Abstract
This communication documents age-associated pathologic changes and final observations on experimental transmission of chronic wasting disease (CWD) by the intracerebral route to raccoons (Procyon lotor). Four kits were inoculated intracerebrally with a brain suspension from mule deer with CWD. Two uninoculated kits served as controls. One CWD-inoculated raccoon was humanely killed at 38 months after inoculation, and 1 control animal died at 68 months after inoculation. Both animals had lesions that were unrelated to transmissible spongiform encephalopathy. Six years after inoculation, none of the 3 remaining CWD-inoculated raccoons had shown clinical signs of neurologic disorder, and the experiment was terminated. Spongiform encephalopathy was not observed by light microscopy, and the presence of abnormal prion protein (PrPd) was not detected by either immunohistochemistry or Western blot techniques. Age- related lesions observed in these raccoons included islet-cell pancreatic amyloidosis (5/6), cystic endometrial hyperplasia (3/4), cerebrovascular mineralization (5/6), neuroaxonal degeneration (3/6), transitional-cell adenoma of the urinary bladder (1/6), and myocardial inclusions (4/6). The latter 2 pathologic conditions were not previously reported in raccoons.
Chronic wasting disease (CWD) is a fatal transmissible spongiform encephalopathy (TSE) or prion disease, which was identified in captive and free-ranging cervids 20 and moose. 3 Affected animals show accumulation of disease associated abnormal form of prion protein (PrPd) in tissues of the central nervous system (CNS) and the lymphatic system. Detection of PrPd in these tissues and characteristic histopathologic changes in the brain are the basis of currently available diagnostic methods for TSEs. 14
CWD has been experimentally transmitted by intracerebral inoculation of affected brain from mule deer into a variety of animal species, including cattle, 12 goat, 20 and sheep. 9 Recently, results of experiments in which raccoons were intracerebrally inoculated with 3 different TSE agents (transmissible mink encephalopathy [TME], sheep scrapie, and CWD) were reported. 11,13 Raccoons inoculated with TME died within 6 months after inoculation (MPI), 13 and those inoculated with scrapie died within 2 years after inoculation (YPI), 11 whereas CWD-inoculated raccoons remained alive and healthy at 3 YPI. 11 The present communication documents age-related findings in the CWD-inoculated raccoons at the termination of the study 6 YPI. The detailed design of this study was previously published. 11,13 Two kits (controls) were not inoculated (Table 1).
Animals were euthanized with a pentobarbital overdose, and a complete necropsy was conducted on each of the carcasses. Representative samples of liver, kidney, spleen, skin, striated muscles (heart, tongue, diaphragm, masseter), thoracic aorta, thyroid gland, turbinates, trachea, lung, tonsils, esophagus, stomach, intestine (duodenum, jejunum, ileum, recto-anal area), anal sacs, salivary gland, adrenal gland, urinary bladder, lymph nodes (retro-pharyngeal, mesenteric), nerves (optic, trigeminal), pituitary gland, Gasserian ganglion, and brain (except for a small portion of frontal cortex that was frozen) were immersion fixed in 10% neutral buffered formalin. One eye (retina) was fixed in Bouin solution. The brain was cut longitudinally; one half was fixed in formalin and the other half was frozen. The formalin-fixed brain was cut into 2–4-mm-wide coronal sections. Sections of various anatomic sites (a minimum of 5 hemisections of brain per animal), rostral cerebrum, hippocampus, superior colliculus, cerebellum, brainstem (at the level of obex) were processed for routine histopathology, embedded in paraffin wax, sectioned at 5 μm, and stained with hematoxylin and eosin. Sections of heart were also stained with periodic acid-Schiff (PAS), with and without diastase pretreatment. The CNS sections were stained by using an immunohistochemical (IHC) method for detection of PrPd as previously described. 11,13 A Western blot method a was used on frozen brainstem samples as previously described. 18
