Abstract
Bovine viral diarrhea virus (BVDV) is one of the most relevant pathogens affecting today's cattle industries. Although great strides have been made in understanding this virus in cattle, little is known about the role of wildlife in the epidemiology of BVDV. While persistently infected cattle are the most important reservoir, free-ranging ungulates may become infected with BVDV as demonstrated by serosurveys and experimental infections. Therefore, free-ranging wildlife may maintain BVDV as the result of an independent cycle and may serve as a reservoir for the virus. Systematic studies on prevalence of BVDV-specific antibodies or frequency of persistent BVDV infection in North American wildlife are sparse, and no information is available from the southeastern United States. The objective of this study was to evaluate blood and skin samples from hunter-harvested white-tailed deer (Odocoileus virginianus) for evidence of BVDV infection. Virus-neutralizing antibodies were detected in 2 of 165 serum samples. Skin biopsy immunohistochemistry (IHC) was performed on samples from 406 deer using a BVDV-specific monoclonal antibody (MAb) (15c5), and BVDV antigen was detected in one sample. A similar IHC staining pattern was obtained using a second BVDV MAb (3.12F1). Viral antigen distribution in the skin sample of this deer resembled that found in persistently infected cattle and in a previously described persistently infected white-tailed deer; thus, the deer was presumed to be persistently infected. Evidence of BVDV infection in free-ranging white-tailed deer should encourage further systematic investigation of the prevalence of BVDV in wildlife.
Infections with bovine viral diarrhea virus (BVDV) are cause for substantial losses to the cattle industries worldwide. The virus may result in respiratory, gastrointestinal, and reproductive tract disease of varying severity, ranging from subclinical to fatal disease. 3 There are 2 forms of infection with BVDV: transient and persistent. Transient infections (TI) occur in postnatal, immunocompetent animals and initiate formation of BVDV-specific immunity that results in clearance of the virus. In contrast, persistent infection (PI) is the result of in utero exposure of the developing fetus to a noncytopathic strain of BVDV before the development of immunocompetence. The virus is recognized as self, does not result in antibody formation, and continues a lifelong replication in most tissues of the host. 6
Infections with BVDV are not limited to cattle, but they can occur in a variety of species of the order Artiodactyla, as has recently been reviewed. 25 White-tailed deer (Odocoileus virginianus) are the most abundant species of wild ruminants in the United States, and over the past 50 years, the occurrence of BVDV infection in this species has been evaluated by different researchers. Previous work has demonstrated that white-tailed deer are susceptible to BVDV infection and may develop clinical disease. 22,27 A recent report demonstrated that experimental infection of pregnant white-tailed deer may result in the birth of PI offspring. 20 Despite these published reports, only 3 studies have evaluated BVDV infection in free-ranging white-tailed deer. In contrast to high BVDV prevalence rates detected in other Cervidae. BVDV seroprevalence in free-ranging white-tailed deer from Anticosti Island, Quebec was 0%, and 2 reports from New York state indicated a prevalence of 3% and 5.75%. 14,18,23 In light of the extensive distribution of white-tailed deer, ranging from southern Canada to South America, extrapolation of results from these serosurveys to other geographic areas may not be appropriate.
Additional studies have been performed in captive or locally confined white-tailed deer populations and cannot provide information on the risk of BVDV transmission from free-ranging white-tailed deer to cattle populations. 8,10 Because BVDV eradication and control programs are being developed and implemented by veterinary and producer organizations in different states, it is necessary to investigate the occurrence of BVDV in wildlife species. Populations of free-ranging wildlife may harbor BVDV and could be a reservoir for the virus. 12,26 The objective of this study was to evaluate free-ranging white-tailed deer in Alabama for evidence of BVDV infection.
Samples were collected during the 2005–2006 Alabama white-tailed deer hunting season. Site visits were made to 23 deer-processing units across the state of Alabama. To increase the diversity of sample origin, a maximum of 40 skin and 20 serum samples were collected from each deer processing facility. Blood and skin biopsy samples were collected from all deer arriving during a visit. When possible, blood samples were collected from the opened heart or major vessels. If a deer had been field-dressed, blood was collected from the thoracic cavity. Additional skin samples were taken from all deer stored in refrigeration that had been processed earlier. Blood was collected into 10-ml serum tubes and placed on ice for transportation to the laboratory. Skin biopsies (25 mm 2 ) were harvested using commercially available ear-notch pliers from the ventral aspect of the ear and placed into 10% neutral-buffered formalin. All samples were delivered to Auburn University within 24 hours of collection for further processing.
