreases due to vascular damage by the cytokines massively released in TSS. 6 A coagulopathy was also present as multiple renal vessels were thrombosed, indicating disseminated intravascular coagulation (DIC), and a thrombocytopenia was apparent on hematology. Impaired renal function seemed present as blood urea nitrogen (BUN). creatinine, and potassium were both elevated and that the glomerular capillaries were extensively thrombosed. As all the clinical criteria are fulfilled to conclude to a TSS but as no superantigen could be demonstrated, this was called a case of streptococcal toxic shock-like syndrome.
The hypothesis of an endotoxic shock instead of a TSS was also considered. Endotoxins released by Gram-negative bacteria found in the lung lesions of this animal may have induced a local coagulopathy (as demonstrated by the thrombi visible in the lungs) and DIC. However, the limited extensiveness of the bronchopneumonia (20% of the whole lung parenchyma) compared with clinical cases of M. haemolytica pneumonia, in which the lung parenchyma is often involved at 75% or more, with generally no evidence of DIC, makes this hypothesis very unlikely. However, coadministration of sublethal doses of lipopolysaccharides (LPS) and superantigens has been demonstrated to cause shock in rodents. 14 The exact mechanism of this phenomenon is unknown, but human monocytes exposed to superantigens have a significantly enhanced TNF-α response following exposure to LPS. 14 So the endotoxins of Mannheimia and Pasteurella could have contributed to the shock in a synergistic manner with streptococci superantigens, if present.
The portal of entry of S. dysgalactiae in this case remains unknown. A skin lesion is the most likely hypothesis: however, no deep skin lesion was found. In about half the cases of cellulitis reported, physical examination failed to detect a breach in the skin, but a history of local nonpenetrating trauma was often present. 15,21 In this case, the cellulitis was very extensive, and careful examination did not reveal a site of a penetrating wound injury. It is possible that the bacteria gained access to the subcutaneous tissue through minor skin trauma caused by the prolonged decubitus or by the standing procedure with the hip lifter. Also, there is a possibility that the syringes and needles used by the owner to deliver subcutaneous calcium borogluconate solution were not sterile. Another possibility to consider is that the bacteria invaded the subcutis by the hematogenous route, as demonstrated in a case of breast cellulitis following a bacteremia to S. agalactiae in a woman. 3 Streptococcus dysgalactiae is often isolated from the bovine mouth, tonsils, and vagina and is often responsible for subclinical and clinical mastitis and endometritis. In this case, the mammary gland and uterus were grossly normal, but no samples were taken for microscopic examination or bacterial culture. However, bacterial cultures of liver, kidney, and brain samples were negative, indicating that there was apparently no bacteremia at the time the necropsy was performed, and the possibility of a past and transient streptococcal bacteremia is very unlikely, as this cow had never received antibiotics prior to its presentation at the Hospital and died less than 1 hour following the intravenous (IV) administration of antibiotics. In conclusion, this cellulitis is unique in regard to its extensiveness, its etiology, and by its association with a toxic shock-like syndrome, a condition that has never been reported before in the bovine species.
Acknowledgements. The authors would like to thank Jacinthe Cardin and Jules Deslandes for their technical assistance; Drs. André Dallaire, Malcolm Gains, and Pierre Hélie for their comments on the manuscript; and John McCormick for doing the PCR testing for streptococcal superantigens and the proliferation experiment.