Findings in control and intracerebrally inoculated raccoons with chronic wasting disease agent from mule deer.∗
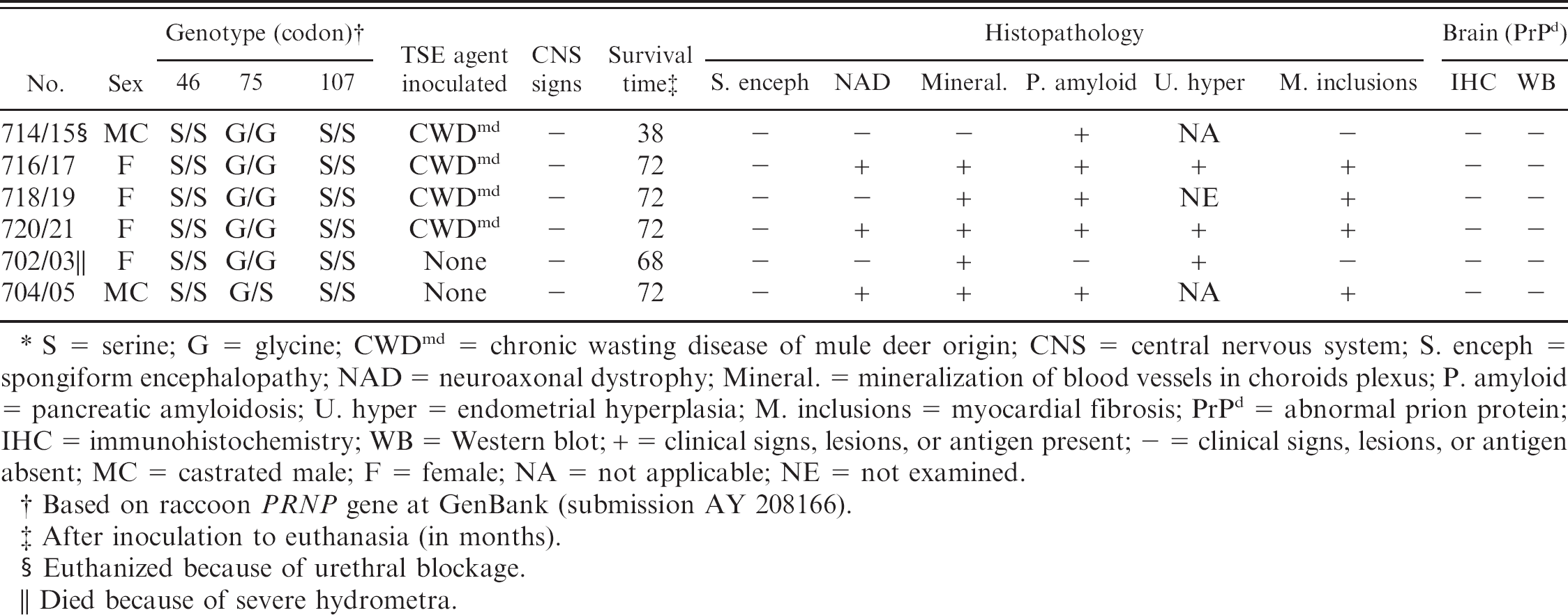
S = serine; G = glycine; CWDmd = chronic wasting disease of mule deer origin; CNS = central nervous system; S. enceph = spongiform encephalopathy; NAD = neuroaxonal dystrophy; Mineral. = mineralization of blood vessels in choroids plexus; P. amyloid = pancreatic amyloidosis; U. hyper = endometrial hyperplasia; M. inclusions = myocardial fibrosis; PrPd = abnormal prion protein; IHC = immunohistochemistry; WB = Western blot; + = clinical signs, lesions, or antigen present; – = clinical signs, lesions, or antigen absent; MC = castrated male; F = female; NA = not applicable; NE = not examined.
Based on raccoon PRNP gene at GenBank (submission AY 208166).
After inoculation to euthanasia (in months).
Euthanized because of urethral blockage.
Died because of severe hydrometra.
Frozen brain tissue homogenates (10% in phosphate buffered saline [PBS]) were used for characterization of the raccoon prion protein (PRNP) gene polymorphism as described previously. 10 The following primers were used for the amplification of PRNP sequences spanning codons 23–243 of the open reading frame; forward primer 5′-CTG CAA GAA GCG GCC GAA GCC-3′ and reverse primer 5′-CAC GGG CGG CGG GGA GAA GAG GAT-3′. Briefly, RNA was isolated from 10% homogenates of frozen brains in PBS, and the reverse transcriptase PCR was performed as previously described. 10 Amplified DNA products were purified, and sequencing was performed by using the ABI prism 3700 DNA sequencer b with Big Dye Terminator b chemistry with the forward and reverse primers. Two raccoons died or were humanely killed before the termination of the experiment at 6 YPI (Table 1). Raccoon no. 714/15 developed sudden anorexia and extreme lethargy, and was euthanized at 38 MPI. A necropsy revealed complete urethral blockage, hemorrhagic cystitis, and mild bilateral hydronephrosis.
At 68 MPI, one of the control raccoons (no. 702/03, Table 1) died. The uterus was markedly distended and appeared to occupy the majority of space in the abdomen (Fig. 1). The uterine wall was uniformly thin (Fig. 2), and the lumen contained approximately 300 ml of clear watery liquid. The oviducts were moderately thickened, and the ovaries appeared small. The stomach was moderately distended (Fig. 1) and contained approximately 50 ml of dark black mucoid material and had multifocal hemorrhages. The intestinal tract appeared compressed (Fig. 1) and did not contain any digesta.
Microscopic lesions were confined to the uterus and the oviducts, and the lesions were bilateral. The uterine wall was diffusely thin (Fig. 3), with endometrium that showed multifocal areas of adenomatous hyperplasia and cystic dilatations, with the presence of variable amount of pink flocculent material within some of the cysts (Fig. 3). Occasional cysts contained small numbers of inflammatory cells. The Fallopian tubes had bilateral diffuse adenomatous proliferation of mucosa (Fig. 4). The proliferated cells were darkly stained, had indistinct cell borders, and were arranged in a 2- to 4-cell-thick layer; mitotic figures were absent. Both animals were negative for lesions of spongiform encephalopathy, and their tissues did not show the presence of abnormal prion protein (PrPd). The 3 remaining experimental animals and the control raccoon were euthanized at 72 MPI (Table 1).
On microscopy, the dorsal medulla of 3 of 6 raccoons had bilateral discrete foci, with a few swollen axons and isolated foci of vacuolation in the neuropil (Fig. 5; Table 1). These were suggestive of neuroaxonal dystrophy (NAD). 15 Also, foci of mineralizations were observed in blood vessels of the choroid plexus in 5 of 6 raccoons (Fig. 6; Table 1). The affected vessels were small and multifocally affected. Some mineralized foci had the characteristic laminated appearance of psammoma bodies. 7