A microtiter virus neutralization (VN) assay was used to detect and quantify antibodies in serum. 11 Sera were tested for neutralizing antibodies to the cytopathic BVDV 1 strain NADL and the cytopathic BVDV 2 strain 125c. Sera were inactivated for 30 minutes in a 56°C water bath. Serial 2-fold dilutions, ranging from 1:4 to 1:4096, were made for each serum sample in 50 μl of minimum essential medium with Earle's salts a supplemented with 10% (vol/vol) equine serum, b sodium bicarbonate b (0.75 mg/dl), L-glutamine c (0.29 mg/ml), penicillin G d (100 U/ml), streptomycin d (100 U/ml), and amphotericin B d (0.25 μg/ml). For each dilution, 3 wells of a 96-well microtiter plate were inoculated with 50 μL of culture media containing 100 cell-culture infectious doses (CCID50) of the respective BVDV strain. The plates were incubated for 1 hour at 37°C in humidified air containing 5% CO. The Madin Darby bovine kidney (MDBK) cells were used as the indicator cells by adding 50 μl of media containing 15,000 cells per well. Each test included a back titration of the virus and a positive and negative serum control. After a 5-day incubation period, antibody titer was defined as the reciprocal of the highest dilution with complete inhibition of cytopathic effect.
Immunohistochemical (IHC) detection of BVDV antigen was performed on formalin-fixed paraffin-embedded skin biopsies using a monoclonal antibody (MAb), 15c5. 16 , e The BVD MAb 15c5 reacts with an epitope of the ERNS protein of BVDV that is shared by, and therefore is suitable for detecting, a wide variety of BVDV strains. The IHC stain distribution in BVDV PI animals is represented by diffuse staining in the epidermis and hair follicle epithelium; stain may also be present in the dermis and adnexal structures surrounding hair follicles. 7
In Alabama, the white-tailed deer population is estimated at around 1.7 million animals. Paired serum and skin samples were collected from a total of 165 animals. Additionally, 241 skin samples were harvested from deer that had already been stored in refrigeration. The majority of samples were obtained from deer harvested in the central, eastern-central, and northern regions of the state. Two of 165 (1.21%; 95% confidence interval [CI]: 0–2.88%) samples were positive by VN. One sample was positive for BVDV 1 (NADL) and BVDV 2 (125c), exhibiting a higher titer against the former (1:128 for NADL; 1:16 for 125c). The second sample only contained neutralizing antibodies against BVDV 2 (strain 125c) with a titer of 1:8 but did not react positively for BVDV 1 (<1:4). BVDV antigen was detected by IHC in only 1 of the 406 skin biopsy samples (0.2%; 95% CI: 0–0.6%), and this IHC-positive deer was a doe sampled from central Alabama (Fig. 1). Positive IHC staining was distributed diffusely in epidermal cells and multifocally in hair follicle epithelium and apocrine gland cells. To reevaluate and ascertain this result, another IHC was performed using a different monoclonal antibody. BVDV MAb 3.12F1, f as described previously. 5 Antigen was similarly distributed with BVDV MAb 3.12F1 as detected with BVDV MAb 15c5.
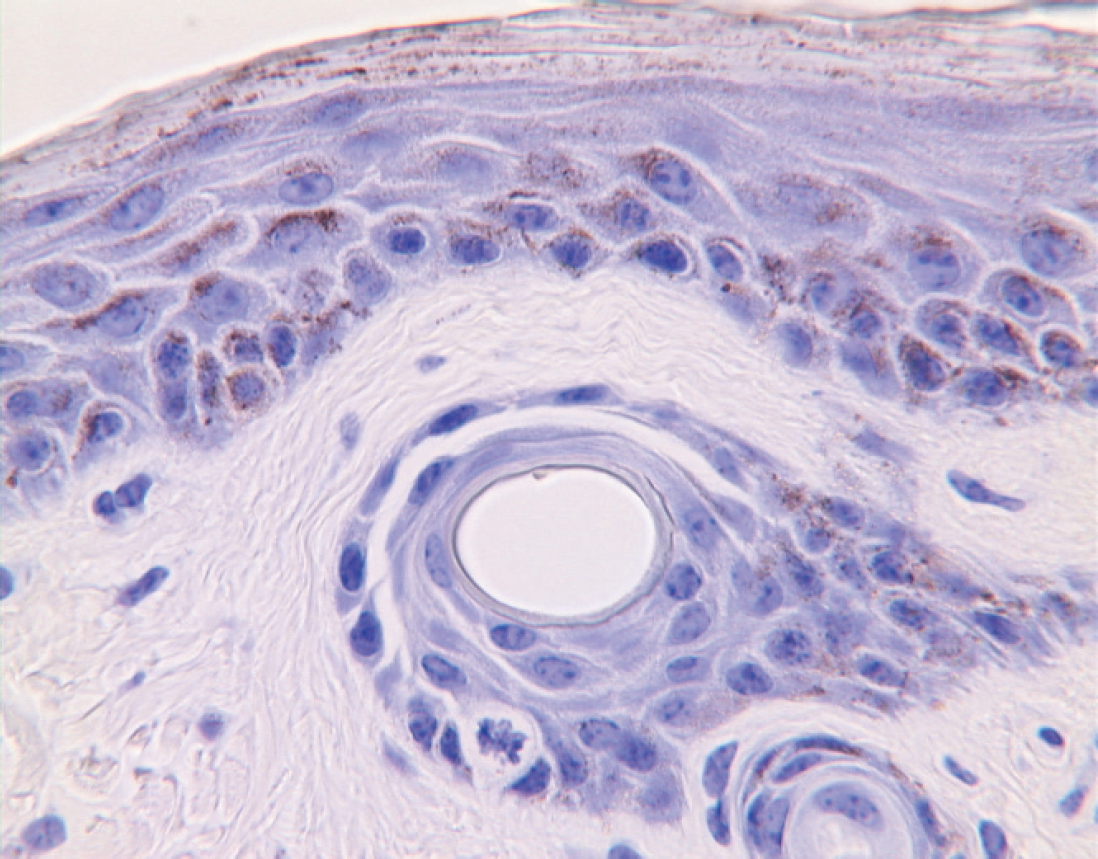
Skin sample from a white-tailed deer positive for bovine viral diarrhea virus antigen by immunohistochemical testing.