Abdominal cavity of raccoon no. 702/03. There is extensive diffuse enlargement of uterus. The stomach (S) is moderately distended and the intestines (IN) appear to be atrophic.

Uterus of raccoon no. 702/03 removed from the animal. There is bilateral diffuse uterine distention from fluid accumulation (hydrometra).

Uterus of raccoon no. 702/03. The endometrium is hyperplastic and has multifocal cysts. Some of the cysts contain flocculent material in their lumina. Hematoxylin and eosin. Bar = 200 μm.

Oviduct of raccoon no. 702/03. The mucosal surface shows marked diffuse adenomatous hyperplasia. Hematoxylin and eosin. Bar = 200 μm.

Dorsal medulla oblongata of raccoon no. 704/05 with neuroaxonal dystrophy. Note the presence of 3 swollen axons (arrows). Hematoxylin and eosin. Bar = 90 μm.

Brain of raccoon no. 716/17. Note the presence of multifocal mineralized foci (psammoma bodies) within the choroids plexus (arrow). Hematoxylin and eosin. Bar 5 25 μm.

Pancreas of raccoon no. 714/15. The 2 islets of Langerhans contain eosinophilic material (amyloid), which has destroyed the normal architecture. Hematoxylin and eosin. Bar = 60 μm.

Urinary bladder of raccoon no. 120/21 showing papillary transitional-cell neoplasm and some normal uroepithelium (arrow). Hematoxylin and eosin. Bar = 250 μm.

Left ventricular myocardium of raccoon no. 704/05 with the presence of eosinophilic homogeneous inclusions (arrows) within cross sections of myofibers. Hematoxylin and eosin. Bar = 35 μm.

Left ventricular myocardium of raccoon no. 704/05 with the presence of an eosinophilic homogeneous inclusion within longitudinal section of a myofiber. Hematoxylin and eosin. Bar =15 μm.
Five of 6 raccoons revealed microscopic lesions in pancreas (Table 1). In all cases, the lesions consisted of presence of eosinophilic amorphous material that separated and effaced normal tissue in the islets of Langerhans (Fig. 7). On Congo red stain, this eosinophilic material revealed apple-green birefringence when examined under polarized light, consistent with presence of amyloid deposits.
A neoplasm was seen in 1 raccoon (no. 718/19). This was in the urinary bladder and consisted of multifocal papillary proliferation of mucosa. Within the proliferated areas, there were a few dilated cystic cavities, some that contained eosinophilic homogeneous material (Fig. 8). Mitotic figures were not evident. The lesion was diagnosed as a transitional papillary adenoma.
Microscopic lesions were seen in the myocardium of 4 raccoons. All had multifocal randomly distributed inclusions (either eosinophilic or basophilic) in cytoplasm of myocardiocytes (Table 1). Occasional inclusions appeared to be located extracellularly. On cross section, they appeared generally round and varied from 20 to 70 μm in diameter (Fig. 9). However, on longitudinal section, they were cigar shaped (up to 100 μm) and their ends appeared to blend with the longitudinal striations of the myofibers (Fig. 10). The basophilic inclusions stained PAS positive and were diastase resistant. In 1 raccoon (no. 05–892) the inclusions were seen within isolated myocytes that were present in areas of myocardial fibrosis. Significant microscopic lesions were not seen in other tissue sections.
The open reading frame (ORF) of the PRNP gene between codons 23–243 of all raccoons used in this study was sequenced. One nonsynonymous polymorphism at codon 75 (Gly/Ser) was detected in the PrP sequence of raccoon no. 704/05, the other animals were homozygous for Gly at this position (Table 1).
Raccoons are found throughout North America. Because they are omnivores and their diet may include carrion, it is very likely that, in the wild, they would be exposed to carcasses of animals with TSEs. 5 Although naturally occurring TSEs have not been described in raccoons, experimental transmission of TME and sheep scrapie were documented via intracerebral 11,13 and oral inoculations. 5
In this study, characteristic microscopic lesions of spongiform encephalopathy 10,11,13 were not seen in any of the CWD-inoculated raccoons. Similarly, PrPd was not detected in the brain tissues by either IHC or Western blot. The same CWD inoculum when given intracerebrally to cattle and sheep resulted in 38% and 25% positive (for PrPd) animals, respectively. 9,12 Therefore, the present findings indicated that raccoons were not capable of amplifying the CWD agent of mule deer origin. This observation and our previous findings 10,11,13 on the incubation periods of other animal TSEs in raccoons (TME within 6 months and sheep scrapie within 24 months) indicate that the raccoon model may be a useful biological assay for differentiating TME, scrapie, and CWD. However, further studies are necessary before raccoons can be recommended as a biological model for TSE strain differentiation. Studies such as experimental inoculation of BSE (classical and atypical) need to be evaluated in this species. Because the BSE agent appears to be more promiscuous than other TSE agents (i.e., easily transmitted to other species, such as sheep, wild ungulates, cats, and primates), it may have a shorter incubation period than scrapie or TME in raccoons. Also, experimental inoculations of raccoons by using CWD isolates from other cervid species, such as elk and white-tailed deer and moose, would be useful. Data from these types of studies will also contribute to our knowledge of the behavior of different strains of TSEs in raccoons.
The presence of mineralization (psammoma bodies) in choroid plexuses of 5 raccoons and the observation of NAD in the brainstems of 2 raccoons were not surprising. The former has been observed in wild-caught raccoons as an incidental finding, 7 and the latter has been observed as age-related change in older (>7 years) raccoons. 15
Cystic endometrial hyperplasia and hydrometra are rather rare in wild free-ranging animals. However, they have been documented in artificially confined wild animals, 1 including a raccoon. 8 It is believed that this condition results from prolonged, noncyclic estrogen stimulation or from excessive levels of circulating estrogens. 1 In some species, nulliparity and aging are associated with progressive proliferative changes in the endometrium, 1 and the presently described case had similar predisposing conditions.
In raccoons, deposition of amyloid in pancreatic islet cells appears to be a rather common finding. 2,17 Therefore, in this study, it was not surprising to find such lesions in 5 of 6 raccoons. Because pancreatic amyloidosis has been observed in relatively young wild raccoons, the cause or causes of this condition remains a mystery.
Neoplastic lesions have infrequently been documented in the family Procyonidae. In a retrospective survey of over 400 raccoon necropsies, only 2 cases of neoplasia, an astrocytoma of the brain and a fibroma of the skin, were found. 4 In an aged population of raccoons (zoological facility), 16 tumors were observed. Thirteen of these involved the thyroid glands and one each involved the liver, kidney, and uterus. 16 The documentation of transitional-cell adenoma in the urinary bladder of the raccoon in the present study appears to be the only reported case in this species.
In this study, myocardial inclusions were observed in 4 of 6 raccoons. The inclusions resembled those observed in inclusion-body myositis as seen in humans. The human condition is age related (seen after midlife) and may be associated with certain metabolic disorders. 6 These inclusions are reported to consist of polysaccharide complexes, and similar inclusions were observed in skeletal muscles. 19 Two cases of such inclusions in skeletal muscles of dogs with sarcomas were recently documented. 19 None of the raccoons in this study had inclusions in skeletal muscles. Because no such cases have previously been described in relatively young raccoons in the wild, it seems that the condition is most likely associated with advancing age and appears to be an incidental finding.
In this study, the PRNP genotypes found for the CWD-inoculated raccoons have been shown previosly to be susceptible to both TME and scrapie. 10 Therefore, it seems likely that, if raccoons are susceptible to CWD, then the mule deer–CWD-inoculated raccoons would have succumbed to the disease within 6 YPI, a length of longevity uncommon in the wild.
Acknowledgements. The authors thank Drs. L. Nusz and J. Laufer for clinical assistance. Expert technical assistance was provided by Martha Church, Dennis Orcutt, Deb Clouser, Kevin Hassall, Sharla Van Roekel, Ginny Montgomery, and animal caretakers at the National Animal Disease Center (NADC). The authors would also like to acknowledge their coauthor Elizabeth S. Williams who passed away on December 29, 2004. This study was carried out under the guidelines of the institutional Animal Care and Use Committee at NADC. Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture.
Footnotes
a.
Prionics-Check, Schlieren-Zurich, Switzerland.
b.
Applied Biosystems, Foster City, CA.