The VN results indicate that BVDV infection occurred in 2 of the 165 sampled deer. The low titer (1:8) found in one of the samples may indicate a false-positive VN; increasing the cutoff for positive samples to a higher VN titer may increase the specificity of the test, while decreasing its sensitivity. VN is a common serologic method to determine BVDV-specific antibodies; it allows quantification of serum-neutralizing antibody titers. In samples obtained from heterologous species, it may be difficult to determine which strain to use for VN as there exists great antigenic heterogeneity among strains of BVDV in cattle, and this may be exacerbated by passage in a different host. Additionally, there is no universally accepted reference strain for BVDV VN, further complicating the choice of the strain used for this procedure. VN cannot detect acute infections before anti-BVDV antibodies have formed, and the low titer found in one of the serum samples could have resulted from a very recent exposure. A low antibody titer could furthermore result from a transient infection that occurred long before VN testing.
Surveys for BVDV-specific antibodies have been performed in a number of countries and many species of wildlife and have identified a highly variable BVDV seroprevalence in different free-ranging wildlife populations. Similarly, serosurveys conducted in North America also demonstrate variable results. Bovine viral diarrhea antibody prevalence in North American ungulates was reported to be 0% on Anticosti Island, Quebec, 23 3% in Alaska, 28 3% in New York state, 18 4% in Alberta and Saskatchewan, 4 34% in New Mexico, 9 59% in selected national parks of the western United States, 1 60% in Wyoming, 26 and 60.7%/69.3% in Quebec. 13 Reasons for this variation among reported seroprevalence rates are unknown. It is plausible that increased contact with cattle causes a greater BVDV seroprevalence in wildlife; however, this was not true in a previous study comparing the BVDV antibody prevalence of wild Cervidae from areas of different cattle densities. 15 Much of the known data on BVDV seroprevalence stems from distinct and often isolated areas and populations of wildlife, such as from wildlife refuges and national parks. Exact epidemiologic implications for statewide or national BVDV-control programs and the role of wildlife in BVDV maintenance and transmission may therefore be difficult to extrapolate from these studies. However, evidence of BVDV occurring in free-ranging wildlife and high prevalence rates reported for some wildlife populations, indicating intrapopulation maintenance of BVDV, 26 should encourage further systematic prevalence surveys.
The importance of PI animals in the epidemiology of BVDV has prompted the development and evaluation of different assays for antigen detection. IHC on skin biopsies (i.e., ear notch) has been successfully used since the 1990s to identify PI cattle. The test shows high agreement with results of microtiter virus isolation. 7 Immunohistochemical detection of animals PI with BVDV is not affected by colostral antibodies, and TIs only rarely cause false-positive results. 7 In a recent study, IHC was performed on a skin biopsy collected from a white-tailed deer fawn, known to be PI with BVDV. 20 Immunohistochemical stain distribution in the skin sample from this PI fawn resembled that found in PI cattle. Viral antigen was identified in only one skin biopsy in the present study and the stain distribution was similar to that found in PI cattle and PI white-tailed deer. Unfortunately, a blood sample was not available from the deer from which the ear notch had been taken, making further testing by VN or virus isolation impossible. However, given the usefulness of IHC for identifying PI and its high agreement with microtiter virus isolation in cattle, it is presumed that this white-tailed deer was PI with BVDV.
In cattle, TI may rarely cause positive antigen staining in ear notch samples. 7 In samples from TI cattle, BVDV antigen is distributed multifocally in the epidermis and infundibula of hair follicles, 19 rather than diffusely as found in this deer. Experimental TI in calves did not result in positive IHC in one study. 21 The likelihood of positive IHC in TI cattle increases when high doses of BVDV inoculum are administered to calves experimentally 19 or when a large number of PI cattle are in a herd. 7 The exposure of the IHC-positive deer to high titers of BVDV is unlikely, indicating PI rather than TI BVDV in this animal.
To assess the possibility of cross-reactivity of M Ab 15c5 with non-BVDV antigen, resulting in a false-positive result, another BVDV M Ab (3.12F1) was used in a second IHC on the suspect ear notch. Interestingly, this MAb resulted in identification of the same antigen distribution within the suspect specimen, underscoring the correct identification of BVDV antigen by MAb 15c5. Further research is needed to evaluate BVDV antigen distribution in skin of white-tailed deer and the value of IHC for identifying PI animals in heterologous species.
In cattle herds, presence of a PI animal results in seroconversion in a large percentage of in-contact herd mates. In white-tailed deer, a similarly high seroprevalence would be expected when a PI animal is present within a sampled population. In contrast, a previous study detected a 60% BVDV seroprevalence rate in mule deer sampled from the same geographic area as a clinically ill BVDV isolation-positive mule deer. 26 The low seroprevalence rate detected in the present study may be the result of more widespread sampling from across the state of Alabama rather than sampling within a limited population containing a BVDV-positive animal. It is unlikely that multiple deer from the same group were presented to the deer processor on the same sampling day. No other samples obtained from deer at the location where the IHC-positive sample was collected were positive for BVDV or seropositive to BVDV; therefore, the IHC-positive deer may represent a locally contained infection that has not spread to other populations of white-tailed deer within the region.
Although it has been demonstrated that white-tailed deer can be infected with BVDV, the main route of transmission in free-ranging animals has not been elucidated. 20,27 While some authors, 1,4 presumed that close proximity to cattle caused high seroprevalence rates in wildlife, others hypothesized that an independent cycle is responsible for intrapopulation persistence. 12,15 Transmission of BVDV among individuals of a white-tailed deer population may be dependent on a variety of factors, including amount and degree of individual contact, human interaction in the form of wildlife management, and survivorship of persistently infected animals. With seasonal variation, a female white-tailed deer normally has limited contact with other deer in her home range, except for those within her matrilineal group, 2,17 which may preclude BVDV transmission to a large number of other animals.
Recently, white-tailed deer in Michigan have been identified as a reservoir for Mycobacterium bovis. 24 The maintenance of tuberculosis in free-ranging white-tailed deer resulted largely from increasing deer densities in the state of Michigan and the practice of baiting and winter feeding for hunting purposes, which resulted in closer contact of animals than in populations where supplemental feeding is less prevalent. 24 In Alabama, supplemental feeding during the hunting season is illegal, and the harsh winters that force deer to commingle around limited feeding areas in northern regions are not as influential. Therefore, BVDV may be contained within distinct social groups in white-tailed deer in Alabama.
The low BVDV seroprevalence rate detected in white-tailed deer in Alabama corroborates previous work in this species but contrasts with reports of high anti-BVDV antibody rates detected in other wildlife species. Therefore, determining the PI prevalence rather than seroprevalence may more accurately assess the role of white-tailed deer in BVDV control or eradication programs.
Acknowledgements. This study was supported by Food Animal Research Funds administered by Animal Health Research, Auburn University. The authors thank the participating students of the School of Forestry and Wildlife for devoting time to collecting and transporting samples. In addition, the authors appreciate the efforts and expertise provided by the technical staff of the BVDV laboratories at Auburn University, especially Ms. Anne Cochran, Ms. Terri Hathcock, and Ms. Terri Woods.
Footnotes
a.
Minimum Essential Medium, Sigma Chemical Co., St. Louis. MO.
b.
Equine Serum, Sigma Chemical Co., St. Louis, MO.
c.
L-glutamine (100X), Gibco BRL, Life Technologies, Grand Island, NY.
d.
Penicillin-streptomycin solution (100X), Sigma Chemical Co., St. Louis, MO.
e.
BVD MAb 15c5, Syracuse Bioanalytical, East Syracuse, NY.
f.
BVDV MAb 3.12F1, Oklahoma Animal Disease Diagnostic Laboratory, Stillwater, OK.
